Abstract
Spine fusion is used to treat traumatic or degenerative lumbar instability in the cervical or lumbar spine. Although degenerative radiological changes in discs adjacent to a fusion have been well-recognized, histopathological changes in adjacent discs have not been studied and are poorly understood. An economical small animal model for lumbar fusion would be a useful research tool. Study objectives were to: (1) develop a model of non-instrumented spine fusion in the sand rat, a rodent with spontaneous, age-related disc degeneration; (2) use radiological–histological analyses to study fusion and disc degeneration in cranial and caudal discs adjacent to the fusion. Studies were approved by our Institutional Animal Care and Use Committee. A small segment of outer annulus tissue was surgically removed from lumbar discs, radiographs obtained and the animal allowed to recover and age. At surgical harvest, radiographs of 28 spine fusion specimens were scored and statistically analysed for adjacent disc space narrowing, wedging, endplate sclerosis and irregular disc margins. At harvest, the incidence of these radiological indices of disc degeneration were significantly greater than at time of surgery. Pilot studies presented here indicate that this model provides a novel addition to basic science approaches studying the clinically important topic of spinal fusion and adjacent segment changes. The resulting fusion site can be assessed statistically with radiological scoring to determine development/progression of disc degeneration in adjacent segments, and correlative histological features can be examined. The sand rat is well-suited for use in spine fusion studies because of reliable/reproducible progression of disc degeneration and the favourable economics of small rodent studies.
Spine fusion is increasingly used to treat traumatic or degenerative lumbar instability in the cervical or lumbar spine. 1 Patients have reported pain following spine fusion; such pain has been attributed to either an extended range of motion in the adjacent, non-fixed vertebrae, or to degenerative changes in adjacent discs or facet joints caused by mechanical loading after fusion. Although the degenerative radiological changes in discs adjacent to a fusion have been well recognized, the histopathological changes that develop in the adjacent discs have not been studied and are not well understood. An economical small animal model for lumbar fusion would therefore be a useful research tool in investigations of lumbar fusion and adjacent segment changes.
Recently, there has been increased discussion and controversy concerning the potential for long-term adjacent segment degeneration cephalad or caudad to the fused site. Although there is a growing body of literature on this topic, opinions remain diverse.
The true frequency of degeneration in adjacent discs, and the clinical implications of such radiological changes, are unknown. 2,3 Some reports suggest that adjacent segment disease may not merely be a complication of lumbar fusion. 1,4,5 Other investigators have suggested that the individual's specific genetic predisposition to develop degenerative spondylosis overshadows the effect of fusion itself. 6 It has also been suggested that the effects from fusion may in fact be minor compared with the progression of the natural history of degenerative disc disease. 7
Long-term clinical studies include a 10-year follow-up, 1 and an evaluation of 374 patients in a follow-up over 21 years that reported an overall prevalence of adjacent segment disease of 14.2% with a progression rate of 2.9% annually. 8 This latter study was not able to identify any relationship between initial radiological degenerative features in the adjacent levels and subsequent development of adjacent segment disease. This work supports the interpretation that adjacent segment disease results from continuing disc degeneration and is not a late complication of fusion. 7
Another retrospective study found no adverse impact on outcome when the lumbar fusion was adjacent to a degenerative site. 3 In this evaluation, 80% of the patients had disc degeneration at a level adjacent to the fusion. The authors emphasized the complexity of this problem, and noted the importance of considering the length of follow-up post-fusion and the underlying complication that normal physiological aging might produce similar symptoms over time.
In contrast are the findings of a retrospective study of adjacent segment degeneration in the lumbar spine which found that 27.4% of 215 patients showed evidence of degeneration at adjacent levels, with new disease at an adjacent level developing at a rate of 2.9% per year. 9 This analysis, however, was not able to show a relationship between preoperative status of the adjacent segments and outcome.
In summary, it appears from the available clinical and sparse basic science information that a combination of mechanical factors and genetic predisposition to degenerative disc disease plays a role in the development of adjacent segment disc disease. Park et al. 10 have provided a concise review of the literature on adjacent segment disease, and major risk factors included instrumentation, fusion length, sagittal malalignment, age, facet injury and pre-existing degenerative changes. It has also been pointed out that most evidence currently points to a number of factors in the development of adjacent segment disease, including increased stress on facets and the adjacent disc, increased adjacent segment mobility and increased intradiscal pressure. 11
In this study, we present pilot data on a new small animal model for the study of fusion and adjacent segment changes. The sand rat is an excellent model for such studies because the radiological signs of disc degeneration are clear, reliable as the animal ages and are well-documented. 12–15 The modest costs associated with rodent housing are also an economic advantage.
Methods
All animal studies were performed following approval by the Institutional Animal Care and Use Committee. Psammomys obesus was obtained from our sand rat colony (which is maintained in a separate room as an exotic species), and were housed one to two per cage (using a high-impact transparent plastic cage autoclaved between changes). Bedding was 3.175 mm corncob; a 12:12 h light–dark cycle was utilized. The housing room was maintained at 30% humidity and 22.2°C. Animals were provided tap water and Purina sand rat diet #5L09 ad libitum. This diet is a special low caloric formulation with 10132.056 J metabolizable energy, which prevents the development of diabetes seen with high caloric ad libitum feeding. Animals were identified by IMI-10000 Implantable Micro Identification chips (BioMedic Data Systems, Inc, Maywood, NJ, USA).
Surgical procedure to induce lumbar vertebral fusion
The sand rat was sedated using a 3:2:1.5 solution NaCl/ketamine (Fort Dodge, Fort Dodge, IA, USA)/xylazine (Phoenix Pharmaceuticals Inc, St Joseph, MO, USA) injected i.p. at a dose of 0.75 mL/kg. Following sedation, a lateral X-ray was taken to determine normal spinal anatomy and to determine the lumbar disc to be operated upon to induce fusion. The ventral side of the sand rat was shaved and the animal attached to an anaesthetic machine using a rodent circuit. A flow of 2 L isoflurane (Abbott Laboratories, North Chicago, IL, USA) and 1 L of O2 was maintained throughout surgery. Following betadine and alcohol scrubs, a 2.5–3.8 cm incision was made down the centre of the abdomen and the internal organs gently pulled aside using Freer elevators to provide access to the spine. The aortic bifurcation was located and the vessels gently pulled aside to expose disc sites L4–5, L5–6 or L6–7. Muscle was gently scraped away using a Lempert elevator and any excess fluid removed using absorption spears. A ‘V’-shaped slice of annulus was cut using a micro blade (1.5 mm) and disc tissue removed by curettage. A vascular clip was clamped onto nearby muscle to mark the surgery site for future radiological studies. The abdominal cavity was then gently flushed with sterile saline, and organs gently repositioned. The abdominal muscle layer was closed with 4-0 Vicryl (Ethicon, Somerville, NJ, USA) and the skin layer closed with 5-0 Vicryl (Ethicon), and then stapled using a sterile Visistat stapler (Weck, Research Triangle Park, NC, USA). A postsurgical lateral X-ray was taken. The animal was placed in a rodent cage on top of a heating pad and allowed to recover for 24 h. Food and water were placed on the bottom of the cage. Surgical animals were returned to the colony and individually housed. Following surgery, the animals were treated with ketoprophen (2 mg/kg) (at least once at the end of surgery and then every 6–12 h as needed) and buprenorphine (0.05 mg/kg, administered once) at the end of surgery for pain control. A final lateral X-ray was obtained immediately prior to animal sacrifice. Euthanasia was performed using pentobarbital administered i.p., 300 mg/kg (concentration 50 mg/mL).
At tissue harvest, lumbar vertebral specimens were fixed in 10% neutral buffered formalin, vascular clips removed, the lumbar spine decalcified, embedded in paraffin, sectioned in the sagittal plane and stained with Masson-trichrome.
General experimental design
These studies were designed to provide initial pilot data on the formation of spine fusion following removal of small segments of disc tissue as described above. The study design consisted of evaluation of lateral radiographs obtained at the time of surgery, evaluation of lateral radiographs obtained at euthanasia and morphological studies on the fusion and adjacent sites. The ages of animals tested in this design ranged from one to 10 months of age, and averaged 5.05 months (Table 1). Various time intervals were used postsurgery (Table 1); the average number of months posturgery was 10.9, and ranged from one to 26 months (Table 1).
Demographic features and total radiographic score of the sand rat fusion study population
Scoring of radiological features and statistical analysis
Surgical and harvest radiographs of 28 spine fusion specimens were scored and statistically analysed for development or progression of disc space narrowing, wedging, endplate sclerosis and irregular disc margins. Absence of a feature was recorded as 0; presence was recorded as 1. A total score value was then calculated which summed-up the scores for narrowing, wedging, sclerosis and irregular disc margins. Cranial and caudal adjacent discs were each evaluated in this manner. In order to test whether cranial or caudal sites showed more degeneration after fusion, data were analysed with the paired Wilcoxon signed rank test. Standard statistical methods were performed using SAS software (version 8.2, SAS Institute, Cary, NC, USA). Data are presented as means ± SD (n). A P value of 0.05 was considered statistically significant. To assess radiological features at surgery vs. harvest, analysis of these categorical data (presence or absence of the feature) used the McNemar test for paired data.
Results
To present this new fusion model, 28 fusions in the sand rat lumbar spine were analysed here for morphological and for radiological features of the adjacent segments. Table 1 presents the demographic features of the study population. Mean animal age at surgery was five months, and the mean number of months postsurgery to harvest of the fused spinal segment was 10.9 months.
Radiological and histological findings
Radiographs obtained postsurgery and at spine harvest were scored and statistically analysed for development or progression of disc space narrowing, wedging, endplate sclerosis and irregular disc margins as described above. Absence of a feature was recorded as 0; presence was recorded as 1. Cranial and caudal adjacent discs were each evaluated for their total summed-up scores (Tables 1 and 2). In order to test whether cranial or caudal sites showed more radiological signs of degeneration after fusion (Table 2), analysis of these categorical data (presence or absence of the feature) used McNemar test for paired data. (Additional analysis found no significance differences in cranial vs. caudal data based on either age at surgery or the number of months between surgery and harvest.)
Analysis of radiological features of disc degeneration at time of surgery vs. time of harvest*
*To assess these radiological features at surgery vs. harvest, analysis of these categorical data (presence or absence of the feature) used McNemar test for paired data
In all radiology and histology images presented below, the ventral face of the spine is on the bottom, and the dorsal face on the top of the illustration. Cranial direction is to the right in the figure. Histology images show Masson-trichrome stain in which bone and denser connective tissue are stained blue; cartilage and the nucleus pulposus appear white.
The study design employed here utilized a lateral radiograph at the time of disc surgery, an interval postsurgery and a final lateral radiograph obtained at the time of euthanasia and harvest of the spine for histological studies. The radiological and histological features of a fusion formed following the surgical procedure on a four-month-old sand rat. Spine radiological features at the time of surgery at age three months in a sand rat, and a vascular clamp placed in soft tissues near the surgical disc site (Figure 1A) are compared and contrasted with radiological findings at euthanasia and harvest of the fusion site 26 months later (Figure 1B). A large bony fusion mass was present at the site of surgery (Figures 1B and C). Histological examination of the excised spine documented that the ventral portion of the fusion mass consisted of a bone, which was relatively solid in the ventral portion. The dorsal portion of the fusion site showed remnants of fused vertebral growth plates and only a small markedly degenerated segment of disc tissue. Annulus tissue present in the cranial adjacent disc showed prominent wedging (Figure 1C, disc on the right). In the adjacent caudal disc (marked by the arrow), the area of nucleus pulposus is reduced (Figure 1C).
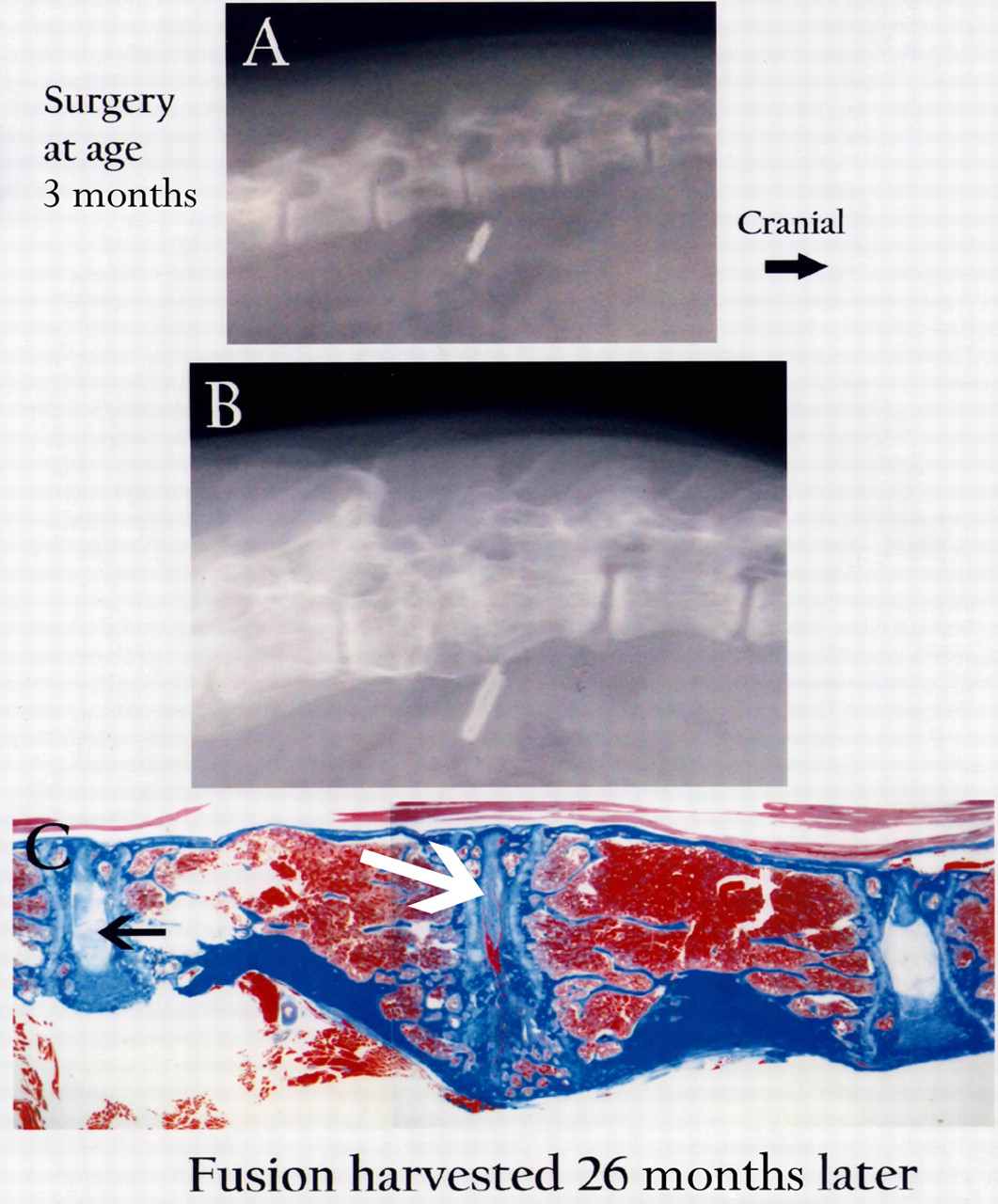
Radiological features at time of surgery at the surgical site (marked by a vascular clamp) and adjacent discs in a three-month-old sand rat (A). At harvest 26 months later, a large fusion mass is present as seen by radiological (B) and histological (C) examination. Only a small region of disc space remains at the fusion site (C, arrow; Masson-trichrome, 6×)
Findings observed in an animal older (8 months of age) at the time of disc surgery, with a shorter interval to euthanasia (8 months) are shown in Figure 2A. The resulting fusion mass at harvest (Figures 2B and C) shows some cartilage remodeling that is still ongoing in the fusion mass as reflected by white-appearing cartilaginous tissue in the ventral region of the fusion site. Note that a bony margin covers the perimeter of the fused area (Figure 2C). The final radiological image shows that dorsal portions of the fused disc show remnants of closed vertebral physes, and a narrow, degenerated remnant of the disc. The disc caudal to the fusion shows prominent wedging and endplate calcification (sclerosis) (Figure 2B).
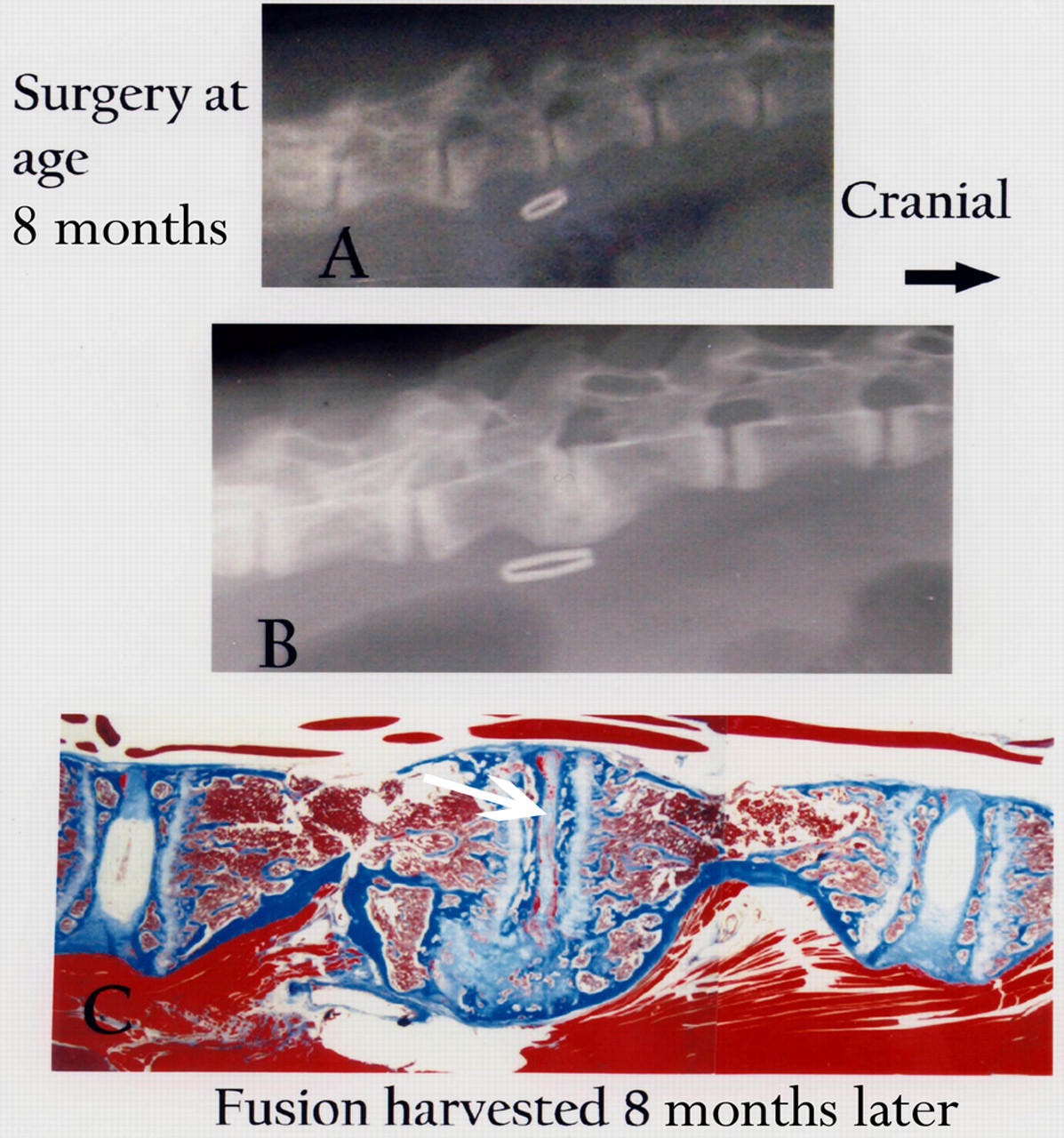
Radiological features at time of surgery at the surgical site (marked by a vascular clamp) and adjacent discs in a sand rat aged eight months. At harvest eight months later, only a thin region of degenerated disc material remains in the fusion site (C, arrow), and the adjacent caudal disc shows prominent wedging and endplate calcification (B) (C, Masson-trichrome, 6×)
Histological images presented to illustrate longer surgical to harvest intervals (4–16 months) are shown in Figure 3 (for animals aged 2 months at surgery [Figures 3E and F], 4 months old at surgery [Figures 3A and B] and animals 9 or 10 months at surgery [Figures 3C and D]). With increasing time postsurgery, the disc tissue present in the site that becomes the fusion mass becomes reduced in size. Cartilaginous tissue is present in the early ventral fusion site (Figures 3A and B). Only rudiments of cartilage, however, remain in the fusion sites apparent in Figures 3C–F. Prominent disc degeneration, with loss of the nucleus pulposus, frequently characterized findings in discs adjacent to large fusion masses (Figures 3C and F).
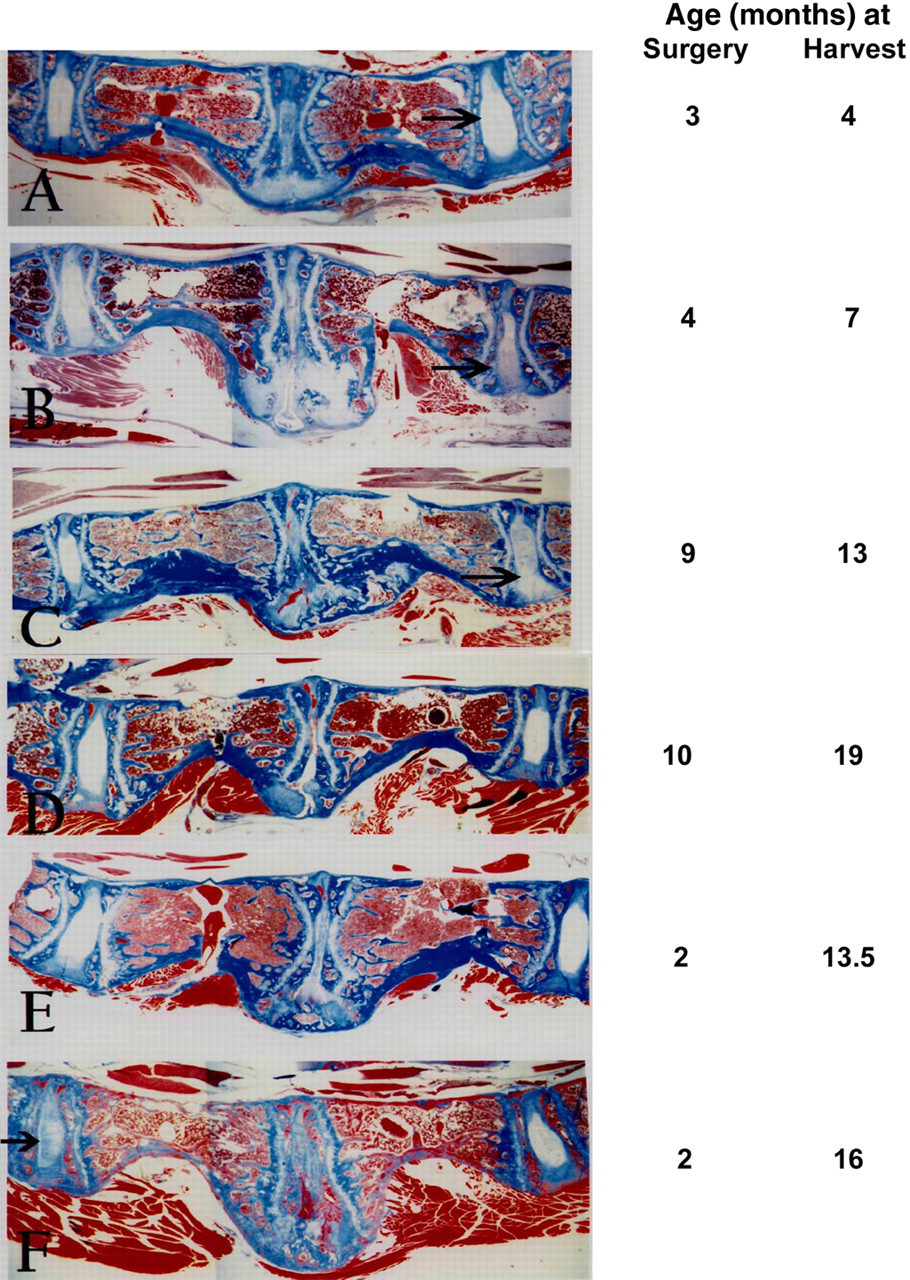
Masson-trichrome-stained histological views of mid-sagittal sections through the lumbar spine at the fusion site. Cranial is to the viewer's right of the fusion mass. Information to the right of each panel indicates the age at disc surgery and the age at harvest (Masson-trichrome, 4×)
Figure 4 illustrates a fusion site formed three months after surgery performed on a four-month-old animal. Prominent wedging is present in the adjacent cranial disc evident in the radiograph. Some cartilage remodelling is still ongoing in the fusion site (the white tissue in the fusion site); a solid bony margin is present with numerous trabecular struts in the ventral fusion body. In the dorsal area, only a narrowed remnant of the disc remains bordered by the calcified blue-stained vertebral endplate. Fused vertebral physes are also present.
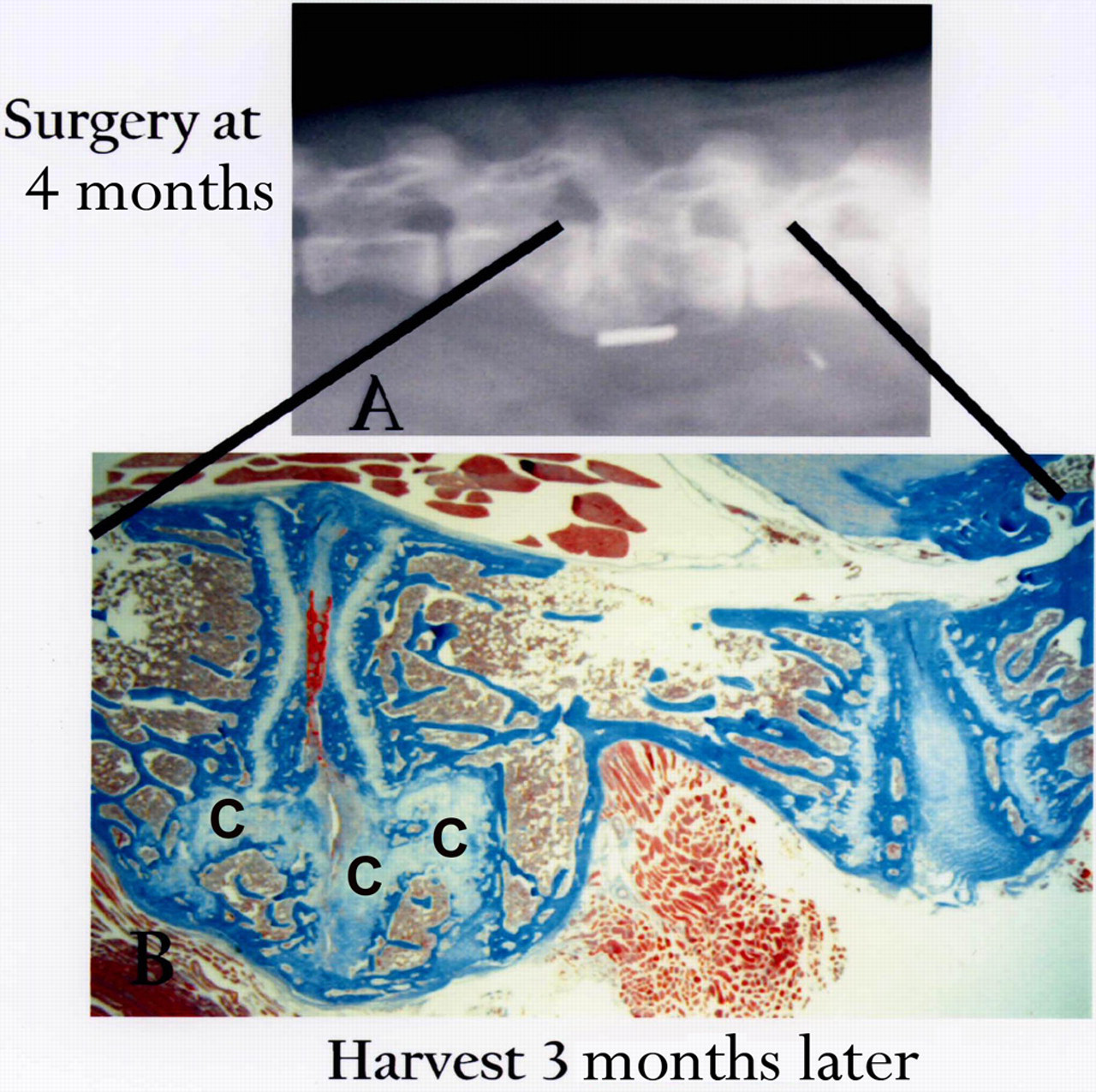
Fusion present three months following surgery performed on a four-month-old animal. Note prominent ongoing cartilage remodelling (C) and wedging in the cranial disc site (right-hand disc) apparent in the radiograph and in corresponding histological image (Masson-trichrome, 11×)
Discussion
The value of animal models in understanding the biology of spine fusion has been previously recognized. 16 Rats, guineapigs, rabbits and canines have all been used in fusion studies. The canine model is quite commonly utilized in experimental spine fusion studies, 17–20 but use of this species has the drawbacks of high purchase price (especially for pure-bred beagles) and high maintenance per diem fees. The study presented here demonstrated that the sand rat is well-suited for use in spine fusion studies because of reliable/reproducible progression of disc degeneration, the opportunity to avoid the use of canines and the favourable economics of small rodent studies.
The sand rat, with age-related spontaneous disc degeneration, is an attractive, well-characterized model 12–15,21–25 that avoids high animal costs and the need to use chemonucleolysis or surgical injury to induce disc degeneration.
In the present study, surgical removal of a segment of annulus tissue resulted in subsequent, non-instrumented fusion in the lumbar spine of the sand rat. Initial radiographs at the time of surgery (where the surgical site was marked by placement nearby a vascular clamp in soft tissue) were compared with changes in segments adjacent to the fusion site at the time of spine harvest. Pilot data presented here were based upon surgeries performed on animals aged two to 10 months, and harvest times ranging from 1 to 26 months postsurgery. Data from our small sample sizes were evaluated with caution. However, analysis of these preliminary data on adjacent segment changes did not find any differences in cranial vs. caudal data based on either age at surgery or time of harvest. Histologically, some variation in cartilage and bone amounts is present in our data. This, however, may represent remodelling which occurs across time as the biomechanical changes in the adjacent segments stabilize.
It is important to note that three previous large studies from our laboratory have examined the spine of aging sand rat with careful radiological criteria; no spontaneous fusions have ever been identified in 134 sand rats aged 10–46 months. 12–14,22,26 Thus, the fusion changes observed in the surgical model here are distinct from the features that characterize normal aging of the lumbar sand rat spine.
The model presented here provides a novel addition to the basic science approach to studying the clinically important topic of spinal fusion and adjacent segment changes. It is important to remember, however, that this model should not be directly compared with human adjacent segment disease, because it is likely that adjacent segment disease seen here would progress more rapidly in this small animal model than it would in humans.
In addition, although human disc degeneration and disc degeneration in the sand rat model share a number of features, including morphological similarities, endplate calcification with aging and loss of notochordal cells in the nucleus pulposus with aging, one must remember that this is indeed an animal model which may have a different underlying pathogenesis of disc degeneration.
This caveat being noted, however, we feel that the data presented here illustrate a model which can be extremely useful for future studies of the effect of fusion on adjacent segments and for the study of potential interventions that might mitigate the changes in adjacent discs. Future studies with this model employing age-matched control groups, specific harvest times and inclusion of sham muscle scraping or disc exposure alone will be especially valuable. Animals should be young at the time of surgery (aged 2–3 months) in order to avoid any confounding issues of age-related disc degeneration in disc sites adjacent to the surgery.
Footnotes
ACKNOWLEDGEMENT
We thank the Carolinas Back Pain Research Endowment for continued support.
