Abstract
At present, no minimally invasive technique exists for the continuous evaluation of the biochemistry of animal and human intervertebral discs, but for research purposes microdialysis may be such a new technique. Thus, the aims of this study were to (1) evaluate if microdialysis can be used successfully in lumbar porcine disc and (2) develop a suitable procedure for the application of microdialysis in the human disc. Conventional specific pathogen-free pigs were used to evaluate cannulation (n = 2) and then insertion of a 10 mm microdialysis membrane, including recovery of a dialysate from the nucleus pulposus (n = 6). The procedure was performed immediately after euthanasia and aided by fluoroscopy to guide the needle and confirm catheter placement. Access to lumbar porcine disc was obtained with an 18 G 2 in. needle applied at a 35–45° angle from the sagittal plane, and took less than 8 min to perform. At a 0.5 μL/min flush rate, dialysates could be recovered and analysable amounts of glucose, lactate and pyruvate were obtained. In one human cadaver, the L4–L5 disc was accessed by a 19 G 3 in. needle inserted at a 35–40° angle. It was possible to apply 10 mm as well as 30 mm microdialysis membranes in the nucleus. In both species the position of the membranes was verified by direct fluoroscopy and with contrast fluid. The results obtained from porcine and human cadavers are promising, and encourage further in vivo studies using microdialysis technique on intervertebral discs.
Human intervertebral disc disorders are among the most common ailments in modern industrial societies. 1,2 Although degenerative disc conditions rank first among musculoskeletal disorders and have serious effects on the individual as well as on society, the pathogeneses of these conditions are not fully understood. 3–5 The morphology of the disc is well described, and consists, in short, of an outer annulus fibrosus, which is organized to resist tensile loads and contain the inner nucleus pulposus. The nucleus pulposus is a gelatinous tissue that contains primarily collagen type II and proteoglycans. However, the physiological and the pathophysiological biochemistry of the disc remain relatively unknown. 6–8 The lack of knowledge concerning normal disc biochemistry in combination with the long time period over which the degenerative process develops further add to the complexity of this understanding.
A degenerated disc can be described as a disc with structural failure combined with accelerated or advanced signs of ageing. 9 In addition to mechanical structural failure, factors such as ageing, genetics, compressive loading, repetitive injury, as well as metabolic and toxic influences, can lead to the degeneration of the disc through biochemical reactions. 10,11
Human tissue necessary to perform studies to evaluate the physiology or pathophysiology of the intervertebral disc is understandably limited due to ethical considerations. Studies on cell cultures and various animal models have contributed valuable data; however, there are significant differences between animal species and humans with respect to disc and spine anatomy, biomechanical factors as well as biochemistry, 12 and care has to be taken when extrapolating these data to human conditions. Thus, more effort is needed to facilitate research on the human disc. 13–15
One technique for biochemical measurements that might enable studies on human structures that are hard to access, such as the disc, is microdialysis. This technique has been proven successful for analyses of metabolites, hormones and growth factors. In addition to the routine clinical use in neurosurgery, microdialysis is also used in a number of other tissues such as skeletal muscle, tendons, fat, skin and liver. 16–18 The microdialysis technique enables continuous tissue monitoring for several days. This is performed by insertion of a microdialysis membrane in the tissue with subsequent recovery of molecules from the interstitial fluid. In this way, components from the interstitial environment which are smaller than the pores of the membrane can be analysed and monitored over time.
For this study, conventional cross-bred specific pathogen-free (SPF) pigs (Yorkshire × Swedish Landrace) were chosen as an animal model because of their similarities to humans, especially with respect to skin, skeletal muscle and weight. 19,20 Pigs have been subjected to microdialysis in a previous study concerning gentamicin concentration in cortical bone. 21
The aims of this cadaveric study were to evaluate if the microdialysis membrane could be inserted into a porcine disc and examine if an adequate dialysate could be retrieved from the nucleus pulposus, given a successful outcome of the pig study, develop a suitable procedure for application in the human disc.
Materials and methods
Microdialysis
One study on pigs and one on a human cadaver were performed with microdialysis equipment (CMA/Microdialysis, Solna, Sweden). In both studies the catheter used was the CMA 71, available in three membrane sizes, all with 100 kDa cut-off and a fluoroscopically visible gold thread in the catheter membrane. The 10 mm membrane version was used in the porcine study and the 10 and 30 mm versions in the human cadaver study. The catheter device consists of an inlet tube, a shaft which in turn consists of a concentric inner and outer tube, a membrane, an outlet tube and microvials for collection of samples. The perfusion fluid is pumped through the inlet tube (600 mm) into the shaft inner tube (60 mm); flows to its distal end; exits the tube and enters the space between the inner tube and the outer dialysis membrane. The dialysis takes place in the space between the inner tube and the outer membrane and the dialysate flows out through the shaft outer tube (60 mm) via the outlet tube (220 mm) into the distal vial. The set-up can be seen in Figure 1. The dead volume of the catheter outlet is 5.1 μL from the membrane to the microvial. In the animal study, the CMA 107 microdialysis pump was used with an adjustable flow rate of 0.1–5.0 μL/min, and the T1 perfusion fluid (CMA/Microdialysis) consisting of an isotonic sterile solution for use in peripheral tissue (Na+ 147 mmol/L, K+ 4 mmol/L, Ca2+ 2.3 mmol/L, Cl− 156 mmol/L). Analyses of porcine dialysates obtained were performed using an ISCUS Clinical Microdialysis Analyzer (CMA/Microdialysis).
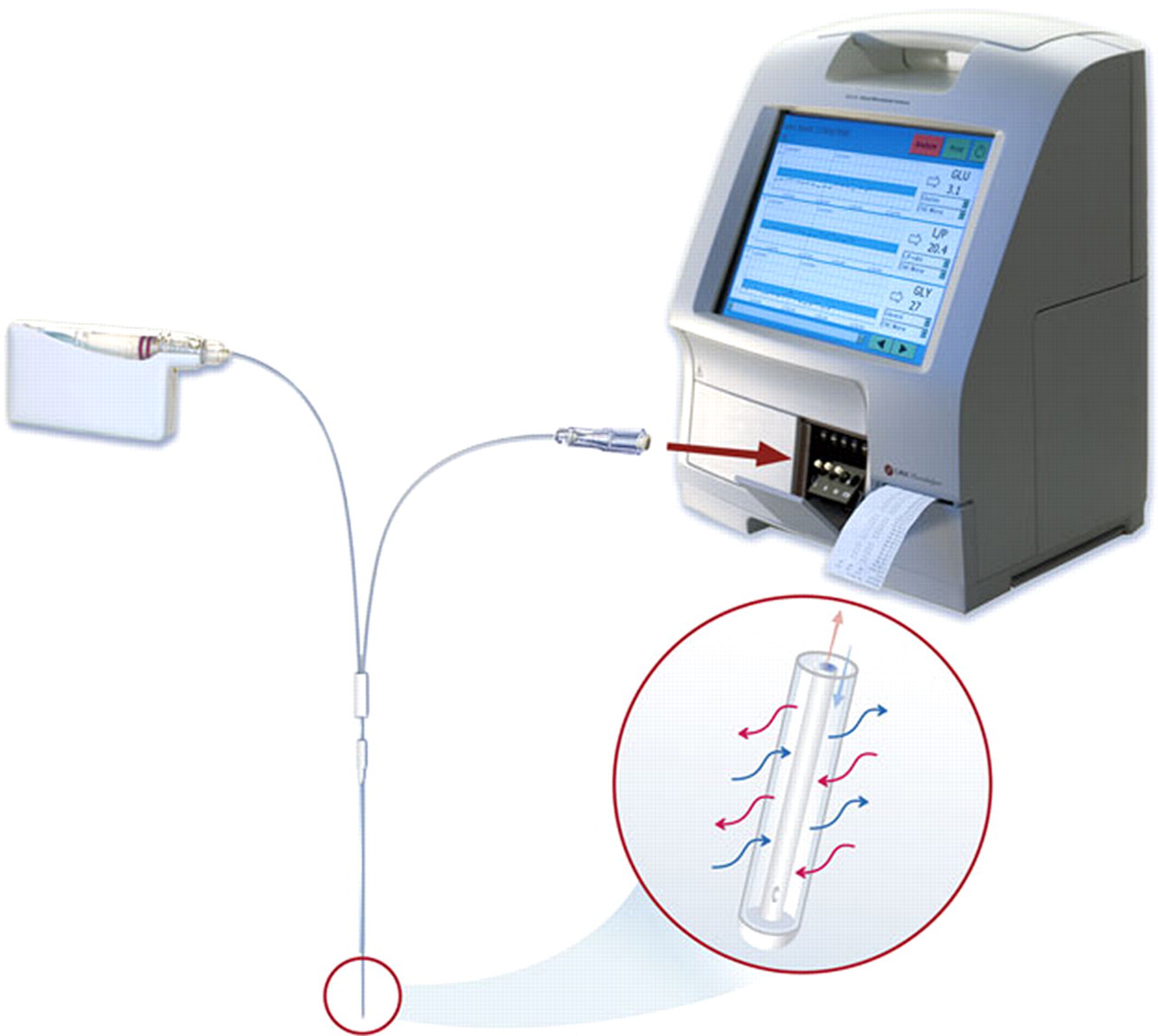
The microdialysis set-up. The catheter semi-permeable membrane placed in the disc nucleus pulposus works similarly to a capillary, allowing a selective diffusion of substances in both directions
Animals
One litter, consisting of eight cross-bred (Yorkshire × Swedish Landrace) SPF female and castrated male pigs, was used. The pigs were born at the Department of Clinical Sciences, Swedish University of Agricultural Sciences, and raised in a stable separated from the other animals. They were kept in a pen measuring approximately 12 m 2 with a concrete floor and straw as bedding. The temperature was maintained at 20 ± 2°C and a 12:12 light/dark schedule (lights on at 07:00 h) was used. An infrared lamp was provided in one corner of the pen. After weaning at six weeks, the pigs were fed ad libitum on a commercial diet without growth promoters (Solo, Klara 130, Lantmännen, Uppsala, Sweden) and had free access to water. Clinical examinations and weighing were performed weekly and no clinical signs of disease were observed throughout the study. The experimental procedures on pigs were approved by the Ethical Committee for Animal Experimentation, Uppsala, Sweden.
Human cadaver
One fresh-frozen cadaver, which was thawed for 24 h prior to the experiment, was used. The female specimen was thinly built, around 70 years old, dried out and had thoracolumbar scoliosis. The experiment using the cadaver was performed at the Institute of Anatomy and Musculoskeletal Research, Paracelsus Medical University, Salzburg, Austria, in accordance with the national legislations for use of human cadavers in Austria.
Experimental design
Animal study
The study was conducted in two steps. First, in order to make sure that the method for positioning of the needle into the nucleus pulposus was correct, three different needles, a 17 G 6 in. Crawford Tip needle, a 19 G 3 in. Trocar Tip needle (ArthroCare Corp, Austin, TX, USA) and an 18 G 2 in. Crawford Tip needle (BD, Morristown, NJ, USA), were inserted at different angles. Two animals weighing approximately 30 kg were used for this needle insertion optimization. As measured from the sagittal plane, angles between 35° and 55° were evaluated (Figure 2). The pigs were sedated with medetomidine (Domitor vet. 1 mg/mL, Orion Pharma Animal Health, Sollentuna, Sweden) intramuscularly at a dose of 0.03 mL/kg body weight. They were subsequently euthanized with an overdose of pentobarbital sodium intravenously (Avlivningsvätska, Apoteksbolaget, Sweden). Immediately after death, the animals were shaved and positioned prone on an adjustable elevation table that could be raised to an adequate position to perform the insertion of the three different needles at the previously described angles. The disc between the lumbar vertebral bodies L3 and L4 was used for the procedure.
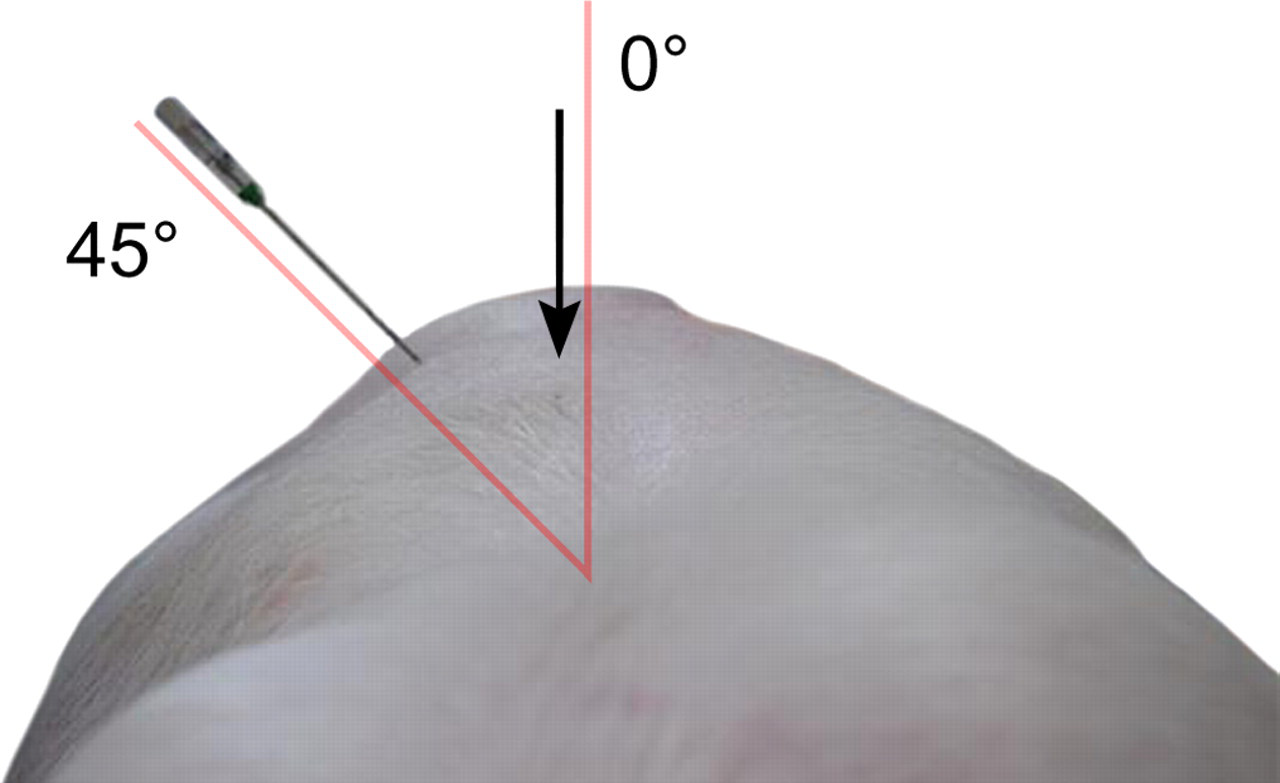
Different approaches of insertion of the different spinal needles for access to the porcine disc were evaluated. The photo showed a 17 G 6 in. Crawford Tip needle inserted at a 45° angle in a 30 kg animal. The animal disc nucleus pulposus in a 60 kg specimen could be accessed with an 18 G 2 in. needle, inserted at an angle of 45° as measured from the sagittal plane. Arrow indicating the processus spinosus
Under fluoroscopic guidance (Exposcop CB 745, Ziehm, Mediel AB, Västra Frölunda, Sweden) the following technique was used. The disc was approached at an oblique angle. The angle was between 35° and 40°. The needle was introduced into the disc, keeping the endplates parallel. A typical resistance was felt at the penetration of the annulus fibrosus. The needle was then forwarded 10 mm into the nucleus pulposus. In one pig, Evans blue dye (Apoteksbolaget, Stockholm, Sweden) was injected and the positioning was further confirmed at postmortem examination (Figure 3).
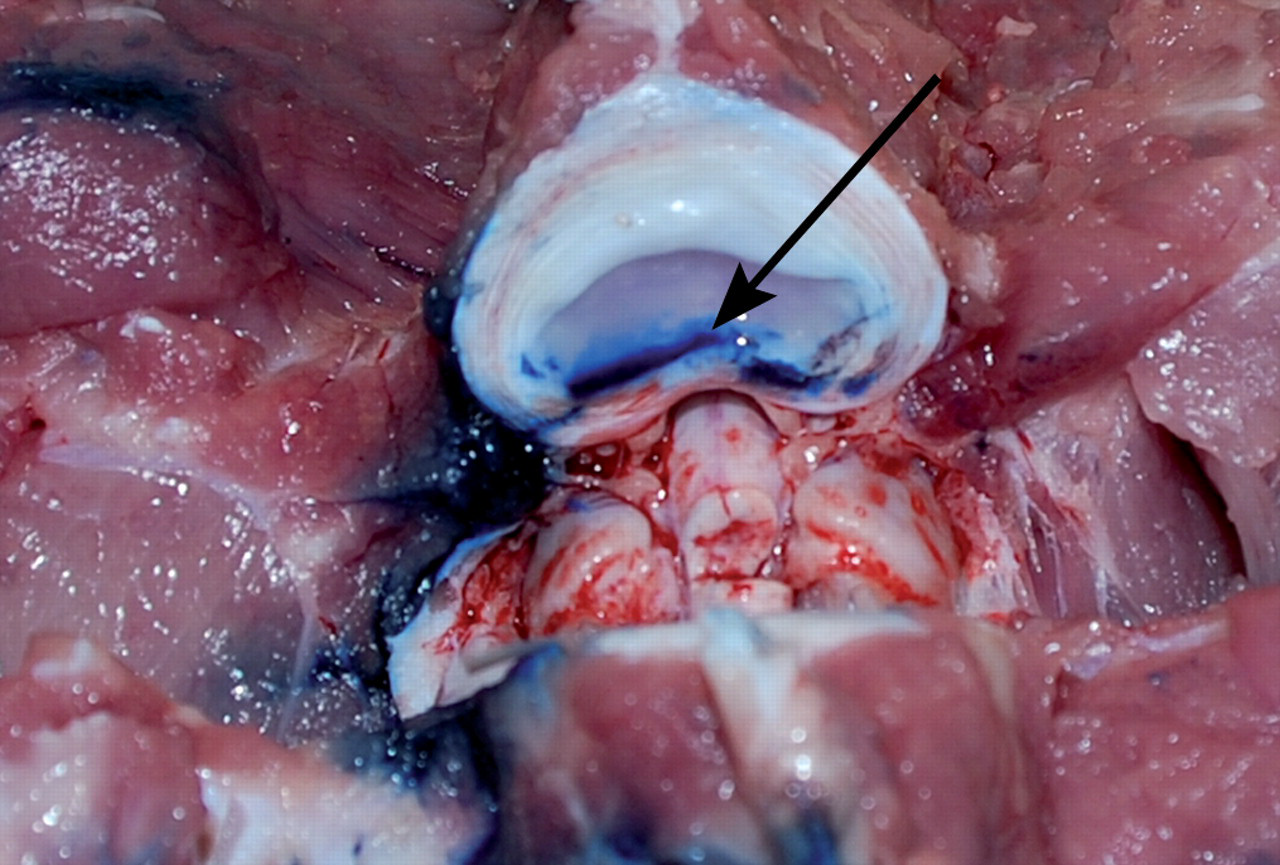
At dissection of the animal it could be confirmed that with the evaluated technique for needle insertion, the injected Evans blue dye was confined to the disc nucleus pulposus (arrow)
In the second step of the study, i.e. the microdialysis procedure, a decision was made to use six heavier animals (60–70 kg body weight) with larger musculus longissimus dorsi and larger cranial to caudal width of the discs, in order to better resemble an adult human being. After euthanasia and cannulation of the animals, performed according to the above procedure, the microdialysis membrane was inserted through the needle. The gold thread in the membrane was visible on the fluoroscope and was used to confirm the position in the nucleus pulposus. The microdialysis catheter was then flushed with isotonic perfusion fluid, and the following procedure to obtain dialysates was initiated. A flow rate of 0.5 μL/min was used for a total of 45 min to collect three consecutive samples of intradiscal dialysates from the nucleus pulposus. The samples were kept in vials on ice and analysed within 7 h after harvest to confirm that three metabolites routinely measured in microdialysis, glucose, pyruvate and lactate, had been obtained in analysable concentrations.
Human cadaver study
On the basis of results from the animal study, two different needles, a 17 G 6 in. Crawford Tip needle and a 19 G 3 in. Trocar Tip needle (ArthroCare), were tested under fluoroscopic guidance. The tip of the introducer needles was slightly bent prior to insertion. With the body in prone position, the needles were introduced using an oblique approach with the endplates remaining parallel and under fluoroscopic control. The L4–L5 intervertebral disc was used for the procedure. The catheter was inserted into the nucleus through the needle and the position of the gold threaded membrane was fluoroscopically confirmed. A further verification of the correct position of the needle was achieved by an injection of Omnipaque contrast fluid (Omnipaque 180 mg/mL, 10 mL vial, GE-Healthcare, Oslo, Norway).
Results
Animal study
The pig was found to be a suitable animal model since the anatomy, specifically that of the heavier animals, closely resembled the human disc size and surrounding tissues. The surgical cannulation technique used was also similar to that used in humans for diagnostic purposes such as discography or for therapeutic reasons, e.g. microdiscectomy and steroid injection. From the technique evaluation, it was concluded that the 18 G needle and an angle of 35–45° were preferred, with the blunter angle suitable for the heavier animal (Figure 2). The needle application took no more than 6 to 8 min. A 10 mm microdialysis membrane could be fully inserted in the nucleus pulposus without folding or creasing of the membrane. The gold thread marker of the microdialysis membrane was fluoroscopically verified and visible approximately 12 mm from the tip of the needle. The position was further confirmed macroscopically at gross examination of the disc where the Evans blue dye was contained in the nucleus pulposus area (Figure 3).
In all six animals, the chosen flush rate of 0.5 μL/min was adequate to obtain dialysates with sufficient concentration of the chosen metabolites – glucose (approximately 0.5 mmol/L), lactate (approximately 4 mmol/L) and pyruvate (approximately 25 μmol/L) – to enable analysis.
Human cadaver study
The cadaver was slightly built, with additional postmortal changes such as dehydration resulting in smaller dimensions compared with a standard clinical situation. Similar to the animal study, access to the nucleus pulposus was obtained by insertion of spinal needles from the sagittal plane, at an angle of 35–40°. The disc was easily accessible on the cadaver and insertion of the microdialysis membrane in the nucleus was feasible. Through fluoroscopic control it was found that the shorter 19 G needle precisely reached the annulu-nuclear junction when fully inserted. Taking the postmortal changes into consideration, the longer 17 G needle would be preferable in vivo. When inserted through the 19 G needle, the microdialysis membrane easily penetrated the nucleus tissue. However, neither of the needles had dimensions that were compatible with the catheter and as a result the available 60 mm catheter shaft was too short for the membranes to be fully positioned in the nucleus.
When using the 19 G needle, the gold thread of the catheter membrane was fluoroscopically visible and it was observed that both membrane dimensions (10 and 30 mm) were suitable for positioning into the nucleus of the L4–L5 disc of the specimen. Nevertheless, it was recognized that the 30 mm membrane would be 2–3 mm too long in a smaller disc. Apart from fluoroscopic verification of the microdialysis membrane gold thread, positional verification of the needle in the nucleus was also confirmed postprocedure through contrast fluid injection.
Discussion
To assess if the microdialysis technique could be used for obtaining dialysates suitable for biochemical analyses from the nucleus pulposus, we evaluated the technique in both porcine and human cadavers. The morphology of the intervertebral disc is well described and has been studied by conventional microscopy as well as other imaging techniques such as magnetic resonance, plane radiographs and lateral full spine X-rays. 22 Significantly less information is, however, available on the disc biochemistry, specifically over time, during physiological as well as pathophysiological conditions. Knowledge about the biochemistry will contribute to the understanding of the pathogenesis of disc diseases. Until now, data have primarily been obtained from material consisting of loose bodies or displaced discal material from patients undergoing surgery. 23 For diagnostic purposes, biopsies have also been harvested from suspected tumours or inflammatory conditions of the disc. 24
The experiments in this study were performed in the pig to verify that a dialysate could be obtained by microdialysis, and in a human cadaver to establish the appropriate catheter dimensions for use in humans. The commercial pig was shown to be an adequate model, and even though the porcine disc is smaller than the human disc with regard to height and width, the proportions and surrounding structures, as well as the consistency of the nucleus pulposus, are similar to those in humans. This is important since commercially available microdialysis devices have not been used previously for studies of the intervertebral disc in animals or humans where the tissue consistency, with regard to the water content, determines whether or not microdialysis can be used successfully. 25 The validity of the microdialysis technique is generally established, as it has been used for several years in a number of clinical specialties for monitoring tissue biochemistry. 16–18,25–29 From a regulatory standpoint, the specific microdialysis product used in this setting has been approved for sales in the US, and has also obtained a CE mark. The number of research fields where the technique is applied are also constantly increasing, and it has been used in several animal models. 30 However, this is the first time microdialysis has been applied to monitor intervertebral discs in situ.
The procedure was quick and easy to perform, and the preferred angle of the needle during cannulation was similar in the pig and the cadaver. However, the length of the catheter shaft was too short to allow complete positioning of the dialysis membrane in the cadaver, thus the catheter would have to be custom-made for use in humans.
The needle insertion to access the disc for performing microdialysis will be similar to other procedures involving cannulations, e.g. discographies and percutaneous nucleotomy. 31–34 Consequently, it may be possible to perform the microdialysis procedure under local or general anaesthesia. For porcine research, the microdialysis technique can probably be applied in live pigs if they are kept in single pens.
Side-effects associated with performing microdialysis clinically or in an animal model will primarily be associated with (1) the cannulation to access the disc and (2) the placement of the dialysis membrane.
Correctly performed, disc cannulations are associated with few adverse events such as nerve root and vascular injury, insertion site soreness, haemorrhages, dura penetration and discitis. 31,32,35
Microdialysis is minimally invasive not only in the sense of limited trauma at insertion, but also due to the fact that no tissue or interstitial fluid are harvested; only molecules are collected by the diffusion taking place over the membrane into the neutral dialysis fluid. 25 As a result, the method is appropriate in animal research for evaluation of sensitive structures such as the mouse brain. 36 However, as always when introducing a biomaterial for any length of time, infection is a potential risk. In a study of 97 patients using 122 implanted cerebral microdialysis catheters during treatment of cerebral injuries, no device-related infections were registered. 29
Microdialysis combines significantly less invasiveness with continuous sampling in the same individual compared with single tissue biopsies representing a static condition. 21 In orthopaedic research evaluating the antibiotic concentration in bone, microdialysis has been suggested as a superior alternative to bone biopsies. Once the catheter has been inserted in a tissue, dialysates can be obtained every 15–20 min, once the recovery of the system has been stabilized. 25 Microdialysis is also suitable in sensitive clinical applications such as neurosurgery and neonatal care where it enables continuous monitoring for up to 16 days. 26–28,37 A limitation with the technique is that it cannot detect morphological changes or supply material for tissue culture.
Lumbar discs are susceptible to spontaneous disc degeneration in humans and non-chondrodystrophic dogs, and this region is affected in the absolute majority of disc herniations, over 90%, while fewer than 5% occur in the cervical or thoracic regions. 38 Consequently, the L4–L5 disc was chosen for intervention in the human specimen, and L3–L4 in the pigs. Pigs do not develop disc diseases; therefore, when using pigs as an animal model for disc degeneration, the injury has to be induced by stabbing techniques for example. Since the cannulation technique in the present study is similar to that used in other clinical settings, there should be no problem using this technique to access other disc spaces.
The dialysate obtained from the porcine discs was sufficient to be used for further analyses of three metabolites that are routine in microdialysis analyses: glucose, lactate and pyruvate. 39 For such metabolite analysis, a 20 kDa membrane is usually sufficient. When deciding the technical set-up, one should consider that the recovery is dependent on the velocity of flow through in the system, the membrane length and cut-off, and also on the configuration of the molecules to be analysed. In the present study, we chose microdialysis membranes with a 100 kDa cut-off, which with similar flow rates have been shown to also enable detection of larger molecules such as cytokines, which are of great interest when inflammatory processes are studied. 18
A velocity of 0.5 μL/min results in a larger volume of the dialysate, compared with 0.3 μL/min, without significant decrease of metabolite recovery. Even though the results obtained reflected that the process was performed in postmortem material, we were still able to obtain detectable, and considering the circumstances, reasonable concentrations of metabolites. We chose to collect three dialysates from each investigated disc during a period of 45 min, to confirm that the nucleus pulposus properties were such that concentrated dialysates could be obtained. However, due to the relatively slow diffusion rate of the disc, continuous measurements would need to be performed for a longer period of time to evaluate intradiscal changes. 40 Gadoteridol and other non-ionic media have been shown to have higher diffusion rates in the disc. 40 In this study dialysates were obtained from euthanized animals and we have not compared the results of the biochemical analysis to reference values for live animals. The next step will be to perform the procedure in live animals over a longer period of time to evaluate the procedure thoroughly, and also to study potential complications and adverse effects of the procedure in vivo.
In addition to measuring metabolites, other markers such as proinflammatory mediators and cytokines can give new insights into the disc degenerative process and provide a better understanding of the physiology and pathophysiology of the disc. Microdialysis can also be a tool for evaluation of new treatments, and different pharmacological effects on the disc biochemistry can be studied. In addition to pathological processes, it might also be possible to detect physiological effects of normal ageing, diet and physical activity.
There has been a recent surge in interest for minimally invasive techniques for studies of the intervertebral disc. The present study concludes that microdialysis is a possible new tool for biochemical and physiological evaluation of the animal and human intervertebral disc and is encouraging for further in vivo studies.
Footnotes
Acknowledgements
The authors wish to acknowledge Karin Peterson (CMA/Microdialysis, Solna, Sweden) for her expertise in the microdialysis technique and David Skog for providing technical assistance.
Author contributions: EL, GS and MJW conceived and designed the experiments; GS, HS and MJW performed the experiments; and EL, GS, HS and MJW wrote the paper.
