Abstract
Objective:
The aim of this study was to investigate the role of glutathione peroxidase 7 (GPX7) in mitigating oxidative stress-induced cellular ageing and its contribution to intervertebral disc degeneration (IVDD).
Introduction:
Human nucleus pulposus (NP) cells in degenerated intervertebral discs (IVDs) show signs of ageing, such as telomere shortening, DNA damage, and mitochondrial dysfunction. GPX7, known for its ability to protect against ageing in cancer cells, may also play a role in NP cell ageing.
Methods:
Two datasets (GSE34095 and GSE147383) were analysed to compare GPX7 expression in normal and degenerated IVDs and used KEGG analysis to identify related pathways. An H2O2-induced cell model and a natural ageing model were used to simulate ageing. GPX7 was transfected into and overexpressed in NP cells, and its protective effects were examined. Molecular docking identified GNF-5837 as a potential compound to prevent GPX7 cleavage, which was tested in an IVDD rat model.
Results:
The expression of GPX7 was increased in degenerated IVDs and H2O2-induced models. GPX7 overexpression reduced ageing in NP cells. GNF-5837 alleviated IVDD in rats by upregulating GPX7.
Conclusion:
Our study demonstrates that GNF-5837 reduced the expression of ageing markers by upregulating GPX7, suggesting it could serve as a potential treatment for IVDD.
Introduction
Ageing is an irreversible process in organisms whereby tissues and organs undergo functional decline, disrupted stabilisation of homeostasis and degeneration of physiological stress responses.1–3 As the trend of global ageing intensifies, low back pain (LBP) has emerged as the most common musculoskeletal (MSK) ailment. In 2019, LBP accounted for approximately 47.2% of the global burden of MSK disorders, with an estimated prevalence of around 170 million individuals affected. 4 Mainly affecting middle-aged and elderly individuals, it imposes heavy medical and economic burdens on both individuals and society.5,6 IVDD and its associated conditions, such as spinal stenosis and disc herniation, are major contributors to LBP.7,8 IVDD is a progressive condition characterised by biochemical, structural and mechanical changes within the IVDs. The process begins with the breakdown of the extracellular matrix (ECM), primarily involving a reduction in proteoglycans and collagen alterations, which leads to loss of hydration and decreased disc height. This ECM degradation impairs the disc’s ability to absorb mechanical stress. Over time, oxidative stress, inflammation and cell senescence further exacerbate tissue damage, resulting in fibrosis, reduced cellularity and compromised disc function. These changes finally contribute to chronic pain and reduced mobility. 9
Current treatment options for IVDD are limited, with mainstream pharmacotherapy or surgical interventions often associated with significant side effects or high complication rates, putting the development of biological treatment on the agenda.10,11 The IVD comprises a NP that is abundant in proteoglycans, encompassed by a fibrocartilage-rich annulus fibrosus (AF) and the upper and lower cartilaginous endplates.12,13 The NP, located at the centre of the disc, is a gel-like structure that is rich in proteoglycans and water, which provides the disc with the ability to resist compressive forces. The degeneration of the IVD, particularly the NP, is closely associated with the accumulation of oxidative stress, which is driven by the excessive generation of reactive oxygen species (ROS). During IVDD, upregulated inflammatory cytokines, such as IL-1β and TNF-α, increased cellular senescence and mitochondrial dysfunction promote ROS production. Additionally, the hypoxic conditions typical in IVDD, combined with weakened antioxidant defences, exacerbate oxidative stress, leading to a harmful buildup of ROS within NPCs microenvironment. This oxidative stress-mediated degeneration is characterised by the breakdown of proteoglycans and a reduction in water content, compromising the biomechanical properties of the disc and contributing to the pathogenesis of LBP.14–16 Biomarkers such as p53, p21, p16 and IL-6 are widely utilised to evaluate ageing-related pathologies, including IVDD, and their dysregulation has been linked to oxidative stress and ECM degradation in degenerated discs.17,18
The glutathione peroxidase (GPX) family, comprising eight subtypes, metabolises ROS within cells, thus maintaining cellular homeostasis. 19 GPX catalyses the reduction reaction and eliminates peroxides in the body, thereby preserving normal physiological functions and delaying ageing.20,21 GPX7 is a newly discovered non-selenium-containing member of the GPX family. 22 A growing body of research shows that GPX7 plays a protective role against oxidative stress in cancer cells. 23 In breast cancer, knockout of GPX7 promotes cell death induced by polyunsaturated fatty acids. 24 GPX7 significantly enhances the survival rate of oesophageal cancer cells after hydrogen peroxide treatment and reduces apoptosis. 25 Deficiency of GPX7 may affect ageing-related diseases via NF-κB activation. 26 However, whether GPX7 participates in the PI3K‒Akt pathway and the underlying mechanisms of its role in the pathogenesis of IVDD remain unclear.
GNF-5837 is a selective pan-tropomyosin receptor kinase (TRK) inhibitor that has been investigated primarily in the context of cancer research. TRK family proteins, including TRKA, TRKB and TRKC, play critical roles in regulating cell growth, differentiation and survival, making them attractive targets in oncology. Upon activation, TRKs initiate several key signalling pathways: MAPK/ERK, PI3K/AKT and PLCγ pathway. These pathways collectively contribute to the regulation of cell survival, proliferation and differentiation, especially in neural tissues. 27 GNF-5837 has demonstrated significant antiproliferative effects by inhibiting these receptors, leading to cell cycle arrest, apoptosis and reduced cell viability in various cancer models. 28 GNF-5837 can suppress the viability of renal carcinoma cells and induce G0/G1‑phase arrest and apoptosis in a concentration‑dependent manner. 29 However, studies investigating the correlation between GNF-5837 and cellular senescence are still at an early stage.
In this study, our objective was to explore whether GPX7-mediated antioxidation could obstruct oxidative stress-induced cellular ageing and its underlying signalling pathways in vivo and in vitro. We hypothesised that the expression level of GPX7 decreases irreversibly with natural ageing, resulting in the loss of antioxidant capacity in NP cells and thereby further contributing to IVDD. In addition, we predicted and identified effective compounds that could bind to GPX7 through molecular docking, providing novel drug candidates for the biological treatment of IVDD.
Materials and methods
Harvesting, cultivation and treatment of nucleus pulposus cells
Human intervertebral disc nucleus pulposus cells (NPCs) were purchased from Life Science & Technology Co., Ltd. (Cat No. CP-H097, China). NPCs were maintained in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, USA) and 1% penicillin‒streptomycin (Gibco, USA) under standard incubation conditions (37°C, 5% CO2). The culture medium was completely replaced every 3 days. When the cells reached 80% confluence, they were passaged using trypsin with 1% ethylene diamine tetraacetic acid (Trypsin-EDTA; Gibco, USA) for up to 10 generations.
To establish an in vitro oxidative stress-induced ageing culture model, the optimal concentration and exposure time were determined by treating the cells with 0, 50, 100, 200, 300 or 500 μM H2O2 for 0, 1, 2, 4, 6 or 8 h before the experiments. GNF-5837 (Cat No. HY-13491; MCE, USA) was dissolved in DMSO (dimethyl sulfoxide) at a concentration of 5 mM. The cells were treated with 500 nM GNF-5837 for 72 h before performing further experiments following the manufacturer’s instructions.
Microarray datasets and bioinformatics analysis
To explore the differences of expression profiling and relevant biological processes in IVDD patients, we searched datasets from the GEO database with the keywords ‘intervertebral disc degeneration’ [All Fields] AND ‘Homo sapiens’[porgn] AND ‘gse’[Filter] AND ‘2021/01’[Update Date]. The inclusion and exclusion criteria were as follows: datasets should be the whole-genome expression data of mRNA; expression data should be obtained from human annulus disc tissue of IVDD patients and healthy controls. Finally, two mRNA expression datasets: GSE34095 (a total of six samples (three degenerative discs and three non-degenerative discs), generated using the Affymetrix Human Genome U133A Array); GSE147383 (eight samples from two groups of individuals (young and non-degenerated group and an aged and degenerated group), covering both annulus fibrosus and nucleus pulposus tissues, was analysed using Affymetrix Human Genome U133 Plus 2.0 Array) were obtained from the Gene Expression Omnibus (GEO) database (http://www.ncbi.nlm.nih.gov/geo/) were incorporated into our studies.
Overall gene expression was examined using R software version 4.3.2 (http://www.r-project.org). In brief, the limma package was used to identify differentially expressed genes (DEGs) with statistical significance between groups, and a false discovery rate (FDR) threshold of 0.05 was used to determine significance. The intersection of upregulated genes was taken from two gene expression microarrays and then were visualised using a Venn diagram. The complete list of DEGs in GSE34095 and GSE 147383 is shown in Supplemental Data 1.
Weighted correlation network analysis (WGCNA) was subsequently used to analyse the microarrays to identify clusters (modules) of highly correlated genes, with a soft-thresholding power of 10 and a minimum module size of 30, selecting modules highly correlated with phenotypes. The targets of GPX7 were compiled with 232 genes identified through WGCNA. The signalling pathways that correlated with GPX7 were subsequently investigated via Kyoto Encyclopaedia of Genes and Genomes (KEGG, https://www.kegg.jp/) analysis using the ‘ClusterProfiler’ package, with significant pathways identified by FDR <0.05 and Bonferroni-corrected p-value thresholds.
Total protein extraction and western blotting
The cultured primary human NP cells were washed twice with prechilled phosphate-buffered saline (PBS; Gibco, USA) and lysed with 200 μL of a mixture of RIPA buffer (Beyotime, China) protease, phosphatase inhibitors and phenylmethyl sulfonyl fluoride (PMSF) on ice for 20 min. The lysed cells were subsequently centrifuged at 4°C for 15 min at 12,000 rpm. A BCA protein assay reagent kit (KeyGEN BioTECH, China) was used to determine the protein concentration. Protein extracts (20 µg per well) were loaded and separated on 10% sodium dodecyl sulphate−polyacrylamide gels (SDS-PAGE, Epizyme Biotech, China) before being transferred onto 0.22 µm polyvinylidene difluoride (PVDF) membranes (Millipore, USA). After blocking with 5% skim milk in TBS-T for 1 h, the membranes were incubated with primary antibodies against GPX7 (diluted 1:1000, Cat No. 13501-1-AP; Proteintech, USA), β-tubulin (diluted 1:1000, Cat No. 10094-1-AP; Proteintech, USA), P53 (diluted 1:1000, Cat No. 21891-1-AP; Proteintech, USA), P21 (diluted 1:1000, Cat No. 10355-1-AP; Proteintech, USA), P16 (diluted 1:1000, Cat No. 10883-1-AP; Proteintech, USA) and IL-6 (diluted 1:1000, Cat No. 21865-1-AP; Proteintech, USA) at 4°C overnight. The membranes were subsequently incubated for 2 h at room temperature with HRP-conjugated goat anti-rabbit immunoglobulin G (IgG; diluted 1:10000, Cat No. SA00001-2; Proteintech, USA). Enhanced chemiluminescence (ECL) reagents (Millipore, USA) were used to visualise the protein bands, and ImageJ software was used to quantify the results.
RNA extraction and qPCR
TRIzol Reagent (Invitrogen, USA) was used to isolate total RNA from cultured NP cells according to the manufacturer’s instructions. A Nanodrop (Thermo Scientific, USA) was used to analyse the RNA quantity. Total RNA (1 μg) was reverse transcribed into complementary DNA (cDNA) using a reverse transcription kit (Takara, Cat. #RR047A, Japan) for qPCR. qPCR was performed using RealStar Fast SYBR qPCR Mix (A301, GenStar). Relative expression levels were calculated using the 2−ΔΔCT method. GAPDH was used to normalise the expression levels of the target genes. Primers used for qPCR were as follows: GPX7 (forward: 5′-CGACTTCAAGGCGGTCAACATC-3′, reverse: 5′-TCGGTAGTGCTGGTCTGTGAAG-3′) and GAPDH (forward: 5′-GTCTCCTCTGACTTCAACAGCG-3′, reverse: 5′-ACCACCCTGTTGCTGTAGCCAA-3′).
Senescence-associated β-galactosidase (SA-β-gal) staining and quantification
The cells were washed in PBS and fixed with PBS supplemented with 2% formaldehyde and 0.2% glutaraldehyde for 15 min at room temperature. The cells were subsequently incubated with SA-β-gal staining solution (Beyotime Institute of Biotechnology, China) for 24 h at 37°C following the manufacturer’s instructions. The cells with blue-stained cytoplasm were considered positive. Three fields of view were chosen at random for each group, the percentage of SA-β-gal-positive cells was calculated by counting the number of blue-stained cells and dividing it by the total number of cells in the same field, and the mean was calculated. 30
Immunofluorescence
The NP cells were treated as indicated and then fixed with 4% paraformaldehyde in PBS for 15 min at room temperature. After permeabilisation with 0.2% Triton X-100 in PBS for half an hour and washed with PBS, the cells were blocked with 10% goat serum (Thermo, USA) for 1 h at 37°C. The cells were subsequently incubated with a GPX7 antibody (diluted 1:100, Cat No. 13501-1-AP; Proteintech, USA) overnight at 4°C. The next day, the cells were incubated with a goat anti-rabbit recombinant secondary antibody (diluted 1:200, Cat No. RGAR002; Proteintech, USA) for 2 h at room temperature, and the cell nuclei were counterstained with DAPI. Images were acquired using a fluorescence microscope (Leica TCS SP8) and quantified using ImageJ software.
Cell transfection
NP cells were transfected with lentiviral particles (Gene Pharma, Inc., China) following the manufacturer’s instructions. Specifically, NP cells were seeded in six-well plates for 24 h (5 × 105/well). After reaching 30%−40% confluence, the cells were infected with lentivirus carrying the GPX7 gene or control lentiviral vector at a MOI of 30 for 48 h using Lipofectamine 3000 (Invitrogen, USA). After 48 h of treatment, the cells were selected with 2.0 μg/mL puromycin (Sigma-Aldrich, USA).
MitoSOX Green staining
Mitochondrial ROS were detected with the MitoSOX Green indicator (Cat No. M36006; Thermo, USA). Briefly, NP cells were seeded in six-well plates for 24 h (5 × 105/well). Next, the NP cells were washed in PBS and incubated with 2 mL of 5 μM MitoSOX reagent solution for 10 min at 37°C. The cells were washed, and images were captured by fluorescence microscopy. Total cells and MitoSOX Green-positive cells were manually counted. The MitoSOX Green-positive cell percentage was calculated as follows: MitoSOX Green-positive cell percentage (%) = MitoSOX Green-positive cells/total NP cells × 100%.
Molecular docking simulation
Molecular docking simulation was performed using AutoDock 4.2, an open–source software suite designed for computational docking and virtual screening of small molecule binding to a receptor with a known 3D structure. Briefly, the 3D crystal structure of GPX7 was obtained from the protein data bank website (http://www.rcsb.org) (PDB ID: 2P31). The AGFR1.2 programme was used to determine the active site of GPX7 according to the binding site of the crystallised ligands. The binding free energy (ΔG) and inhibition constant (Ki) values between the effector compounds that provide the most appropriate conformational fit to the 3D structure of GPX7 were subsequently estimated (Supplemental Data 2).
Rat IVDD model and treatment
Sprague–Dawley (SD) rats (Shanghai Laboratory Animal Center, China) with an initial weight of approximately 200 g and aged 12 weeks were used in this study. Ethics approval for these procedures was obtained from the Medical Ethics Committee of The First Affiliated Hospital of Fujian Medical University. All SD rats were housed in the animal facility of The First Affiliated Hospital of Fujian Medical University and were kept pathogen free. The Animal Care and Use Committee reviewed and approved the procedures involving the care and use of animals.
All the SD rats were housed at 22 ± 2°C and 50 ± 10% humidity with a 12 h light/12 h dark cycle. The SD rats were randomly sorted into three groups: the control group (CTR): rats in this group received no intervention, serving as the baseline control for comparison; annulus fibrosus (AF) puncture group (AFP): rats in this group underwent annulus fibrosus puncture without further treatment, simulating intervertebral disc degeneration and AF puncture with GNF-5837 treatment group (AFP+GNF): rats in this group also underwent annulus fibrosus puncture, followed by daily oral administration of GNF-5837 at 50 mg/kg to assess its therapeutic effects on disc degeneration. The SD rats were anaesthetised with 2% pentobarbital (50 mg/kg) and then placed in the supine position. A 1 cm midline ventral longitudinal incision was made to visualise the position of the IVDs. A 27 G limited depth needle was subsequently inserted into the IVD with a 360° rotation for 30 s. After all, the internal organs were carefully repositioned, and the peritoneum was sutured using 4-0 coated sutures. The skin incision was subsequently closed with 4-0 sutures, and the animals were monitored continuously while kept warm until they fully recovered from anaesthesia. 31
The SD rats in the GNF group were administered GNF-5837 daily via oral gavage at a dosage of 50 mg/kg until they were euthanised.
After 8 weeks, the SD rats were euthanised via 100% CO2 overdose at a displacement rate of 20% of the chamber volume per minute. Subsequent to lumbar magnetic resonance imaging (MRI) examinations, the IVDs were collected for further experimentation.
In accordance with ethical standards, specific humane endpoints were established to ensure animal welfare throughout the study. Animals were immediately euthanised if they exhibited any of the following criteria prior to the scheduled 8-week endpoint: significant weight loss exceeding 20%, signs of severe distress or lack of mobility, and any overt signs of unmanageable pain despite analgesic treatment.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 9.0 software (GraphPad, USA). For comparisons between two groups, unpaired Student’s t-test was applied. For multi-group comparisons, one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test was used to assess significance. A p < 0.05 was considered statistically significant.
Results
Association of GPX7 with ageing
To validate our hypothesis that GPX7 plays an important role in the progression of IVDD by regulating oxidative stress-induced cellular ageing, we separately analysed GSE34095 and GSE147383 and intersected the results to identify upregulated genes between normal and IVDD tissues. A criterion was established where a mean fold change >5 or <0.2, along with p values <0.01, was considered significant. Consistent with our hypothesis, the results demonstrated that GPX7 was differentially expressed in both datasets (Figure 1(a)). All the DEGs, including TGFBI, SCRG1, LTBP1, CCN6, IFI16, ADK, ZNF207, CHCHD3, GPX7, TFDP1, EVI2A, CASP4, CCN4 and CEP76 were subsequently subjected to WGCNA. Using a soft-thresholding power of 10 and a minimum module size 30, we constructed co-expression networks from the two datasets. The analysis revealed that the turquoise module (r = 0.89, p < 0.001) exhibited the strongest positive correlation with IVDD severity, and GPX7 was identified as a hub gene within this module. These genes were clustered into 22 modules, and the results revealed that GPX7 played a significant role in the progression of IVDD (Figure 1(b) and (c)).

Association of GPX7 with ageing. (a) Venn diagram illustrating up-regulated genes in GSE34095 and GSE147383. (b) Module-trait relationship of GPX7 in weighted gene co-expression network analysis (WGCNA). (c) Cluster dendrogram depicting the relationship between GPX7 and other DEGs in WGCNA. (d and e) Western blotting and quantitative analysis of GPX7 expression in NP cells under different concentrations of H2O2 treatment. (f and g) Western blotting and quantitative analysis of GPX7 expression in NP cells under different durations of H2O2 treatment. (h) RT-qPCR results showing GPX7 expression was increased after H2O2 (200 μM and 4 h) treatment. (i and j). Representative images of SA-β-Gal staining in NP cells and quantitative analysis under H2O2 (200 μM and 4 h) treatment (Scale bar = 50 μm).
To further verify our findings, we established an H2O2-induced IVDD cell model and examined the expression level of GPX7. Surprisingly, the western blotting and quantitative analysis results indicated that the expression level of GPX7 gradually increased in H2O2-treated NP cells in a dose- and time-dependent manner (Figure 1(d)–(g)). Therefore, the optimal treatment concentration and duration of H2O2 were determined (200 μM and 4 h). The RT-qPCR results further confirmed that the mRNA expression level of GPX7 was significantly increased in H2O2-treated NP cells (Figure 1(h)).
Research has shown that H2O2 can induce cellular oxidative stress, activate sensory proteins and initiate cellular senescence, making it broadly applicable in the establishment of cellular senescence models.32,33 Thus, SA-β-gal staining was performed to evaluate the ageing status of H2O2-treated NP cells. The results revealed that the β-gal density significantly increased in the H2O2-treated group (Figure 1(i) and (j)).
In summary, the expression of GPX7 was positively correlated with IVDD, and the expression level of GPX7 was increased during H2O2-induced cellular ageing.
Upregulation of GPX7 in response to H2O2 treatment, not natural ageing
We observed increased GPX7 expression in IVDD patients in the microarray datasets, whereas GPX7 expression was significantly increased in the H2O2-induced IVDD cell model. To decipher the underlying mechanisms of NP cell ageing in IVDD, we subsequently introduced the natural ageing cellular model in our studies. Research has shown that continuous passage can induce senescence in NP cells. Consistent with these reports, we observed a transition in the morphology of the NP cells from triangular and polygonal shapes at passage 0 (P0) to fibrocartilage-like fusiform shapes at P3. The cell volume and the time required for cell adherence, proliferation and passage gradually increased. To explore the reaction of ageing NP cells to ROS in vitro, we examined the effect of H2O2 in the natural ageing cellular model. Consistently, western blotting and quantitative analysis revealed that the expression level of GPX7 gradually decreased with increasing passage number of NP cells. In agreement with our findings in the H2O2-induced IVDD cell model, the protein level of GPX7 was significantly elevated in response to H2O2 treatment in the natural ageing cellular model (Figure 2(a) and (b)). Of note, the cells at P9 exhibited a reduced ability to respond with an increase in GPX7 expression upon oxidative stress stimulation, which aligns with our hypothesis that, as cells age, the regulatory capacity of GPX7 expression diminishes, leading to a reduced ability to resist oxidative stress. Therefore, we proceeded with further experiments using the ninth passage of NP cells as the natural ageing cellular model. These observations were further corroborated by RT-qPCR (Figure 2(e)). SA-β-gal staining revealed that the β-gal density of NP cells was significantly greater at P9 than at P0, and H2O2 treatment further promoted the ratio of ageing NP cells in both the P0 and P9 groups (Figure 2(c) and (d)).

Up-regulation of GPX7 in response to H2O2 treatment, not natural ageing. (a and b) Western blotting and quantitative analysis of GPX7 expression in different generations of NP cells under H2O2 treatment. (c and d) Representative images of SA-β-Gal staining and quantitative analysis under H2O2 (200 μM and 4 h) treatment in different generations of NP cells (Scale bar = 50 μm). (e) RT-qPCR results depicting GPX7 expression in different generations of NP cells under H2O2 (200 μM and 4 h) treatment. (f–k) Western blotting and quantitative analysis of classic ageing-related protein markers, such as GPX7, P53, P21, P16 and IL-6, under H2O2 (200 μM and 4 h) treatment in different generations of NP cells. (l–m) Representative images of cellular fluorescence indicating GPX7 expression under H2O2 (200 μM and 4 h) treatment. (Scale bar = 50 μm). (n–p) Representative images of ROS(Green) and p16(Red) fluorescence under H2O2 (200 μM and 4 h) treatment in NP cells of the 0 th and 9 th generations. (Scale bar = 50 μm). (q–r) Representative images of AGG (Aggresome) fluorescence in NP cells. (Scale bar = 25 μm).
During cellular proliferation, incomplete replication of DNA leads to telomere shortening, predominantly activating the p53-p21-Rb and p16-Rb signalling pathways to promote cellular ageing. 34 In turn, ageing cells secrete proinflammatory factors (including IL-1, IL-6, IL-8 and TNF-α) that induce adjacent cell senescence and immune infiltration. 35 Therefore, western blotting and quantitative analysis were used to further detect the senescence markers p53, p21 and p16 and the inflammatory cytokine IL-6 in an H2O2-treated natural ageing cellular model. IL-6 and the expression levels of p53, p21 and p16 were significantly increased in ageing NP cells with or without oxidative damage. However, the protein level of GPX7 was significantly elevated in response to H2O2 treatment in the control group but was not different in the natural ageing cellular model (Figure 2(f)–(k)). These findings were further corroborated by IF and quantitative analysis results (Figure 2(l)–(r)).
These data indicated that H2O2-induced oxidative stress, not natural ageing, upregulated GPX7 expression.
GPX7 overexpression mitigates cellular ageing
To decipher the role played by GPX7 in H2O2-induced cellular ageing and to investigate the underlying mechanism by how GPX7 participated in the progression of IVDD, we performed KEGG analysis. Previous studies have shown that the PI3K-Akt pathway plays crucial roles in cell survival, proliferation and metabolic regulation, thus influencing the ageing process.36,37 Consistent with this reports, neuroactive ligand−receptor interactions, the PI3K-Akt signalling pathway, human papillomavirus infection, the MAPK signalling pathway and the cAMP signalling pathway, among others, were enriched in current study (Figure 3(a)).

GPX7 overexpression mitigates cellular ageing. (a) KEGG analysis of GPX7 in the database reveals a significant up-regulation of ligand-receptor interaction in NP cells. (b) Western blotting analysis of classic ageing-related protein markers, such as GPX7, P53, P21, P16 and IL-6, with H2O2 (200 μM and 4 h) treatment in NP cells overexpressing GPX7. (c and d) Representative images of SA-β-Gal staining and quantitative analysis under H2O2 (200 μM and 4 h) treatment in NP cells overexpressing GPX7 (Scale bar = 50 μm). (e) RT-qPCR results illustrating GPX7 expression in NP cells overexpressing GPX7 under H2O2 (200 μM and 4 h) treatment. (f–g) Representative image of cellular fluorescence indicating GPX7 expression in NP cells overexpressing GPX7 under H2O2 (200 μM and 4 h) treatment (Scale bar = 50 μm). (h–j) Representative image of ROS probe (Green) and P16 (Red) staining in NP cells overexpressing GPX7 under H2O2 (200 μM and 4 h) treatment. (k–l) Representative image of AGG (Aggresome) staining in NP cells. (Scale bar = 25 μm).
We subsequently generated lentiviruses overexpressing GPX7 and transfected them into NP cells (P0 and P9), fluorescence microscopy images were obtained to confirm the successful transfection (Supplemental Figure S1A). The western blotting results indicated that the expression level of GPX7 was significantly increased in the GPX7-overexpressing group (OE-GPX7) in both generations of NP cells. Interestingly, we found that the upregulation of GPX7 downregulated the expression of senescence markers (p53, p21 and p16), and IL-6 and even inhibited the H2O2-induced increase in these proteins both in normal NP cells and in the natural ageing cellular model (Figure 3(b)). SA-β-gal staining and quantitative analysis results indicated that compared with the H2O2 treatment group, the β-gal densities were significantly decreased in H2O2-treated NP cells pre-transfected with GPX7 overexpression lentivirus (Figure 3(c) and (d)). RT-qPCR results confirmed that the expression level of GPX7 was significantly increased in the GPX7 overexpression group, almost reaching the level observed in the H2O2-treated group (Figure 3(e)). MitoSOX Green and IF co-staining was used to assess the level of mitochondrial ROS in NP cells, and its correlation with P16 expression. The results demonstrated that the H2O2-induced increase in ROS levels was suppressed by the overexpression of GPX7, and the expression of P16 and AGG have a positive correlation with GPX7 expression in NP cells (Figure 3(f)–(l)). Collectively, these findings suggested that GPX7 might mitigate H2O2-induced cellular ageing by regulating ROS.
GNF-5837 alleviates IVDD by upregulating GPX7 expression
To identify potential compounds that might alleviate ageing-related IVDD by regulating GPX7, we performed molecular docking simulations. We used an online protein database to predict the structure of GPX7 and AGFR1.2 to determine its active site according to the binding site of the crystallised ligands (Supplemental Figure S1B). The binding free energy between the effector compounds that provided the most appropriate conformational fit to the 3D structure of GPX7 was subsequently estimated, and five compounds were screened for further analysis (Figure 4(a)). Consistent with the predicted free energy of binding results, western blotting analysis revealed a significant increase in the expression of GPX7 in NP cells treated with GNF-5837 (Figure 4(b)).
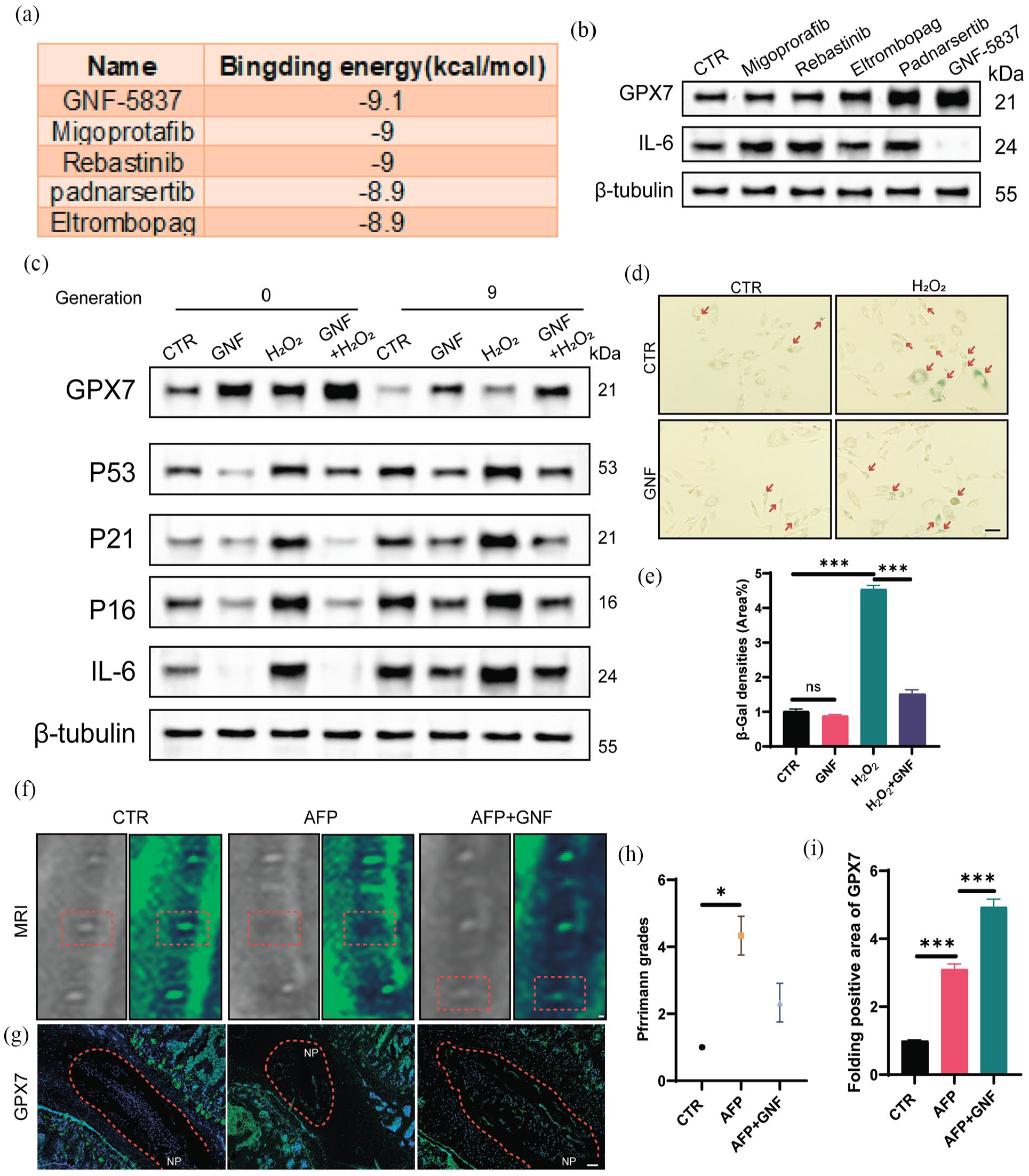
GNF-5837 alleviates IVDD by upregulating GPX7 expression. (a) The top five drugs and their corresponding binding energies (kcal/mol) predicted as potential ligands for GPX7 (PDB id: 2P31). (b) Western blotting screening results of GPX7 and IL-6 expression after treatment with GNF-5837, Migoprotafib, Rebastinib, Padnarsertib, Eltrombopag following H2O2 pretreatment. (c) Western blotting analysis of classic ageing-related protein markers, such as GPX7, P53, P21, P16 and IL-6, with H2O2 (200 μM, 4 h) and GNF-5837 treatment in different generations of NP cells. (d–e) Representative images of SA-β-Gal staining and quantitative analysis of NP cells in different generations with H2O2 (200 μM, 4 h) and GNF-5837 treatment (Scale bar = 50 μm). (f and h) MRI images and Pfirrmann grade analysis of rats from the CTR, AFP and AFP plus GNF-5837 groups, obtained 8 weeks after surgery. (g and i) Representative IF staining images depicting GPX7 expression in rat IVDs from the CTR, AFP and AFP plus GNF groups, conducted 8 weeks post-operation. (Original magnification 200×, scale bar = 200 µm).
To determine whether GNF-5837 could attenuate cellular ageing by upregulating GPX7 in vitro, we pretreated NP cells with GNF-5837 followed by subsequent H2O2 treatment. Western blotting and quantitative analyses suggested that pretreatment with GNF-5837 significantly increased the protein level of GPX7, and the phosphorylation of AKT compared with NP cells treated with or without H2O2, both in normal NP cells and the natural ageing cellular model (Figure 4(c) Supplemental Figure S1C). Consistent with the results of the GPX7 overexpression experiments, we found that GNF-5837 treatment also downregulated the expression of senescence markers (p53, p21, p16), and IL-6 and even inhibited the H2O2-induced increase in these proteins, both in normal NP cells and in the natural ageing cellular model. SA-β-gal staining and quantitative analysis results indicated that compared with H2O2 treatment group, the β-gal densities were significantly decreased in H2O2-treated NP cells pre-treated with GNF-5837 (Figure 4(d) and (e)).
Previous studies have demonstrated that AFP accelerates IVDD, recapitulating key features of ageing-associated IVDD, including oxidative stress, senescence and extracellular matrix degradation.38,39 To investigate the potential efficacy of GNF-5837 as a therapeutic intervention for IVDD in vivo, we administered GNF-5837 in an AF puncture-induced IVDD rat model. The MRI results revealed significant improvements in IVD height and water content in the GNF-5837 treatment group, indicating that oral administration of GNF-5837 significantly alleviated IVDD in the rat model (Figure 4(f) and (h)). Moreover, IF and western blot results demonstrated that the expression levels of GPX7 were significantly increased in the GNF-5837-treated group than in the AF puncture group (AFP; Figure 4(g) and (i), Supplemental Figure S1D). Collectively, these findings suggested that GNF-5837 attenuated cellular ageing, consequently alleviating IVDD through the upregulation of GPX7 expression both in vitro and in vivo.
Discussion
IVDD is a common degenerative disease of the musculoskeletal system and a major cause of chronic LBP. 1 It is estimated that approximately 80% of individuals experience back pain symptoms at some point in their lives. 3 Previous studies have suggested that IVDs undergo degeneration with increasing age and are closely associated with the ageing of NP cells.40,41 However, the underlying mechanisms of ageing-related IVDD and the genes involved in this process still need to be further studied.
An increasing number of studies have indicated that H2O2-generated ROS lead to an oxidative stress environment in cultured NP cells and subsequently activate the p53-p21-Rb and p16-Rb pathways to promote cellular ageing.33,42 SA-β-gal activity can also rise in non-senescent conditions, such as cell confluence, serum starvation and oxidative stress. GPX7 has been associated with cellular ageing due to its role in regulating redox balance, protecting proteins from oxidative damage, and reducing the accumulation of damaged or misfolded proteins. This protective mechanism underscores GPX7’s potential involvement in slowing age-related cellular deterioration. 43 To explore the role played by GPX7 in the pathogenesis of IVDD, we treated human primary NP cells with H2O2 and found that the expression of GPX7 was significantly increased and that the percentage of senescent NP cells was increased. Consistent with our findings, Wang et al. 26 reported that alginate oligosaccharide alleviates vascular ageing and reduces ageing-related vascular remodelling via the GPX7 antioxidant pathway and that GPX7 depletion disrupts its geroprotective effect. Moreover, our bioinformatics analysis results indicated that GPX7 expression was upregulated in IVDD patient samples. Thus, we hypothesised that GPX7 upregulation occurred in response to H2O2 treatment, that is, oxidative stress, but not natural ageing. Our GPX7 overexpression experiments confirmed its protective role in NP cells under oxidative stress. Overexpression reduced key senescence markers (p53, p21 and p16) and inflammatory cytokines, notably IL-6, while lowering mitochondrial ROS levels. These findings highlight GPX7’s potential in mitigating oxidative damage, supporting its therapeutic relevance in countering NP cell ageing and IVDD progression.
Cell senescence refers to a phenotype of functional decline that results in damage with age. 44 It can be regarded as a process in which cells undergo a permanent proliferation arrest state driven by sustained internal and external stress and damage, along with the physiological dysfunction that occurs throughout their lifespan. 45 It is well accepted that human primary NP cells have a limited capacity for proliferation under in vitro conditions. 46 A previous study revealed that human NP cells exhibit a finite lifespan in culture. 47 With increasing culture passage number, NP cells exhibit typical characteristics of cellular senescence, including telomere shortening, DNA damage, mitochondrial dysfunction and nutrient deprivation. Therefore, in this study, we established a natural ageing cell model by continuous passage to simulate natural ageing. The present study revealed that the cellular response to oxidative stress was weakened in the natural ageing cell model (P9) and that the capacity of the cells to regulate the expression of GPX7 was restrained. H2O2 treatment resulted increased senescence marker (p53, p21, p16) and inflammatory cytokines expression levels and an elevated senescent ratio. Therefore, we hypothesise that, TRK-activated pathways like PI3K/AKT may support GPX7’s function in maintaining cellular integrity under stress by promoting cell survival and reducing apoptosis. This interaction between TRK and GPX7 pathways in NPCs underscores a combined protective mechanism, potentially slowing degeneration in IVDs through coordinated regulation of cellular stress responses.
Excessive ROS-mediated oxidative stress accelerates IVDD by inhibiting the PI3K-Akt pathway. 48 Moreover, several studies have shown that cancer cells can promote glycolysis and lactic acid secretion by activating the PI3K-AKT-mTOR pathway, thus increasing ATP production and promoting proliferation.49,50 To decipher the underlying mechanisms of GPX7-mediated cellular ageing, KEGG analysis was performed. We identified several pathways, including the PI3K-Akt signalling pathway, which were involved in the process of IVDD.
The utilisation of molecular docking to identify compounds as novel treatment methods for IVDD has become increasingly prevalent in recent studies.51–53 In this study, we predicted the ability of GNF-5837 to bind with GPX7 and subsequently validated its ability to increase GPX7 expression in vitro and in vivo. Interestingly, our results demonstrated that GNF-5837 treatment notably attenuated the degeneration of IVDs, as evidenced by increased signals on T2-weighted MR images and elevated expression levels of GPX7. Taken together, the findings of this study indicated that the upregulation of GPX7 could repress the progression of IVDD and supported our hypothesis that regulating GPX7-mediated cellular ageing is a promising strategy for the biological treatment of IVDD.
However, this study also has several limitations. We did not include Ki67 and pRPS6 staining in our current study, which limits our ability to distinguish senescent cells from quiescent cells. Specifically, Ki67 is used as a marker for cell proliferation, while pRPS6 identifies active protein synthesis, differentiating senescent cells from quiescent ones. Cells characterised by low Ki67 and high SA-β-gal activity, along with active RPS6, are identified as senescent. 54 We identified the involvement of the PI3K-Akt pathway in IVDD, but the mechanism underlying its deactivation and whether it was correlated with GPX7-regulated cellular ageing remain unclear. GNF-5837 is currently regarded as a potent pan-TRK inhibitor. Previous studies have reported that TRK family members can inhibit cell apoptosis and promote cell cycle progression by activating the PI3K-Akt pathway, which suggests a potential function of GNF-5837 in inducing G0/G1‑phase arrest and apoptosis.55,56 Although our results confirmed that GNF-5837 could alleviate IVDD by increasing GPX7 expression, its side effects still require further examination. As such, before it can be administered to humans to treat ageing-related diseases, several issues and hurdles must be addressed, including determination of the optimal dose and duration of treatment.
Conclusions
This study demonstrates the critical role of GPX7 in mitigating oxidative stress-induced cellular ageing and its contribution to IVDD by regulating senescence markers and inflammatory factors. GPX7 overexpression was shown to counteract oxidative damage, while the small molecule GNF-5837 effectively upregulated GPX7 and alleviated IVDD in vivo. Our findings provide a foundation for developing novel therapeutic strategies for IVDD.
Supplemental Material
sj-csv-2-iji-10.1177_03946320251343365 – Supplemental material for GNF-5837 alleviates intervertebral disc ageing by upregulating glutathione peroxidase 7
Supplemental material, sj-csv-2-iji-10.1177_03946320251343365 for GNF-5837 alleviates intervertebral disc ageing by upregulating glutathione peroxidase 7 by Yangkai Xu, Rongsheng Chen, Yan Zhuang and Weihong Xu in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-csv-3-iji-10.1177_03946320251343365 – Supplemental material for GNF-5837 alleviates intervertebral disc ageing by upregulating glutathione peroxidase 7
Supplemental material, sj-csv-3-iji-10.1177_03946320251343365 for GNF-5837 alleviates intervertebral disc ageing by upregulating glutathione peroxidase 7 by Yangkai Xu, Rongsheng Chen, Yan Zhuang and Weihong Xu in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-tiff-1-iji-10.1177_03946320251343365 – Supplemental material for GNF-5837 alleviates intervertebral disc ageing by upregulating glutathione peroxidase 7
Supplemental material, sj-tiff-1-iji-10.1177_03946320251343365 for GNF-5837 alleviates intervertebral disc ageing by upregulating glutathione peroxidase 7 by Yangkai Xu, Rongsheng Chen, Yan Zhuang and Weihong Xu in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
The authors would like to express their gratitude to everyone for assisting in the preparation of this manuscript. We also appreciate the support of The First Affiliated Hospital of Fujian Medical University.
Author contributions
Yangkai Xu and Weihong Xu designed the study, Yan Zhuang collated the data, Yangkai Xu and Rongsheng Chen drafted the manuscript, carried out the data analyses, produced the initial draft of the manuscript, and contributed to collecting the specimens and tissue sections. All the authors read and approved the final manuscript.
Data and materials availability
All data of this study are included within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: 1. The Natural Science Foundation of Fujian Province (2023J011535); 2. Fujian Provincial Clinical Medical Research Center for First Aid and Rehabilitation in Orthopaedic Trauma (2020Y2014).
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the First Affiliated Hospital of Fujian Medical University. No human samples were used, and ethics approval about human is not applicable. All the methods were carried out in accordance with the approved guidelines, and the full experimental procedures were carried out under the guidance of the Institutional Animal Care and Use Committee of The First Affiliated Hospital of Fujian Medical University (FZS-2021136). All methods are reported in accordance with the ARRIVE guidelines.
Patient consent for publication
Not applicable, this study does not include human tissue.
Animal welfare
The present study followed international, national and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
All the methods were carried out in accordance with the approved guidelines, and the full experimental procedures were carried out under the guidance of the Institutional Animal Care and Use Committee of The First Affiliated Hospital of Fujian Medical University (FZS-2021136). All methods are reported in accordance with ARRIVE guidelines.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
