Abstract
We investigated electroencephalographic (EEG) responses to tail clamping in lightly anaesthetized rat pups (5–22 days) in order to determine the ontogeny of EEG activity and at what age they may be capable of experiencing pain. Median frequency (F50) and spectral edge frequency (F95) of the power spectrum in the range of 1–30 Hz were determined before and after the application of a noxious stimulus and power spectra were compared by multivariate analysis. There was a postnatal increase in EEG power as, before clamping, pups aged 5–7 days exhibited isoelectric traces, whereas those aged 12–14 days and 21–22 days had intermittent EEG activity where the power in all frequencies was significantly lower at the former than at the latter age. Pups aged 5–7 days exhibited no EEG response to clamping in view of their isoelectric traces. Pups aged 12–14 days showed a significant decrease in F95 (P = 0.002), whereas those aged 21–22 days showed highly significant reduction in F50 and F95 (P = 0.028 and P < 0.001, respectively) as well as changes in EEG power of specific frequencies after clamping. The results and related literature suggest that rat pups aged 5–7 days and younger are not likely to perceive pain and that the ability to perceive pain develops gradually between postnatal ages 12–14 days and 21–22 days.
For an animal to experience any sensation, including pain, two prerequisites have to be met. 1 First, the animal must be sentient. This means that its nervous system must have developed sufficiently to relay sensory impulses from peripheral organs and tissues to the higher centres of the brain where they can be interpreted in terms of the sensory modality involved. Secondly, the brain must be in a functional state of consciousness, because unconsciousness nullifies perception. As the neurological maturity of mammalian young at birth shows wide variation, ranging from exceptional immaturity (e.g. marsupial joeys) through moderate immaturity (e.g. rat pups) to maturity (e.g. lambs), the timing of the onset of the capacity for pain perception may be expected to vary between species. 2,3 This timing is important for the humane management of newborns, especially in experimental settings (e.g. Henare et al. 4 ). The rat pup is the focus of the present paper as it has been used in previous work exploring the ontogeny of peripheral pain mechanisms. 5–12
The cerebral cortex and associated structures are known to be involved in the perception of pain. 13–16 While the general ontogeny of such higher brain centres of the rat pup is well understood, little is known about the maturation of nociceptive pathways in the brainstem, thalamus and cortex, 9,10 or about the time at which there is conscious perception of pain.
Changes in cerebral activity associated with the cognitive perception of pain are thought to be reflected in electroencephalographic (EEG) spectra. 17,18 A study of ontogenic changes in the EEG of newborn rats and associated differences in EEG spectra in response to painful stimuli may therefore aid our understanding of the onset and development of conscious perception of pain in these animals.
The literature is equivocal regarding the onset of EEG activity in rat pups. Some researchers report weak EEG activity from days 1 to 4 after birth. 19–21 Others report that EEG activity is present at 5–7 days of age, being initially characterized by continuous, but irregular, low-voltage slow waves. 22 Studies of the development of rapid eye movement (REM) and non-REM sleep-like EEG patterns show that during the first 10–12 days after birth rat pups show signs of undifferentiated sleep-like states of unconsciousness. 23–26 REM–non-REM differentiation begins from about 10–12 days after birth and sleep–wake cycles also appear during this period. Whether or not the wakefulness associated with developing sleep–wake cycles can be said to be ‘conscious wakefulness’ 27 is not apparent from the literature available. Adult-like sleep–wake patterns, which presumably include conscious wakefulness, have been reported to appear from about 18–20 days after birth. 23,24,28 Although these findings do not provide evidence that newborn and young rats are able to experience pain, they do provide a basis for judging when pain experience is unlikely (i.e. before electrical activity is present in the cortex), when it is more likely (i.e. once cortical electrical activity is present and EEG differentiation into REM–non-REM sleep cycles begins to develop) and when it is present (i.e. once the EEG exhibits fully differentiated REM, non-REM and conscious waking cycles).
The aim of this study was to investigate cerebral responses to a noxious stimulus in young rat pups before, during and after EEG differentiation to determine the age at which they are likely to experience pain. A minimal anaesthesia model was used on the basis that the EEG responses to potentially painful stimulation in conscious and anaesthetized animals 29,30 and in conscious humans 18 are broadly similar. This model relies on noxious stimuli evoking changes in EEG spectra in animals that are lightly anaesthetized with halothane. 30–32 Halothane is used on the basis that it has no analgesic properties and causes less depression of the cerebral cortex than other anaesthetic agents. 33 The use of this model allows the ontogeny of nociception to be investigated in a humane manner. Moreover, the contamination of EEG records by movement artefact is minimized. EEG responses to tail clamping were examined in lightly anaesthetized rat pups at 5–7, 12–14 and 21–22 days after birth and the results are reported and interpreted in the Result section.
Materials and methods
Animals
Outbred Sprague-Dawley rat pups of either gender were obtained from the small animal production unit at Massey University. Beforehand, adult rats and their pups were housed in a controlled environment (12:12 light/dark cycle with dawn and dusk transitional periods, room temperature 22±1°C). The pups were weaned at 20 days and subsequently maintained on a commercially available pelleted diet with access to water ad libitum. The rat pups were assigned by postnatal age to three groups: 5–7-day pups (n = 15), 12–14-day pups (n = 14) and 21–22-day pups (n = 14). This study was approved by the Massey University Animal Ethics Committee (Protocol 06/12).
Experimental procedure
Experiments were conducted during daylight hours between 09:00 h and 16:00 h. The weighed pups were each anaesthetized in a chamber with halothane (Merial, Parramatta, Australia) in oxygen to effect and were subsequently maintained on 2% halothane in oxygen delivered via a facemask. Heat loss was minimized by a heat lamp (250 W) during induction and subsequently by a heated pad. Rectal temperature was monitored in pups in the two older groups using a temperature probe (Dual Input Digital Thermometer, Dick Smith Electronics, New Zealand), whereas, in the younger 5–7-day pups, the temperature between the animal and the pad was recorded.
Needle electrodes (Medelec, Oxford Instruments Medical Systems, Surrey, UK) were placed transdermally (12–14 and 21–22-day pups; EEG recordings) or at a shallow angle through the cranium (5–7-day pups; electrocorticographic [ECoG] recordings). A three-electrode montage was used with the non-inverting electrode in the midline over the frontal sinus, the inverting electrode over the left mastoid process and the ground electrode caudal to the occipital process. The animals were stabilized for 2 min following electrode placement, prior to recording a 5 min baseline EEG/ECoG. The tail was then stimulated mechanically at its mid-point by application of a haemostat closed to the first ratchet and held for 10 s (supramaximal stimulus). Recordings were taken during and for 5 min following this stimulus. The pups, while still anaesthetized, were subsequently euthanized by intraperitoneal overdose of pentobarbitone sodium (National Veterinary Supplies, Auckland, New Zealand).
Pup behaviour
A note was made of any body movements or vocalizations following the placement of electrodes and the clamping of the tail.
EEG recordings
The EEG/ECoG was recorded continuously using suitably isolated biological amplifiers (World Precision Instruments Inc, FL, USA) set to a gain of 1000 and a pass band of 0.1–0.5 kHz. Data were digitized using an analogue to digital recording system (PowerlabTM, AD Instruments Ltd, Bella Vista, Australia) at a sampling rate of 1 kHz. Analysis of EEG data was conducted offline following completion of each experiment.
EEG/ECoG analysis
Isoelectric periods were present in the EEG/ECoG traces of all pups. The percentage of time occupied by isoelectric periods of 300 ms or longer during the last minute before (baseline recording) and the first minute after tail clamping, together with the number and average duration of isoelectric periods, were determined. ECoG traces for 5–7-day pups were largely isoelectric with regular electrical excursions of too brief a duration to allow fast Fourier transformation (FFT) analysis. Mean features of 10 of these excursions immediately before and after clamping were recorded. Parameters included amplitude from lowest to highest points (μV), duration (ms) and interval between excursions (ms). Excursions containing movement artefact were excluded from analysis.
For each 12–14 and 21–22-day pup, 10 continuous epochs (5 before clamping and 5 after clamping) of non-isoelectric EEG longer than 0.5 s were selected and subjected to spectral analysis (sample size, 1024 samples; cosine bell, 50% overlap; bin width, 0.97/0.98 Hz; filtered at 30 Hz); uneven samples were fitted using zero padding. EEG epochs shorter than 10 s were assumed to be stationary. Longer epochs were tested for stationarity using specialized software (FFT programme, CB Johnson, Massey University, New Zealand). Epoch lengths for 12–14-day pups were 1.794 ± 0.178 s and 2.002 ± 0.250 s before and after clamping, respectively, and for 21–22-day pups were 2.391 ± 0.317 s and 3.249 ± 0.675 s before and after clamping, respectively. Epochs containing movement artefact were excluded from analysis. Median frequency (F50) and 95% spectral edge frequency (F95) were calculated from frequency spectra (1–30 Hz) using standard statistical techniques. The F50 is the frequency below which 50% of the total power of the EEG is located, whereas the F95 is the frequency below which 95% of the total power of the EEG is located. 34
Statistical analyses
Statistical analyses were undertaken using SPSS 11.0 for MAC OSX (SPSS Inc, Chicago, USA). Data are presented as mean ± standard error of the mean. Differences were considered significant at P = 0.05 or less. Data were tested for normality using the Shapiro-Wilks statistic (if n < 50) or the Kolmogorov-Smirnov statistic (if n > 50) and normal probability plots.
Body temperature
Body temperature data were analysed by one-way analysis of variance (ANOVA) and differences in body temperature between the three groups of rat pups were assessed by Tukey post hoc test.
Isoelectric periods, features of electrical excursions (5–7 days) and EEG epochs (12–14 and 21–22 days)
The effect of clamping on each of the EEG/ECoG parameters was assessed by Wilcoxon signed rank test. Between-group comparisons were conducted using the non-parametric Kruskal-Wallis test followed by the Mann-Whitney test for pairwise comparisons to detect differences among groups.
Spectral analysis
Age differences in F50 and F95 were assessed using the Mann-Whitney test and independent-sample t-test, respectively. The effect of clamping on F50 and F95 was assessed using the two-related sample t-test. Where necessary logarithmic transformation was performed (F50, 21–22-day pups). Subsequently, log-transformed sums of frequency spectra blocks for each frequency in the range 1–30 Hz, as well as means of log-transformed frequency spectra for all groups (12–14 vs. 21–22-day pups; before and after clamping) were graphed and evaluated (Figures 1–3). Differences between frequencies were investigated using multivariate statistics (principal component [PC] analysis) to describe the orthogonal axes of variation of log-transformed data (frequencies 1–30 Hz, 10 EEG periods per animal – 5 before and 5 after clamping). The PC loadings were subsequently evaluated and the PC scores were plotted against each other in reduced dimensions to explore variation with treatment and age group. The scores were also analysed, where relevant, by one-way ANOVA with the Tukey post hoc test to assess the significance of any differences between the various groups.
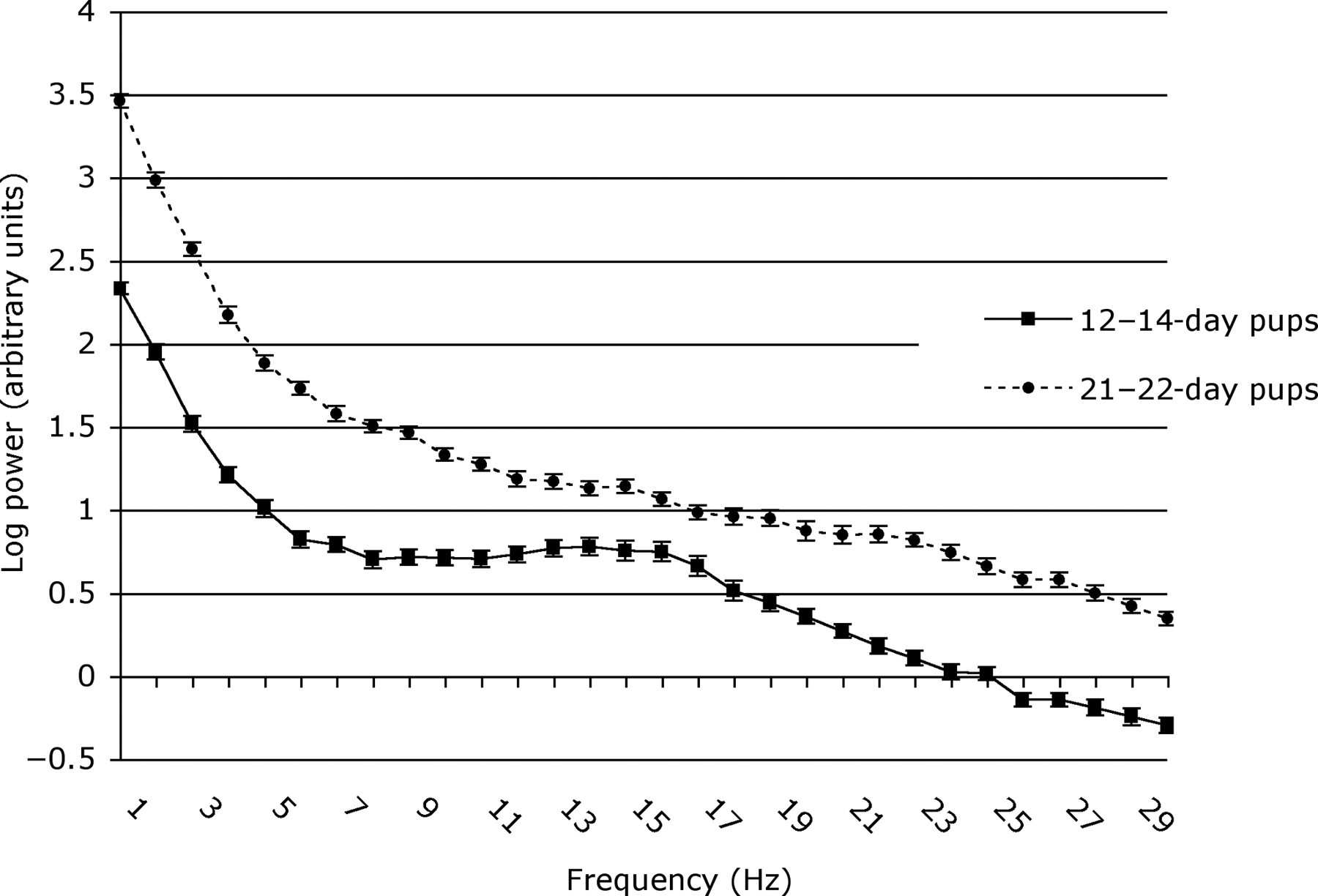
Mean power (log) (±SEM) in frequencies of the EEG spectrum during baseline observations for 12–14 and 21–22-day pups
Results
General information
Mean body weight and temperature (skin and rectal) are shown in Table 1. Temperature was stable throughout the period of observation. Due to methodological differences, the small between-age differences shown were not anticipated to have any relevant biological significance. Heart rate, derived from electrocardiographic artefact in the EEG signal, was also stable in all pups throughout the study (321–328, 334–338, 359–383 beats per minute, respectively for 5–7, 12–14 and 21–22-day pups).
Details of weight and rectal/skin temperature of rat pups of the three ages
ST: skin temperature; RT: rectal temperature
*Mean
†Number of animals
§Standard deviation
‡Standard error
Behaviour
Body movements, recorded as present or absent but not characterized, occurred in 64–69% of the pups at each age in response to electrode placement and in 93, 46 and 50% of 5–7, 12–14 and 21–22-day pups, respectively, in response to tail clamping. There were audible vocalizations in response to electrode placement and clamping, respectively, in 15 and 69% of 5–7-day pups, but none in the older pups.
Isoelectric periods, characteristics of brief electrical excursions and EEG epochs
5–7-day pups
ECoG traces for 5–7-day pups were largely isoelectric with regular electrical excursions that were of too brief a duration to permit FFT analysis. The percentage of time occupied by isoelectric ECoG periods and the average duration of these periods did not differ before and after clamping (percentage: 78 ± 3 and 75 ± 2%, df 1, 29, Z = −0.909, P = 0.363; duration: 1.9 ± 0.7 and 1.7 ± 0.5 s, df 1, 27, Z = −0.659, P = 0.510). The amplitude of electrical excursions was lower (42 ± 5.5 vs. 63 ± 11.5 µV; df 1, 27, Z = −2.731, P = 0.006) and were shorter (1.4 ± 0.4 vs. 1.7 ± 0.5 s, df 1, 27, Z = −2.229, P = 0.026) for the 10 excursions observed before compared with after clamping. Although the intervals between electrical excursions were similar before and after clamping for the 10 excursions investigated, the intervals before clamping had a tendency to be shorter than those after clamping (394±35 ms vs. 459 ± 38 ms; df 1, 27, Z = −1.195, P = 0.056).
12–14-day pups
The percentage of time occupied by isoelectric EEG periods and the average duration of these periods did not differ in 12–14-day pups before and after clamping (percentage: 55 ± 4 and 51 ± 5%, df 1, 25, Z = −1.608, P = 0.108; duration: 1.7 ± 0.2 and 1.7 ± 0.2 s, df 1, 25, Z = −0.314, P = 0.753). The frequency of isoelectric EEG periods (periods/1 min) before and after clamping did not differ significantly, but it tended to be higher before clamping (21 ± 1.9 vs. 19.3 ± 1.7 periods/1 min, df 1, 25, Z = −1.721, P = 0.085). There was no difference between the durations of epochs of EEG activity before and after clamping (1.8 ± 0.2 and 2.0 ± 0.3 s, df 1, 25, Z = −1.642, P = 0.101).
21–22-day pups
The percentage of time occupied by isoelectric EEG periods did not differ in 21–22-day pups before and after clamping, although it tended to be higher before clamping (23 ± 3 and 19 ± 2%, df 1, 27, Z = −1.789, P = 0.074). Isoelectric EEG periods were longer before than after clamping (709 ± 40 vs. 614 ± 30 ms; df 1, 27, Z = −2.606, P = 0.009). Again, the frequency of isoelectric EEG periods before and after clamping did not differ (18.6 ± 1.6 and 18.7 ± 1.3 periods/1 min, df 1, 27, Z = −0.491, P = 0.624). The difference in duration of epochs of EEG activity before and after clamping approached significance (P = 0.056), indicating a tendency for EEG epochs to be shorter before clamping (2.4 ± 0.3 vs. 3.3 ± 0.7 s).
Between-group comparison
The percentage of time occupied by isoelectric EEG/ECoG periods differed between the groups (df 2, 41, χ2 = 33.27, P < 0.001). The post hoc Mann-Whitney tests showed that this difference was highly significant between all groups (5–7 vs. 12–14-day pups: df 1, 27, Z = −3.801, P < 0.001; 5–7 vs. 21–22-day pups: df 1, 28, Z = −4.583, P < 0.001 and 12–14 vs. 21–22-day pups: df 1, 26, Z = −4.270, P < 0.001), with 5–7-day pups having the highest, 12–14-day pups intermediate and 21–22-day pups the lowest values (78 ± 3, 55 ± 4 and 23 ± 3%, respectively). The duration of isoelectric periods before clamping was significantly different among the three age groups (df 2, 40, χ2 = 23.791, P < 0.001). Mann-Whitney post hoc tests showed that the duration of isoelectric periods before clamping in 5–7 and 12–14-day pups was longer (1.9 ± 0.7 s and 1.7 ± 0.2 s, respectively) compared with that of 21–22-day pups (0.7 ± 0.04 s) (5–7 vs. 21–22-day pups: df 1, 27, Z = −3.997, P < 0.001; 12–14 vs. 21–22-day pups: df 1, 26, Z = −4.125, P < 0.001), but did not differ between each other (df 1, 26, Z = −1.698, P = 0.094). The frequency of isoelectric EEG periods (periods/1 min) before clamping did not differ significantly between the 12–14 and 21–22-day pups (21 ± 1.9 and 18.6 ± 1.6 period/1 min, df 1, 26, Z = −0.195, P = 0.867). Epochs of EEG activity tended to be shorter in 12–14-day pups than in 21–22-day pups, although this was not significant (1.8 ± 0.2 vs. 2.4 ± 0.2 s, df 1, 26, Z = −1.893, P = 0.061).
Spectral analysis
The absence of epochs of ECoG activity in 5–7-day pups precluded spectral analysis. Thus the analysis was restricted to EEG epochs of 12–14 and 21–22-day pups that varied in duration from 0.5 to 27 s.
Median frequency (F50) and spectral edge frequency (F95)
The F50 values of 12–14-day pups before and after clamping were similar (4.87 ± 0.38 vs. 4.66 ± 0.33 Hz, df 1, 12, t = 0.468, P = 0.648), but were lower in 21–22-day pups after clamping (3.35 ± 0.16 vs. 2.99 ± 0.07 Hz, df 1, 13, t = 2.471, P = 0.028). The F95 decreased in response to clamping in pups of both ages (12–14-day pups: 19.37 ± 0.49 vs. 18.57 ± 0.53 Hz, df 1, 12, t = 4.061, P = 0.002; 21–22-day pups: 15.65 ± 0.80 vs. 11.61 ± 0.80 Hz, df 1, 13, t = 5.652, P < 0.001). Overall, baseline (i.e. preclamping) F50 and F95 decreased with age between 12–14 and 21–22 days (F50, 4.87 ± 0.38 vs. 3.35 ± 0.16 Hz, df 1, 129, Z = −4.790, P < 0.001; F95, 19.37 ± 0.49 vs. 15.65 ± 0.80 Hz, df 1, 129, t = 5.355, P < 0.001).
Graphic comparisons
Direct graphical comparison of standardized frequency spectral sums and means of log-transformed data showed differences in the profiles of the curves between the two age groups (Figure 1) as well as before and after clamping (Figures 2 and 3). The differences in the features of these graphs were explored by multivariate analysis (PC analysis). The analysis identified five orthogonal PCs with eigenvalues >1 that contributed 78.5% of the overall variation in the data (Table 2). The component loadings in PC1 were all high (≥0.5) (Table 2), indicating that this axis of variation represented overall power and possibly the duration of epochs of EEG activity. The pattern of component loadings for PC2 contrasted powers of frequencies between 1 Hz and 12 Hz (negative) with those between 14 Hz and 25 Hz (positive). The pattern of component loadings for PC3 contrasted powers of frequencies 9–17 Hz (positive) with those between 19 Hz and 30 Hz (negative). The pattern of component loadings for PC4 contrasted the power of frequencies between 1–7 Hz and 16–19 Hz (negative) with those between 12–14 Hz and 22–29 Hz (positive) The pattern of component loadings for PC5 contrasted the power of frequencies between 1, 7–10, 14 and 19–25 Hz (positive) with those of 2, 12, 16, 17 and 26–30 Hz (negative). However, these loadings were smaller than 0.4 in the majority of cases (Table 2).
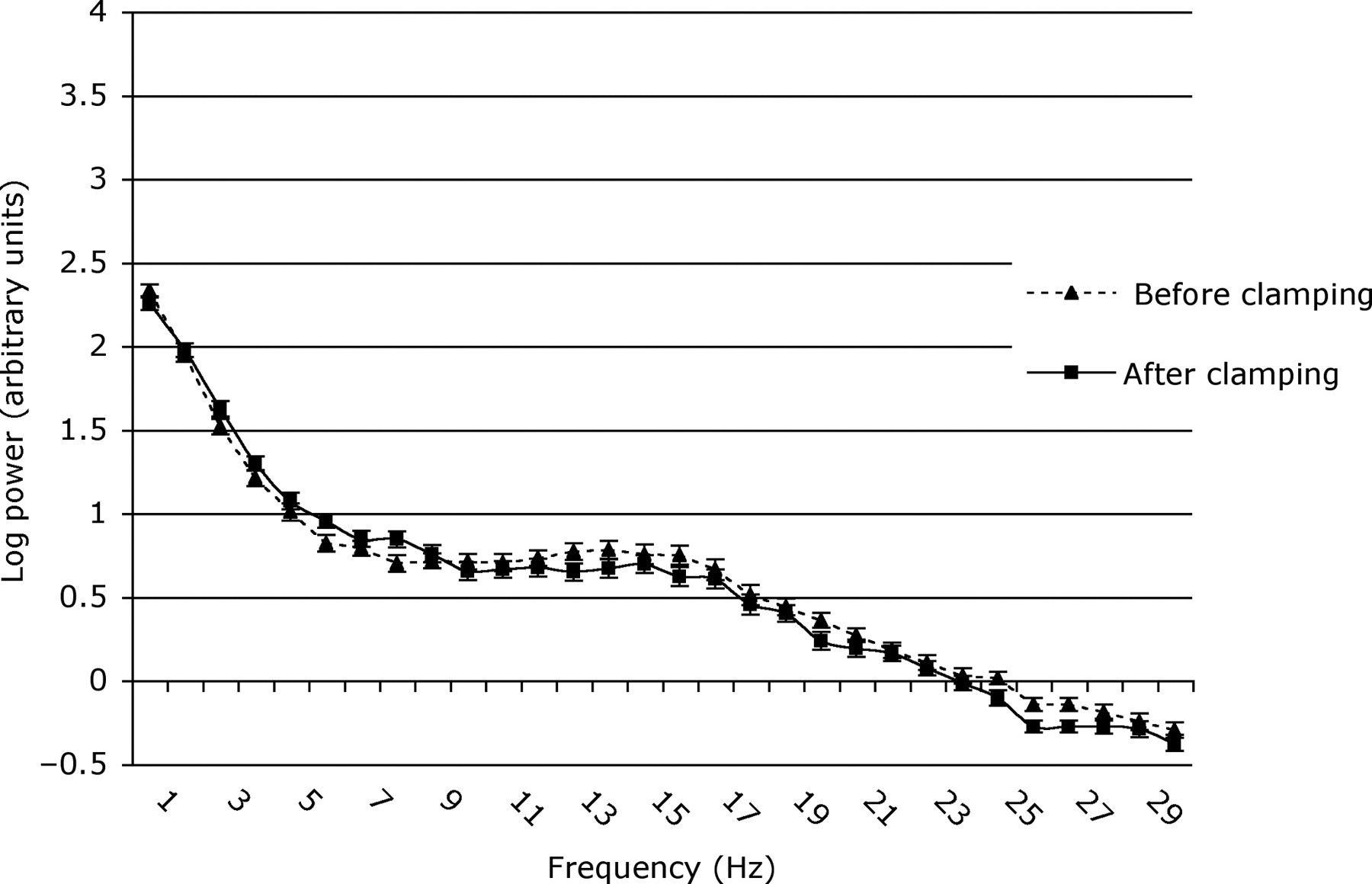
Mean power (log) (±SEM) in frequencies of the EEG spectrum of 12–14-day pups before and after clamping for all epochs of EEG activity
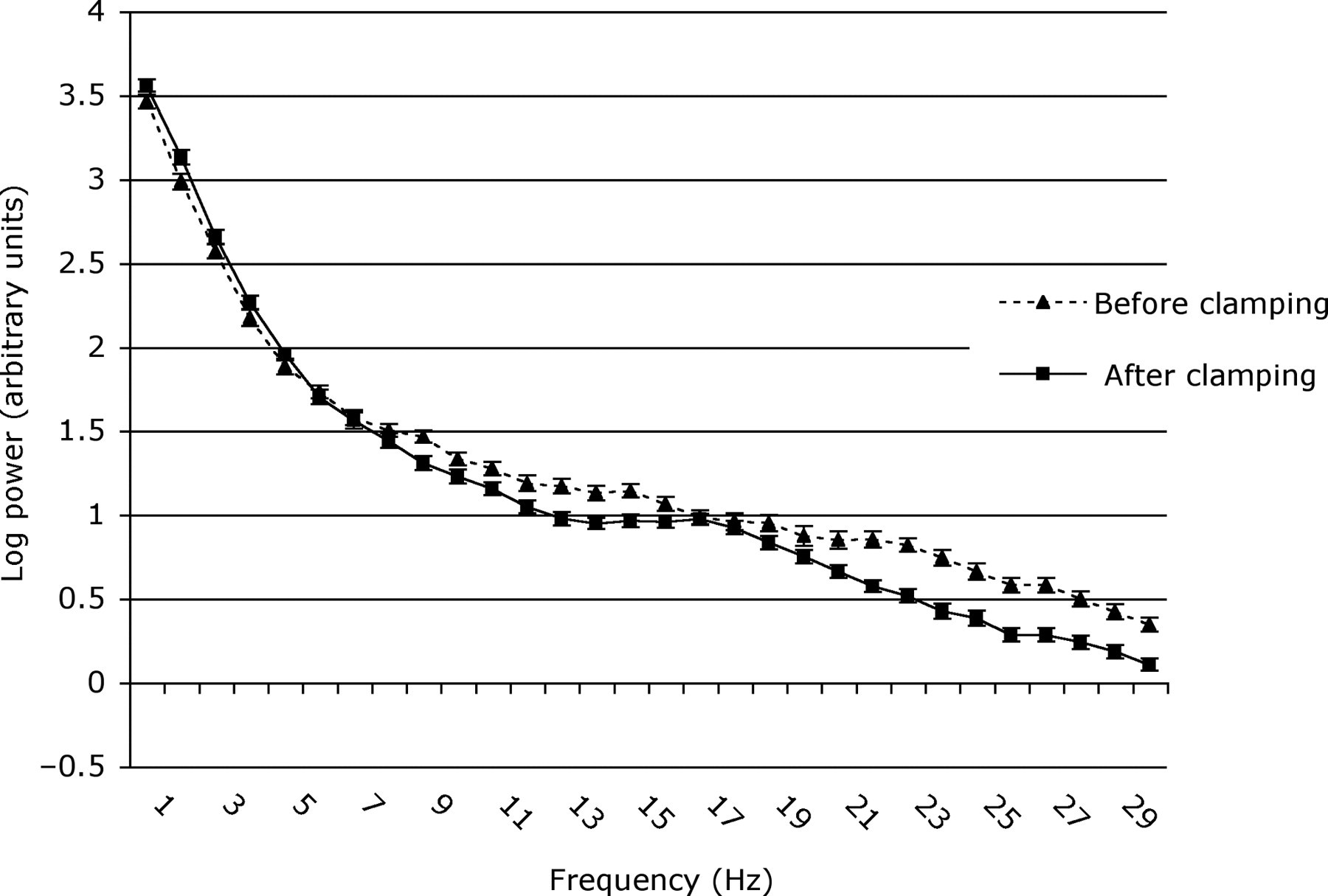
Mean power (log) (±SEM) in frequencies of the EEG spectrum of 21–22-day pups before and after clamping for all epochs of EEG activity
Eigenvalues and component scores of all frequencies for principal components (PCs) 1–5 calculated by PC analysis
It was possible to distinguish between the two age groups and the two treatments on the basis of the PC scores along the first and fourth axes of variation (Figure 4). Hence 12–14-day pups had significantly lower PC1 factor scores on ANOVA than those aged 21–22 days (df 3, 266, F = 189.01, P < 0.001), indicating that the overall EEG power was lower in the younger group. The separation of data according to the fourth PC showed that no distinction was possible on the basis of PC scores of EEG frequencies of 12–14-day-old pups before and after clamping. However, this was possible for 21–22-day pups in which the PC4 scores for the EEGs were significantly higher on ANOVA before clamping than after clamping (df 3, 266, F = 25.45, P = 0.001). Thus, EEG power in 21–22-day-old pups decreased in frequencies of 12–14 Hz and 22–29 Hz and increased in frequencies of 1–7 and 16–19 Hz after clamping.
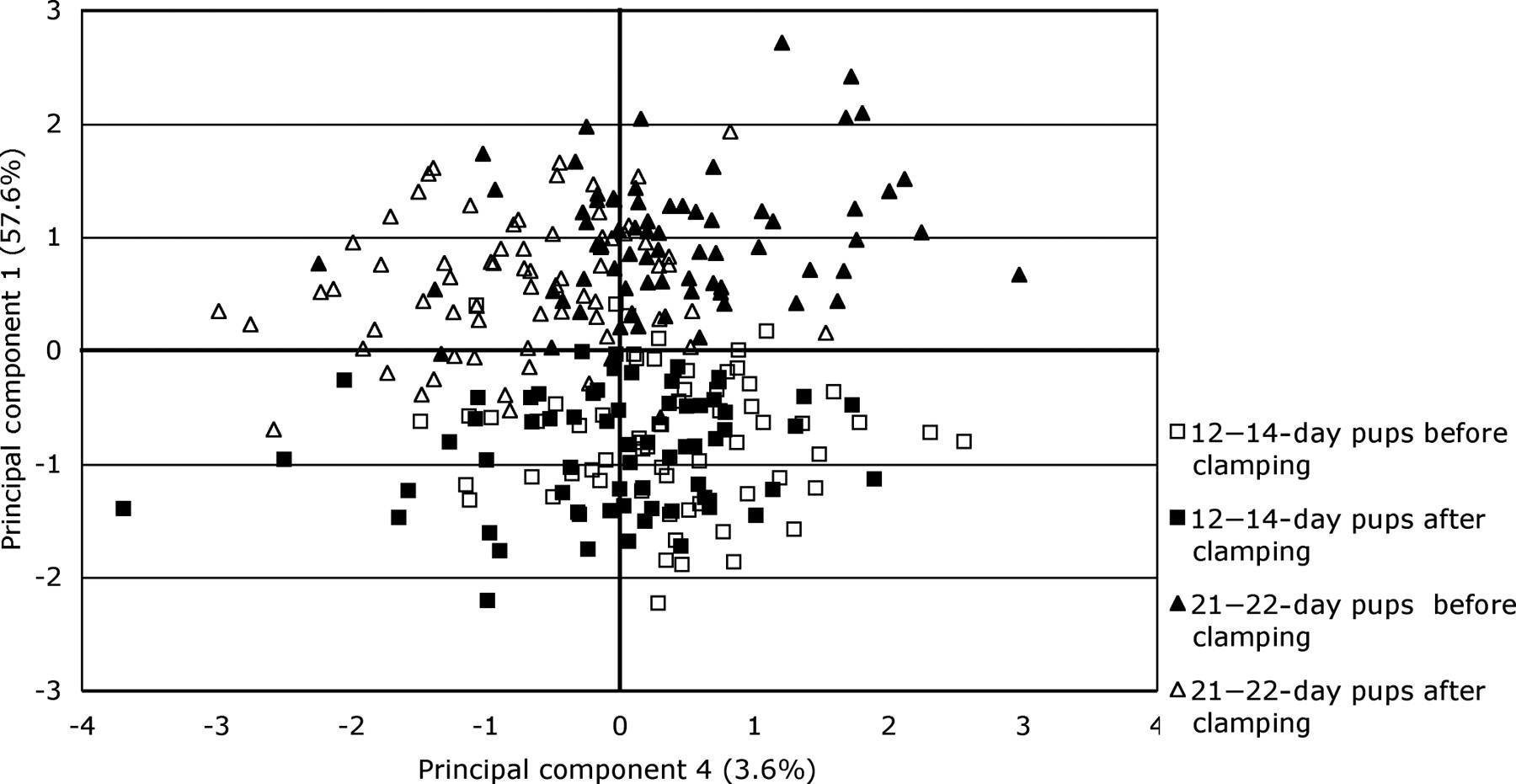
Factor scores plotted against the first and fourth principal component axes showing separation according to age (12–14 vs. 21–22-day pups) and tail clamping (12–14 and 21–22-day pups before vs. after clamping)
Discussion
As far the authors are aware, this is the first investigation of the cerebral responses of young rat pups to noxious stimulation, in particular, EEG/ECoG responses of anaesthetized pups to tail clamping at ages up to 21–22 days after birth. The main findings were as follows. There was little or no ECoG activity in the rat pups at 5–7 days, but prolonged periods of active EEG were present by 12–14 days. The percentage of time occupied by isoelectric periods decreased and that of EEG activity increased between 12–14 days and 21–22 days. There was an age-related progression in response to clamping, with no response in 5–7-day pups due to the absence of ECoG activity, a marginal EEG response in 12–14-day pups and a significant EEG response in 21–22-day pups. Finally, behavioural and physiological responses to electrode placement and clamping were evident at all ages.
These findings presumably relate to well-established developmental changes in the central nervous system during the same postnatal period. These include myelination, proliferation of neurites, increases in synaptic vesicles and cortical synaptogenesis, 35 as well as the appearance of somatosensory evoked potentials from 1–5 days after birth, 36,37 auditory evoked potentials from 10–13 days, 38 and thereafter until 21 days after birth, a rapid maturation of the morphology of these potentials. 38–40
EEG characteristics and pup behaviour also exhibit developmental changes. A continuous or intermittent EEG consisting of irregular slow waves of low amplitude can be detected in rat pups at days 5–7. 19–21,23,41–44 Such pups huddle with littermates, suck from their mother, crawl, stretch, yawn and show rudimentary grooming. 45 The initially undifferentiated EEG begins to exhibit REM–non-REM differentiation and sleep–wake patterns between 12 and 18 days after birth, with an adult pattern being attained around 18–20 days. 23,24,28 Behaviourally from days 9–10, 45 walking, cage exploration and orientation away from the mother and nest are evident and these activities become more vigorous or prolonged in days 12–13. Eye-opening around day 14 is associated with increased attention to objects and, at day 19, the first obvious awareness of the investigator. Pups aged 17–20 days are often physically active, with fighting between littermates predominant. Physical activity decreases for about one day when the pups are weaned at day 21 by removal of the mother. 45
During the preclamping control period, the progressive postnatal decrease in the proportion of time occupied by isoelectric periods (5–7-day pups, 78%; 12–14-day pups, 55% and 21–22-day pups, 23%) and the concomitant increase in EEG activity presumably also reflected developmental changes in cerebral function in the anaesthetized rat pups studied here. Interestingly, isoelectric periods have not been reported in most previous studies of unanaesthetized rat pups of a similar age. This difference may be due to experimental methodology or to improvements in EEG recording equipment and analytical techniques during the intervening 30–40 years. Halothane has not previously been reported to induce EEG burst suppression, 33 but this possibility cannot be ruled out in rat pups without further study.
Developmentally, the overall increase between postnatal days 12–14 and 21–22 in the power of all frequencies (1–30 Hz) during control EEG recording (Figure 1) may reflect progressive maturation with age of the ascending reticular activating system (ARAS). The ARAS evidently participates in cortical arousal and sleep–wake cycles by integrating inputs from many other brain centres 46 and functional maturation of many of its components occurs during the first three weeks after birth. 47–51 Although the present pups were anaesthetized, structures of the ARAS will nevertheless exert effects on the cerebral cortex, as can be seen by the cortical responses to tail clamping in rat pups at 12–14 days and especially at 21–22 days (Figures 2–4).
As ECoG activity was mainly absent in the 5–7-day pups, no cerebral response to tail clamping could be observed. This, and the fact that the pups were under general anaesthesia throughout, suggest that the movement and/or audible vocal responses of these pups to electrode placement and tail clamping were due to subcortical activation of the autonomic nervous system and motor and vocal reflexes via nociceptive pathways. As EEG responses of older pups were not associated with audible vocalizations or more marked movement responses, it is likely that the greater behavioural responses observed in 5–7-day pups were due to immature descending inhibition. 9,10,12
It is possible that noxious stimulation may cause arousal sufficient to induce an onset of EEG activity. However, this was not observed here in response to the supramaximal stimulus. The increased amplitude observed in ECoG excursions in 5–7-day pups in response to tail clamping is difficult to explain. The authors speculate that these electrical excursions in 5–7-day pups were more likely to be breathing movement artefact than ECoG activity, so that the increase in amplitude in response to tail clamping may represent an increase in autonomic nervous system activity.
In contrast, the older pups responded to clamping both with body movements and significant changes in EEG spectra. As these spectral changes were more pronounced in the 21–22-day than in the 12–14-day pups (Figures 2–4), this presumably reflected maturation of cerebral functionality during this period and/or differences in the maturation of peripheral and central nociceptive pathways. In addition, it is possible that an increase in tail size/diameter may have contributed to the more pronounced changes in EEG parameters in the 21–22-day compared with the 12–14-day pups. Therefore, interpretation in terms of the potential for conscious perception of pain is not straightforward.
Previous studies investigating the cerebrocortical responsiveness of adult animals anaesthetized with halothane have observed an increase in F50 and F95, also known as EEG desynchronization/arousal. 32,34,52,53 A decrease in F50/F95, as observed in the present study, is commonly referred to as synchronization or paradoxical arousal. This has also been observed in anaesthetized infants and children in response to skin incision. 54 The causes of these differences between adult and young animals and humans apparently remain unclear. 55 An additional complication is that in newborn and young lambs, which are neurologically mature at birth, the EEG responded to noxious stimulation with desynchronization. 31,56 Further studies will be necessary to elucidate the causes of these differences.
Several studies have shown that young rats are in some respect more responsive to noxious stimulation than older rats. 5,6,9,12 These studies have relied mainly on behavioural parameters for pain assessment such as withdrawal reflexes, tail flick responses and paw lifting or licking. However, behavioural data may not definitively elucidate whether newborn animals experience pain and the character of such pain in comparison with that in the adult animal. In contrast, various studies have shown that EEG responses to noxious stimulation occur in anaesthetized animals, 30–32 including newborn and young lambs, 56 and that these are similar to EEG responses to painful stimulation in conscious animals. 29 Accordingly, the EEG responses observed here might be more informative than behaviour with regard to the potential for pain experience.
The structural links between the neuronal centres which generate REM–non-REM sleep cycles and conscious perception suggest that it is not likely that animals could be conscious prior to REM–non-REM differentiation during development. 27 In rat pups, this differentiation is not apparent before 10–12 days and is complete by about 18 days, by which age sleep–wake cycles are well established. 23–26,57 The lower EEG responses to noxious stimulation in the present 12–14-day pups compared with the 21–22-day pups are consistent with these observations.
The onset of sleep–wake cycles in rat pups is associated with an increase in level and complexity of behaviour including manipulation of objects, interactions with littermates and exploration of the environment. 45 Conscious perception may develop gradually over this period, rather than as an on–off phenomenon. Once the capability for consciousness is present, levels of conscious perception may increase as brain function matures. Overall, not enough is known about the physiological basis of conscious perception to go beyond this speculation.
Although the results of the present study do not inform us directly about whether or not rat pups are able to experience pain, they do suggest that there may be differences in the intensity and nature of the pain experienced by rat pups of different ages. The absence of REM–non-REM differentiation, together with neurological and behavioural evidence for cerebral immaturity, strongly suggest that consciousness and consequent perception of pain in rat pups younger than 10–12 days are doubtful. The marginal response to tail clamping in 12–14-day pups, together with their behavioural repertoire when not anaesthetized, 45 suggest that pups may be capable of experiencing pain from this age onwards, although this experience may be qualitatively different from that of mature animals. The cerebral responses to noxious stimulation of pups aged 21–22 days, the presence of sleep–wake cycles and pup behaviour at 18–20 days suggest that animals older than about 18 days are normally capable of conscious perception and pain experience.
Footnotes
Acknowledgements
We would like to thank Drs Paul Chambers and Joanna Murrell for their advice on anaesthesia and other matters, and Debbie Chesterfield, Corrin Hulls and Sheryl Mitchinson for their practical assistance. Support for related research projects from the Ministry of Agriculture and Forestry is gratefully acknowledged, as is the Doctoral Scholarship for TJD from the Agricultural and Marketing Research and Development Trust.
