Abstract
Summary
The golden hamster (
Hydrocephalus is one of the most frequently encountered neurological disorders of the brain and can be encountered in many species (Harrington et al. 1996). It is the result of a circulation disturbance of the cerebrospinal fluid and is more often congenital than acquired. In addition to the classical neurological forebrain symptoms, it can cause behavioural changes in affected animals (Lorenz & Kornegay 2004, Jaggy 2005).
In golden hamsters,
Golden hamsters are frequently used in behavioural studies as well as in studies on the environmental influences on the occurrence of hydrocephalus. Therefore, it is of great importance to introduce a clinical diagnostic procedure to eliminate hydrocephalic animals from the breeding programmes. It should be minimally invasive, economic, fast and easy to operate. In human as well as in veterinary medicine, different diagnostics have been used in the past. One of them is electroencephalography (EEG), the recording of spontaneous electrical activity of the brain (Lorenz & Kornegay 2004, Jaggy 2005).
The typical electroencephalogram seen in hydrocephalic dogs has a high-voltage slow wave activity (HVSA; 25–200 µV, 1–6 Hz) in all leads. In most cases, there is a fast superimposed activity (10–12 Hz) (Lorenz & Kornegay 2004). However, this pattern is not pathognomonic for hydrocephalic animals, but may also be seen with other chronic encephalopathies that destroy cortical parenchyma (Harrington et al. 1996, Jaggy 2005).
The purpose of this study was to investigate the usefulness of EEG in the screening for congenital hydrocephalus in golden hamsters.
Materials and methods
Animals
No specific selection took place in the composition of the study population with a total of 25 animals from the breeding programme of the Institute of Animal Genetics, Nutrition and Housing, Division of Animal Housing and Welfare, Vetsuisse Faculty, University of Berne, Switzerland (progeny of Crl: LVG [SYR]). The group consisted of 12 females and 13 males. None of the animals had any phenotypical signs of hydrocephalus. They were reared using standard practices and all handling followed animal welfare guidelines approved by the Cantonal Office for Agriculture and Nature. Their mean age at the time of the experiment was 49.7 days (standard deviation [SD]: 3.08) and their mean body weight (BW) was 66.3 g (SD: 8.5). The animals from this colony were chosen due to their known high morbidity for congenital hydrocephalus. A preceding study showed that 88.6% of the population were affected. Previous screenings had not shown any teratogenic levels of metals or mycotoxins in liver or food. In addition, the sera from several animals were negative for antibodies against Sendai virus, reovirus 3 and lymphocytic choriomeningitis virus. Test matings had confirmed that the hydrocephalus was hereditary, and it was presumed to be an autosomal recessive trait (Edwards et al. 2006).
The control group came from a colony of wild-derived hamsters (Gattermann et al. 2001, 2002) of the Institute of Zoology, University of Halle, Germany. In a previous neuropathological survey, six animals from this colony were examined and all were found to be free of hydrocephalus. There were a total of 12 healthy randomly chosen animals in the control group with an equal distribution of females and males. Their husbandry differed from the Berne colony in that their cages were smaller (55 × 33 × 20 cm), they had a different artificial light regime (light–dark cycle of 14–10 h), their relative humidity was higher (60–65%) and they were fed a different diet (one part altromin 7014 and two parts altromin 7024; Altromin GmbH, Lage, Germany). Their mean age at the time of the experiment was 48.7 days (SD: 2.31) and their mean BW was 80.1 g (SD: 10.0). They were brought to Berne four days before the launch of the experiment, and from the time of their arrival they were kept under the same living conditions as the animals from the Berne group.
Housing
Each hamster was held separately in its own cage, which was made up of a plastic bottom and a wire top (95 × 45 × 57 cm). All of them were equipped with a wooden shelter, a carton tube, a hazel branch, a sand bath, paper tissue as nesting material and a running wheel (30 cm diameter, 10 cm wide). Wooden shavings were used as litter (Allspan®, D-76151 Karlsruhe, Germany). The whole group was kept in a closed room where an artificial light regime (light–dark cycle of 12–12 h) was meticulously followed. Dusk began at 13:00 h (CEST), and the light decreased from 280 lux to 5 lux over a period of 30 min. The room temperature was kept between 21 and 23°C, and the relative humidity varied from 30–55%. Fresh fruits and vegetables were provided on a daily basis. In addition, the animals were supplied with grain feed (Hamster Food, Witte Molen®; NL-Meeuwen, Product number: 3005301) twice and curd cheese as a protein source once a week. Fresh tap water was offered
Experimental design
The study was performed at the Department of Clinical Veterinary Medicine, Vetsuisse Faculty, University of Bern, Switzerland, and in accordance with the
Anaesthesia
A plexiglas chamber (10 × 20 × 10 cm) in combination with an isoflurane vaporizer (Vapor®, Dräger Medical Deutschland GmbH, Lübeck, Germany) were utilized for the anaesthesia. Isoflurane was administered as 5% of partial pressure and delivered in 100% oxygen. First, the chamber was flooded with isoflurane before the animal was placed inside. Then the hamster received an isoflurane–oxygen flow of 3 L/min for 90 s, which was then reduced to 1.5 L/min and continued for another 60 s. After this all gas flow was discontinued. Now the lid was opened halfway and the depth of anaesthesia was determined. For this purpose, the loss of righting reflex as well as the effect of toe pinching were evaluated. For the assessment of the righting reflex the animal was placed in an unphysiological position. A failure to correct this position was defined as absence of the reflex. The toe pinching was standardized and performed with mosquito forceps. These tests are commonly employed in laboratory animal science and their absence indicates that a surgical level of anaesthesia has been reached. Thus, a comparable depth of anaesthesia was obtained in the tested animals. Subsequently, the electrodes were positioned onto the hamster's head as described below. These procedures were performed in approximately 60 s. The recording of the EEG was initiated immediately and continued until the animals started to awaken. During the recording, the anaesthesia was sustained by the oxygen–isoflurane mixture that remained inside the chamber. The animals remained inside the anaesthetic box throughout the whole procedure. Thus, we attempted to let the box empty gradually, to slow the waking of the animals. Everything was performed under an extractor hood, to prevent human exposure to the anaesthetic gases.
Electroencephalography
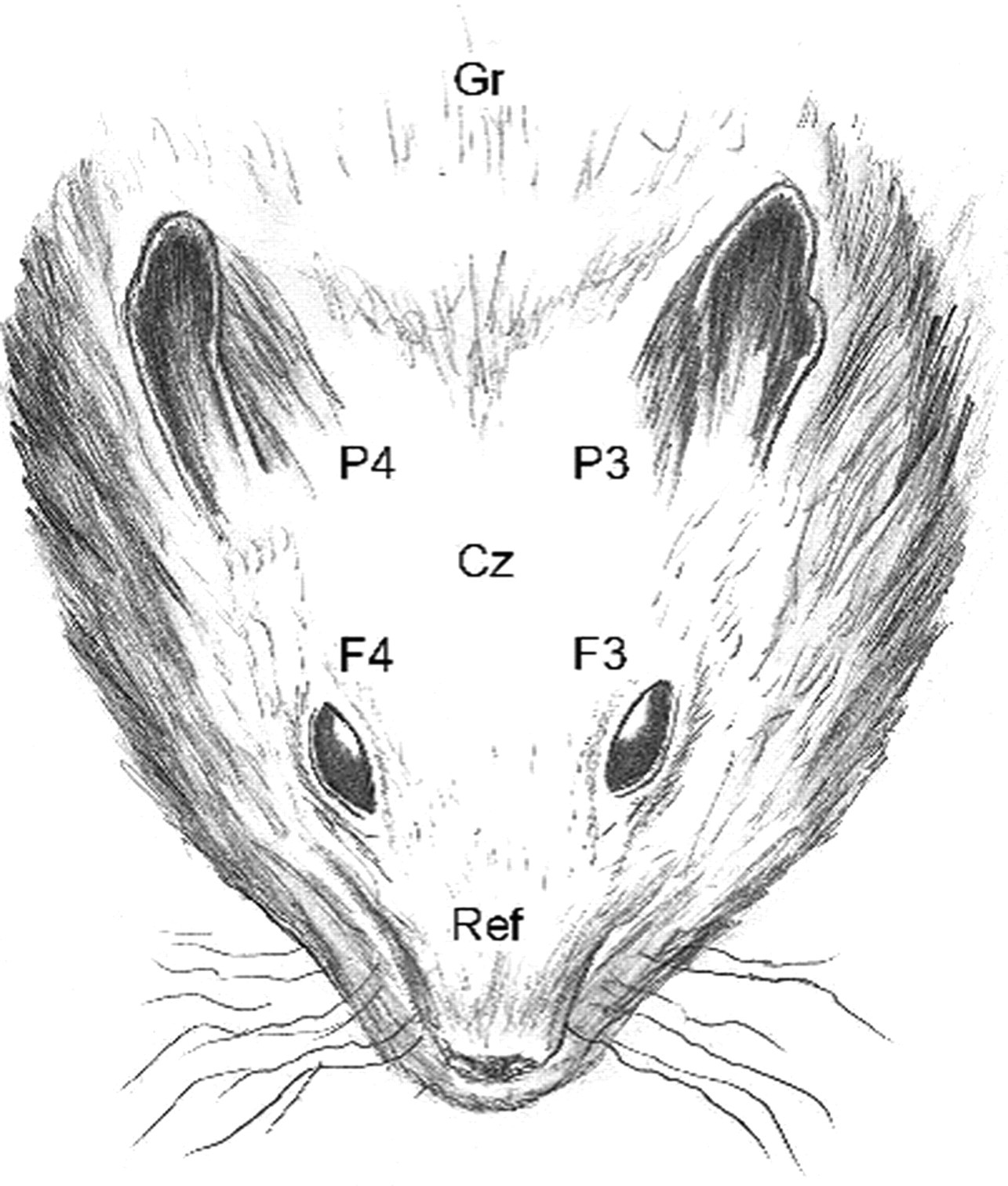
An eight-channel montage (F4-Cz, F3-F4, F3-Cz, F3-P3, F4-P4, P4-Cz, P3-P4, P3-Cz; speed = 30 mm/s; sensitivity = 70 µV/cm; band-pass filtered between 0.016 and 30 Hz; impendance: 10 Ω; Oxford Instruments Medical, Oxfordshire, UK, MedelecValor32 R) was applied to record bioelectrical activity. Seven EEG needles (18 mm monopolar stainless steel needle electrodes; camed GmbH, Switzerland) were used as subdermal active, reference and ground electrodes. Their placement together with a prior determination of anaesthetic depth were performed within 60 s. The active electrodes were arranged over the left and right frontal, parietal as well as central cranium area. The reference electrode was placed on the bridge of the nose between the eyes and the ground electrode caudally of the external occipital protuberance (Figure 1). The EEG was set for the standard lead system to be used and electrode resistances were tested. Bipolar EEG recordings were performed until the animals started to awaken, which was defined as the moment movement of the animal became apparent. This protocol was devised in a preliminary test several months prior to the experiments, where we conducted EEGs on two hamsters that were known to be affected. It is a slight modification of the method described for dogs (Srenk & Jaggy 1996). The evaluation of the EEGs was performed double-blind by a single examiner (MB). First, each recording was analysed visually and subsequently amplitude (µV) and frequency (Hz) were measured with the specific EEG analysis software (Oxford Instruments Medical, MedelecValor32). Six measurements (epochs), each with a duration of 1 s, of every single lead, were determined in the last minute of the recording. These were standardized at 60, 59, 40, 39, 20 and 19 s prior to the awakening of the animal.
Pathology
After the EEG recording, the animals were re-anaesthetized with isoflurane and immediately decapitated according to the guidelines of the Swiss Federal Veterinary Office (Bundesamt für Veterinärwesen 1993). The brains were removed from the skulls within 3 h of the euthanasia. Each brain was fixed in 10% phosphate-buffered formalin for 48 h and subsequently cut into coronary segments of approximately 4 mm. All of these were processed for paraffin embedding after which they were cut into 5 µm sections. From each segment, one section was mounted on a glass slide and stained with haematoxylin and eosin. A board-certified pathologist (CB), specialized in neuropathology, performed all macroscopic as well as histological examinations. The presence of hydrocephalus was further characterized using the following criteria: unilateral or bilateral, symmetric or not symmetric and the degree was subjectively assessed as minimal, mild, moderate or severe.
Statistical analysis
Descriptive statistics (mean, SD) and box plots were used to assess the distribution of measurements in normal and abnormal animals. For statistical comparison of measurements between the groups, a one-way analysis of variance (ANOVA) was used. The overall level of statistical significance was set to
Results
Animals
There were no significant differences in age or gender, but the normal animals (mean: 80.1 g; SD: 10.0) were significantly heavier (
Electroencephalography
Altogether 33 EEGs were evaluated. From the original 37 animals, three (2 from the Berne group and 1 from the control group) had to be excluded because of too many movement artefacts in their EEG leads. Another control animal was excluded, because the anaesthetic protocol could not be maintained due to technical problems. Because of minor movement artefacts, one lead had to be eliminated in six animals and between one and three epochs had to be shifted within the last minute of the EEG in seven other animals. We compared amplitude and frequency between the group of animals with clean EEGs to those missing one lead (6 animals) and those with epoch shifts (7 animals) using one-way ANOVA, but no significant differences were encountered. Thus, all the 33 EEGs were pooled together according to the neuropathological findings.
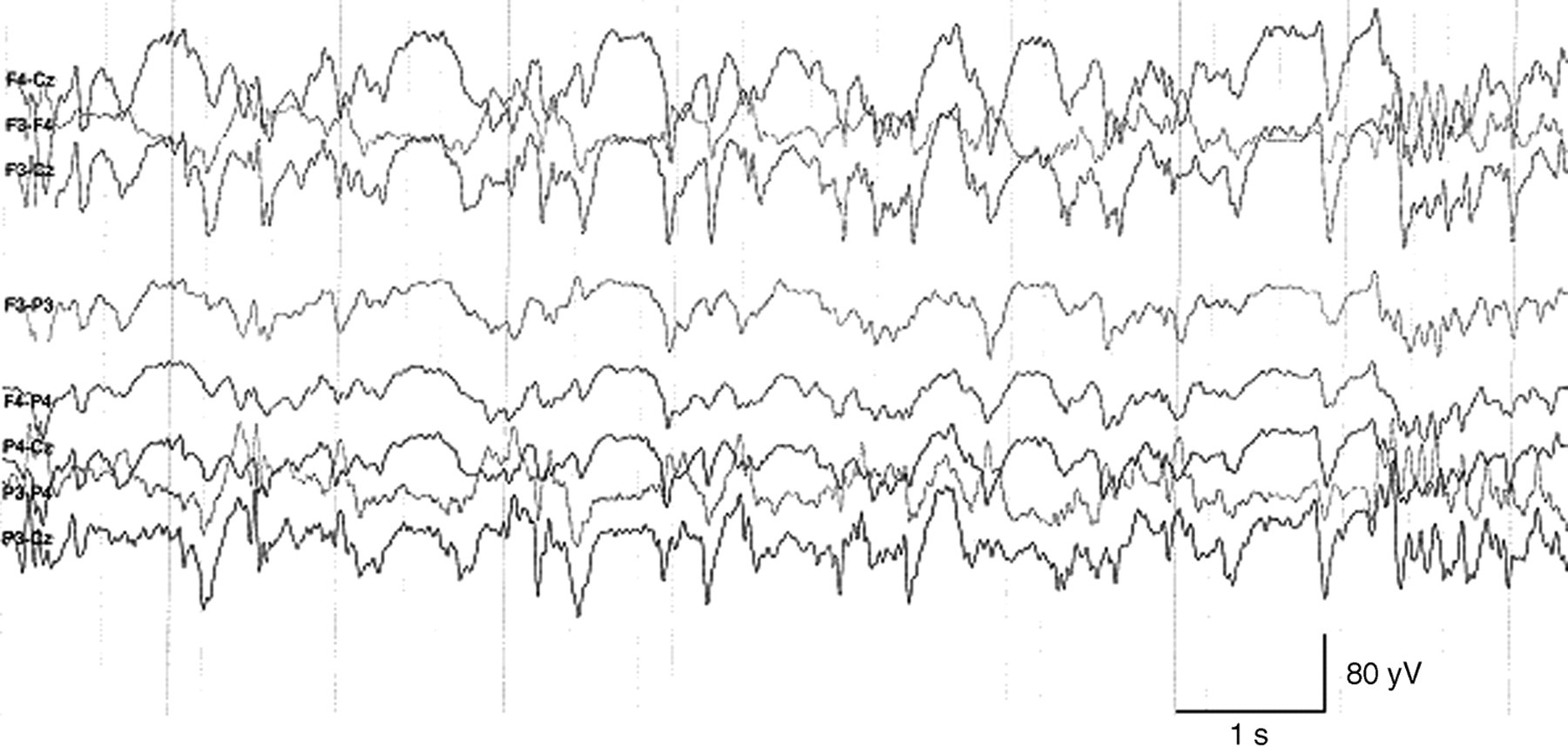
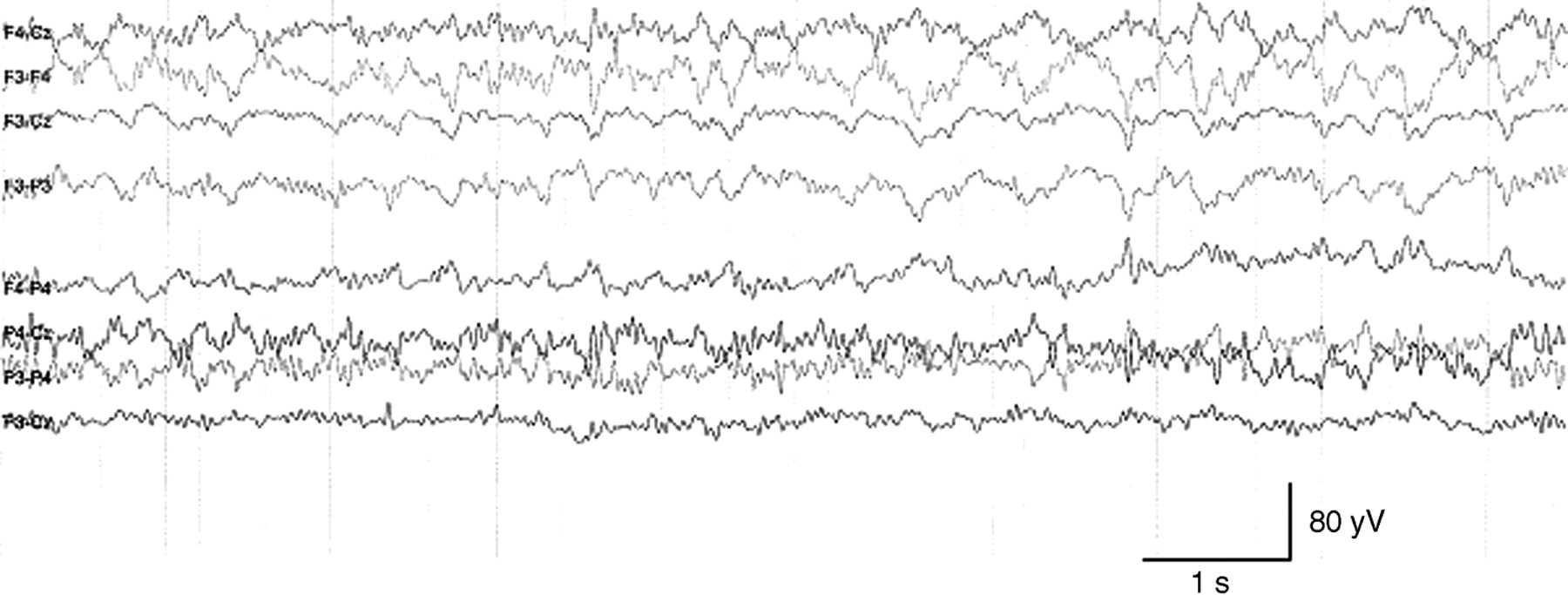
When we analysed our EEG measurements, we found a HVSA in most of the animals from the hydrocephalic group. These findings differed significantly from the results of the normal group (Table 1). The 23 hydrocephalic animals showed a high amplitude (mean: 60.64 µV; SD: 15.27), low frequency (4.07 Hz; 0.52) main activity with a low amplitude (9.94 µV; 2.52), high frequency (14.66 Hz; 1.13) activity superimposed onto it (Figure 2). Only five (21.7%) affected hamsters had an amplitude of <50 µV, with two (8.7%) <40 µV. All the 23 affected animals had a frequency of <5 Hz. The normal animals showed a low amplitude (30.78 µV; 6.16), high frequency (6.49 Hz; 0.58) activity with a low amplitude (7.54 µV; 1.91), high frequency (11.95 Hz; 0.96) activity superimposed onto it (Figure 3). Only one normal animal (4.4%) had an amplitude of over 40 µV and none showed a frequency of <5 Hz. Amplitude (
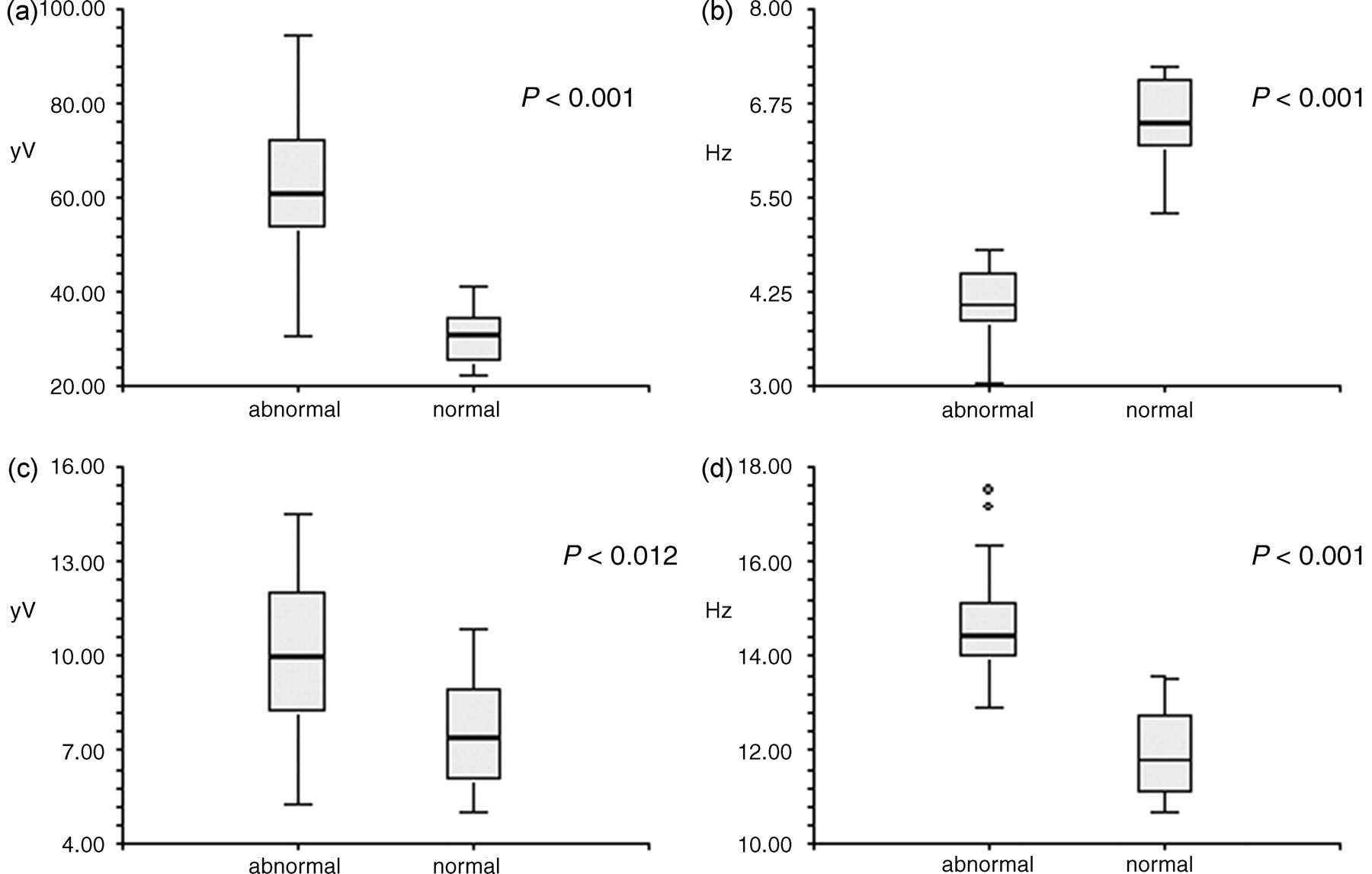
Data are means ± SD
Pathology
The pathology showed that all 23 hamsters from the Berne group had a macroscopic apparent hydrocephalus and that all 10 from the control group had normal ventricular dimensions. The affected animals showed an enlargement of the lateral ventricles, which except for one animal was bilaterally symmetric (22/23; 95.7%). Most animals had a severe dilation (15/23; 65.2%), while six animals only showed a mild bilateral enlargement (26%), one with a mild unilateral (4.4%) and one with a minimal bilateral dilation (4.4%). We compared the EEGs of these different groups but found no significant differences (
Discussion
In the present experimental study, EEG recordings were performed on 25 golden hamsters that were suspected to be hydrocephalic, as well as on 12 control animals. Isoflurane was used for anaesthesia, and after the EEG the hamsters were euthanized and neuropathology was performed on their brains. The objective was to investigate the usefulness of EEG in the screening for congenital hydrocephalus in golden hamsters.
In hydrocephalic dogs, a number of studies have stated comparable findings, including a HVSA with a fast activity superimposed onto it (deLahunta & Cummings 1965, Prynn & Redding 1968, Klemm & Hall 1971). This resembles the results of our study. Our hydrocephalic hamsters mostly had an amplitude of over 50 µV (18/23; 78.3%) and always an activity of <5 Hz. The normal animals almost always had an amplitude of <40 µV (22/23; 95.7%) and always an activity of >5 Hz. Discrepancies in our study were the five hydrocephalic animals that showed a low voltage amplitude (<50 µV). One of the quoted studies included two hydrocephalic dogs that did not show the typical HVSA, but a low-voltage fast wave activity. In both cases, the hydrocephalus was complicated by a coexisting encephalopathy: one a subcortical tumour and the other an otitis media/interna (Klemm & Hall 1971). Unlike in the mentioned study, the neuropathological examination gave no indication of a concomitant encephalopathy in our animals. Furthermore, our five hamsters only showed the low-voltage amplitude and not the fast wave activity the dogs expressed, as their frequency did not differ significantly from the other hydrocephalic hamsters. As these findings cannot be assigned to another disease process, it must be surmised that they may represent the normal variance in the EEG of the hydrocephalic golden hamster. However, the HVSA is not pathognomonic for hydrocephalic animals alone and can also be seen with other chronic encephalopathies that destroy cortical parenchyma (Klemm & Hall 1971, Harrington et al. 1996, Jaggy 2005). In our study, pathology did not reveal any evidence of another concurring encephalopathy. It also seems less likely for such young animals to be suffering from an encephalopathy other than an anomaly. Therefore, an amplitude of over 50 µV in combination with an activity of <5 Hz seems to be a good evidence of hydrocephalus in young golden hamsters.
Pathology rendered similar results as the original study on this milder form of hereditary hydrocephalus (Edwards et al. 2006). None of our hydrocephalic brains showed signs of over-distension, absence or obstruction of the third or fourth ventricle or the mesencephalic aqueduct. Histologically, we saw slight periventricular oedema, flattening of the ependyma and sporadic narrowing of the mesencephalic aqueduct. This stands in contrast to the findings in hamsters affected by the phenotypically identifiable form of hereditary hydrocephalus. These show an almost complete destruction of the septal area as well as extensive damage to nerve cell bodies and fibres. Furthermore, a marked distension of the third ventricle is seen, together with signs of compression and distortion of the fourth ventricle (Yoon & Slaney 1972). The lack of clinical symptoms in our hydrocephalic animals probably reflects the more subtle nature of histological changes.
Another difference between the hydrocephalic and the normal group was their weight. The normal animals were distinctly heavier than their hydrocephalic counterparts. Hamsters that are affected by the phenotypically identifiable form of hereditary hydrocephalus, are usually smaller and weigh less (Yoon & Slaney 1972). The hamsters with the milder form of hereditary hydrocephalus have not yet been compared against normal animals, but are supposedly asymptomatic (Edwards et al. 2006). As the husbandry in our study differed slightly between the hydrocephalic and the control groups (Gattermann et al. 2002), it remains unclear whether the hydrocephalus had any consequence on the weight gain of the animals.
There have been numerous studies in mice and rats that showed how early husbandry can affect neurodevelopment. Impoverished living environments lead to impaired brain development (Balcombe 2006). We can exclude the possibility that this factor may have played a role in our study, as both the affected as well as the control groups were reared under comparable living conditions, with a high environmental complexity.
Several diagnostic techniques may be employed in the diagnosis of hydrocephalus. Survey radiographs of the skull may suggest the presence of hydrocephalus through characteristic radiographic findings (Few 1966), but usually cannot give a definitive diagnosis, especially if the hydrocephalus develops after the skull is formed (Harrington et al. 1996). Ultrasonography is another routine procedure. Its main drawback is that it necessitates a persistent bregmatic fontanelle (Rivers & Walter 1992). Pathology showed that this was not the case in our population (Edwards et al. 2006). The methods that are mainly used today are the computed tomography (CT) and the magnetic resonance imaging (MRI). Both offer immediate diagnoses and are minimally invasive. MRI is the method of choice, because of its excellent soft tissue contrast. There have already been two papers published on the use of MRI in hydrocephalic hamsters (Narayana et al. 1987, Wahnschaffe et al. 1990) and both offered very good results. The main drawback for both MRI and CT are the costs and their poor availability in laboratory animal research. In addition, their examination time exceeds all other diagnostics and both require general anaesthesia under injectable agents, as an isoflurane chamber cannot be used inside the MRI or CT. EEG is a comparably cheap diagnostic procedure, is minimally invasive and relatively easy and quickly performed. It does not give as good a diagnosis as the MRI or CT, but as the aim is to exclude potentially affected animals from the breeding programmes, it seems to be a legitimate option.
Isoflurane anaesthesia is known to affect the EEG. With increasing concentrations of the inhalant, the EEG may become isoelectric, indicating electrical silence of the brain or exhibit burst suppression. The latter refers to isoelectric periods interrupted by brief intervals of high amplitude activity (March & Muir 2005). These changes were not observed in our study during the standardized measurements in the last minute of recording. A possible explanation could be that at this point, the plane of anaesthesia was not deep enough to produce these changes. At moderate degrees of sedation, isoflurane has been reported to produce a continuous high-voltage low-frequency activity (March & Muir 2005). We therefore cannot exclude the possibility that the isoflurane may have altered our EEG measurements. But as the protocol was identical in both groups and followed meticulously, it is unlikely that the isoflurane had any influence on the outcome of our study.
From the initial 37 hamsters, four had to be excluded from the study, one because of technical problems and the other three because of excessive movement artefacts that made it impossible to obtain six epochs from the last minute of the recording. There were also minor movement artefacts in the recordings of the other hamsters. Yet, these did not impair the results. A possible explanation for the relatively high number of artefacts might be that smaller animals generate more movement artefacts during anaesthesia than larger ones. Despite these artefacts most of the EEGs could be evaluated.
In conclusion, the EEG may be a very good diagnostic aid in the screening of larger hamster populations for hydrocephalic animals. An amplitude of >50 µV in combination with a frequency of <5 Hz will give a good indication that the animal is hydrocephalic. There might still be a chance that the animals suffer from an encephalopathy of an unknown aetiology, but as the aim is to exclude potentially affected animals from the breeding programmes, the EEG may still be a valuable tool. A goal for future research could be to reduce the number of artefacts in the EEG leads.
Footnotes
Acknowledgement
This research was supported by the Frauchiger Foundation.
