Abstract
The tammar wallaby joey is born extremely immature and most of its neurological development occurs in the maternal pouch. It is not known at what in-pouch age functions such as conscious sensory perception commence. We determined the electroencephalographic (EEG) responses to noxious stimulation in lightly anaesthetized tammar wallaby joeys. Baseline median (F50) and spectral edge (F95) frequencies, total power (Ptot) and frequency spectra between 1 and 30 Hz of the EEG power spectrum were determined. Joeys aged less than 127 days showed little or no EEG activity. Prolonged periods of spontaneous EEG activity were present by 142 days. This activity increased, as did the power in all frequencies, while the duration of any intervening isoelectric periods decreased with increasing in-pouch age. EEG responses to a noxious stimulus (toe clamping) changed with increasing in-pouch age as there was no response from joeys aged 94–127 days (no EEG), a minimal decrease in the F50 in those aged between 142 and 181 days (P = 0.052) and a greater decrease in the F50 in those aged between 187 and 261 days (P < 0.001). The pattern of these changes, which presumably reflects anatomical and functional maturation of the cerebral cortex, is similar to, but develops more slowly than, that reported in the rat. The opening of the eyes and development of the pelage are discussed as markers of when brain development may be sufficient for joeys to consciously perceive noxious sensations including pain.
The age at which newborn and young animals can experience good welfare or the opposite, suffering, depends on the acquisition of two key neurological characteristics. 1,2 Firstly, the animal must be sentient, i.e. its nervous system must be sufficiently developed to relay sensory inputs to the higher centres of the brain, including the cerebral cortex, for appropriate interpretation. 3,4 Secondly, the brain must be functionally capable of inducing consciousness, as this is a prerequisite for the animal to experience any sensation including pain. 5 There is evidence that the cerebral cortex is involved in the human experience of pain perception. 6–9 Moreover, changes in electroencephalographic (EEG) spectra in humans have been shown to reflect changes in cerebral activity associated with the cognitive perception of pain. 10,11 Hence, EEG responses to noxious stimulation may be useful in determining the time at which the neurological apparatus required for conscious perception of sensations, including pain, becomes functional.
The fact that the ontogenic stage and chronological age at which the key neurological features necessary to perceive pain have developed varies widely between species, 1,2,12 precludes any generalization regarding the welfare of newborn mammals. At one extreme the neonates of a number of species, which include cattle, deer, goats, sheep and horses, are neurologically mature at birth and exhibit the behavioural and physiological signs indicative of conscious responses to sensory inputs within minutes or hours of birth. 3,13,14 The young of other species, which include rats, mice, rabbits, cats and dogs are neurologically relatively immature, 12 and the onset of consciousness and the ability to perceive sensations is apparently delayed for several days. In rat pups, for example, postnatal development of the behavioural repertoire, the EEG and the EEG responses to noxious stimuli, together suggest that pups are not likely to perceive pain during the first week after birth, and that the ability to perceive pain develops gradually between postnatal ages 12–14 days and 21–22 days. 15
The young of some species, including marsupials, are neurologically exceptionally immature at birth. 16 The tammar wallaby joey (Macropus eugenii) is born after a 28-day pregnancy with a cerebral cortex comprising only two layers of cells and resembling that of a 40-day human or 26-day sheep embryo. 17 After entering the pouch the joey remains permanently attached to a teat for about 100 days, and intermittently thereafter, leaving the pouch permanently after approximately 250 days. 16 It is evident that most neurological development occurs postnatally in macropods but it is not known precisely when neurological development is sufficient to permit conscious sensory perception.
A greater understanding of the ontogeny and chronology of pain perception in this group of animals would be useful for minimizing suffering in joeys during pest control programmes 18–20 and would also be relevant to their humane use in developmental research. For instance, the unique developmental pattern of the marsupial nervous system is of great interest in its own right 16,21–29 and provides models for aspects of neurological development in eutherian mammals. 16,30 In addition, including joeys in comparative neurophysiological investigations, such as that currently being undertaken in our laboratory, 1 allows responses of neurologically exceptionally immature young (the wallaby joeys reported on here) to be compared with those of young that are neurologically moderately immature (e.g. rat pups 15 ) and mature (e.g. lambs 31 ) at birth.
Thus, the purpose of the current work was to assess when cerebral cortical processing of noxious sensory input may begin in the tammar wallaby joey using an approach, previously used in rat pups, 15 and lambs, 31 which evaluates EEG responses to potentially painful stimulation (in this case toe clamping) in anaesthetized animals. This minimal anaesthesia model is based on observations that EEG responses to noxious stimulation in conscious and anaesthetized animals are similar to those of conscious humans exposed to painful stimuli. 11,32,33 The model has the advantage of being humane as it avoids any conscious perception of pain, and it also minimizes EEG artefacts by markedly reducing spontaneous muscular activity. Although the minimal anaesthesia model does not demonstrate that animals are capable of conscious perception, it does allow inferences to be drawn regarding whether or not the cerebral cortex is capable of responding to systemic stimulation at the different ages investigated. Placing these observations in the context of general neurological development, as well as development of EEG patterns and related behaviour, has allowed broader inferences to be drawn about the likely timing of the onset of conscious perception. 15
Accordingly, the ontogeny of EEG activity and EEG responses to noxious stimulation in halothane anaesthetized wallaby joeys aged between 94 and 261 days were investigated and the results are reported and interpreted here. The Massey University Animal Ethics Committee approved this study (Protocols 04/94 and 06/11) which was part of a wider programme that also included observations on musculoskeletal and gut development in wallaby joeys. Thus, once the above EEG observations had been made, the joeys were euthanized, dissected, measured and selected tissues collected. The data generated were pooled with that from other joeys, and were then analysed, interpreted and published elsewhere. 34–36
Materials and methods
Animals
Animals were obtained from a colony of feral South Western Mainland Tammar Wallabies (Macropus eugenii eugenii) maintained at Massey University, New Zealand. They were kept in a controlled environment (12:12 h light/dark cycle, room temperature ranged on average between 15°C in winter and 23°C in summer), fed a pelleted diet supplemented with Lucerne hay and fresh fruit and given access to water ad libitum. Females were allowed to mate year round to provide a continuing supply of in-pouch joeys. Overall, 20 joeys of varying age and gender were used in the present study.
The in-pouch ages of wallaby joeys were inferred from the relationship between head length (mm) and age (days) 37 and subsequently checked against unpublished data obtained from the Massey University tammar wallaby colony relating weight to in-pouch age. The joeys were assigned to three groups on a basis of postnatal age: those aged between 94 and 127 days (n = 6); those between 142 and 181 days (n = 5); and those between 187 and 261 days (n = 9). A cut-off point of 127 days was used as joeys younger than this principally had isoelectric EEGs. The cut-off point of 185 days was used as a series of major nutritional and physiological changes occur at this age including the achievement of homeothermic control, elaboration of locomotive behaviour and changes in nutrient intake which are mainly due to changes in composition of maternal milk. 16,38,39
Experimental procedure
All experiments were conducted during daylight between 09:00 and 16:00 h. Joeys were anaesthetized in a 25 L chamber with 4% halothane (Merial, Parramatta, Australia) in oxygen delivered at 5 L/min. They were intubated with the aid of a guidewire once loss of the righting reflex and absence of general body movements were achieved. Anaesthesia was maintained with halothane in oxygen delivered via a calibrated vaporizer with intermittent positive pressure ventilation (Small Animal Ventilator, Vetronics, West Lafayette, IN, USA). Endtidal halothane and carbon dioxide partial pressures were measured using an anaesthetic agent monitor (Hewlett Packard M1025B, Hewlett Packard, Hamburg, Germany). Endtidal halothane was titrated to 1.0% by vaporizer adjustment. Endtidal carbon dioxide was titrated to maintain normocapnoea by adjustment of the ventilator. This represented a very light plane of anaesthesia equivalent to those used in other studies. 15,31 Heat loss was minimized by a heated pad and a small sheep wool blanket placed over the joeys.
Stainless steel needle electrodes (Medelec, Oxford Instruments Medical Systems, Surrey, UK) were placed transdermally. A three-electrode montage was used with the non-inverting electrode placed in the midline over the frontal sinus, the inverting electrode over the left mastoid process and the ground electrode caudal to the occipital process. The animals were allowed to stabilize for 5 min following electrode placement, prior to recording a 5 min baseline EEG. The fourth phalange of the right hind leg, subsequently referred to as the toe, was then stimulated mechanically between its base and midpoint by a single application of a haemostat (overall length, 18 cm; jaw dimensions: 6.5 cm long, 3–5 mm wide) closed to the first ratchet and held for 30 s, as validated in previous studies in the rat. 15,40 The force delivered was not measured but was obviously sufficient to represent a supramaximal stimulus because the haemostat caused significant tissue damage, which was indicated in each case upon release by a clearly visible imprint of the jaws. This stimulus was obviously sufficient to cause significant pain if applied to a human finger. EEG recordings were taken during and for 5 min following the application of this stimulus. At the end of the study and before recovery from anaesthesia, the joeys were euthanized by intracardiac or intraperitoneal overdose of pentobarbitone sodium given to effect (National Veterinary Supplies, Auckland, New Zealand).
EEG recordings
The EEG was recorded continuously using suitably isolated biological amplifiers (World Precision Instruments Inc, Saratosa, FL, USA) set to a gain of 1000 and a pass band of 0.1–500 Hz. The data were digitized using an analogue to digital recording system (Powerlab™, AD Instruments Ltd, Bella Vista, Australia) at a sampling rate of 1 kHz. Analysis of EEG data was conducted offline following completion of each experiment.
EEG analyses
Isoelectric periods were common in the EEG traces of joeys of the two younger age groups, i.e. joeys younger than 185 days (e.g. Figure 1). An isoelectric EEG was classified as a stable trace consisting of background noise with an amplitude of less than one-eighth of normal EEGs, 41 i.e. those recorded from joeys older than 200 days. The percentage of time occupied by isoelectric periods of 300 ms or longer during the first 3 min of the baseline period and the first 3 min immediately after toe clamping, together with the number and mean duration of isoelectric periods, were determined.
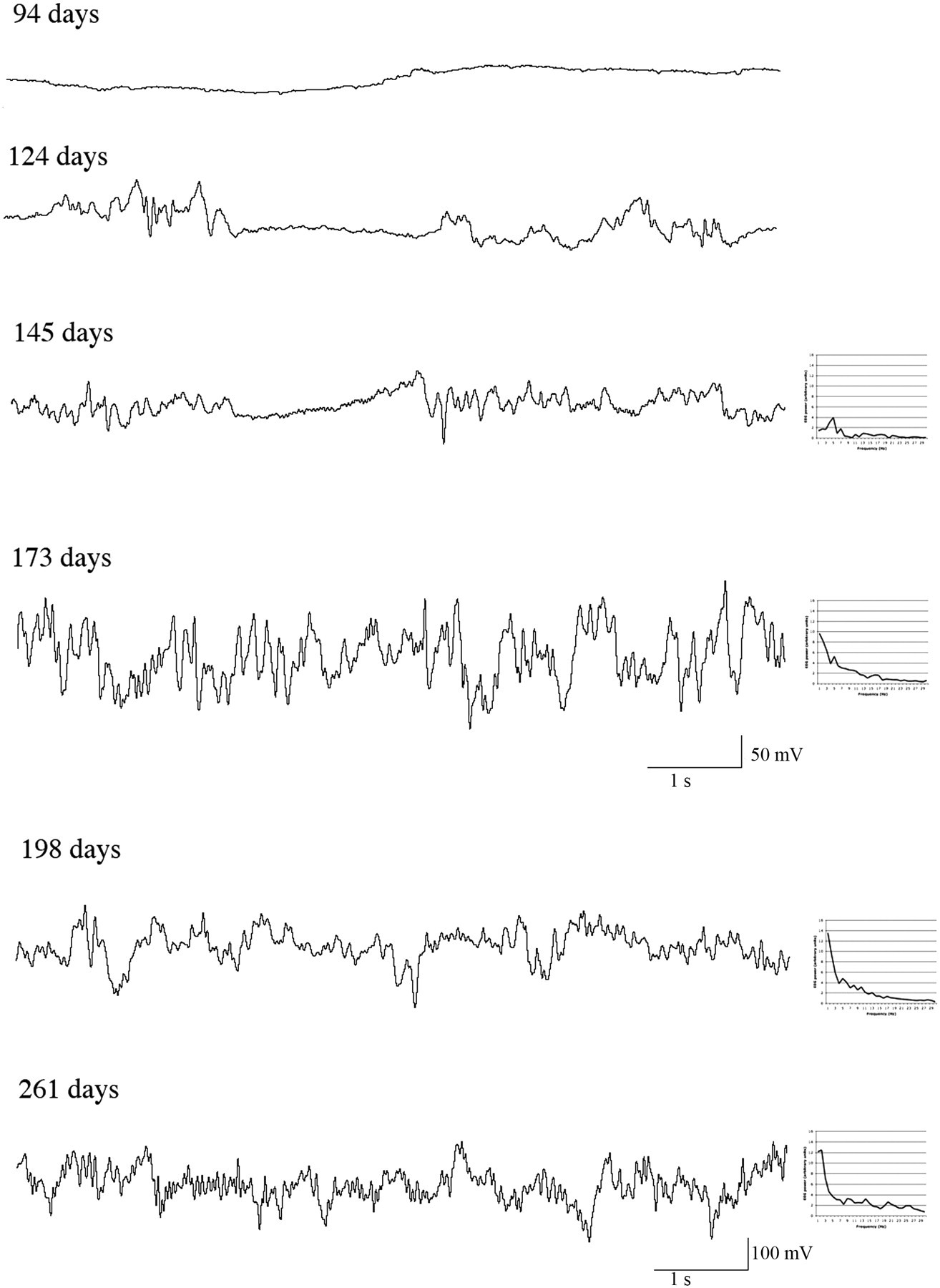
Electroencephalographic (EEG) traces of joeys at different in-pouch ages, showing an isoelectric EEG at 94 days and isoelectric epochs for joeys at 124 and 145 days of in-pouch age. Note the different scale for the two oldest joeys – the first scale shown relates to the four traces above it and the second to the two traces above it. Power spectra drawn to the same scale are illustrated for the traces from joeys aged 145–261 days
Continuous epochs of EEGs, each 20 s in duration, were obtained from the beginning of baseline, during clamping and immediately after clamping and each was subjected to spectral analysis. In five joeys, isoelectric periods occurred too frequently to allow this approach. In these cases, five continuous epochs of non-isoelectric EEGs longer than 3 s duration were selected at equivalent recording periods as those described above, and subjected to fast Fourier transform (FFT) analysis.
Median frequency (F50), 95% spectral edge frequency (F95) and total power (Ptot) were calculated from frequency spectra (1–30 Hz) using a customized FFT program (Spectral Analyser, CB Johnson, Massey University, New Zealand, 2006) based on a commercial FFT algorithm. 42 The F50 is the frequency below which 50% of the total power of the EEG is located, the F95 is the frequency below which 95% of the total power of the EEG is located and Ptot reflects the overall area under the power spectrum curve. 43,44 Quantitative changes in these EEG variables have been previously shown to be associated with noxious sensory input in mammals. 31–33,43,44
Statistical analyses
Statistical analyses were undertaken using SPSS 11.0 for MAC OSX (SPSS Inc, Chicago, IL, USA) and Prism 5 for MAC OSX (GraphPad Software, Inc, San Diego, CA, USA). Data are presented as mean ± standard error of the mean (SEM). Differences were considered significant at P < 0.05. Data were tested for normality using the Shapiro-Wilks statistic (if n < 50) or the Kolmogorov-Smirnoff statistic (if n > 50) and normal probability plots. Isoelectricity and spectral data were not normally distributed and non-parametric tests were employed for statistical evaluation.
Age differences in the percentage of time occupied by isoelectric periods were assessed by the Kruskal-Wallis test with Dunn's post hoc test. Taking all joeys into consideration, the Wilcoxon test for two related samples was used to determine whether clamping had an effect on the mean duration or number of isoelectric periods present in the EEG traces. Age differences in baseline spectral parameters were assessed using the Mann-Whitney test for two unrelated samples. The effect of clamping on F50, F95 and Ptot was assessed using the non-parametric Friedman test with Dunn's post hoc test.
The means of the log-transformed frequency spectra (1–30 Hz) from all groups before, during and after clamping were graphed and evaluated. Variation in frequency spectra with age and clamping were further explored using multivariate methods (principal component [PC] analysis) to describe the orthogonal axes of variation of log-transformed data (1–30 Hz). The PC loadings were subsequently evaluated and the PC scores were plotted against each other in reduced dimensions to explore variation with treatment and age group.
Results
Isoelectric periods
The proportion of time occupied by isoelectric EEG periods (%), as well as the number of isoelectric periods and their average durations during the 3 min periods before and after clamping, are presented in Table 1. Overall, the incidence of isoelectric EEG epochs decreased with age (χ2 statistic = 16.319, df 2, 18, P < 0.001). Clamping had no discernable effect either on the duration or on the number of isoelectric periods (duration, 3950 ± 2011 ms versus 3087 ± 1762 ms; df 1, 19, Z = −1.070, P = 0.285; number, 9.5 ± 3.25 versus 10.6 ± 3.29; df 1, 19, Z = −1.265, P = 0.206).
Percentage of time occupied by isoelectric electroencephalographic (EEG) periods (%), their average duration (ms) and the number of them present (n) during 3 min of EEG recordings during the baseline and post-clamping phases
Spectral analysis in response to clamping
The absence of sufficiently long epochs of EEG activity in joeys aged 94 to 127 days precluded spectral analysis. Thus the analysis was restricted to EEG epochs of joeys aged between 142 and 181 days and between 187 and 261 days.
Median frequency (F50), spectral edge frequency (F95) and total power (Ptot)
Statistical results and means ± SEM values are presented in Table 2. There were significant overall changes in F50 in response to clamping in the 187–261-day-old joeys (P < 0.001). Changes in F50 and Ptot in 142–181-day-old joeys in response to clamping approached significance (F50, P = 0.052; Ptot, P = 0.085). The post hoc Dunn's test indicated that the significant changes in F50 in joeys aged between 187 and 261 days resulted from an immediate and significant decrease in F50 on clamping (before versus during clamping) and a subsequent and significant return (increase) to preclamping values once the clamp had been removed. While in the younger joeys there were no significant overall changes in F50 in response to clamping, there was an immediate significant decrease in F50 upon clamping (before versus during clamping) while Ptot tended to increase in immediate response to clamping (Table 2).
Results of the non-parametric Friedman test, including P values, χ2 statistic, degrees of freedom (df), means and standard error of the mean (SEM) for electroencephalographic parameters from anaesthetized joeys comparing data obtained before, during and after clamping
Means denoted with the same letters indicate differences within an age group with significant levels as follows: a P < 0.001; b P = 0.001–0.01 and c P = 0.01–0.05
F50 = median frequency; F95 = spectral edge frequency; Ptot = total power; χ2 = Friedman statistic
Overall, baseline (preclamping) values of F50 did not change significantly with age (P = 0.207; df 1, 279; Z = −1.262), although they tended to be higher in the older joeys (5.60 ± 0.14 Hz, 142–181 days versus 5.96 ± 0.17 Hz, 187–261 days). In contrast, baseline values of both F95 and Ptot increased significantly with age (F95, 23.52 ± 0.21 versus 24.18 ± 0.13 Hz, df 1, 279, Z = −2.455, P = 0.014; Ptot, 47.4 ± 2.4 versus 89.4 ± 1.8, df 1, 279, Z = −10.780, P ≤ 0.001).
Graphic comparisons
The pattern of variation of the mean of log-transformed power with frequency differed between the two age groups prior to clamping, on clamping and after removal of the clamp (Figure 2). The significance of these differences was further explored by multivariate (PC) analysis. The analysis identified three orthogonal PCs with eigenvalues >1 that together contributed 52.7% of the overall variation in the data. The component loadings in PC1 were all positive and high (>0.5) (Table 3), indicating that this axis represented variation in overall power. The loadings of frequencies varied in accordance with the form of the spectral density curves (Figure 2). Power spectra for some individual joeys are illustrated beside their traces in Figure 1. Their numerical relationship thus reflected to some extent F50 and F95 values. It was possible to distinguish age group on the basis of PC1 scores (Figure 3), younger joeys having lower scores than older joeys, i.e. less overall EEG power.
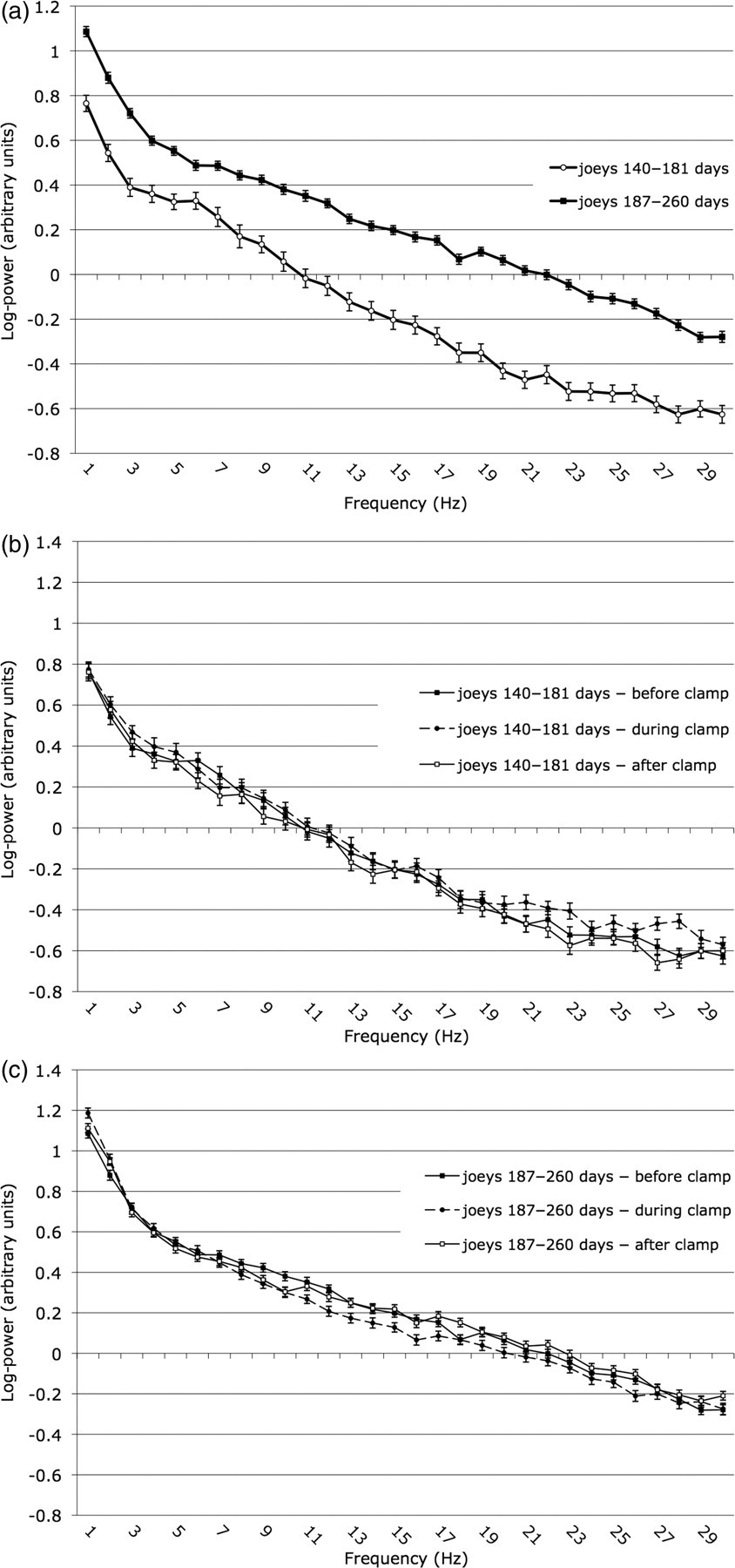
Mean power (log) (±SEM) in frequencies of the electroencephalographic spectrum during baseline observations for joeys aged 142–181 days and 187–261 days (a) and before, during and after clamping for joeys aged 142–181 days (b) and 187–261 days (c)
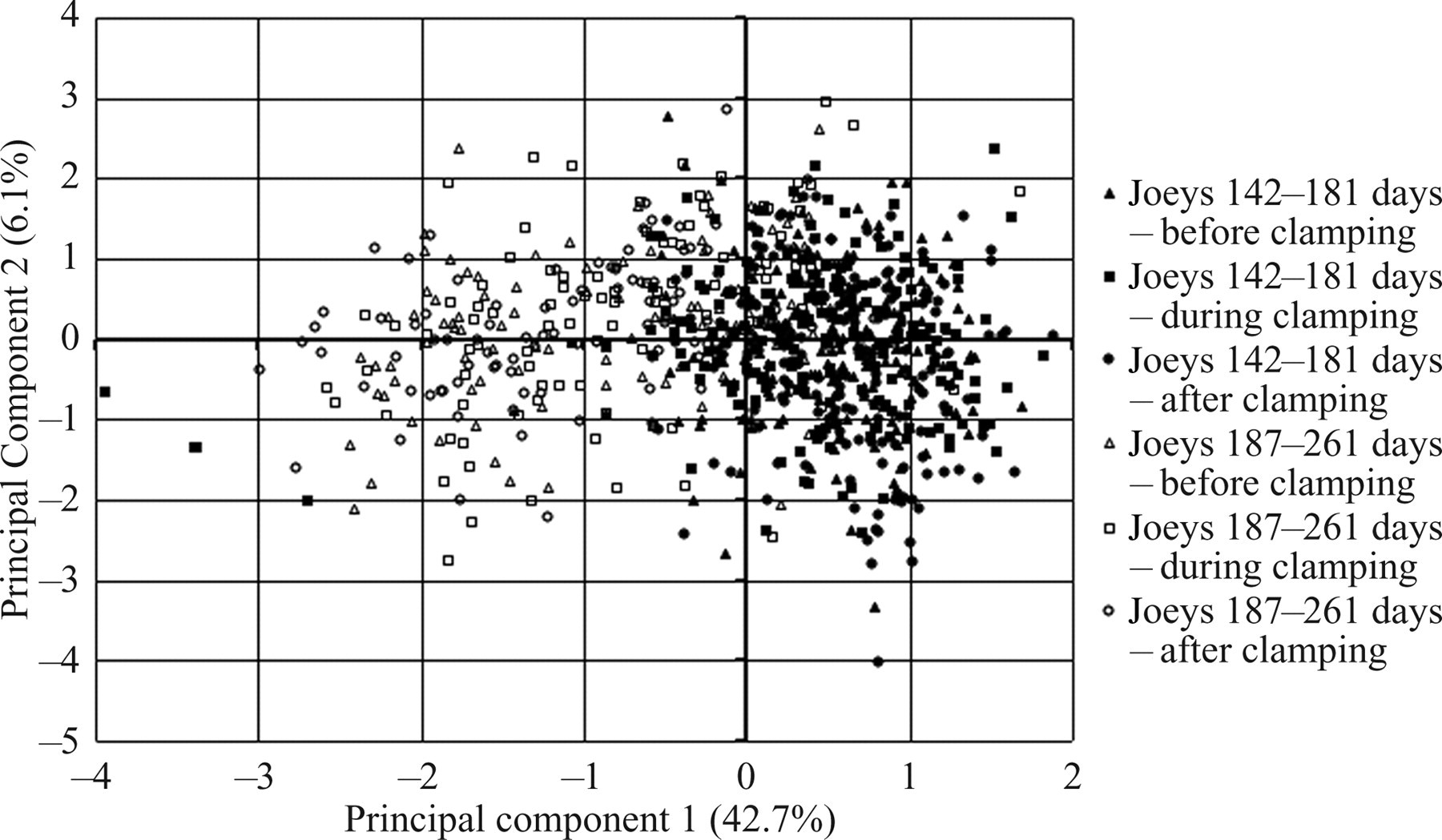
Factor scores plotted against the first (PC1) and second (PC2) principal component axes showing separation according to age (142–181 days and 187–261 days) and treatment (i.e. before, during and after clamping)
Eigenvalues and component scores of all frequencies for principal components (PCs) 1–3 calculated by PC analysis
The pattern of component loadings for PC2 contrasted the magnitudes of the frequency components of 1 to 12 Hz (positive) with those of 16 to 30 Hz (negative) (Table 3). The pattern of component loadings for PC3 contrasted the magnitudes of the frequency components of 1 to 5, 24 and 28 to 30 Hz (positive) with those of 7 to 16 Hz (negative) (Table 3). Hence these two axes each represented contrasts in the power of particular frequencies within the power spectrum (Figure 2). It was not possible to distinguish either by treatment or by treatment with age on the basis of variation in PC2 or PC3 scores. Variations in the relative proportions of the frequency components therefore did not occur ontogenically and reflected individual variation. Thus, although the frequency distribution in the power spectrum of an individual varied, presumably due to differences in the activity of the various cerebral components during the sampling period, it did not vary with nociceptive stimulation.
Discussion
This is the first report of the developmental changes in the EEG of an in-pouch macropod. The developmental changes in the EEG of the tammar wallaby joey are broadly similar to those reported to date in placental mammals and birds 4,12 as well as in one marsupial species, the Virginia opossum (Didelphis virginiana). 45 All these species exhibit isoelectric EEG traces in the early stages of brain development, subsequently exhibit isolated short epochs of activity, and go on to develop continuous mixed sleep-like patterns, classic alternating rapid-eye-movement (REM) and non-REM patterns, and finally repetitive sleep–wake cycles. In anaesthetized tammar joeys, the initial isoelectric phase lasted until about 114 days, becoming continuous after about 173 days (Table 1). These changes were accompanied by a progressive increase in EEG power over all frequencies (Figure 1).
These general changes presumably reflect local increases in neuronal complexity due to maturation of the cerebral cortex as it increases in thickness from two cells to six multicellular layers, 17,46 and also to increases in the connectivity of the thalamus and cortex 22,27 and in sensory neuronal communication arising from differentiation of the somatosensory and visual cortices. 22,29,47–49 Hence the period of change in EEG from isoelectric to continuous discharge (Table 1) occurs synchronously with the establishment of the neural foundations for the operation of sensory modalities. Behavioural evidence for the appearance of consciousness, including the age when the joey first looks out of the pouch, 16 is consistent with this timing.
It is noteworthy that the EEG was isoelectric at the time when electrical potentials have been reported to be evokable in the primary somatosensory cortex by peripheral stimulation (about 72 to 85 days of in-pouch age) 29,47 and at the time that auditory responses can be evoked in the brainstem by bone conduction (85 to 94 days). 50 Hence some early elements of the neural circuitry associated with sensation appear to be established before the onset of spontaneous electrical activity in the cortex. This accords with other circumstances where evoked potentials can be recorded from cerebral cortices in the absence of spontaneous EEG activity. 51
While the sequence of EEG development is similar to that of placental mammals, the timing differs significantly in the marsupial. Hence the disappearance of the isoelectric phase occurs more promptly after birth in anesthetized rat pups in which prolonged periods of activity develop by 21–22 days. 15 The much slower rate of tammar wallaby EEG development continues to be evident if we calculate the time points of this sequence in terms of days from conception given that intrauterine gestation of the rat lasts about 21 days while that of the tammar wallaby lasts about 28 days. This slower rate reflects the relative delay in marsupial brain ontogeny 52 and accords with suggested constraints inherent in sustaining joey cerebral development via lactation. 53
The limited extent of brain development exhibited by tammar wallaby joeys aged under 100 days, as indicated by the isoelectric EEG (Table 1, Figure 1), is reflected in their limited interaction with their environment within the mother's pouch because the joey remains permanently attached to the teat during this period. 16 More complex behavioural interactions become increasingly evident between 120 and 140 days of age. During this period the number and duration of isoelectric EEG periods diminishes (Table 1), the external ear canals become patent (125 to 130 days) and the eyes open (around 140 days), 50 and at about 180 days, the joey first looks out of the pouch. 16 It is noteworthy that the period of transition to more sustained EEG activity and behaviour also coincides with growth of the pelage (around 140 days), 16 and the development of thermostasis (between about 140 and 149 days). 54 As reported in possums, 55 the onset of pelage development and eye opening coincide in wallaby joeys. 16,50 In the present study, the onset of sustained EEG activity followed pelage development and eye opening (Table 1, Figure 1). One joey exhibited low voltage activity for about 45% of the time at 124 days of age. No power spectrum was derivable from this trace as the periods of EEG activity were too short to allow this. Such EEG activity as seen in this joey is not likely to be able to support conscious awareness in unanaesthetized animals. 56
The continuing increase in power over all EEG frequencies after 180 to 190 days (Table 1, Figure 3), the stage when the joey first looks out of the pouch and then begins to temporarily exit from it, 16 indicates that full brain development has not yet been attained so that the joey is likely to be less adept at responding to physical challenge (e.g. predation) during the period soon after its emergence from the pouch. Hence laboratory or field studies aimed at characterizing or utilizing the behavioural competence of joeys need to take into account the joey's age in relation to the timing of its permanent emergence from the pouch.
The development of complex behaviours appears to be correlated with general increases in EEG activity that are reflected in significant effects on the PC scores on axis 1 (Figure 3), but not on local variations in the power spectrum as reflected in the PC2 and PC3 scores (Table 3). It is noteworthy that similar axes accounting for local variations in the power spectrum have been obtained from EEGs in developing rats, but scores on these axes differed with postnatal age, i.e. between 12–14 and 21–22 days. 15
Joeys aged 187–261 days exhibited a biphasic response to the noxious stimulus, i.e. a decline in F50 (P = 0.001–0.01) during stimulus application and a recovery to pre-existing levels after its removal (P = 0.01–0.05) (Table 2). The absence of any coincident change in the PC2 axis scores associated with clamping at this age indicates that no specific local activation occurred in immediate response to nociceptor input. Hence neither the application nor the removal of a noxious stimulus induced local changes in frequency distribution of the EEG. It is noteworthy that this contrasts with findings in rat pups in which both a general decrease in F50 and F95 and a change in the local frequency distributions occurred after application of a noxious stimulus, these effects being more pronounced in 21–22-day than 12–14-day-old pups. 15
In summary, these general and/or local changes in the EEGs of joeys and rat pups indicate that noxious sensory input elicits responses within the cerebral cortex at these developmental stages. However, the EEG responses to noxious stimulation in joeys were less pronounced than those previously observed in rat pups. 15 This is likely to reflect lower levels of neuronal communication as a consequence of the slower rate of brain development in joeys. For example, axon numbers in the anterior commissure of the tammar wallaby do not peak until around 150 days and only 4% are myelinated by 216 days compared with around 60% in the adult. 57 In contrast, myelination of axons of the corpus callosum in rat pups begins between 10 and 13 days after birth and achieves its maximum rate by about one month after birth. 58
All of the above conclusions are conditional on there being few confounding effects of halothane anaesthesia. We consider it unlikely that the use of halothane anaesthesia delayed the appearance of continuous EEG activity as this agent has not previously been reported to suppress burst activity at similar endtidal partial pressures to those used here. 59 Likewise, we consider it unlikely that other anaesthesia-related factors such as hypoxaemia, hypercapnia or altered thermal status would have affected cerebral functioning as anaesthetic practices that minimize such effects were used. Moreover, the development of severe hypoglycaemia was not likely as it would be expected to have induced seizure activity 60 rather than isoelectric epochs.
In conclusion, the results presented here do not indicate directly whether tammar wallaby joeys are able to experience pain. However, they do provide evidence of changes in cerebrocortical responsiveness to noxious stimulation and thereby allow inferences to be drawn regarding possible pain experience. Accordingly, they suggest that after an initial period lasting to about 127 days, during which EEG activity is largely absent and pain experience is doubtful, tammar wallaby joeys may develop the capacity to experience pain in response to noxious stimulation, and that this experience would change in intensity and nature as the joey's age further increases. The authors therefore suggest the following guidelines would be useful for avoiding pain and distress in laboratory or wild populations of macropods. Firstly, joeys without pelage and with permanently closed eyelids are likely to be incapable of consciously experiencing pain because of their neurological immaturity. Secondly, during the subsequent period of neurological transition that coincides with the development of the pelage and opening of the eyes, macropod joeys may be capable of experiencing pain. These guidelines are consistent with the marginal EEG response to noxious stimulation in tammar joeys aged 142–181 days. Thirdly, the greater cerebrocortical responses to noxious stimulation in older joeys (e.g. tammars age 187–261 days), coupled with their degree of neurological development and increased behavioural repertoire, together suggest that such tammar and other marsupial joeys are highly likely to be capable of conscious perception and pain experience. Accordingly, in line with the principle of refinement, we suggest a cautious approach of assuming that marsupial joeys with open eyes and with pelage evident be provided with adequate analgesia and/or anaesthesia to safeguard their welfare whenever potentially noxious procedures are to be undertaken.
Footnotes
Acknowledgements
We would like to thank Drs Paul Chambers and Joanna Murrell for their advice on anaesthesia and other matters, and Corrin Hulls and Sheryl Mitchinson for their practical assistance. Support for related projects from the Ministry of Agriculture and Forestry is gratefully acknowledged, as is the Doctoral Scholarship for TJD from the Agricultural and Marketing Research and Development Trust of New Zealand. This research was funded by the Geoffrey Gardiner Foundation Australia, the Palmerston North Medical Research Foundation New Zealand, and the Riddet Centre and the Animal Welfare Science and Bioethics Centre at Massey University.
