Abstract
Red cedar-derived bedding materials cause changes in cytochrome P450-dependent microsomal enzyme systems in laboratory animals. We examined the effect of essential oil of red cedar (EORC), as well as the effect of bedding from which it had been removed, on the hepatic expression cytochrome P450s in mice. EORC was obtained from liquid extracts of red cedar bedding by a soft-hydrothermal process and was administered orally to mice. Between days 1 and 2 after administration, hepatic P450s were significantly induced as follows: CYP3As, 7.1×; CYP1As, 1.6×; CYP2E1, 1.5×; CYP2Cs, 1.6×. A housing study of mice indicated that red cedar bedding increased the levels of these P450s in mouse liver, whereas mice housed in cedar bedding from which EORC had been removed (ST-cedar bedding) showed significantly lower levels of P450s, especially CYP3As, CYP1As and CYP2E1. Soft-hydrothermal processing partially removed many components of EORC. In particular, several volatile sesquiterpenes, naphthalene-derived aromatics and 4,4-dimethyl-13α-androst-5-ene were decreased in the ST-cedar bedding, suggesting that these may be responsible for P450 induction. This study demonstrated that the removal of these volatile compounds by soft-hydrothermal processing can decrease the hepatic P450-inducing effect of red cedar bedding.
Keywords
The Japanese cedar, Cryptomeria japonica D. Don, is a widely distributed conifer, known as ‘sugi’ in Japanese, which has been extensively planted since the 1960s. Even though the planting of Japanese cedar has played a very important role in maintaining the balance of ecological distribution and meeting the need for building materials for Japanese-style house, 1 recently cedar growth has become excessive and has caused serious environmental problems. Cedar pollen is now spread over most areas of Japan in early spring and more than 10% of Japanese suffer from ‘cedar pollen allergen’, and there is no effective medical treatment yet. 2–4 Therefore, new usages of cedar besides timber or fuel are desirable. Coincidently, the consumption of the bedding materials derived from wood is very high in Japan; according to the questionnaire data surveyed by Miyamoto in 2004, woody bedding comprised 80% of the total amount of bedding materials, and most was imported from abroad. 5 Although the main imported bedding material is spruce bedding due to its low content of volatile organic compounds, we propose that cedar also should be used as bedding for laboratory animals as a cost-effective use of local timber resources. Therefore, we considered whether these cedar resources can be converted into safe bedding for laboratory animals using a practical technique.
Hepatic microsomal enzymes are important in the metabolism of pharmacological agents. Thus, factors that affect the activities of hepatic microsomal enzymes can markedly influence experimental results by altering the metabolism of pharmacological agents.
Bedding materials or their extracts have been reported to cause various changes in the activities of hepatic microsomal enzymes in laboratory animals. 6–13 Many previous studies have shown that volatile organic chemicals in red cedar-derived bedding materials caused changes in P450-dependent microsomal enzyme systems in laboratory animals. 7,10 Mice kept on red cedar bedding exhibited higher activities of hexobarbital oxidase, ethyl morphine N-demethylase and aniline hydroxylase and had markedly decreased barbiturate-induced sleep times compared with mice kept on hardwood bedding. 6,14 Barbiturate sleep time is inversely proportional to hepatic microsomal enzyme activity, because the rate of barbiturate metabolism is controlled almost entirely by these enzymes. 6,15 Autoclaving and heat treatment had been examined to determine whether they would change barbiturate sleep times of mice housed on cedar bedding, but these processes made little difference. 9,10
The strong induction effects on the activities of hepatic microsomal enzymes were suggested to involve the volatile terpene hydrocarbons and aromatic compounds present in cedar bedding. 16,17 However, little work has been reported describing the effects of removing such volatile compounds from bedding materials on the activities of hepatic microsomal enzymes in laboratory animals. In a previous study, we developed soft-hydrothermal processing (SHP), which is based on the use of a steam phase at a temperature of 100–200°C and a pressure below saturated vapour pressure, to improve the bedding for laboratory animals. 18 SHP effectively removed volatile organic compounds from fresh bedding.
In the present study, we examined the effect that a single oral administration of essential oil of red cedar (EORC), derived from bedding had on the induction of hepatic P450 in mice. We also removed volatile organic compounds from Japanese red cedar bedding by SHP and used this SHP-treated red cedar bedding in a housing study focused on the induction of hepatic P450.
Materials and methods
Materials
Antibodies against rat CYP1A2, CYP2C6 and CYP2E1 were purchased from Daiichi Pure Chemicals (Tokyo, Japan). The anti-CYP3A4 antibody has been previously described. 19 An electrochemiluminescence-Plus (ECL-Plus) Western blotting detection reagent system was obtained from GE Healthcare (Piscataway, NJ, USA).
Animals
Ten-week-old male ICR mice (Slc: ICR) (Japan SLC, Hamamatsu, Japan) were allowed to habituate to their new housing on spruce bedding for one week, with five mice per cage. The bedding was autoclaved at 121°C for 20 min and used within one week after autoclaving. It was changed twice a week. The animals were maintained under specific pathogen-free conditions at 23 ± 3°C and 55 ± 10% humidity and with a 12 h light/12 h dark cycle. The mice were given tap water and standard chow (Labo MR stock, Nihon Nosan Kogyo KK, Yokohama, Japan) ad libitum. All animal experiments conformed to the Guide for the Care and Use of Laboratory Animals of Tohoku University.
Bedding and extraction of essential oil of red cedar by soft-hydrothermal processing and hexane
Two types of bedding, Japanese red cedar (Cryptomeria japonica D. Don) (cedar bedding) and spruce (Picea sitchensis) (spruce bedding), were obtained from Douourika Inc (Sapporo, Japan). Both beddings consisted of shaved rectangular chips, measuring approximately 10 × 15 × 0.3 mm. Fresh cedar bedding was treated using SHP to remove EORC, as previously described, 18 and was designated ST-cedar bedding. Briefly, it was treated with dry steam at a pressure of 0.35 MPa and a temperature of 140°C for 60 min in the reactor of a large-scale SHP apparatus. The steam flowed into a reservoir for cooling and for separating the gas and liquid phases. The liquid phase was further separated by letting it stand for 12 h at room temperature, and the upper oil layer was collected as EORC. To remove any remaining organic compounds from the ST-cedar bedding, it was extracted with n-hexane, a non-polar solvent, in a capped bottle (2.0 g bedding in 15 mL hexane for 72 h), and the compounds in the extract were compared with those in a hexane extract of fresh cedar bedding.
Chemical analysis of essential oil of red cedar and the hexane extract
The chemical compositions of EORC and the hexane extract were analysed using gas chromatography-mass spectrometry (GC-MS) and a gas chromatograph equipped with a flame-ionization detector (GC-FID). GC-MS analyses were carried out on a Mass Selective Detector (MSD) 5971 mass spectrometer (ion source 200°C, 70 eV) connected to a Hewlett-Packard GC-5890 II gas chromatograph with a HP-1 capillary column (30 m × 0.25 mm i.d., film thickness 0.25 μm) (all from Agilent Technologies, Wilmington, DE, USA). The column temperature was held at 50°C for 2 min and programmed to increase to 150°C at 6°C/min, then to 250°C at 4°C/min, and then to 290°C at 7°C/min, after which it was held isothermally for 5 min. The temperatures of the injector and detector were maintained at 250°C and 280°C, respectively. EORC (1 μL) in hexane (1:20) or the hexane extract of the bedding was injected in a splitless way and allowed to flow with helium (0.7 mL/min) as the carrier gas. Compounds were identified based on GC retention times relative to those of known compounds and computer matching of the mass spectra with data from the WILEY138.L mass spectral library. GC-FID analysis was performed under the same conditions, on a Hitachi 663-50 GC-FID and an electronic integrator, Hitachi D-7500 (both from Hitachi Ltd, Tokyo, Japan). The percentage compositions of the oil samples were computed from GC peak areas, without correction for response factors.
Oral administration of essential oil of red cedar
EORC was administered orally to mice (5 per time point) at a dose of 2000 mg/kg after a 16 h fast. The mice were weighed and killed by cervical dislocation at 0.5, 1, 2, 4 and 8 days after oral administration. Control mice were orally given the same volume of distilled water and were killed at 0.5 day after oral administration. The livers were immediately excised and frozen at −80°C for later analysis.
Housing study on SHP-treated bedding
In order to examine the effect of the improved cedar bedding with SHP technique, the hepatic P450 level of mice housed on cedar bedding and ST-cedar bedding were compared by referring to the shared control mice. After one week of habituation on spruce bedding, five mice were randomly assigned to the shared control mice and killed, and the other mice were randomly divided into two groups and housed on different beddings (5 mice in a cage), one with cedar bedding and another with ST-cedar bedding, and maintained for 2, 7 and 14 days. After the designated periods, mice were weighed and killed by cervical dislocation, and the livers were immediately excised and frozen at −80°C for later analysis.
Microsome preparation and Western blots
A microsomal fraction was prepared from mouse liver as previously described,
18
with minor modifications. Briefly, livers were thawed, finely chopped and homogenized in three volumes of ice-cold 1.15% KCl solution, using a motor-driven Teflon-glass homogenizer. The tissue homogenate was centrifuged (9000
Western blots were performed to immunoquantify P450 apoprotein levels. Microsomal proteins (1-10 μg) were separated by 10% SDS-polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride (PVDF) membrane (Hybond-P; GE Healthcare). The PVDF membrane was blocked with 5% skim milk in PBST (0.05% Tween-20 in phosphate-buffered saline) for 1 h with shaking and then incubated with the primary antibodies and appropriate horseradish peroxidase-labelled secondary antibodies. Signals were detected using ECL-Plus reagent, according to the manufacturer's protocol, and were quantified by densitometry, using the Fuji Film Las-1000 Program (Fuji Film Inc, Japan), to estimate hepatic P450 protein levels.
Statistical analyses
Results are expressed as the means ± standard deviation. Statistical significance was assessed at P < 0.05 using the Tukey test for the EORC oral administration data and a two-way analysis of variance (ANOVA) for the housing study data.
Results
Chemical analyses
The chemical compositions of EORC and the hexane-soluble fractions of cedar bedding and ST-cedar bedding are listed in Table 1. The EORC comprised sesquiterpene hydrocarbon compounds (55.8%), naphthalene-derived aromatics (20%), diterpene alcohols (12.9%), phenanthrene-derived aromatics (4%) and a small amount of steroids (2%). The hexane-soluble fraction of cedar bedding primarily contained sesquiterpenes (20.6%), naphthalene-derived aromatics (7%), diterpene alcohols (52.3%) and steroids (5.2%). The ST-cedar bedding contained diterpene alcohols (81.6%), steroids (7.6%) and some phenanthrene-derived aromatics (4.4%). Thus, treating red cedar bedding with the SHP reduced the total amounts of the chemicals remaining in the bedding by over 50% and removed 20–55% of the diterpene alcohols and almost all sesquiterpenes and naphthalene-derived aromatics.
Components of the extracts from Japanese cedar shaving
Compounds listed in order of elution from HP-1 column. Rt, retention times are relative to α-cubebene; Hs1, hexane soluble-fraction from cedar bedding; Hs2, hexane soluble-fraction from ST-cedar bedding. Loss rate: The extent of removal in percentages by soft-hydrothermal processing through calculating the reduction of peak area of each component from Hs1 to Hs2 determined by gas chromatography. The total loss rate (54%) was obtained from the loss of total area of all peaks from Hs1 to Hs2
*Figures represent the percentage of each component in essential oil of red cedar (EORC) based on the peak area determined by gas chromatography
†Not present in detectable amount
Oral administration of essential oil of red cedar
One of the 30 mice died on the first day after oral administration of EORC. Hepatic microsomal fractions were prepared from the remaining mice. Western blots were performed to determine P450 levels in the liver. The antibodies against CYP3A4, CYP2C6 and CYP1A2 could not distinguish mouse P450 forms in the corresponding subfamilies (i.e. CYP3A, CYP2C and CYP1A, respectively); therefore, we describe the sum of the immunoreactive proteins as CYP3As, CYP2Cs and CYP1As, respectively.
Western blots demonstrated that, at the first and second days after EORC administration, the apoprotein level of CYP3As was significantly increased (maximum of 7.1× at 1 day, compared with control mice), whereas the levels of CYP1As, CYP2E1 and CYP2Cs were only slightly increased (approximately 1.5×) (Figure 1). These proteins returned to initial levels before day 4 and were stable thereafter. Thus, orally administered EORC increased the activities of hepatic enzymes, especially reactions catalysed by CYP3As, in mice.
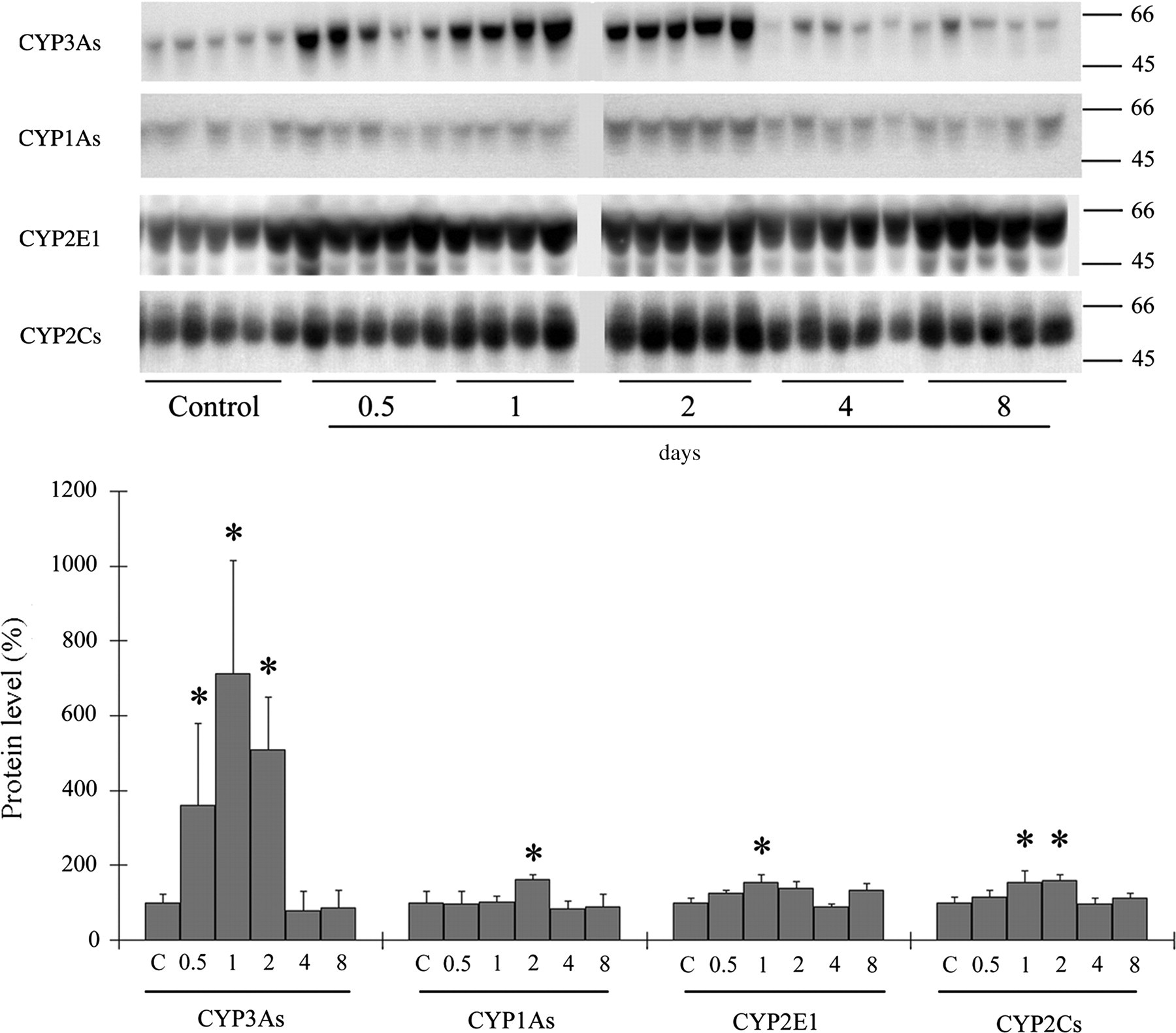
Hepatic P450 protein levels in mice after oral administration of essential oil of red cedar (EORC). Male mice were treated p.o. with EORC (2000 mg/kg) and killed 0.5, 1, 2, 4 and 8 days later. Hepatic P450 apoprotein levels were then determined. Molecular weight markers (kDa) are shown on the right of gel bands. The value for control mice (C) was set at 100%. Data represent the mean ± standard deviation (n = 4 or 5). *P < 0.05 vs. control mice, analysed by Tukey's method
Housing study
The levels of hepatic P450 apoprotein in the mice maintained in cages with ST-cedar bedding did not differ significantly from those of control mice during the experiment (Figure 2). In contrast, the levels of CYP3As, CYP1As and CYP2E1 in the livers of mice housed on cedar bedding were significantly increased (by 2.3-fold, 1.4-fold and 1.5-fold, respectively) at day 2 (Figures 2A–C). In addition, CYP2E1 was significantly increased at days 7 and 14 (Figure 2C), and CYP3As were significantly increased at day 14 (Figure 2A). A two-way ANOVA demonstrated that the protein levels of CYP3As, CYP1As and CYP2E1 were significantly decreased in the livers of mice maintained on ST-cedar bedding, compared with the levels in mice maintained on cedar bedding. These results indicate that the EORC in cedar bedding induced hepatic P450s, especially CYP3As, CYP1As and CYP2E1, and that the removal of EORC by SHP significantly reduced the ability of cedar bedding to induce hepatic P450.
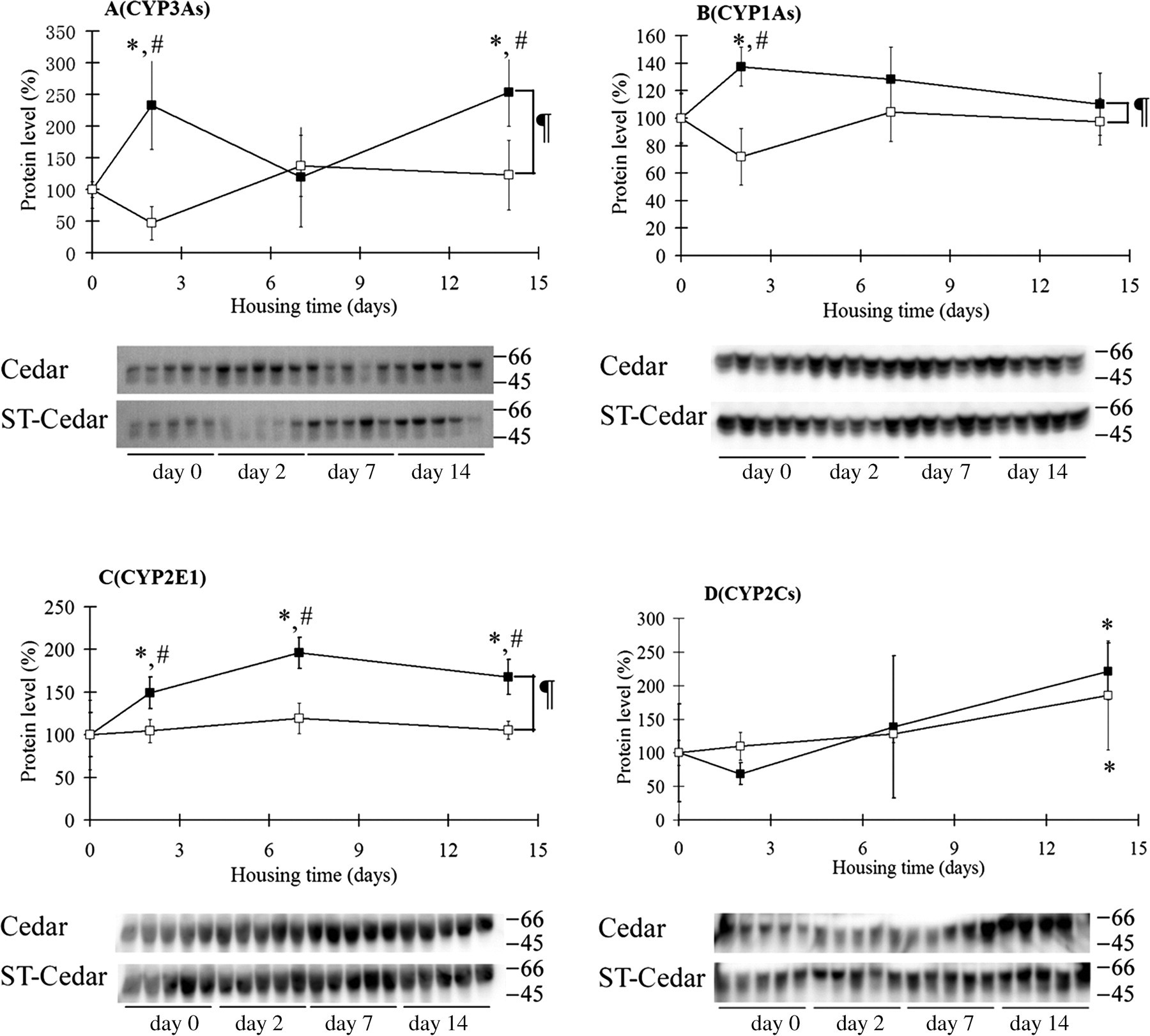
Hepatic P450 apoprotein levels in mice housed on cedar bedding or ST-cedar bedding. After one week of habituation on spruce bedding, five mice were assigned to the control mice and killed. The other mice were housed on cedar bedding (closed symbol) or ST-cedar bedding (open symbol) for 2–14 days. Hepatic P450 apoprotein levels were then determined. The same microsomal samples from the control mice were used for both cedar and ST-cedar groups. Molecular weight markers (kDa) are shown on the right of the gel bands. The value for control mice (day 0) was set at 100%. Data represent the mean ± standard deviation (n = 5). *P < 0.05 vs. control mice; # P < 0.05, vs. mice housed on ST-cedar bedding, analysed by Tukey's method. ¶ P < 0.01 between cedar bedding and ST-cedar bedding, analysed by two-way analysis of variance
Discussion
In this study, we demonstrated that EORC can induce P450s in mouse liver and that SHP removes this property from bedding made of a softwood, such as Japanese red cedar. The oral administration of EORC at a high dose resulted in strong induction of CYP3As, as well as induction of CYP1As, CYP2E1 and CYP2Cs P450 enzymes. In addition, the housing study indicated that cedar bedding increased hepatic P450 levels and that ST-cedar bedding, which had been treated by SHP, significantly decreased the levels of P450s, especially CYP3As, CYP1As and CYP2E1. Volatile sesquiterpenes and naphthalene-derived aromatics present in cedar bedding, but not in ST-cedar bedding, might be responsible for the induction of P450, although this alone does not explain the apparent differences in P450 selectivity between the oral administration and bedding experiments. Thus, it is possible that another component in EORC selectively induced CYP3As. Less volatile and higher molecular-weight diterpene alcohols (e.g. ferruginol [1R, 3S]–cembra-4,7,11,15-tetraen-3-ol) and the steroid 4,4-dimethyl-13α-androst-5-ene may be candidates. These were present in EORC, cedar bedding and ST-cedar bedding. Owing to their low volatility, these compounds may have exhibited little or no effect on P450 levels in the bedding study, despite their considerable amounts in both cedar and ST-cedar bedding.
The nuclear receptor known as pregnane X receptor (PXR) plays a key role in the xenobiotic induction of CYP3As. 20 As suggested by its name, PXR was first shown to be activated by pregnane steroids. Subsequently, other classes of steroids, including dexamethasone, pregnenolone 16α-carbonitrile, oestradiol and androstanol, have been shown to be ligands for human and/or mouse PXR. 21,22 Interestingly, EORC contained a steroid with an androstane structure, i.e. 4,4-dimethyl-13α-androst-5-ene (Figure 3), which may be a potential PXR ligand, although further studies are necessary to examine this.
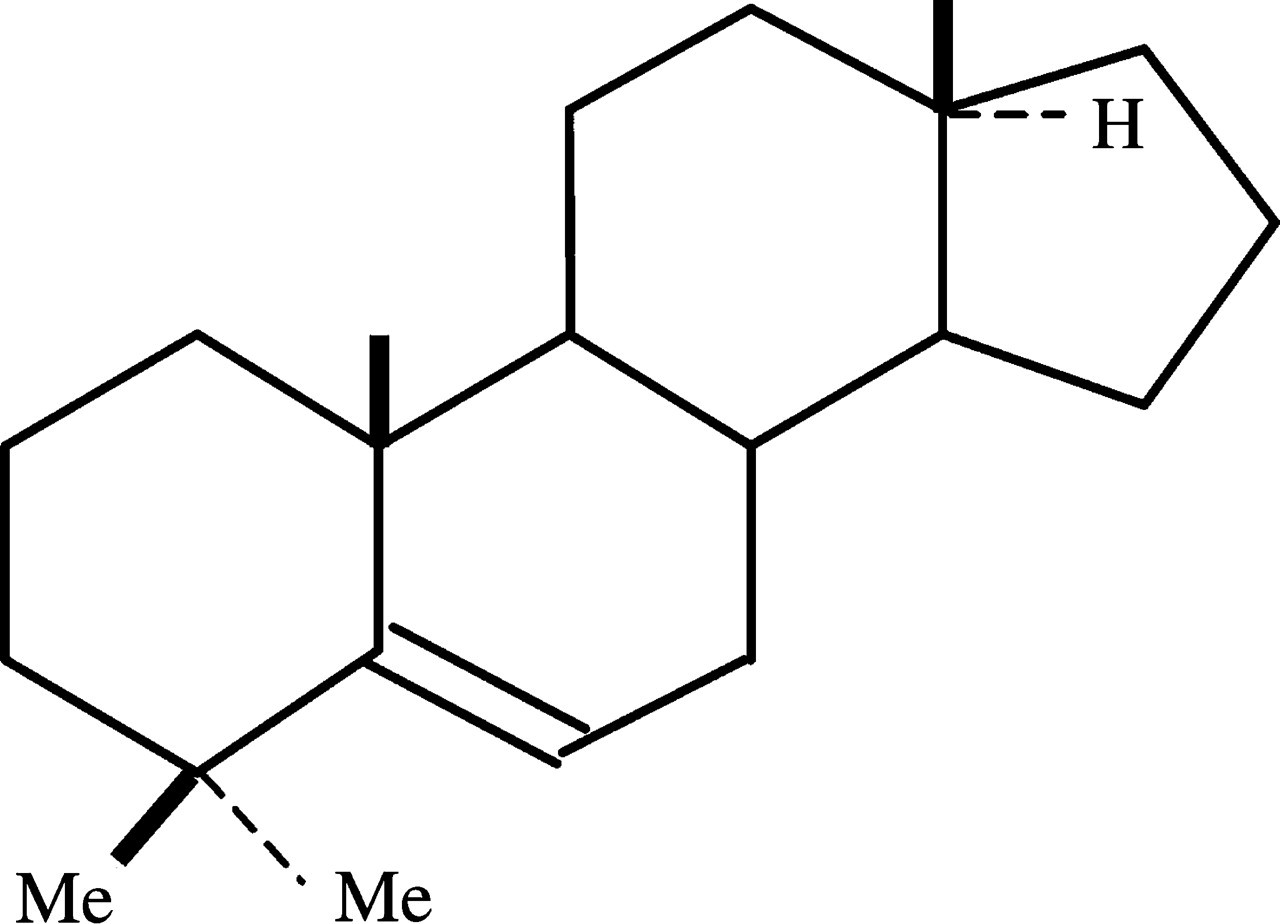
Chemical structure of the steroid 4,4-dimethyl-13α-androst-5-ene
According to previous reports, polycyclic aromatic compounds in cedar bedding seem to be responsible for the induction of CYP1As in animal livers, through aryl hydrocarbon receptor (AhR). 23–26 EORC contains many naphthalene- and phenanthrene-derived aromatics and 7,12-dimethyl-benzen[a]anthracen-4-ol, which could be agonists of AhR and could result in increased expression of CYP1As induced by oral EORC administration and cedar bedding. ST-cedar bedding did not cause obvious induction of CYP1As, although it contained considerable amounts of 7,12-dimethyl-benzen[a]anthracen-4-ol; this may be the result of the low volatility of this compound.
Treatment of bedding by SHP removed not only most volatile sesquiterpenes and naphthalene-derived aromatics but also much high molecular-weight diterpene alcohols and steroids. The SHP conditions of 0.35 MPa and 140°C for 60 min have been shown to be optimal for softwood bedding and useful for improving its physical properties such as absorption of ammonia, as described in our previous study. 18 In the present study, SHP was also demonstrated to be a practical technique for the removal of organic chemicals such as hepatic P450-inducing compounds from bedding, improving the quality of red cedar shavings as a laboratory animal bedding material.
Footnotes
ACKNOWLEDGMENTS
We thank Dr Nakamichi Yamasaki (Osaka University, Osaka, Japan) for advice on soft-hydrothermal processing. We are also grateful to Ms Teruko Sueta and Ms Tomomi Kibushi (Tohoku University, Sendai, Japan) for their assistance in the animal experiments.
