Abstract
The intravenous injection of the plant lectin concanavalin A (ConA) is a widely used model for acute immune-mediated hepatitis in mice. In contrast to several other models for acute hepatic damage, ConA-induced injury is primarily driven by the activation and recruitment of T cells to the liver. Hence, the ConA model has unique features with respect to its pathogenesis and important similarities to immune-mediated hepatitis in humans, such as autoimmune hepatitis, acute viral hepatitis or distinct entities of drug toxicity leading to immune activation. However, the ConA model has considerable variability, depending on the preparation of the compound, genetic background of the mice, sex, age and microbial environment of the animal facility barrier. This standard operating procedure (SOP) comprises a detailed protocol for the ConA application, including preparation of ConA working solution, handling of the animals, choice of the appropriate conditions and endpoints, as well as efficient dose-finding.
Historic background of the model
Lectins, also known as phytohemagglutinins (PHAs), were described more than 100 years ago, and their potential to act as mitogens, toxins and activators of the immune system was shown. 1 In general, they are a family of carbohydrate binding proteins with a high specificity for distinct sugar moieties. Some lectins such as the mannan-binding lectin (MBL) have been shown to be part of the innate antimicrobial defense in higher organisms. Lectins have also been shown to act as stimulators for lymphocytes.2,3 The lectin concanavalin A (ConA) (Figure 1) has been used in biological research for more than 40 years, e.g. as an insulin receptor agonist, 4 but also in general T cell biology. 2 The outstanding popularity of ConA in biology and biochemistry has arisen from the fact that this protein was the first of the lectins to be purified in the pure, crystalline form5,6 and therefore the first that was available on a commercial basis. Together with PHA, ConA has been shown to induce haemagglutination and also an ability to crosslink cellular receptors, therefore leading to both activation and cellular adhesion. 2 In 1992, Tiegs et al. described a ConA-induced model of acute immune-mediated liver inflammation, showing the specific involvement of T cells in promoting such liver damage. 7 This enabled the model for the first time to selectively address T cell functions in inflammatory liver disease.
Other models such as staphylococcal enterotoxin B (SEB) + D-galactosamine (GalN) 8 or GalN + lipopolysaccharide (LPS) mediated liver disease 9 have been shown to be mediated by a GalN-induced hypersensitivity of the liver to tumour necrosis factor (TNF)-α,9,10 and therefore only resembling a secondary response following the Toll-like receptor-mediated or superantigen-induced cytokine release. 8 Both models trigger innate and adaptive immune responses and are less specific regarding the target organ. By contrast, ConA has been shown to be deposited specifically in the liver, e.g. by insulin receptor binding. 4
Other models such as acetaminophen (APAP)-induced liver injury 11 , dietary non-alcoholic steatohepatitis 12 or carbon tetrachloride-induced liver inflammation13,14 are triggered by primary damage to the hepatic parenchymal tissue with subsequent leukocyte activation and secondary immune-mediated damage 15 . Consequently the ConA model serves as a valuable method, non-redundant to other models, for investigating the specific mechanisms of lymphocyte activation and immunity in acute liver disease.
Pathogenic mechanisms of liver damage
ConA-induced acute liver inflammation, together with APAP-induced liver injury, are the two most commonly used models for acute liver injury. The outcome of both diseases is somewhat similar, leading to severe liver inflammation, tissue necrosis accompanied by highly elevated serum transaminases and possible terminal organ failure, nonetheless the underlying disease aetiologies show fundamental differences. While in APAP-induced liver injury the primary damage is caused by the toxic metabolite N-acetyl-p-benzoquinone imine (NAPQI) leading to interference with the mitochondrial metabolism, ConA primarily leads to recruitment and activation of T cells7,16 and natural killer T (NKT) cells.
17
Although ConA has also been reported to be at least partially toxic to hepatocytes in vitro,
1
the majority of the in vivo studies show that lymphocytes are the key players in the induction and progression of ConA-induced liver disease.6,18,19 Various mechanisms have been previously identified that drive the liver damage that follows ConA challenge (Figure 2). Interference with the cytokine TNF-α by administration of inhibitory anti-TNF-α antibodies protects the liver against ConA-induced injury.7,20,21 Also, treatment with recombinant interleukin-6, which has been shown to be counter-regulated with TNF-α, ameliorates hepatocyte damage in acute ConA hepatitis.
16
TNF-α is directly capable of inducing hepatocyte apoptosis via TNF-receptor signalling inducing caspase-8 leading to mitochondrial cytochrome c and caspase-3 activation.
22
However, in hepatocytes TNF signalling can also trigger alternate pathways activating downstream signals such as NF-κB essential modulator (NEMO) and NF-κB21,23 which have been shown to counterbalance apoptosis via induction of Bcl-XL and inhibition of c-Jun N-terminal kinase (JNK) activation.
24
Consequently TNF-α can only induce hepatocellular damage if NF-κB signalling is sufficiently inhibited.
25
Three dimensional structure of the concanavalin A (ConA) monomer. The ConA protomer of the jack bean (Canavalia ensiformis) has a unique arrangement of β structures forming a dome-shaped, ellipsoidal structure. The backbone is dominated by the presence of two anti-parallel pleated sheets, a twisted sheet of seven strands and a bowed sheet of six strands forming the back surface of the monomer.48,49 In the molecule two metal-binding sites for manganese or calcium depicted as spheres are bound close together at the top of the molecule. The structure depicted here was generated by using the Ribbons XP software (Version 3.0)
50
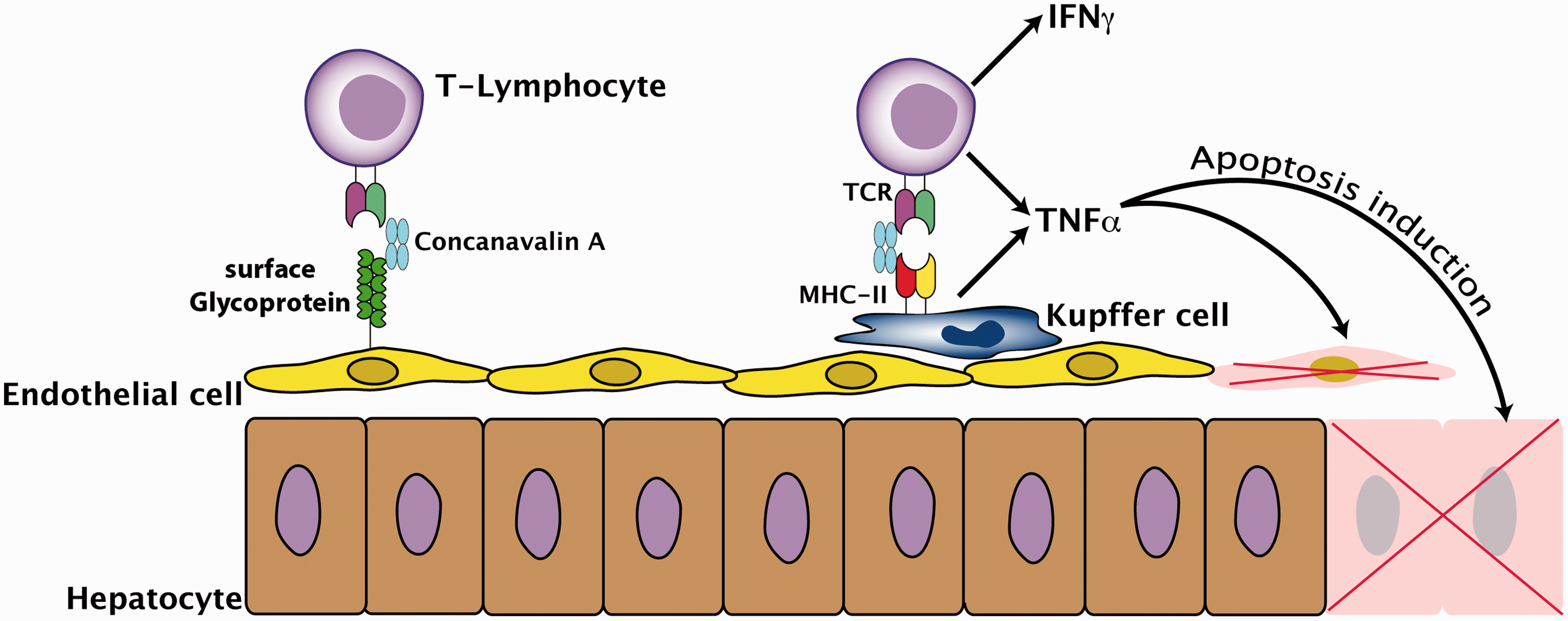
and the crystal coordinates of ConA that were taken from the RCSB Protein Data Bank repository (PDB, access. no. 2CNA) that can be found at http://www.rcsb.org. Induction of concanavalin A (ConA)-mediated liver damage by activation of T cells and macrophages. ConA binds to mannose-rich glycoproteins on liver sinusoidal endothelial cells (LSEC) and Kupffer cells (liver macrophages). T cells become activated by crosslinking their T cell receptors (TCR), leading to a secretion of immunogenic cytokines such as interferon (IFN)-γ and tumour necrosis factor (TNF)-α which subsequently induce apoptotic cell death in LSEC and hepatocytes. MHC-II: major histocompatibility complex class II.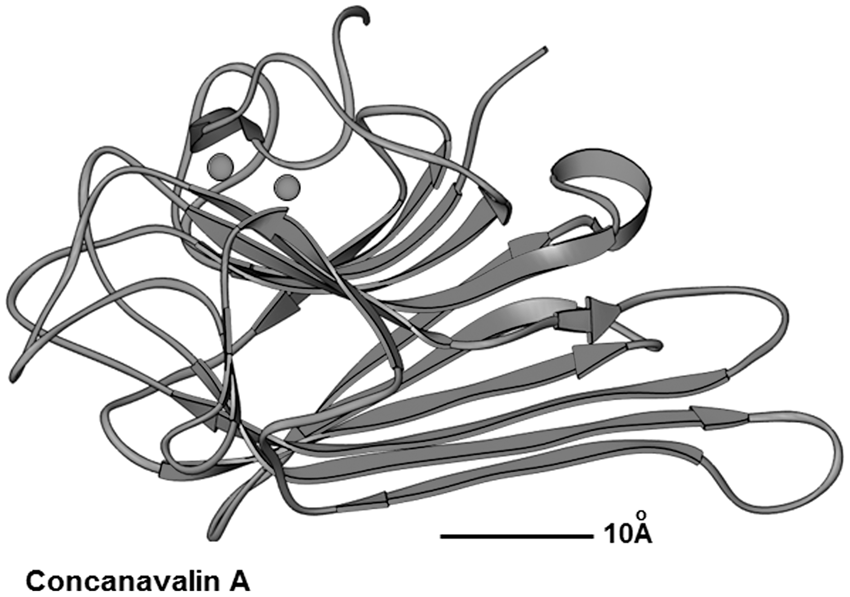
However, DNA fragmentation and formation of apoptotic bodies was visible as early as 5 h after ConA injection in hepatocytes, preceding serum transaminase elevation (Figure 3),
20
clearly suggesting programmed cell death mechanisms. Other signals such as TNF-related apoptosis inducing ligand (TRAIL) or Fas ligand have been shown to be involved in hepatocyte damage in the ConA model26,27 and are readily capable of inducing hepatocyte cell death, e.g. via TRAIL-induced necroptosis and receptor-interacting protein 1 (RIP)1/RIP3 kinase signalling.
28
Necroptosis shares several features of regulated cellular degradation such as breakdown of the mitochondrial potential and also nuclear fragmentation, but it ultimately leads to an uncontrolled degradation of the cells resulting in the release of cytoplasmic components and therefore triggering inflammatory responses.25,29
Liver damage after concanavalin A (ConA)-mediated liver damage. (a) Serum transaminase activities (alanine aminotransferase [ALT]) determined 2 h and 8 h after intravenous injection of 25 mg/kg ConA into C57BL/6 mice. Saline-injected animals were used as controls. (b) Representative hematoxylin and eosin (H&E) stainings of livers from mice injected with saline solution (left) and ConA (right) 8 h after injection. Scale bar: 500 µm. Black arrows indicate representative areas of injury (apoptosis and necrosis), white arrows indicate leukocyte adhesion to vascular endothelium.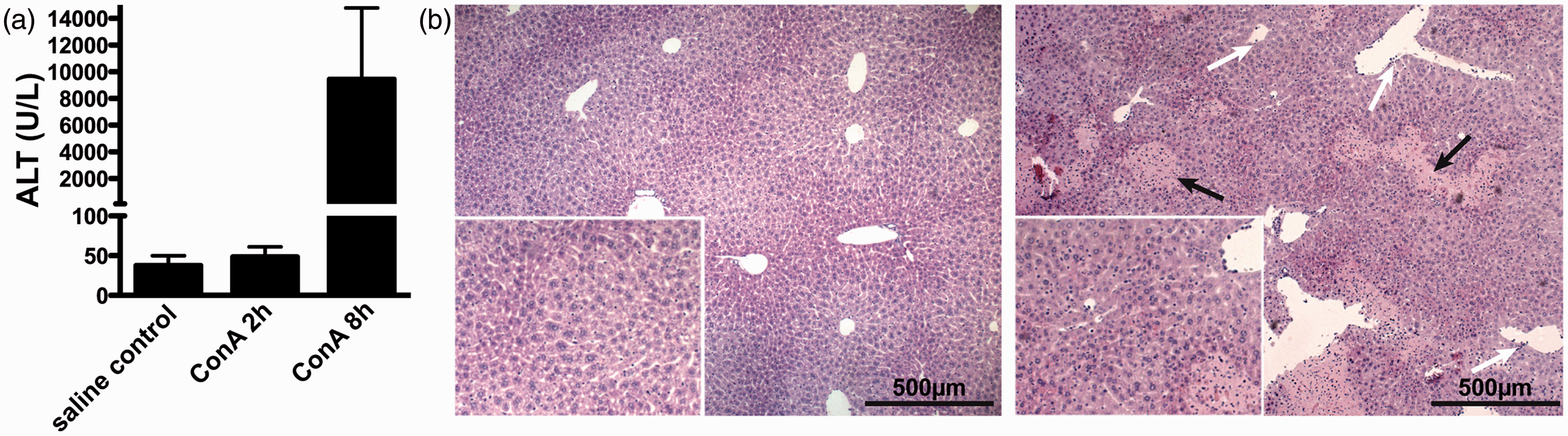
Furthermore, the T cell activating cytokine interferon (IFN)-γ acts as a necessary signal for the induction of liver inflammation by ConA. 19 The role for cytotoxic CD8 T cells (cytotoxic T lymphocyte [CTL]) has remained controversial, but at least one study has shown a direct role for perforin-induced hepatocyte apoptosis by CTL. 30 Although ConA, like various other lectins such as PHA, can in principle induce blood clotting and red blood cell aggregation, 31 the pathological effects are limited to the liver and spleen. However, depletion of platelets is not protective against ConA-induced liver disease, arguing against passive organ damage, e.g. due to impaired circulation or hypoxia. 32 It can be shown that ConA is mainly accumulating in the liver after intravenous injection, binding primarily to liver sinusoidal endothelial cells lining the small hepatic blood vessels. 33 Therefore, it can act as a bridging component, inducing firm cellular arrest of passenger T cells, leading to their activation.33,34 This view is further supported by the finding that mice carrying knock-outs for adhesion molecules such as lymphocyte function-associated antigen (LFA)-135 or P-selectin 36 are protected from ConA liver damage.
Experimental procedure
General considerations
Genetic background
Susceptibility to concanavalin A (ConA)-mediated liver injury in different inbred mouse strains.
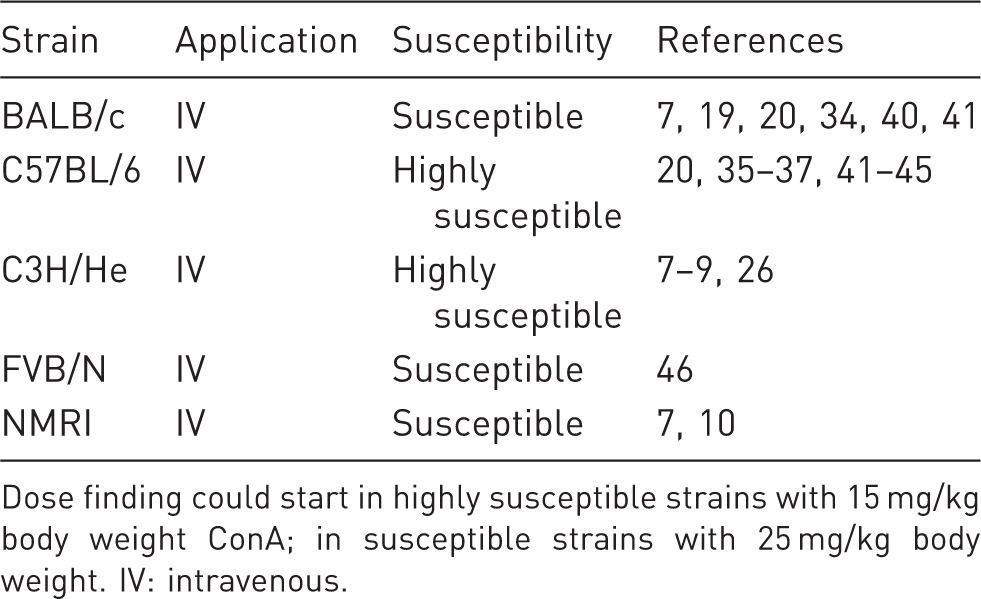
Dose finding could start in highly susceptible strains with 15 mg/kg body weight ConA; in susceptible strains with 25 mg/kg body weight. IV: intravenous.
Representative examples of established concanavalin A (ConA) application protocols.
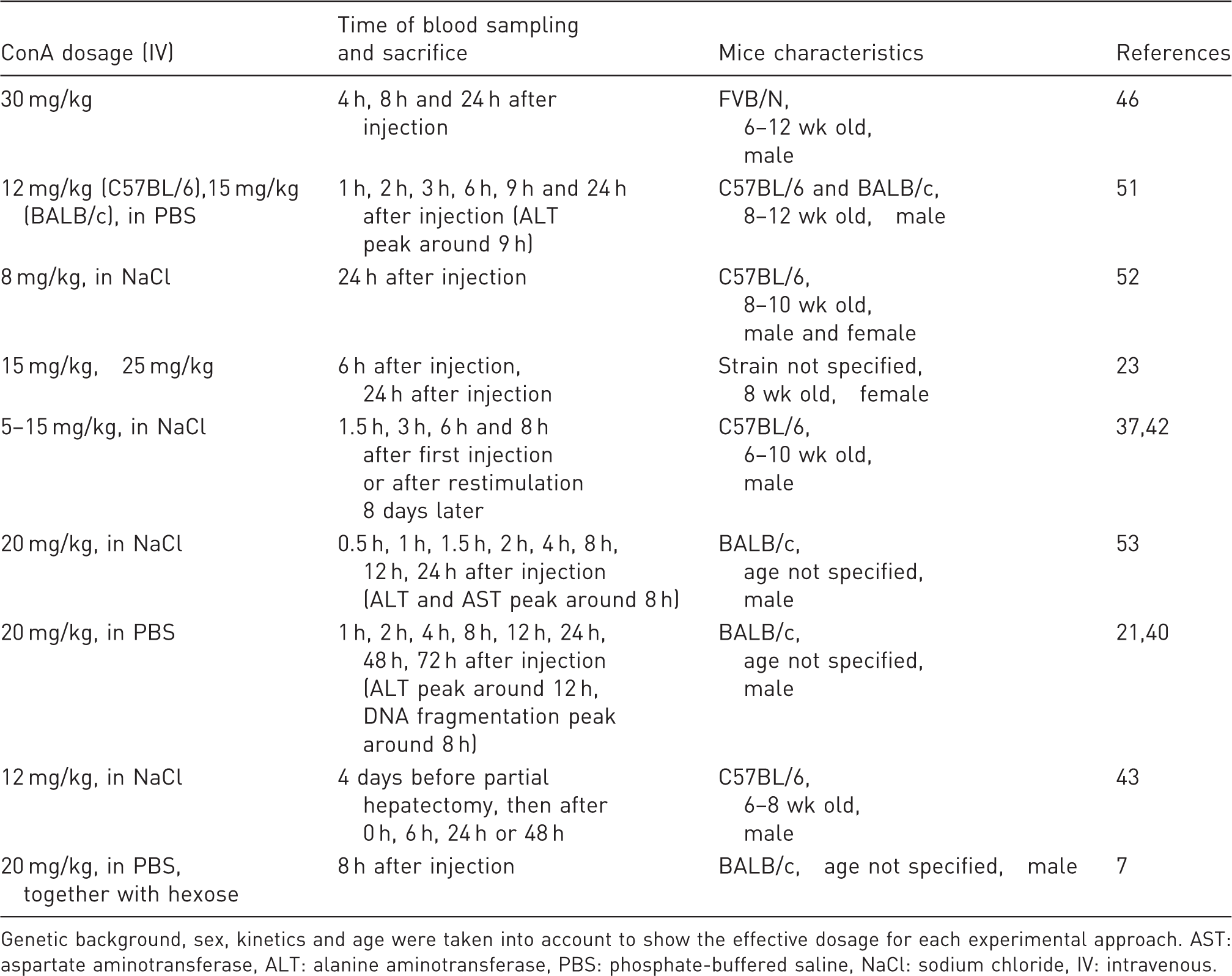
Genetic background, sex, kinetics and age were taken into account to show the effective dosage for each experimental approach. AST: aspartate aminotransferase, ALT: alanine aminotransferase, PBS: phosphate-buffered saline, NaCl: sodium chloride, IV: intravenous.
Duration of treatment/experimental time course
Dosage calculation for concanavalin A (ConA) application into mice adapted to body weight.
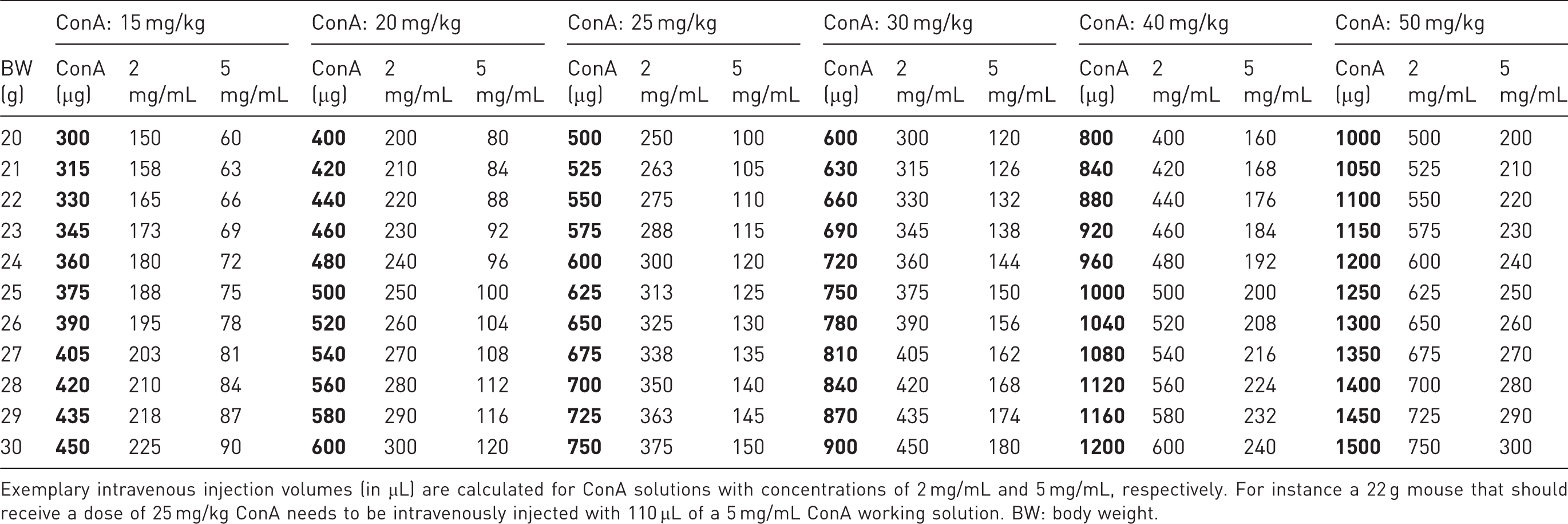
Exemplary intravenous injection volumes (in µL) are calculated for ConA solutions with concentrations of 2 mg/mL and 5 mg/mL, respectively. For instance a 22 g mouse that should receive a dose of 25 mg/kg ConA needs to be intravenously injected with 110 µL of a 5 mg/mL ConA working solution. BW: body weight.
Gender preference
The effect of ConA is highly dependent on the hormonal state of the animal. Female mice may show a higher susceptibility to ConA, but also a greater variation in the disease outcome as shown in a comparative study performed in BALB/c mice. 38 Therefore, it is preferable to perform the experiments solely using male mice, if possible, to reduce the number of animals needed to reach statistically significant results.
Dose finding
Since ConA is extracted and purified from jack beans, its biological activity shows batch-dependent variations. Therefore, the dose necessary for inducing liver disease has to be tested for each batch before an experimental series, and typically varies between 10 and >30 mg/kg for inducing severe acute liver inflammation. Due to variations in ConA susceptibility based on the genetic background, sex, age and microbial environment (see Table 2), we strongly advise dose-finding experimentation before a large series of experiments is started in a new research project. A typical dose-finding experiment would sequentially test 15 mg/kg, 20 mg/kg and 25 mg/kg in n = 3 (wild-type/control) mice per condition; and the readout is the alanine aminotransferase (ALT) level at 8 h. The minimal dose that reliably induces severe, but sublethal, liver injury (e.g. ALT > 1000 IU/L) should be subsequently employed for the experiments. Previous studies have shown that even dosages as low as 1.5 mg/kg are already capable of inducing mild immune-mediated hepatitis with slightly elevated transaminase levels but no further clinical manifestation of liver disease. 7
ConA as a tetrameric protein complex is sensitive to environmental decay and needs to be stored under protected conditions at either –20℃ or –80℃. However, ConA solutions prepared in sterile 0.9% sodium chloride (NaCl) or phosphate-buffered saline (PBS) are usually stable up to several months under proper conditions. Freeze–thaw cycles should be avoided at all costs so as to circumvent protein degradation.
Practical implementation of ConA application
Intravenous application of ConA
It is important to ensure that the compound is completely dissolved to avoid complications like embolism. Also, excessive pipetting or even vortexing should be avoided to prevent foam formation. To perform the single injection into the lateral tail vein, mice are put into a restrainer unit. Furthermore, mice are warmed up with an infrared lamp prior to injection and tails are dipped into 35–37℃ warm water to enhance vasodilation. Injection is commonly performed using a 26 G or 27 G needle.
Dose calculation
ConA is readily soluble in aqueous solution; therefore, the concentration of the injection solution can be adapted to match the requirements according to EU Directive 2010/63 on the protection of animals used for scientific purposes. Administration of ConA has to be performed in adaptation to the body weight of the mouse.
Example calculations are provided in Table 3 to show the injection volume range for the most common dosages of ConA.
Animal burden/side-effects
Regarding clinical observations from patients with acute liver failure, primary liver damage can be considered a painless process per se. However, as the disease progresses the possible side-effects make it necessary to closely monitor the animals starting at 5–8 h after injection. If necessary, mice that show severe signs of fatigue, hypothermia (<35℃), or illness should be sacrificed, thus preventing them dying from complications caused by acute liver failure such as cerebral oedema, encephalopathy, coagulopathy, renal failure and circulatory failure. 39
In a typical experiment intending to analyse established ConA-mediated liver injury, mice are inspected one hour after application and sacrificed 8 h after ConA injection by an overdose of isoflurane followed by cardiac puncture and withdrawal of blood. Mice that are intended to be analysed at 12 h or 24 h after ConA are inspected every 6–12 h and sacrificed prematurely should they show severe signs of distress. To monitor development of liver disease, blood samples can be taken 8 h after ConA application to determine serum transaminase (ALT and aspartate aminotransferase [AST]) levels.
To minimize handling-associated stress to the animals related to the fixation and intravenous injection, laboratory personnel should receive extensive training in order to be able to perform ConA administration by the intravenous injection procedure reliably and with minimal impact on the mice.
Biometrical calculation of required cohort size
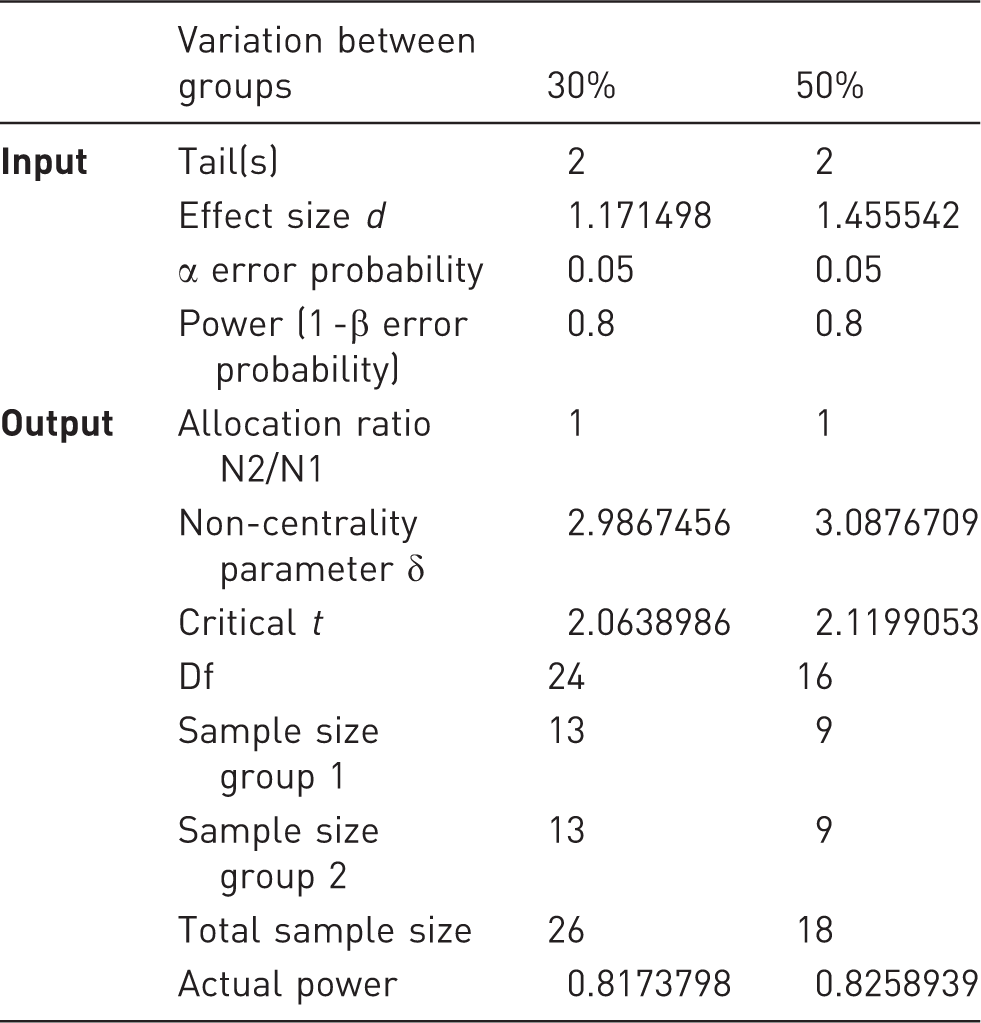
Liver damage is routinely assessed by qualitative and quantitative histology and by quantification of serum transaminase activities (AST and ALT). Mice treated with ConA show an average standard deviation of about 33% regarding the transaminase levels. To determine the number of animals required to generate statistically significant data, power calculations were performed for exemplary differences of 30% and 50% reductions in mean transaminase levels. The values were chosen as a result of our observations, taking into account that a reduction of 30% in serum transaminases would be considered a moderately liver-protective intervention and a reduction of 50% is seen in clinically highly effective interventions. The effect sizes and sample group sizes were calculated using G*Power V.3.1.9 for Mac, to achieve a statistical power of 80% and a specific α error probability of 0.05. The calculated effect sizes were 1.1715 and 1.4555, respectively, with a total sample size of 26 animals for a difference of 30%, and a total sample size of 18 animals for a 50% difference in mean transaminase measurement (Table 4, Figure 4).
Biometrical power plot to determine the total sample size (= animals). Based on a statistical power of 80% (blue line) calculations were performed (a) for a 30% difference in mean values of serum alanine aminotransferase (ALT) levels (e.g. pharmacologically active compound in concanavalin A (ConA) hepatitis compared with mice treated only with ConA) or (b) for a 50% difference in mean values, respectively. Representative biometric calculation of sample size determined by G Power
47
for 30% and 50% biological effects determined by differences in statistical mean values.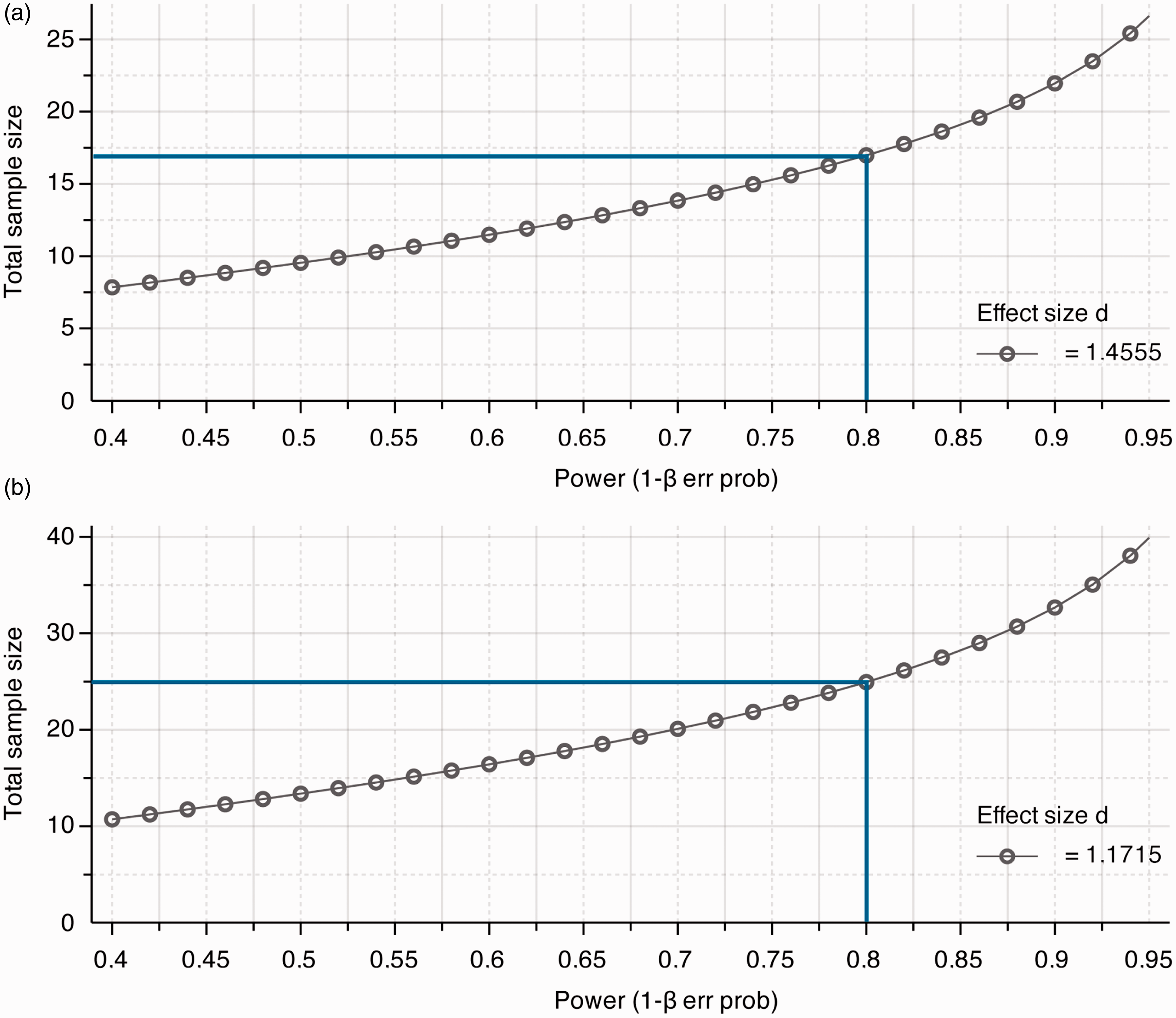
Classification of severity of procedure
According to Article 15 of the EU Directive 2010/63 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF) the estimated degree of pain, suffering, distress or lasting harm to the animals subjected to ConA application should be classified using the scores ‘non-recovery’, ‘mild’, ‘moderate’ or ‘severe’.
Details about the classification criteria that underlie this assessment were established by the Expert Working Group on severity classification of scientific procedures performed on animals. These can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf.
The application of ConA to induce liver failure with no major impairment of liver function is classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63. The induction of liver failure and death due to typical complications of acute liver failure such as cerebral oedema and encephalopathy, coagulopathy, renal, haemodynamic and cardiorespiratory failure is classified as a severe procedure. According to the 3R principle, the procedure should undergo a refinement, and humane endpoints must be implemented with frequent observation points to restrict pain, suffering, distress or lasting harm to the animals. Then the procedure can be reclassified as moderate.
Concluding remarks
ConA-induced liver disease is an important tool in liver inflammation research for several reasons. First, the ubiquitous and highly specific activation of T cells provides a suitable model for directly investigating the mechanisms of T cell-mediated hepatitis, which shares similarities to human autoimmune or acute viral hepatitis as well as with immune-mediated drug hepatotoxicity and may therefore help to develop new treatment options. Secondly, since virtually all forms of acute and chronic liver injury are linked at some stage to immune cell activation and immune-mediated liver damage, the ConA model enables us to specifically address the involvement of T cells in liver disease due to its unique activation and recruitment of T lymphocytes into the liver tissue. This guideline is intended to summarise the most important features, including technical as well as biometrical issues for performing standardized and reproducible animal experiments in line with the new European Animal Welfare Regulations.
Ethical statement
The experiments were performed in accordance with the German legislation governing animal studies following the ‘Guide for the care and use of laboratory animals’ (NIH publication, 8th edition, 2011) and the Directive 2010/63/EU on the protection of animals used for scientific purposes (Official Journal of the European Union, 2010). Official permission was granted from the governmental animal care and use office (LANUV Nordrhein-Westfalen, Recklinghausen, Germany).
Footnotes
Acknowledgements
The authors thank all members of the Tacke Lab and the Department of Medicine III for helpful discussions.
Declaration of conflicting interests
The authors disclose no conflict of interests.
Funding
This work was supported by the German Research Foundation (DFG SFB/TRR57).
