Abstract
Preface
Whenever animals are used in research, minimizing pain and distress and promoting good welfare should be as important an objective as achieving the experimental results. This is important for humanitarian reasons, for good science, for economic reasons and in order to satisfy the broad legal principles in international legislation. It is possible to refine both husbandry and procedures to minimize suffering and improve welfare in a number of ways, and this can be greatly facilitated by ensuring that up-to-date information is readily available. The need to provide such information led the British Veterinary Association Animal Welfare Foundation (BVAAWF), the Fund for the Replacement of Animals in Medical Experiments (FRAME), the Royal Society for the Prevention of Cruelty to Animals (RSPCA) and the Universities Federation for Animal Welfare (UFAW) to establish a Joint Working Group on Refinement (JWGR) in the UK. The chair is Professor David Morton and the secretariat is provided by the RSPCA. This report is the ninth in the JWGR series. The RSPCA is opposed to the use of animals in experiments that cause pain, suffering, distress or lasting harm and together with FRAME has particular concerns about the continued use of non-human primates. The replacement of primate experiments is a primary goal for the RSPCA and FRAME. However, both organizations share with others in the Working Group, the common aim of replacing primate experiments wherever possible, reducing suffering and improving welfare while primate use continues. The reports of the refinement workshops are intended to help achieve these aims.
This report produced by the British Veterinary Association Animal Welfare Foundation (BVAAWF)/Fund for the Replacement of Animals in Medical Experiments (FRAME)/Royal Society for the Prevention of Cruelty to Animals (RSPCA)/Universities Federation for Animal Welfare (UFAW) Joint Working Group on Refinement (JWGR) sets out practical guidance on refining the husbandry and care of non-human primates (hereinafter primates) and on minimizing the adverse effects of some common procedures. It provides a valuable resource to help understand the physical, social and behavioural characteristics and needs of individual primates, and is intended to develop and complement the existing literature and legislative guidelines. Topics covered include refinements in housing, husbandry and common procedures such as restraint, identification and sampling, with comprehensive advice on issues such as primate communication, assessing and facilitating primate wellbeing, establishing and maintaining social groups, environmental and nutritional enrichment and animal passports. The most commonly used species are the key focus of this resource, but its information and recommendations are generally applicable to other species, provided that relevant individual species characteristics are taken into account.
1 Introduction and aims
This report, produced by the UK Joint Working Group on Refinement (JWGR), provides practical guidance on how to refine the husbandry and care of non-human primates (hereinafter referred to as primates), and to minimize the adverse effects of some common procedures. The guidance is based on the existing literature, current good practice and the professional experience and views of the authors. It is intended to complement and interpret the existing legal and professional guidelines (e.g. Council of Europe 1986, 2006, Institute for Laboratory Animal Research 1998, National Research Council 1996, 1998, International Primatological Society 2007), and the advice provided in standard reference texts, such as those listed in Box 1. It also reflects the fact that in many primate units in recent years there has been a cultural change in the way animals are managed, with a shift towards closer human–animal interaction. This has resulted in significant reductions in suffering, improvements in animal welfare and benefits for science. This report provides examples of how these benefits can be achieved.
Useful general reference texts
Useful general reference texts
The Psychological Well-being of Nonhuman Primates (National Research Council 2003), www.nap.edu/catalog/4909.html UFAW Handbook on the Care and Management of Laboratory Animals. 7th edn (Poole 1999) Comfortable Quarters for Nonhuman Primates in Research Institutions. 9th edn (Reinhardt 2002), www/awionline.org/pubs/cq02/Cq-prim.html The Welfare of Non-Human Primates Used in Research (Scientific Committee on Animal Health and Welfare 2002), www.europa.eu.int/comm/food/fs/sc/scah/out83_en.pdf Handbook of Primate Husbandry and Welfare (Wolfensohn & Honess 2005)
Specific recommendations are made throughout the text, with the over-arching principles summarized at the end. The report focuses on the most commonly used primate species, but the information and recommendations are generally applicable to other species, provided that species characteristics (e.g. morphological, behavioural and social) are taken into account. The report should be useful for a wide range of personnel, in particular scientists, animal technicians and veterinarians who are responsible for caring for laboratory primates and/or carrying out procedures. Much of the information is relevant to those involved in designing experiments or managing studies, and it is hoped that they too will embrace the concepts in the report. It should provide guidance for members of ethical review processes (ERPs), institutional animal care and use committees (IACUCs), and other such bodies, and demonstrate the benefits of refinement to those who provide the financial and other resources.
Members of the Working Group would like to dedicate the report to Dr Sylvia Taylor of the United States Department of Agriculture, Animal and Plant Heath Inspection Service (Animal Care), who was a highly valued contributor until her untimely death in 2005.
There are four guiding principles for designing and implementing refinement strategies encompassed in this report.
First, it is important to gain a comprehensive understanding of the biology and behaviour of the primates worked with, and to recognize how their health and psychological wellbeing affects the quality of scientific research. Data from experiments carried out on well-balanced, healthy and calm animals are more likely to be consistent and meaningful, whereas poor welfare can lead to a variety of physiological and psychological responses that will affect experimental results, and can even lead to the premature termination of an experiment and wastage of animals (Poole 1997, Russell 2002, Reinhardt 2004). Refining laboratory animal husbandry, care and use, therefore not only improves animal welfare, but also improves the scientific integrity of the results. In addition, less-stressed animals should interact more positively with humans and be more responsive to training, which will also reduce stress for staff. These issues are addressed in Section 2 of the report.
Second, this understanding should be used to consider every aspect of the animals' lifetime experience, to identify all potential harms, and to factor these into harm/benefit assessments 1 of research programmes. It is important to include the full, cumulative effects of contingent harms from non-experimental factors and procedures associated with housing, husbandry and care, as well as those from experimental procedures. The contingent harms are likely to be greater for some primates (e.g. imported animals) than others. Sections 3–5 of the report provide examples of how refinements can be achieved in primate husbandry and care, and in common procedures. Long-term maintenance and euthanasia are discussed in Sections 6 and 7. The sourcing of primates, including the use of wild-caught animals, husbandry conditions in breeding and quarantine facilities, and transport, are addressed in Section 8. These are relevant to primate users as well as breeders and suppliers, and dialogue between both parties can facilitate many aspects of refinement.
The weighing of potential harms to animals (pain, suffering, distress or lasting harm) against the potential benefits of the research.
Third, an open-minded approach to possibilities for refinement should always be maintained. If there appear to be constraints on changing established practice in any area, it is important to identify what these are, evaluate whether they are real or perceived, and see how they can be overcome. Resistance to changing traditional practices may be due to concerns about the impact on the animals, the science, or animal house management. For example, it may be thought that animals will fight if group-housed, that enrichment items will promote aggression, that a varied diet will affect a study, or that a client's requirements preclude certain refinements. While such concerns may be genuine and should be acknowledged and respected, they may also be unfounded, or based on incorrect or out-of-date information. It is therefore important to explore the reasoning behind any constraints or limitations and to challenge them. Many problems are surmountable with commitment, good communication, up-to-date information, and support from all interested parties. The local ERP or IACUC can be very helpful in this respect.
Fourth, there should be a refinement implementation programme with a key individual empowered to take overall responsibility for effecting refinement in practice and for reviewing and monitoring progress.
2 Understanding primates as a basis for refinement
Understanding the biology and behaviour of primates bred and used for research at a species and individual level, and of how aspects of the laboratory environment (such as husbandry, care and experimental use) are likely to impact on these, is probably the single most important factor in the design and implementation of all types of refinement. This will, for example, help personnel recognize and interpret primate signals and facilitate the design of successful habituation and socialization programmes, which help staff and primates to interact positively. It will also help in training animals to actively cooperate with scientific, veterinary and husbandry procedures so that stress and/or distress for both animal and handler is reduced.
There is a considerable body of literature on primate biology, ecology and behaviour stemming from both field and laboratory studies that can help in this respect (e.g. Coimbra-Filho & Mittermeier 1981, Smuts et al. 1987, Mittermeier et al. 1988, Lindburg 1991, Rylands 1993, Fa & Lindburg 1996, Fleagle 1998, Roder & Timmermans 2002). However, many of these publications are highly specialized and, for those who are not primatologists, their content may be difficult to interpret and apply to the laboratory environment and experimental use. Obtaining specialist advice from primatologists on the biology, ecology and behaviour of individual primate species can help in this respect (see International Directory of Primatology, http://pin.primate.wisc.edu/idp/). Some general texts that are based on the specialist literature, but provide a good introduction to primate biology, ecology and behaviour in the laboratory context are listed in Box 1.
A number of types of primates are used in scientific procedures including prosimians, New World monkeys, Old World monkeys and apes. Families and species may be adapted to different habitats in the wild and may have different physical and behavioural characteristics, patterns of social organization, and temperament styles (Table 1). As a result, their responses to the laboratory environment can vary considerably. It is important to recognize and address such differences, otherwise animals may be kept in a manner inappropriate to their needs. For example, marmoset (Callithrix) and tamarin (Saguinus) species are often housed in exactly the same way, despite differences in behaviour (e.g. in aggression, arboreality and cage use) and ecology (e.g. home range size and daily path length). This can have serious consequences on their welfare and breeding success (Prescott & Buchanan-Smith 2004). There are also implications for the quality of science in that differing responses of species and individuals to the laboratory environment, and to the stresses to which individual animals are subjected, can affect scientific data and its interpretation. For example, rhesus, long-tailed, bonnet and pig-tailed macaques show distinct and consistent differences in behavioural and adrenocortical responses to the stress induced by confinement in a transport cage and by a change in cage or room (Clarke et al. 1988a,b, Crockett et al. 1995, 2000). Thus, for both welfare and scientific reasons, husbandry and experimental protocols always need to be designed, such that they are appropriate to the species and, where relevant, to the individual animals (Buchanan-Smith et al. 2004).
Characteristics of the common laboratory primate species (adapted from Roder and Timmermans 2002, Rowe 1996 and Primate Info Net factsheets at http://pin.primate.wisc.edu/factsheets [secondary sources])
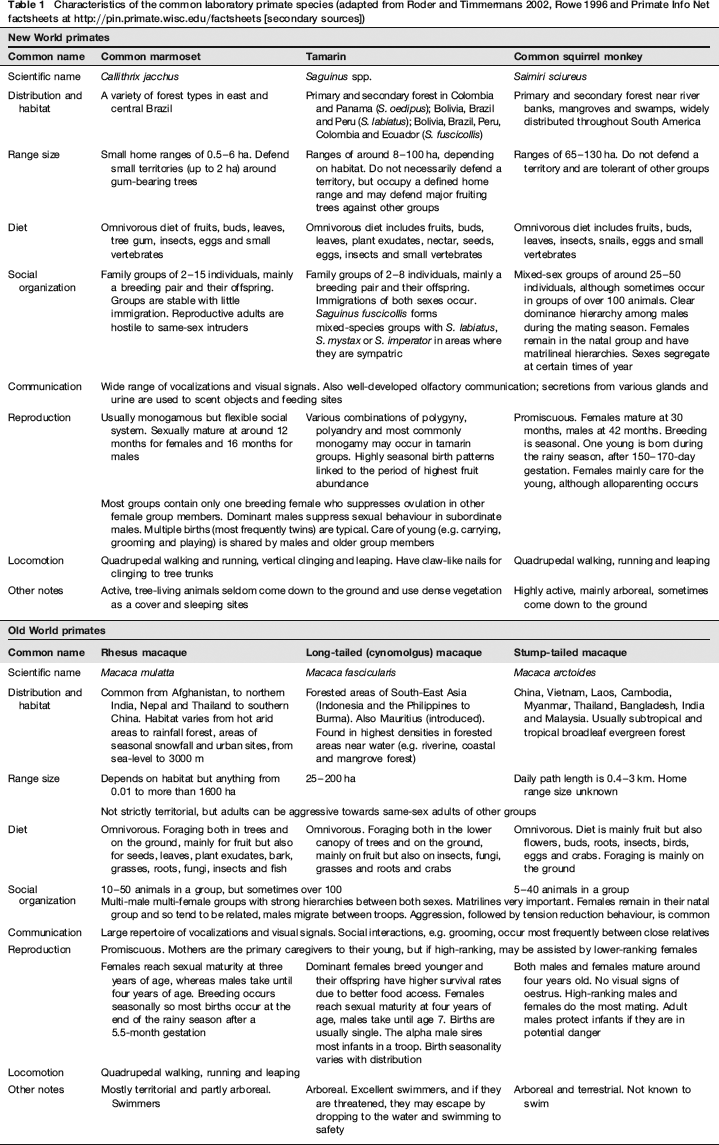
Characteristics of the common laboratory primate species (adapted from Roder and Timmermans 2002, Rowe 1996 and Primate Info Net factsheets at http://pin.primate.wisc.edu/factsheets [secondary sources])

The senses and communication abilities of primates, together with their capacity to experience pain and negative and positive emotions, determine how individual animals perceive and respond to environmental stimuli, including conspecifics and humans. Of all animals, the perceptual world of non-human primates is assumed to be most similar to humans, in particular because of the shared refined visual capabilities. Nonetheless, there are important differences between the sensory capabilities of human and non-human primates, and between genera and some species too.
Understanding these factors is critically important when designing housing and husbandry systems, enrichment strategies, experiments and refinement strategies generally. For example, if a species is highly dependent on its sense of smell and scent marking is an important part of its natural behaviour (as is the case with marmosets), then removing scent marks by frequent cleaning of cages may cause stress. Once this is recognized, cleaning programmes can be designed to minimize any adverse effects on normal behaviour. 2 A summary of key points to consider is given below (see Prescott 2006b for further information).
It is necessary to differentiate between normal (species-typical) behaviour and that which is normally seen, which may be abnormal.
Vision.
Vision is considered to be the dominant sensory modality for primates with the exception of prosimians. Monkeys, apes and humans demonstrate high visual acuity (i.e. ability to distinguish between closely-spaced visual stimuli), surpassed only by large, diurnal raptors, such as eagles (Kirk & Kay 2004). Forward-facing eyes with overlapping visual fields give excellent binocular vision, and together these capabilities enable primates to detect potential predators or harmful situations in their complex three-dimensional forest environment, and to judge depth and distance when moving at speed between trees and branches. They also enable the accurate hand–eye coordination required for, say, capturing fast moving insect prey or manipulating plant material.
Most primates have excellent colour vision that is quantitatively and qualitatively superior to that of all other mammalian groups (Jacobs 1996). Colour vision is important for selecting ripe fruits from unripe and semi-ripe ones, and may also be advantageous for the detection of insect prey and predators (Regan et al. 2001, Buchanan-Smith 2005). It is also important for communication between primates; for example, male and female rhesus macaques show reddening of the facial and anogenital skin during the mating season (Waitt et al. 2003).
Old World monkeys and apes, like humans, have trichromatic vision (based on three classes of cone receptors). Most diurnal New World monkeys, including marmosets, tamarins, squirrel monkeys and capuchins, can have either trichromatic or dichromatic vision (based on two classes of cone receptors, like humans with red–green ‘colour blindness’) (Jacobs et al. 1993). Within each species, some females are trichromatic but other females and all males are dichromatic. Yellows, greens, browns and reds may look alike to individuals with dichromatic vision and it is important to bear this in mind when designing behavioural experiments and choosing colour cues for positive reinforcement training.
Smell and taste.
Both Old and New World primate species have a well-developed sense of smell (Laska et al. 2000). This is important, not only for food identification and selection, but also in social behaviours. Prosimians and New World monkeys possess odour-producing skin glands and demonstrate conspicuous marking behaviours, while many species use olfactory cues to obtain social information such as individual identity, rank and reproductive status (Epple et al. 1993). Scent marking has a range of other functions including territorial defence, preparing males to help deliver and care for infants, reproductive suppression of subordinate females and advertising individual ‘quality’ for mate attraction.
Because of their role in modulating reproductive physiology and regulating social interactions, it is important that familiar scents are not totally removed from the captive environments of marmosets and tamarins during cleaning. Alternate cleaning and sanitation of enclosures and enrichment devices help to retain scent and has beneficial effects on the psychological wellbeing of the animals by reducing overstimulated scent marking. A small amount of their own used litter carried to a new enclosure for primates can help to settle them in more quickly.
Taste is important for choosing appropriate foods. Most primates prefer sweet, sugary foods and avoid bitter or astringent tastes (Hladik et al. 2002). This is an adaptation to ensure that food is palatable enabling, for example, fruit to be selected at the right stage of ripeness, since appearance and touch are not always reliable indicators.
Hearing.
Primates have good low-frequency hearing (below 125 Hz) and can hear higher frequency sounds than humans (Heffner 2004). At 60 dB sound pressure level (SPL), the typical noise level in which people live, the highest audible frequency for a human is 20 kHz, whereas it is 30 kHz for the common marmoset and around 40 kHz for the rhesus macaque (Pfingst et al. 1978, Clough 1982). Laboratory sounds at ultrasonic frequencies could therefore present welfare problems (Clough 1982) and analysis of noise levels in the laboratory should include ultrasonic frequencies.
Vocalization.
Vocalizations are an important mode of communication for most primate species, especially where visual contact is precluded. Repertoires of vocalizations are relatively distinct between species and consist of a wide array of acoustic signals that can be defined by their frequency, intensity, spectral composition and duration. Examples of sounds produced by primates include the high-pitched, birdlike whirrs, chirps and twitters of marmosets and tamarins (Snowdon & Soini 1988, Stevenson & Rylands 1988) and the grunts, barks, coos, geckers and screams of macaques (Hauser 1991, Wheatley 1999). Vocalizations of various primate species, including cotton-top tamarins and rhesus macaques, can be listened to on the Primate Info Net website (http://pin.primate.wisc.edu/av/vocals/).
Some of the functions of vocalizations in primates are to attract the attention of group members and to maintain a certain level of awareness among the group. Both New and Old World monkeys produce contact calls, allowing individuals to keep track of the general whereabouts of other group members. This helps to maintain intragroup cohesiveness and permit cooperative ventures, such as vigilance or transferring an infant (Masataka 1989, Caine & Stevens 1990). Infants of many species produce isolation calls after becoming separated from their caregivers (e.g. isolation ‘phee’ in marmosets, ‘peep’ in squirrel monkeys and ‘whoo’ in macaques). Calling reflects the infant's emotional state, and attracts caregivers and induces them to retrieve the caller.
Many primates also produce calls, which are louder in amplitude and longer in duration than those used in resting contact. These calls are thought to have a variety of functions depending on the species, including territorial defence, to reunite separated group members and to attract mates (Miller & Ghazanfar 2002, Wich & Nunn 2002). Primates can also make non-vocal sounds, such as cage banging, to express their emotions.
Primates can readily distinguish between quiet, calm and loud, forceful human voice tones and words, and this can be useful for training.
Touch and grooming.
Primates have numerous kinds of sensory receptors in their epithelial and connective tissues that are responsive to sensations such as touch, heat, cold, pressure and pain. They make behavioural choices based on these sensations. For example, marmosets prefer to use wooden and plastic nest boxes as opposed to metal ones and this may be related to comfort and temperature (Rumble et al. 2005). They will respond to soft materials (e.g. fleece) by rubbing their bodies against them.
Tactile contact is very important for primates, especially early in life (Seay et al. 1962, Suomi 1986). Infants cling to their mothers, and many species huddle together to rest as adults, which probably helps to promote social cohesion as well as reduce heat loss. Grooming is a very important affiliative behaviour in primate societies and some species spend up to 20% of each day engaged in this activity (Dunbar 1991). Grooming relationships are extremely valuable in helping primates to cope with the stresses and strains of group life and individual animals will make great efforts to maintain these relationships in the face of other demands on their time. Grooming also plays a role in keeping the hair free from parasites and detritus (Hutchins & Barash 1976).
It is essential that primate housing is designed to facilitate these behaviours and includes adequate wide, flat resting surfaces for all the animals to occupy simultaneously in such a way that they can huddle and groom each other. Changes in grooming behaviour (e.g. decreased self-grooming leading to an unkempt coat, or increased grooming attention from conspecifics leading to hair loss) may be indicative of a welfare problem (Honess et al. 2005b).
Being able to recognize, accurately interpret, and respond appropriately to primate communication signals (both vocalizations and body language) provides a good basis for developing many different aspects of refinement. It helps in understanding primate behaviour and interactions between individuals and groups. It is essential for the proper assessment of welfare states, in the recognition and reduction of any pain or distress, and for facilitating appropriate staff-primate interactions. The easiest signals to recognize are visual signals (e.g. body postures, facial expressions) and vocalizations. These provide information on a primate's emotional and physical state, relative social status within the group, and the animal's likely response to, and ability to cope with, any given situation or interaction.
Primate facial expressions, body language and behaviours can appear to be similar to those of humans, but they may have very different meanings and must be interpreted correctly and not in an anthropomorphic or anthropocentric way. For some signals and behaviours the meaning should be obvious. An animal that screams, is physically resistant, urinates or passes faeces during a procedure is clearly frightened, and the procedure should not be continued unless and until the animal has been given additional habituation, or more specific training, to minimize the fear responses. Other behaviours are more difficult to interpret. For example, one of the most common expressions seen among primates is the ‘fear grimace’ or ‘fear grin’, in which the mouth is open and lips retracted. The facial expression is superficially similar to a human smile, and can be misinterpreted as conveying happiness. In fact, it is a reaction given by a tense or frightened animal to appease more dominant or aggressive individuals (including humans) and diffuse tension. Another common source of confusion is the human ‘stare’. A person may stare at an animal out of interest, but most primates will interpret this as an aggressive threat and respond accordingly. Similarly, facing an animal square on can be taken as a threatening posture. This ‘mis-communication’ is a common problem when people who have no experience of interacting with primates visit animal facilities.
Primate communication signals are context-specific, and not just simple, predictable responses. Therefore, it is not possible to attach one rigid meaning to each signal, and inappropriate to adopt inflexible responses to signals that are given. For example, allogrooming can reflect several different states. It may indicate that animals are affiliative and relaxed (e.g. Stevenson & Poole 1976). Alternatively, since allogrooming is known to increase at high population densities and following conflict in some species, it may indicate that aggression has occurred and be linked to tension release (e.g. Schino et al. 1988, de Waal 1989, 2000). Allogrooming may also be used to compete for social contact with, for instance, high-ranking individuals, which may increase tension (Seyfarth 1977). The posture of the animals in each situation can help to interpret the context. It is therefore important not to underestimate the knowledge and empathy that is required to communicate well with primates. The advice of a primate behaviour specialist, perhaps as an integral part of staff-training, is invaluable in helping staff and animals to interact positively (Box 2).
Role of a primate behaviour specialist
Role of a primate behaviour specialist
Primate behaviour specialists can advise on the following:
species-typical behavioural needs behaviour–physiology interactions enclosure design environmental enrichment natural/normal behaviour (e.g. ethogram, behavioural time budgets, communication signals, social interactions within the enclosure environment, foraging) abnormal behaviour and behaviour indicative of poor welfare behavioural monitoring techniques controlling aggression socialization, habituation and training techniques staff–animal interactions primate welfare publications staff training
Primates are highly intelligent, sentient and social animals with a complex range of emotional as well as physical needs. There is no question that they have the capacity to experience pain, and unless the contrary is established, it should be assumed that procedures that cause pain and distress in humans are likely to cause pain or distress in other primates (see Organisation for Economic Co-operation and Development 2001, Soulsby & Morton 2001, National Institutes of Health 2002). It is also generally accepted that they experience a range of negative emotions (e.g. anxiety, fear, frustration, boredom, mental distress and, in some species, grief) as well as a range of positive emotions (e.g. interest, pleasure, happiness, excitement). Primate welfare therefore encompasses the state of both physical health and psychological wellbeing. Knowledge of the ‘normal state of good health and psychological wellbeing’ for the species and the individual, taking both aspects into account, is a prerequisite for the recognition of animals whose welfare is compromised.
The different types of measure that can be used to evaluate animal welfare are set out below (see also the Zoos Forum Handbook [http://www.defra.gov.uk/wildlife-countryside/gwd/zoosforum/handbook/index.htm]). Some of these measures are influenced similarly by both positive and negative events. For example, heart rate can increase either through positive excitement or through fear. The best estimate of an individual primate's true welfare state will therefore be obtained using a combination of measures (Novak & Suomi 1988) including:
Information from routine health management, including coat and body condition as well as physical values such as longevity, growth rate, susceptibility to disease, reproduction and infant care, individual nutritional requirements and wound healing: These are fairly basic measures and will need to be complemented at least by those in (ii) below. Indicators of welfare from postmortem examination (e.g. occurrence of enteric pathology), and from regular review of clinical and postmortem records, can also help to identify trends or case clusters that otherwise may not be apparent. Information requiring structured behavioural assessment, for example, documentation of the behavioural repertoire and activity budgets (including grooming, sleeping, play, social and aggressive behaviours, facial expressions and vocalizations): Other than overt physical clinical signs, ‘behaviour’ is the most readily accessible and commonly used index for assessing an individual animal's welfare, but perhaps the easiest to misinterpret without contextual and specialist knowledge. Information requiring clinical measurement, such as heart rate, blood pressure, body temperature, serum levels of various stress hormones, such as cortisol and immunological functions, including rates of lymphocyte proliferation or suppression of lymphocyte activity: Measurement of many of these parameters requires invasive monitoring techniques that will need careful justification if carried out solely for monitoring purposes. However, where instrumentation, such as a heart rate transmitter, has already been implanted for experimental purposes, data that can help to assess welfare can be obtained with no additional adverse effects for the animal.
Ways of assessing the physical health of primates, including how to observe and approach the animals, are described by Honess and Finnemore (2005), Wolfensohn and Honess (2005) and Honess et al. (2005a,b) (see also National Research Council 1998, 2003, Wolfensohn & Lloyd 2003). Additional information on clinical examination for primates, and the type of measures in (i) and (iii) above, is available in the specialized veterinary literature; so these issues are not covered in detail in this report (see Owen 1992, Taylor-Bennet et al. 1995, 1998, Hrapkiewicz et al. 1998, Federation of European Laboratory Animal Science Associations 1999, Fortman et al. 2002, Ludlage & Mansfield 2003, Steinmetz et al. 2006).
All adverse effects, not just presenting signs, must be taken into consideration when making an assessment of the effect of a procedure and the degree of pain, distress or lasting harm, which may be experienced by an animal. Many early indications of ill health are masked successfully in primate species, especially in the presence of humans. Keen observation of the animal as well as evaluation of daily or weekly body weights, as appropriate, is essential for early diagnosis of mild to moderate pain or distress. Initially, it can be difficult to define what the problem is and whether the cause is related to the environment or pain or distress. Nevertheless, in order to provide the correct treatment, it is important to try to understand and record what is wrong with the animal, even if this does not seem to be objective or quantifiable. Pain tolerance varies widely between individual primates, so each animal should be individually monitored with respect to the need to administer analgesics. For information on techniques for pain assessment, including downloadable score sheets that can be adapted to specific procedures, see Hawkins (2002) and Morton (2000).
Humane endpoints must be set and documented before the start of an experiment, in consultation with the veterinary and animal care staff, and kept under constant review with the aim of further reducing the level of suffering. A person needs to be identified who will be ultimately responsible for decisions on when to humanely kill animals. In some countries this person is defined by legislation and is usually the veterinarian or researcher. All relevant staff members need to be aware of the legal requirements and who the responsible person is for the projects they are involved in. However, all members of the research team bear a moral responsibility for ensuring that humane end-points are not exceeded and that animals are euthanized at an appropriate time.
Reporting levels of suffering retrospectively, for example within the ERP or IACUC, can act as a driving force in the further development of refinement and the implementation of refined techniques in subsequent studies. Sharing information on how pain and distress were recognized, avoided, prevented or alleviated in an experiment can help others use similar techniques and promulgates good welfare and good science (Morton 1992). Such information should always be included when publishing research results.
In order to use behaviour as an indicator of animal welfare, normal behavioural patterns for the species and the individual need to be defined first as a reference point. Animals can then be monitored for any changes in behaviour. However, it is difficult to describe what is normal behaviour for a primate in a laboratory situation, partly because behaviour is context-specific, but also because relatively few clear behavioural indicators of good welfare in the laboratory have been defined and evaluated. Generally, primates in a good laboratory environment will appear relaxed, even if there are social tensions related to bids for dominance. They will spend much time grooming, in body contact or foraging, and the group will not appear tense or nervous.
In addition to the relevant literature, a useful baseline for understanding the normal range of behaviours of a species can be obtained by observing video footage of the species in its natural environment (see Berdoy 2002 for how this has been done for rats). There are many televised wildlife documentaries that can be used as training materials in this respect. A useful DVD teaching resource specifically for primates in a laboratory situation is produced by Wolfensohn and Finnemore (2006) (see Section 2.6). Such materials will help show how the animals interact with each other and their environment, for example through grooming and play, agonistic interactions, foraging and exploration. The Zoos Forum Handbook also has a useful chapter on behavioural assessment of welfare.
Indicators of abnormal behaviour.
Abnormal behaviours can be qualitative (those that occur in captivity but not in natural settings) or quantitative (those that occur more or less often in captivity than in nature) (Erwin & Deni 1979). Behavioural changes that may indicate poor welfare resulting from the present (or past) environment and/or experiences of the animal include:
a restricted behavioural repertoire; an abnormal time budget; inappropriate social behaviour; other abnormal behavioural patterns.
Examples of behaviours in each category are given in Table 2.
Behaviours that may indicate poor welfare in macaques, marmosets and tamarins (NB not all behaviours are applicable to all species)

Stereotypy: the performance of unusual motor acts, repeatedly and often invariably, which serve no apparent purpose; often indicative of an inadequate or inappropriate environment
Some of the behaviours in Table 2 would not be seen in the wild (e.g. stereotypies). Some, such as grooming or scratching and picking at scabs, are natural behaviours that are only a cause for concern when they become excessive. The difficulty in the latter case is defining the level at which a particular deviation from normal becomes a concern. It is also important to recognize that doing nothing (inactivity) may also be an abnormal behaviour.
Although there are well-established scientific methods for measuring animal behaviour (see Martin & Bateson 1993 for the basic principles), there is inevitably a subjective element to recognizing and classifying behaviour as indicative of either good or poor welfare. It is important to try to ensure that behavioural observations and the interpretation of these are reliable and reproducible between observers. All staff should understand the need for close monitoring of animals for detection of abnormal behaviours, know the kinds of behaviours to look for, recognize that behavioural changes may be subtle, and be prepared to seek expert advice if they have any concerns. Animals are likely to modify their behaviour in the presence of humans, particularly if they are not habituated and socialized to humans, so it can be helpful to assess unprovoked behaviour through remote monitoring using closed-circuit television (CCTV) or one-way viewing panels, and then review the behaviour when the animal is first approached and handled by a familiar person.
Abnormal behaviour should not be confused with the normal reactions of animals to experiences they find painful or frightening. For example, an animal unused to handling is likely to be frightened of someone reaching into a cage and of being touched, and may respond by biting and struggling. This behaviour cannot be classed as abnormal. Nevertheless, it should be recognized as undesirable, for both the animal and the handler, and prevented by appropriate habituation, socialization and training of both primate and human handler (see Sections 2.5 and 2.6).
Causes and prevention of abnormal behaviour.
All species of primates used in the laboratory are social animals that live in family groups or troops in very complex environments. They require a varied and stimulating physical and social environment for their psychological wellbeing, and therefore it is not surprising that the absence of these factors can influence the development of abnormal behaviours. Social deprivation, in particular, is strongly linked to abnormal behaviour (Harlow & Harlow 1962), as is an impoverished rearing history, including being nursery-reared rather than reared by the mother (Kraemer & Clarke 1990, Lutz et al. 2003). For these reasons, the early weaning practices at some breeding centres are thought to present a welfare problem (see Section 8).
Housing primates individually is widely accepted as a major stressor and seems to be a particularly influential factor in the development of abnormal behaviours. For example, in macaques, the likelihood of such behaviour increases the longer they are singly-housed and is also influenced by the age at which individuals are removed from their social groups and housed singly (Lutz et al. 2003).
Repeated stresses during the juvenile period (e.g. from veterinary procedures) have been implicated in the development of self-injurious and stereotypical behaviour in rhesus macaques (Lutz et al. 2003, Novak 2003). Rigid scheduling of certain animal care events, such as feeding, has also been shown to influence the expression of stereotyped behaviour in a variety of primate species (Krishnamurthy 1994, Bloomsmith & Lambert 1995).
Some species appear to be easier than others to keep in a state of good welfare in laboratory conditions. For example, healthy common marmosets kept group-housed in good laboratory conditions seldom behave abnormally. In a survey of UK establishments conducting regulatory toxicology studies, common marmosets were reported to show no biting or self-inflicted trauma and a very low incidence of circling (0.3–1%) and weaving (1%). When weaving did occur it was associated with single housing in small cages (Hubrecht 1995). There are also differences between males and females in some species; male rhesus macaques in inappropriate housing are more likely to manifest severe behavioural disorders than females (Sackett 1974, Lutz et al. 2003).
Temperament also affects an animal's ability to cope in the laboratory environment (Bowers et al. 1998), and since temperament is heritable, care should be taken not to breed from animals showing marked and/or persistent abnormal behaviour or stereotypies.
In summary, providing primates with a stable social environment in which to develop and live is the single most important factor in avoiding the development of abnormal behaviours (Table 3). Opportunities for tactile social contact with conspecifics should be maximized, especially when animals are young (Crockett 2004). Weaning animals at a suitable age and into appropriate social environments is also important (Goosen 1989, Timmermans 1997, Bellanca & Crockett 2002) and users need to ask breeders and suppliers to wean and group accordingly. Minimum weaning ages in the revised Council of Europe Appendix A to Convention ETS 123 are: eight months for marmosets and tamarins destined for research, and 12 months for breeding animals; six months for squirrel monkeys; and eight months, preferably 12 months, for macaques. The International Primatological Society (2007) similarly recommends that infants should remain with their mothers for 12–18 months in most species. Group dynamics will change with time (e.g. as young males mature in family groups and begin to challenge dominant males), so changes to group structures may need to be made. Good husbandry and care that provides appropriate stimulation, opportunities for exercise and species-specific behaviour throughout life is also important (see Section 3), as is careful monitoring to ensure that all individuals are coping with their environment and have a good level of welfare.
Strategies for avoiding the development of abnormal behaviours

Addressing behavioural abnormalities.
If behavioural abnormalities develop, the underlying cause should be identified immediately by examining housing and husbandry practices, experimental procedures and their outcomes, and all human–primate and primate–primate interactions, modifying these as required using the recommendations throughout this report. For example, positive reinforcement training techniques have been used successfully to reduce the incidence and severity of abnormal behaviour (Cox 1987, Laule 1993, Schapiro 2002). Breeding, stock or experimental animals persistently showing abnormal behaviours that adversely affect their wellbeing should never be kept in the laboratory long-term.
Interacting sympathetically with primates in a manner that is meaningful to them, as described in Section 2.2, will have a positive impact on their welfare and consequently on the quality of science. However, primates are not domesticated animals and contact with humans has the potential to be stressful, especially where they cannot control that contact. This is a particular problem in the laboratory situation (Scientific Committee on Animal Health and Welfare 2002).
Habituating 3 and socializing 4 captive primates to the presence (sight, sound, smell) and behaviour of humans as early as possible in their lives, while still in their family group, is recommended to avoid or reduce fear responses to novel humans or situations encountered later in life. Early habituation to humans can also facilitate handling and restraint (see Section 5.1) and training of animals (see Section 2.5), and may reduce the need for sedation and some personal protective equipment when carrying out procedures (e.g. Heath 1989, Reinhardt et al. 1995a, McKinley et al. 2003). It also allows staff members to observe behaviour patterns that are relatively unaffected by their own presence, which will help them to assess the welfare of the animals more effectively (Laboratory Animal Science Association/Medical Research Council 2004).
Habituation: The waning of a response as a result of repeated stimulation, but not fatigue. This kind of learning is of importance in familiarizing an animal with aspects of the environment to which it is not expected to react. It is of value in the training of primates to ignore non-threatening stimuli (e.g. the sound of clippers, restraint in a sling, confinement in a transport container).
Socialization: The process by which primates learn how to successfully interact with members of their own and other species (e.g. humans).
Useful strategies for habituating and socializing primates to humans are listed in Table 4. The key to implementing these effectively is a formal habituation and socialization programme, based on the principles of positive reinforcement and involving all staff who come into contact with the animals (see Section 2.5; Rennie & Buchanan-Smith 2006c, Prescott & Buchanan-Smith 2007). This needs to be continuous throughout the animal's life providing an ongoing positive human–animal relationship.
Strategies for habituating and socializing primates to humans

All these will be dependent upon local conditions, the nature of the study and the health status of the animals
Training 5 primates to cooperate with scientific, veterinary and husbandry procedures has significant benefits for animal welfare, science and staff, especially when combined with appropriate socialization, habituation and desensitization 6 (Prescott & Buchanan-Smith 2003, 2007). It is recommended as good practice in many legislative and professional guidelines (Home Office 1989, National Research Council 1998, National Centre for the Replacement, Refinement and Reduction of Animals in Research 2006, International Primatological Society 2007). The concept is not new, but is being successfully developed in several countries, notably the UK and the USA (Iliff et al. 2004). Greater effort is also being made to formally evaluate the benefits to animal welfare and quality of science, the costs in terms of time investment and any unwanted side-effects of the training process. The effectiveness and benefits of specific training techniques are also being explored (Bassett et al. 2003, McKinley et al. 2003, Schapiro et al. 2003, Smith et al. 2004). This type of training is additional to that required for primates to carry out specific tasks in some areas of neuroscience and behavioural research.
Training: The shaping of the behaviour of a primate so that it actively responds in a way that is desired by the trainer (e.g. offers a limb for injection, stands on a weighing scale, waits and allows subordinate individuals to feed uninterrupted).
Desensitization: Systematically pairing positive reward directly with an uncomfortable or aversive experience or stimulus in order to reduce any associated fear or anxiety response.
Recommendations on training techniques and on training staff to use these are discussed in Rennie and Buchanan-Smith (2006b) and Prescott and Buchanan-Smith (2007). Resources for developing and implementing primate training programmes are given in Prescott et al. (2005), including a tabulated literature review and a detailed sample training protocol. The key points are summarized below.
Training primates to cooperate with husbandry, veterinary and scientific procedures as a less-stressful alternative to traditional methods, is a potential refinement for all establishments, even when animals are only kept for a short period. Opportunities for integrating training into existing animal management practices should be continually reviewed and training opportunities should always be considered when planning any research project. Training methods should be based on positive reinforcement techniques,
7
which reward desired behaviour, since this method of training is considered to be the most humane (Laule 1999, Pryor 2002, Laule et al. 2003). If negative reinforcement
8
must be used (for example, showing animals a net), it should be used in combination with positive reinforcement (McKinley 2004).
Positive reinforcement: The process whereby the presentation of a pleasant (rewarding) stimulus (e.g. food treat, verbal praise, tactile contact) increases the probability of a behaviour recurring in similar circumstances in the future.
Negative reinforcement: Frequency of a behaviour is increased because something negative (e.g. cage squeeze-back mechanism) is removed in its performance.
Training is easiest when primates are well-socialized and habituated to humans (Scott 1991, Bassett et al. 2003, McKinley et al. 2003), and so should be implemented in conjunction with a formal socialization and habituation programme. As a precursor to training, all animals should be socialized to humans and habituated to accept food treats from the hands of staff.
Primate training programmes should be planned carefully and well-documented. Advice on application of learning processes should be sought from a professional animal trainer or primatologist with expertise in primate training. All staff who work with the animals should receive basic training in positive reinforcement techniques in order to ensure that all of their actions make a positive and consistent contribution towards the training programme.
Appropriate training for all those working with primates is essential in order to ensure compliance with legislation, good animal welfare, full implementation of the 3Rs of Russell and Burch (1959) and good quality science. Special knowledge and practical skills are required in order to be able to competently address the complex behavioural, social and psychological needs of primates and the ethical considerations involved in their use. In some countries, primate-specific training courses have been developed to deliver such knowledge and skills. This enables people to feel confident that they can care for and interact with primates positively, predict, identify and reduce any pain and distress the animals experience, and improve primate welfare. Training is important for all staff, from those who care for the animals, through to those who design experimental studies, but the content, nature and level of training needs to be appropriate for each role.
The courses currently available vary in syllabus content and delivery and there is no commonly agreed approach or list of learning outcomes. A summary list of learning outcomes for primate-specific training recommended by the Working Group is given in Box 3. More detailed learning outcomes for training in laboratory animal care and use have been described by the UK Animal Procedures Committee (2006b) and the Federation of European Laboratory Animal Science Associations (FELASA 2001). The list may need to be tailored to local requirements depending on the nature of the establishment (e.g. whether breeding or experimental), the nature of the work carried out, and the role of individual trainees. A useful DVD-video ‘Refinements in Primate Husbandry’ is available for use in training programmes within establishments. This learning resource acts as a foundation to promote a better understanding of primate welfare (Wolfensohn & Finnemore 2006).
Learning outcomes for primate-specific staff training
Learning outcomes for primate-specific staff training
Be able to identify, understand and respond appropriately to the particular ethical and welfare issues raised by the use of primates in scientific procedures. In particular to:
recognize and respect that there are different views within society on the acceptability of using animals in scientific procedures and that the use of primates causes particular concern for many people; understand the basis for these concerns; identify, assess and minimize all of the harms to primates throughout their lifetime (including adverse effects resulting from breeding, sourcing, transport, housing, husbandry, handling, procedures and their effects and euthanasia). Understand the nature of non-human primates, in particular to:
understand the basic principles of caring for primates, including the need to enhance psychological wellbeing; be familiar with the biology, behaviour and ecology of the relevant species; understand the needs of the species and the potential for suffering if these are compromised; recognize and interpret the normal and abnormal behaviours of relevant species (including communication signals) and understand how these are affected by laboratory housing and procedures; interact with the relevant species in a positive way. Be able to apply this knowledge to reduce suffering and improve welfare, and in particular to:
know the clinical and behavioural signs to look for when monitoring health and welfare; be able to develop and provide effective environmental enrichment; appreciate the benefits of training primates to cooperate with scientific procedures and husbandry routines, and know how to use recommended positive reinforcement techniques; know the most up-to-date methods of handling and restraint and be able to perform these sensitively and proficiently; understand the importance of ongoing application of all 3Rs and know where to go for advice; understand the importance of humane endpoints and the need to constantly refine these; devote time and resources to develop and maintain relevant practical skills. Understand the specific human health and safety requirements for working with primates in particular to:
be aware of relevant zoonotic diseases and appropriate use of personal protective equipment; be aware of the facility's policies for dealing with potential exposure to pathogens, and of those responsible for carrying out actions; be aware of personal actions required if potentially exposed to pathogens, for example, how to report injuries and to whom; demonstrate understanding after completion of training.
It is important to recognize that a short-taught course only provides an introduction to the issues and cannot ensure competency. Staff will need to work under supervision until they achieve a sufficient level of competency. This enables the course objectives to be further developed and is an essential part of the training process. Opportunities for continuous professional development should also be available and supported, to maximize implementation of the 3Rs. For example, changes in primate husbandry from cage systems to group housing in large enclosures, evaluation of environmental enrichment methods and the training of animals to cooperate with husbandry and procedures, all have implications for staff training. The annual NC3Rs Primate Welfare Meeting is one such opportunity for learning and exchange of information on primate welfare and refinement (see www.nc3rs.org.uk/primatewelfaremeeting for further information). In addition to the scientific programme, it plays an important role in bringing together researchers, veterinarians, animal care staff and facility managers to share their experiences with primate care and use and refinement generally. Such initiatives should be further encouraged.
Staff selection, communication, and health and safety are also important issues relevant to subsequent training needs, and advice on these matters is available from Wolfensohn and Honess (2005) and Rennie and Buchanan-Smith (2006b).
3 Refinements in housing and husbandry
Understanding the animals' natural history, sensory world, social structure and physical and behavioural characteristics and needs, is an essential prerequisite to the design of good primate housing and husbandry. Primate species are adapted both physically and behaviourally to their natural habitats; for example, they may be largely arboreal (e.g. cynomolgus macaques, marmosets, tamarins) or terrestrial (e.g. bonnet macaques and baboons) and it is important to consider this when designing housing systems. The degree of territoriality, styles of locomotion (e.g. vertical clinging and leaping, walking, running), sleeping and nesting behaviour (e.g. using tree holes, building nests), and response to predators and climatic conditions are all factors to be taken into account. For example, most arboreal species flee upwards to escape terrestrial predators; and in the laboratory, most primates will flee upwards if they are alarmed when a human approaches. Keeping these animals in small, conventional tiered cages frustrates this behaviour for individuals in the lower tier, and is unacceptable in animal welfare terms.
Much has been written about primate husbandry, and guidance on designing good housing, husbandry and care is provided in the references in Box 1. The underlying principle is to provide safe, comfortable and hygienic housing and husbandry, which allows the animals to perform a wide range of species-typical behaviour and exercise a degree of choice and control in their environment. For example, they should be able to choose to socialize with, or avoid, group mates, or to be seen or not seen by neighbouring primates (Poole 1999, International Primatological Society 2007). Other factors that have to be taken into account include ease of cleaning, capture and monitoring of the animals.
The components of a good primate housing system are shown in Table 5. Most of them are applicable to all species, although the detail of how to interpret and apply each component may vary. Advice from a specialist in primate behaviour and welfare will help to interpret the requirements for individual species. It is also necessary to take into account the procedures that animals undergo and any requirements imposed by them. For example, surgically-prepared animals may need an area in the home enclosure where they can recover from anaesthesia yet retain visual and olfactory contact with the rest of the social group; enrichment for animals with motor impairment needs to take into account the animals' reduced physical capabilities. The enclosure design team should therefore include, and take account of, the views of scientific and animal care staff and others with practical expertise of using the facilities.
Components of a good primate housing system

Components of a good primate housing system

Good housing that provides much of the above is now routinely provided in some establishments. However, many laboratory primates are still housed in small, barren cages that bear little relation to the complex three-dimensional environments that they inhabit in the wild, and which neither allow performance of a range of species-typical behaviours, nor provide space for adequate socialization, enrichment and opportunity to exert choice and control. This affects both the physical and behavioural health of the animals, which in turn can influence the quality of research data (Reinhardt 2004).
Thus, for both animal welfare and scientific reasons, the Working Group considers that housing primates in small, barren cages is unacceptable, and that animals should be provided with more space and more complex environments. Table 5 provides a reference point for the design of any new facility, but the same principles can also be applied to adapting existing accommodation. Additional practical suggestions on how to improve both the physical and social environment and challenge perceived constraints are set out in Sections 4 and 8.3.
The amount of space provided for housing primates is of paramount importance. This dictates not only the animals' ability to perform species-typical behaviours, but also the size of social group possible and the space available for environmental enrichment. Restricted areas limit both the types of locomotion the animals are able to perform and the amount of physical exercise possible. This can lead to atrophy of the muscles and decreased joint mobility (Faucheux et al. 1978, Turnquist 1985). The prevalence of stereotypies and other abnormal behaviours may also be higher in small enclosures than in relatively larger, more complex ones (Kitchin & Martin 1996). Housing in small enclosures may also reduce breeding success (Boot et al. 1985, Snowdon 1989).
Ways of increasing the space provided beyond the minimum specified in legal texts should always be explored. This will enable additional cage furniture and other enrichment devices to be added, increasing the complexity of the environment, stimulating activity and interest, and enabling animals to be paired or grouped. Criteria for determining space requirements should not just relate to floor area and enclosure height per body weight of animal. The amount of space required depends on a number of factors, including species-typical behaviour and body conformation, age, reproductive status, group size and compatibility, and the activities being performed in the enclosure (Buchanan-Smith et al. 2004, Prescott & Buchanan-Smith 2004). For example, active juvenile macaques of 18 months old are likely to need more space than relatively inactive mature animals, to allow the play behaviours that affect their development.
The quality of space is also important, since even large enclosures can be of limited value to the animals if they are unable to make good use of the space available (Paulk et al. 1977). Thus, enclosure design needs to be related to the natural habitat, social structure and behaviour of the species, with provision of suitable areas for climbing, resting, vertical flight, refuge, foraging and exercise.
Extending living space in standard caging
There are several ways to increase space in traditional style caging that will work for all species and some examples are given below.
Incorporating cage extensions.
A variety of cage extensions that are relatively easy to provide are available.
Tunnels made from industrial tubing or mesh can be attached to cage doors to link adjacent or neighbouring cages of small primate species vertically or horizontally. Verandas (mesh boxes) and bow fronts, with or without solid flooring, can be attached to allow animals greater lateral visibility and can be used for foraging provision. These are already established as workable for marmoset caging and are being developed for macaques (e.g. Frost 2004). The behaviour of animals needs to be monitored because there may be occasional aggression between adjacent groups. However, if verandas are not positioned too closely, and/or visual barriers provided, then this should not be a problem. Roof extensions to marmoset caging can provide animals with all-round views of the animal room.
Converting individual units to gang caging.
Many establishments have successfully converted standard metal cages to linked gang-housing simply by removing the partitions between adjacent and tiered units. A flexible design of gang cage allows different size groups of animals to be maintained or run together in a larger communal space. This type of caging has been used successfully in regulatory toxicology studies, particularly chronic studies (e.g. Frost 2004). There are some potential disadvantages but these can usually be overcome or avoided. For example, it is not possible to measure individual food and water consumption, but the reliability of this measure is in any case questionable since animals often scatter and waste their rations. It may be more difficult to record individual clinical signs such as vomiting, but behavioural signs are often easier to detect when the animals are expressing a wider range of behaviours because they are in a group. Capture of individuals may be more difficult, although training animals to cooperate can overcome this problem.
In contract research organizations in the UK, the gang-caging system has generally been accepted and approved by client companies, who appreciate the benefits to both animals and science of the improved environment and the extended range of behaviours this allows. Very little concern has been expressed that the gang-caging system could compromise regulatory studies, and no studies carried out in such housing have been rejected by regulatory agencies.
Providing an exercise and play area.
If animals are to be housed in a restricted space for extended periods of time, a practical way of giving them opportunities for natural locomotion and other behaviours is to provide a large and enriched exercise and play enclosure adjacent to the home cage, and to allow the animals free or rotational access to this (e.g. Rudling 2003). Animals need to be monitored to ensure that they use the space provided, and do not remain in one part of the enclosure. If this happens, then changes to the enclosure or the home cage may need to be made.
Adapting the cage surfaces or structure.
Fitting stainless steel mesh with a grid size of 1 cm2 on the solid sides and roof of marmoset cages can greatly increase opportunities for vertical clinging and leaping, and provides anchorage points for enclosure furniture, including feeding platforms, nesting boxes and toys. The gap between the mesh and solid side should be sufficient for the animals to be able to grip without trapping fingers or toes.
Replacing solid walls and ceilings with steel bars, or fitting these to existing walls or ceilings, is a similar option for macaques, enabling the animals to climb, swing and jump from one area to another. Mesh and bars also assist ventilation in the enclosure and allow increased diffusion of light. Providing easy access to the floor area and multiple routes up from the floor encourages more extended foraging.
Adding a solid floor.
Grid floors are uncomfortable for the animals and seriously limit provision of substrate, so they are not recommended. The insertion of a solid floor (by welding this into mobile units or sealing the floor of a room) enables deep litter to be provided (for example using soft wood shavings) or the floor can be left bare with foraging pits. Either option is better for the animals than grid flooring. If a full solid floor is not possible (e.g. in a metabolism cage), a solid resting area should be provided.
Including cage furniture.
If there is room, a wide selection of fixed, suspended and hanging objects give the animals items to move on and use (including to huddle and groom), and thus help provide the stimulation they need. Platforms and perches are used by all species and can be accommodated in most sizes of cage. Different species use different sizes and orientations of perching or clinging substrates. For example, marmosets and tamarins perform some vertical clinging and leaping as well as quadrupedal walking and running (Buchanan-Smith et al. 2000), so care should be taken to provide substrates with orientation and diameter that allow natural locomotor activity. Shelves and perches with a camber allow urine to run off, thereby helping to keep the animals' coats clean and avoid urine burn. In large gang cages, wood logs can be fixed at different heights to allow greater aerial movement.
One of the many useful items that can be used for marmoset cage enrichment is polyvinyl chloride matting, available in 5 m or 10 m rolls that can be cut to size very easily. This can be attached with cable ties at the front of the cage and provides a good shelf for marmosets to rest and play on. It can also be fixed to the mesh roof, shaped into hammocks and tubes. It is warm for the animals to sit in and provides good grip for climbing. Another idea is simply to hang a flat piece at the back of the cage for the animals to climb on and hide behind when playing. It can also be used as a climbing ladder system within tubing linking cages or outside runs.
Specially designed enrichment structures, such as large moulded plastic hexagonal swings and barrels, that can be placed into larger caging or room systems are available commercially. Other items appropriate to the species, such as nest boxes for marmosets and tamarins can also be added.
Primates benefit from material that they can manipulate and destroy (e.g. cardboard boxes and hemp sacks) as this allows them some control over their environment by providing an element that they can change. Novel materials may be perceived to be a problem with respect to ease of cleaning or potential effects on scientific data (e.g. potential induction of liver enzymes by wood). Nevertheless, various materials have been used successfully in regulatory studies.
A risk assessment should be carried out on cage furniture and additions to ensure that they are safe for both animals and staff. Care should be taken in siting cage furniture, for example, swings should be safely secured so that they cannot hit people or animals, or damage the fabric of the enclosure. There should not be any sharp objects or edges on any of the items placed in an enclosure that could injure animals, and all items should be examined on a regular basis to make sure that they remain safe.
Incorporating visual barriers.
Visual barriers between and/or within enclosures allow the occupants the choice between seeing other primates and humans, or hiding from them. This enables them to have greater control over their social interactions, helps minimize intra- and inter-group aggression and promotes successful pairing and grouping (Goosen et al. 1984, McKenzie et al. 1986, Watts & Meder 1996). Examples of visual barriers are floor-mounted or hanging plastic panels, shower curtains, sacking or large wide pipes. They should be carefully positioned so that they do not block light sources. The barrier need not be complete, as primates are thought to perceive that they are hidden even if a large part of their body is visible.
Adapting enclosure materials
Wood.
The inclusion of wood within the cage is beneficial for all species. It is less cold and noisy than metal, provides a softer textured surface and facilitates a variety of species-typical behaviours, for example, it can be manipulated and chewed. Natural branches and/or wooden dowel perches can be included to provide additional usable space. The type of wood must not be toxic to the animals; maple, beech and oak are all safe. If real wood cannot be used, then plastic wood is a good alternative.
Transparent materials.
Primates are highly reactive to visual stimuli and strongly motivated to obtain visual information about their surroundings. Primate rooms should have windows wherever possible to provide natural light and stimulation by increasing the area of vision. Doors and access corridors should also have windows, so that the animals can see when someone is about to enter the room and who that person is, so that they are not startled. Windows in enclosures should have stickers to prevent the animals running into them when first introduced.
Environmental enrichment
It is best to provide enrichment as part of a formal enrichment programme. Records of the enrichment items provided should be maintained, together with a record of how the animals use them and whether there are any problems with any items. Changing the enrichment items can provide novelty, but if individuals use certain items extensively then these should not be removed. Where items are rotated between groups this should be recorded to ensure that all groups of animals have access to them.
A useful searchable database for ideas on environmental enrichment and husbandry refinement for primates is provided by the Animal Welfare Institute, Washington DC (see http://www.awionline.org/lab_animals/biblio/enrich.htm). Some additional ideas are provided below.
A heated area.
Primates like to bask in sunlight, but where it is not possible to include outdoor runs or windows, areas of additional heat/lighting can be provided. In pilot studies by members of the Working Group, infant common marmosets were particularly interested to move to heated areas and family groups would congregate there as well.
Stainless steel mirrors.
Mirrors have been provided for primates in a variety of studies (e.g. Gallup & Suarez 1991, Lambeth & Bloomsmith 1992, Brent & Stone 1996). Responses ranged from playing with the mirror in an apparently welfare-beneficial manner, to reactions similar to those seen during confrontations with other primates (Simpson et al. 1989). Both rhesus and long-tailed macaques will use adjustable mirrors to observe activity in other areas such as adjacent corridors and this expands the animals' visual environment (see www.nc3rs.org.uk/primatehousing).
Water enrichment.
Several species of primate dive and swim to retrieve food (Suzuki 1965, Malik & Southwick 1988, Agoramoorthy et al. 2000) and Japanese macaques (Macaca fuscata) submerge potatoes and grains in water to remove dirt (Itani & Nishimura 1973). Provision of a small mobile swimming pool for short periods of time has been found to be effective enrichment for macaques (Anderson et al. 1992, 1994, Parks & Novak 1993) and is used widely within contract research organizations in the UK. Water containers used successfully in the laboratory for various species include: an iron-framed glass aquarium, Plexiglas aquarium, stainless steel bowl, polypropylene barrel, large rodent cage, and child's sand pit (of injection-moulded plastic), with a depth of water ranging from 10 cm to 50 cm. Some laboratories have custom-made polypropylene pools built to fit within the enclosure space, with a metal pipe for attaching to a hose and a drain on the outside (see www.nc3rs.org.uk/primatehousing).
The animals show high motivation to manipulate the water surface, immerse themselves, dive, swim and play underwater. Advantages of this enrichment technique are that it is based on a natural behavioural inclination, encourages play, provides exercise, keeps animals and their enclosure clean, and can facilitate thermoregulation in hot weather. Disadvantages include the time taken to fill the pools and supervise the animals, and the need to remove substrate on the enclosure floor so that it does not get wet.
Providing a customized room
Recently, some primate units have moved to housing primates of defined health status in customized rooms rather than standard or modified cages. This makes better use of the available room space with much more space for furnishings, enrichment devices and social interaction, and more room for the animal care staff to work. This change has resulted in less aggressive, more cooperative animals, who are less stressed by capture. They are also calmer because other groups of animals are not constantly alongside them in a restricted space and because the animals can retreat to refuges where they can see the boundaries of the room (Wolfensohn 2004). They display more natural behaviours and breeding success is improved.
Husbandry practices need to be adapted to fit this style of primate management. There are specific staff training needs, as staff members must feel confident and relaxed working closely with primates in a free environment. Identification and capture of some animals can be more difficult in very large enclosures, but good habituation and socialization to humans (see Section 2.4) can overcome such problems. Routine room maintenance and animal handling encourages staff–primate interaction, and since the animals can see human activity more easily, they become more familiar with their carers. This has the added advantage of enabling care staff to observe uninterrupted behaviour patterns. The housing remains cleaner for longer, which saves money on cleaning equipment and releases staff to spend more time on activities, such as positive reinforcement training for the animals.
There are additional health and safety issues associated with this style of management, but these too can be overcome. The best option is to use animals of defined health status, for example, Herpesvirus simiae-free (see Sections 8.2–8.4). Rooms need to be safe for both staff and animals with respect to electrical outlets, lights, drain holes and pipes. There needs to be good observation panels and adequate barriers for preventing escape.
The physical environment
Maintaining the health and welfare of laboratory animals, and obtaining reliable scientific data, requires control of the physical environment to provide optimum ranges for environmental factors such as light, temperature and sound. Details of approved laboratory conditions for primates are given in various publications (e.g. Home Office 1989, 1995, Canadian Council on Animal Care 1993, Wolfensohn & Honess 2005, Council of Europe 2006) and so are not repeated here.
A concern with regard to lighting is that ‘on–off’ lighting (i.e. without a dawn/dusk control) can startle animals and cause them to fall and injure themselves if they are leaping when the lights are switched off. Provision of low level night lighting for the changeover from light to dark allows primates better night vision and takes account of their startle reflex.
Anecdotally, playing music through room speakers helps primates become accustomed to noise over time and reduces the startle reflex. However, few studies have investigated music as environmental enrichment for laboratory primates (Brent & Weaver 1996, Harvey et al. 2000, Howell et al. 2003). Where music is played, the volume should be no louder than human conversation to avoid inhibiting vocal communication between animals and it should only be played during limited periods of the day (i.e. not continuously). Audio systems can be beneficial in relaying different sounds to provide auditory complexity and help with habituation to the laboratory environment (Drewsen 1990, Iliff & Wismer 2003).
Improving the social environment
Primate species used in laboratories are highly gregarious animals and single housing for even short periods can be a significant stressor (Rennie & Buchanan-Smith 2006a). Providing social housing, either as groups or pairs, is fundamental to primate welfare. It enables them to carry out social behaviours, such as grooming and social play, which helps to alleviate boredom. It buffers the effects of any stressful situations such as husbandry, veterinary or experimental procedures, and it usually means that the animals have a larger enclosure size, which facilitates enrichment (Reinhardt 1989, Coelho et al. 1991, Gust et al. 1994, Schaffner & Smith 2005). It also makes better use of room space. Single housing should no longer be considered acceptable for primates, with exceptions requiring compelling veterinary or scientific justification. The goal should be to establish harmonious social groups and maintain these throughout the animals' lives.
Since single housing has such serious consequences for primate welfare, it is also likely to impact on experimental outcomes. If an experimental design appears to require single housing, even on a temporary basis (perhaps for a few hours daily, as in toxicology studies postdosing or in behavioural studies), then ways of avoiding this and mitigating its effects should be sought. For example, by questioning whether individual housing is necessary for quantitative measurement of food and/or water consumption, and whether observing the eating/drinking habits of the animals and closely monitoring their body weights might suffice to identify treatment effects that reduce appetite. Alternatively, animals may be housed in pairs and paired data accepted. For cognitive tests, data from animals tested in pairs may also be acceptable, particularly since distress caused by social isolation can seriously interfere with behavioural data. It may also be possible to carry out cognitive tests in the home enclosure, with test individuals temporarily separated from conspecifics by a mesh barrier, or in a separate area that animals can choose to enter (Crofts et al. 1999, Scott et al. 2003). It may also be possible to house animals together where they are on different studies, but this will require good liaison between the research groups concerned.
If single housing is unavoidable, measures must be taken to ameliorate its negative effects. Consultation with the animal care staff and competent persons charged with advisory duties in relation to animal welfare is very important in this regard. A time limit should be defined in each individual case; the duration should be kept to an absolute minimum and animals should be closely monitored and supervised. Additional resources must be targeted to the care and welfare of the animals to compensate for the absence of social companions. They should be given additional human socialization time with a familiar caregiver and, unless contraindicated on veterinary or welfare grounds, tactile contact (e.g. through enclosure sides) with other compatible primates on a daily basis. Individuals can be provided with a fleece grooming/foraging board to address their need for social grooming and with additional physical enrichment (e.g. foraging devices, materials to manipulate, televisions). The adverse welfare impact of single housing should be included in the harm–benefit assessment of individual projects carried out by the regulating authority, local ERP, IACUC or equivalent body.
Establishing and maintaining social groups
Most primate species form hierarchies, both in the wild and captivity. These take time to establish as the animals test their relationships with each other by repeated confrontation over resources such as food and favourite perches. The outcome of these incidents is determined by the physical and mental attributes of the individuals involved and their position and support within the social group. Hierarchies do not remain stable permanently, but can change with circumstances such as when juveniles mature and challenge their position in the hierarchy, or when a group member is removed and the hierarchy is thus disrupted. The best way to establish and maintain social groups therefore depends on many factors, including the normal social structure of the species, the age of the individual animals and their previous history and housing conditions. Thus, all grouping and regrouping needs to be thoughtfully managed with careful attention to the way animals are introduced or re-introduced to each other (see Section 3.3.2), together with careful observation and monitoring of the animals, recording which individuals are likely to be compatible.
An understanding of the natural social structure of the species helps determine the most appropriate grouping (see Table 1 and Section 2.1), but this also depends on what is practically possible in any given situation and whether the goal is to maintain breeding groups, stock or experimental animals. For example, macaque species naturally live in multi-male, multi-female groups of around 10–50 animals and this structure is ideal for breeding colonies. Stock groups of 10–20 small (2–3 kg) macaques of the same sex are commonly housed together, and experimental animals have been successfully maintained as groups in toxicology, immunology and neuroscience studies.
Marmosets and tamarins naturally live in extended families and, in captivity, groups containing a mother, father and one or more sets of compatible offspring are the most stable. In the case of stock or experimental animals where breeding needs to be prevented, the female can be implanted with a contraceptive, or the male vasectomized.
Pair housing is not a natural social structure for any primate species and will not work in all circumstances, so as a general rule it is always best to aim for larger groups. However, safe pair formation and subsequent pair-housing techniques have been developed for adult male and female rhesus, stump-tailed and pig-tailed macaques, as well as for female long-tailed macaques (Reinhardt et al. 1995b, Turner et al. 2003). For marmosets, the most stable pairs are mixed-sex pairs. Same-sex pairs are less stable and few positive affiliative interactions are observed (Majolo et al. 2003). However, if same-sex pairs are selected from related familiar individuals, they may show more affiliation, and this is therefore recommended rather than pairing unrelated individuals.
Although the ideal is to keep groups stable and constant, most animals are unlikely to remain in the same groups throughout their lives. Disturbance starts at the breeding centre, usually at weaning when animals are removed from the family group and placed in same-sex or mixed-sex groups of similar age. These groups may be disrupted again to supply animals of a particular age or weight. Animals then face a series of disruptions of their social groupings in the journey from the breeding centre to the user establishment. In some centres they are singly housed or paired for ‘conditioning’ (see Section 8.3.2) and transport. Once they arrive at the user establishment, they are kept either individually, in their transport pairs, or grouped for an initial acclimatization 9 or quarantine period. It is then common practice to randomize individuals prior to use. Experiments may require animals to be taken out of their group temporarily or permanently, which means further disturbance. These regroupings add to other stresses on the animals, and are both a welfare and scientific concern, as stress caused by non-compatible groupings can be a confounding variable in experimental studies.
Acclimatization: Adaptation to a new climate or environment.
To reduce the need to disrupt social groups, animals should be transported in compatible pairs and, on arrival at the user premises, established pairs or groups should be retained. This will reduce subsequent conflict and thereby improve veterinary and behavioural management. The process can be facilitated by good communication between the breeder and user establishment. It is also good practice to randomize animals in established, compatible pairs or small groups within the confines of the experimental protocol wherever possible.
Detailed information on factors contributing to the success of primate introductions is given in Watts and Meder (1996) and summarized in Table 6, together with additional contributions from Working Group members.
Strategies for successfully introducing primates to conspecifics
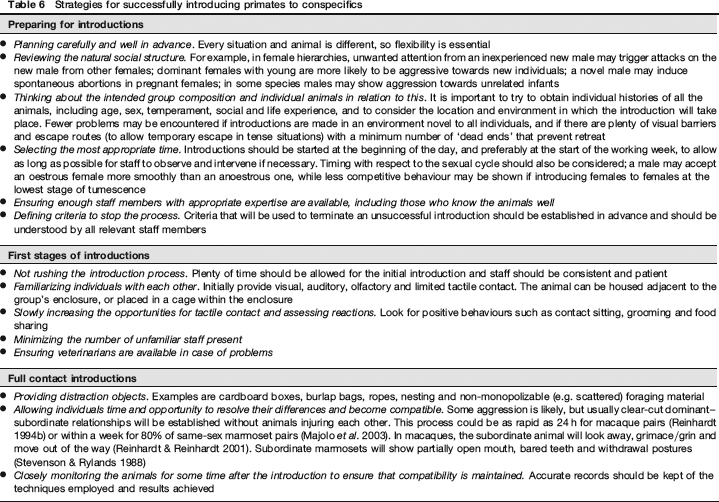
Strategies for successfully introducing primates to conspecifics

The introduction of a primate to unknown conspecifics can generate psychological and physiological stress. Space limitations, lack of refuges and visual barriers may prevent critical social distances being achieved or the ability to move away from a potential aggressor. Individual preferences for specific social companions may not be considered, and time constraints and limited knowledge may prevent staff from carrying out introductions in accordance with sound ethological principles.
Thus, if introductions are not planned and managed carefully there can be serious consequences including bullying or aggression, injury, shock and even death. Introducing a new member to a group may disrupt the settled hierarchy, forcing group members to form a new relationship with the unfamiliar individual, and to reorganize their previous relationships. Despite this, it is always better to attempt a well-managed programme of introductions even if unsuccessful, rather than not to try at all, leaving animals individually housed. However, introducing an entire group of primates to an unfamiliar group should not be attempted because the likelihood of serious fighting is high.
It is impossible to completely eliminate aggression in group-housed animals but the clear benefits of social housing outweigh the risk of minor aggressive incidents. Aggression is a normal behaviour and is usually only a problem if it escalates and causes poor welfare. In well-managed laboratory environments serious aggressive incidents resulting in wounds that require wound suturing are rare, but minor incidents may be seen more frequently depending on colony size. Victims may show small cuts, bites, scratches, bruises, alopecia or a reluctance to socialize with conspecifics or humans. It can be difficult to determine when aggressive behaviour becomes a problem that requires intervention. Misinterpretation and premature or inappropriate intervention in response to an apparently aggressive incident can exacerbate a problem, or even create one where none exists, so advice from someone experienced in primate behaviour may be required.
Aggressive incidents occur most frequently when additional stressors occur, for example, in the breeding season, at puberty, during illness, or when a new individual is introduced or the leadership of a dominant animal is challenged (e.g. due to age or infirmity). Husbandry activities within the pens, such as feeding and cleaning, may also lead to increased levels of arousal and decreased thresholds of tolerance toward other primates. Intergroup aggression may be another issue, where several groups (or pairs) of animals are housed alongside each other. For example, marmosets, tamarins and Old World monkeys can display high levels of territorial threat displays towards other visible groups when housed under crowded colony conditions (Savage 1995, Ely et al. 1998). This may affect breeding success (National Research Council 1998) and may increase aggression within the group as a consequence of displaced intergroup aggression (Savage 1995).
Note that removal of the canine teeth solely for handling or husbandry purposes is not acceptable since this can cause considerable pain and discomfort to the animal and may result in chronic health problems. This procedure is no longer allowed under some national regulations including the Animal Welfare Act in the USA.
Identifying the cause of aggression.
The first step in effective management of aggression is to identify the reason for the behaviour and the stimuli that trigger an aggressive response in a particular animal or group. Some useful questions to consider are listed below.
Is the aggression within or between groups? Is there any particular timing of aggressive incidents throughout the day? Is it linked to any potential event that could increase the level of arousal in the animals such as feeding, moving or weighing animals? Is one particular animal always the aggressor or victim? Does it seem linked to a particular resource such as a perch or nest box? Is it linked to any particular member of staff?
It is important to record details of when the incidents occurred, who was present in the primate unit at the time, what activity was taking place (e.g. cleaning, feeding, weighing), the level of injury to the individual animals involved, and the behaviour of those individuals and other members of the social group before, during and after the incident. A video and sound remote monitoring system can be very helpful in this regard.
Reducing conflict.
Ways to reduce the chances of conflict occurring are listed below.
Identify animals likely to be compatible by careful observation of their behaviour towards each other. Obtain primates in pre-existing groups or compatible pairs and avoid regrouping them on arrival at the scientific establishment. House individuals of different ages and/or body weights together since they are then less likely to compete with each other (Reinhardt 1994b, Majolo et al. 2003). For example, stability in groups of less than 10 young marmosets can be restored by introducing an older animal to the group, but manage this carefully in case the older animal is challenged. Design complex enclosures with escape areas, visual barriers and plenty of enrichment. Increase enclosure size to permit more effective primate signalling of dominance, approach and withdrawal responses, avoidance of aggressive individuals and/or retreat to a safe distance (Erwin 1977, Reinhardt & Reinhardt 1991, Savage 1995, Maninger et al. 1998). Maximize the stability of the group by reinforcing the natural social order within the group. Attend to the more confident individuals first when greeting animals, and allow these individuals to be the first of the group to go through doorways and receive treats or food.
Animal care staff need to be confident in their ability to interpret primate communication and to deal with aggressive primates in an appropriate and safe manner. Depending on the cause of aggressive incidents, they can try to adapt their routines to avoid any circumstances that trigger or exacerbate social conflict. For example, the levels of arousal at feeding times can be decreased by feeding animals without delay, by scatter feeding (see Section 4.3), and by positive reinforcement training for cooperative feeding (e.g. Bloomsmith et al. 1994, Laule 1999).
Dealing with serious and/or persistent aggression.
Where persistent aggressive behaviour cannot be prevented by increases in enclosure size and complexity, animals will need to be separated. Where serious aggression arises in an established group of animals, it may be possible to separate them into compatible subgroups. It is advisable to remove animals to ‘neutral territory’ and put physical space between the subgroups, preferably moving them into separate units, rather than across the aisle of the same unit. This should avoid continuing displays of social conflict.
Alternatively, an individual animal may need to be removed. However, removing one of two fighting individuals can be problematic. Removing the dominant aggressive animal may result in subsequent fighting to reinstate the dominance hierarchy in the enclosure. Removing the subordinate animal may result in the dominant aggressive animal intimidating another animal within the group. Both options have been tried with varying degrees of success, and more information needs to be gathered in order to determine the best approach. Individuals that have to be removed may find the social environment of another group more compatible; for example, a dominant animal may be less dominant in another group.
It has been common practice to house primates individually after surgery in the belief that animals will interfere with sutures or equipments such as cranial implants, or that those animals who are slower to recover than others could be at risk of aggression. However, this is often not the case and surgically-treated animals, including those with cranial implants, can be pair or group-housed soon after recovery (Wolfensohn & Peters 2005).
In the immediate postoperative period, animals usually need to recover separately until they reach full consciousness and alertness, have full motor control and can eat and drink. Recovery housing should be in the proximity (sight and sound) of the social group. With a well-refined technique of general anaesthesia, such as use of sevoflurane with good postoperative analgesia, rapid recovery can be achieved so that the animals can return promptly to their companions in the home environment. This avoids any alterations to the social hierarchy and consequential aggression that may occur if there is a prolonged period of separation. Animals may need longer support if surgery has significant side-effects, as with some brain lesions.
Refinements in suturing, such as interrupted sub-cuticular sutures, with the possible additional use of tissue adhesives, should allow the integrity of a surgical wound to be maintained so that removal of sutures by cage-mates does not occur. Adequate perioperative analgesia and aseptic techniques will also prevent animals from paying untoward attention to their own wound sites due to discomfort or infection leading to irritation for the individual and attracting the attention of group mates.
Primates may start to climb before they are fully recovered from anaesthesia, and may fall and injure themselves. This can be prevented using a purpose-built recovery enclosure, or by temporarily reducing the height of the home enclosure. Recovery enclosures should be constructed such that the animal does not have to be caught to be returned to the home enclosure. Alternatively, animals should be trained to enter and leave a transport box, which can be lined with a soft warm material to prevent injury and heat loss (see Prescott et al. 2005a). If surgery involves any possible long-term side-effects, the home enclosure may need to be adapted to take this into account (Rudling 2003).
Husbandry in biological safety level category 3 and 4 facilities
There are two major types of containment that are commonly used for primates. Isolators, which operate under negative pressure and in which moveable cages can be placed, are used for small primates. Staff members carry out all husbandry and other procedures by entering a half suit from below, which provides them with complete protection and can obviate the need for other protective clothing. For larger primates, rooms can be modified with appropriate barriers and airflow and staff wear protective clothing with respiratory protection. In both these systems, a barrier separates staff from the animals, thus affecting the level and closeness of contact. This could lead to an emotional detachment between the animals and those responsible for them (see Applebee in Hawkins et al. 2003). It may also be thought to be more difficult to provide a variety of environmental or nutritional enrichment in isolators. This can be overcome for rodents (Hawkins et al. 2003) and similar principles can be applied to primate species as set out below.
Improving visibility and staff–animal interactions
Staff need to be able to see the animals and the animals need to be able to see the staff, particularly their faces; the appearance of a person in a full protective suit with only the eyes visible can be a frightening experience for all primates. Always consider whether and how protective masks will affect interaction with the animals. A full visor can be incorporated to both full or half suits to ensure that the animals can see the face of the person, and enable some form of basic communication. Full barriers that separate staff from cages should include a transparent area. Being able to observe animals at all times is a requirement, not least for inspections by national regulatory authorities. Remote viewing equipment should be used if direct access to the facility is difficult for non-essential personnel.
There is no reason arising from the barrier system itself why animals cannot be housed in appropriate social groups. However, the nature of the experiments the barrier is designed to contain may create a problem. For example, when dealing with dangerous pathogens there can often be variations in individual susceptibility, such that one animal may have to be euthanized several days/weeks before others in a pair or group. If this does occur, then particular attention needs to be paid to the surviving animal, with enclosure orientation such that other animals in the isolator can be seen and heard.
Enrichment
Housing design should facilitate foraging, either through provision of substrate or, for example, with forage boxes or activity feeders (if dust is an issue and tray tidies are employed). Strategically placed openings in line with cage shutters or trays will allow for normal husbandry procedures. Where cages are metal, wooden materials should be incorporated to allow chewing and scent marking (especially for marmosets). Engineering solutions may need to be applied to allow ease of placement and removal.
It is possible to train primates to come to the front of a cage to present limbs for blood sampling or administration of substances to avoid the necessity of using a squeeze back (although with some pathogens, staff safety issues will be paramount). It may be easier to do this training before animals enter the containment facility, and time needs to be allowed for this.
4 Nutrition, feeding and enrichment
In the wild, primates spend a variable but significant proportion of their waking hours searching for, procuring, processing and consuming food (Clutton-Brock & Harvey 1977a,b). These activities form a fundamental part of their natural behavioural repertoire and involve complex thought processes such as spatial and temporal mental maps, sampling, decision-making and social cooperation. Primates will voluntarily work for food, even when it is freely available, which indicates that the act of foraging in itself has intrinsic appeal (Anderson & Chamove 1984, O'Connor & Reinhardt 1994, Reinhardt 1994a,b). Feeding primates should not, therefore, just be seen as satisfying a physical need. Providing food in the laboratory as a single meal on a predictable schedule, or as easy to process food items in one location (e.g. pellets in a hopper), ignores the complex behaviour and cognition associated with feeding, and may have adverse consequences for animal welfare. Appropriate nutrition is clearly essential, but providing variety in the content of the diet and in the method of food presentation will enrich the environment of primates in all captive situations.
Content of the diet
Under natural conditions, all primate species used in the laboratory consume a varied diet. For example, common marmosets and tamarins eat fruit, insects, lizards, eggs, small birds and the gum or exudates of trees; macaques eat fruit, flowers, buds, leaves, bark, twigs, roots, pith, resins, gum, sap, grass, fungi and animal prey (National Research Council 2003). As diet generalists, these animals are likely to crave novelty in their environment (Morris 1970).
When designing a diet, it is important to consider the natural diet as well as the nutrient requirements for health and wellbeing. For example, gum-feeding is related to the health of the gut in marmosets (Power 1996) and insects are a major source of protein for many callitrichids (Garber 1984, Terborgh 1993). Commercially available artificial ‘gum trees’ and insects (e.g. locusts, crickets) are therefore used in several laboratories to provide enrichment as well as nutrients for callitrichids. Some insects (e.g. mealworms) can be fatty and it may be necessary to balance the phosphorus content of insects with a calcium intake to maintain an ideal Ca:P ratio (Wakenshaw 1999). Macaques are often categorized as frugivores, so fruit would seem an obvious food choice. However, commercially cultivated fruit bears little resemblance to the fibrous and weakly sweet fruit consumed in the wild; so vegetables, particularly leafy greens high in fibre, are a better option for basic nutrition. In fact, macaques are largely omnivores, often ingesting small birds, fish, crustacean and insects, fungi, tree bark and roots, leaves and tree exudates, and their diet should reflect this variety.
Purpose-made diets
Most feeding programmes for primates in captivity use a purpose-made complete diet, in the form of extruded pellets or biscuits, as the chief source of nutrients, supplemented by natural foods for enrichment. Purpose-made diets contain all the nutrition needed to maintain animals in a healthy state but are less palatable than natural foods for most primates. To ensure their consumption, it is common for such diets to be modified in some way or offered as the first feed of the day when the animals are most hungry, leaving more highly preferred foods until at least 4 h later. For example, many laboratories moisten pellets or biscuits before feeding (e.g. with warm water, fruit juice or flavoured non-milk-fat milkshake), cover them with a porridge preparation, or make a mash with other food items such as raisins or kidney beans. Soaked pellets should not be left out for extended periods of time as this can result in loss or destruction of some vitamins, facilitate spoilage by moulds and bacteria, and increase the incidence of oral health problems (National Research Council 2003). Dry pellets allow for good dental hygiene, which is not afforded by soaked pellets.
Some manufacturers add flavourings and vary the colour, texture, size and shape of their pellets to make them more interesting and palatable. Sweet and fruity tastes generally enhance dietary palatability for most species. Colour preferences probably have a role in food selection in the wild and some colours can influence food acceptance (Barbiers 1985). Pellets should be small enough to be readily held and chewed (extruded pellets are generally easier to bite). There are data that suggest immune sensitization to wheat in callitrichids; so wheat-based pellets should be used with care (Gore et al. 2001).
Variations in diet
Introduction of different colours, odours, flavours, textures and shapes increases sensory, mental and physical stimulation for the animals. This helps compensate for time that is no longer spent on food sourcing, capture, preparation and consumption (e.g. Schapiro et al. 1996, Reinhardt 1997). Providing a variety of food types can also prevent ‘the monotony effect’ (i.e. reduced food intake) that occurs with prolonged feeding of any one food.
In some studies (e.g. regulatory studies) there is a need to control dietary content, but this need not preclude the use of dietary supplements such as fruit and vegetables. Absence of variety in the diet can result in abnormal behaviour and this can affect scientific data; hence the need to limit dietary variation should always be carefully considered in consultation with the veterinarian, person(s) with responsibility for day-to-day care of the animals and the study director. Where diets cannot be varied or supplemented, varying the complexity of the feeding experience by presenting food in different ways can be enriching (see Section 4.3).
Where animals are given a choice between foods, it is important that the amount of each is carefully controlled and that variation in palatability between ingredients is managed in order to ensure consumption of a balanced diet (Kirkwood 1983, Price 1992, Tobin 2003). In addition, when feeding palatable foods, rewards and treats, animals should be monitored for signs of weight-gain, dental problems or resource-related aggression (see Section 4.6). A specialist in primate nutrition should be consulted to help design a feeding programme that allows a combination of a complete diet and supplementary foods in amounts that achieve the goal of providing a balanced and varied diet without risk of nutritional disorders.
Foraging mix (e.g. a mixture of peanuts in the shell, sunflower seeds, pumpkin seeds, pine kernels, flaked maize, dog biscuits, locust beans, banana chips, dried apricots, raisins, rice grains) can be used as a supplement to purpose-made diets, vegetables and fruit. Commercially available preparations or in-house mixes encourage animals to forage for longer periods and reduce hoarding, especially if the mix contains very small food items. Attention should be paid to the levels of fat and sugar in the ingredients that make up the mix and the nutritional content should be included in the make-up of the diet.
Food treats used for positive reinforcement training also need to be counted in the nutritional content of the diet and high-fat or high-sugar food should be avoided. For example, rather than using chocolate, cheese or marshmallow as a reward, a part or whole raisin or grape provides a more nutritious but highly palatable food item. A small New World monkey does not need a whole raisin as a reward, while an Old World monkey may do so. Guidance on the use of food rewards with food control as a motivational tool in neuro-science and behavioural research can be found in National Research Council (2003) and Prescott et al. (in preparation).
Feeding requirements
Feeding requirements will differ according to species, age, sex, body weight, health status, environment and reproductive status. Food should be provided so each animal has enough to maintain good body weight and condition but not become overweight. Regular weighing enables detection of losses or gains that may signal the start of underlying disease problems and is a useful tool for monitoring health and welfare. Body condition scoring can also be used to assess the health of individual animals as well as to determine nutritional adequacy within groups of animals (Clingerman & Summers 2005, Wolfensohn & Honess 2005).
Although, in general, primates eat to meet their energy requirements, individuals of some species (e.g. stump-tailed and rhesus macaques) seem to consistently eat in excess of immediate needs and become obese under certain conditions. Thus, it may be necessary to limit intake of diets that are energy-dense and very palatable. Estimates of daily calorific requirements (and hence the amount of food needed to meet these requirements) can be obtained from several sources (e.g. National Research Council 2003, Wolfensohn & Lloyd 2003), including the manufacturer of the specific food being used. The software Zootrition® is a useful tool in estimating nutritional content of diets (www.stlzoo.org/animals/animal-foodnutritioncenter/zootrition.htm).
Estimates of feeding requirements might need substantial modification because of the variations in metabolic rate/ physiological state of individual primates, the calorific value of their food, opportunities for exercise and the ambient environment. It is important to remember that social hierarchy can play a significant role in the amount of food each individual can acquire. That is, subordinate individuals may have a limited intake if food presentation is not taken into account.
Refining food presentation
Food should be provided in ways that increase the opportunity to express species-typical behaviour patterns by simulating one or more components of foraging. This will stimulate the animals mentally and physically (Gore 1993). Choice of appropriate feeding methods will also increase the time spent foraging and feeding, which may help to alleviate boredom in captivity (Beaver 1989, Scott 1991). It can also inhibit stereotypy (Fajzi et al. 1989, Brent & Long 1995, Holmes et al. 1995, Knapka et al. 1995, Novak et al. 1998) and help disperse animals, thereby reducing tension and the likelihood of aggressive interactions (Boccia 1989, Gore 1993, Honess & Marin 2006). There are many methods of foraging enrichment that will accomplish these objectives, which can be device-based or non-device-based. It is important not to simply use one or two of these methods, but rather to use a good variety over time to provide novelty for the animals, and to regularly and critically review each method for its effectiveness.
Non-device-based foraging enrichments
These provide changes in food distribution, location, complexity or ease of access, but little or no additional or special equipment is needed.
Scattering food (e.g. fruit, forage mix, seeds) on a barren floor increases foraging time and helps satisfy searching instincts for terrestrial species. For macaques, food can also be scattered on the bottom of a swimming pool, to encourage the animals to dive under water (see Section 3.2.4), or on a mesh enclosure roof through which the animals must pull the food (Buchanan-Smith 1995). This will help promote a variety of locomotor postures, muscle use and physical fitness (Britt 1993) and social interaction (Gore 1993). Hiding food in deep substrate (e.g. straw, hay, shredded paper, wood-wool, wood chips, wood shavings, blankets, corncobs) will also increase foraging time for terrestrial species. Food can also be hidden by wrapping it inside paper sacks containing straw or wood shavings, and then tying these up with rope. This will encourage manipulation, provide sensory stimulation and occupy the animals (Gore 1993). Liquid foods (e.g. honey or yogurt), with or without seeds (e.g. sesame seeds), can be smeared on enclosure beams, branches, wooden sticks or artificial turf throughout the enclosure. Suspended food (e.g. fruit skewered onto pieces of wire or wood suspended from the enclosure furniture, roof or sides) is unpredictable in movement, especially when two primates attempt to access it simultaneously, which can be stimulating for the animals. Changes in food size or texture (e.g. diced or whole food, frozen food or juice) can alter foraging and feeding patterns. For example, time spent feeding was increased when long-tailed macaques were presented with whole versus chopped food (Lindburg & Smith 1988). Live food (e.g. insects) allows primates to hunt and can encourage use of the enclosure floor (McKenzie et al. 1986). Change in timing of feeds and amount of food allow a more natural foraging regime. The amount of food at each mealtime is varied on a scheduled basis to ensure that adequate and appropriate amounts of food are provided (Gore 1993).
Foraging devices provide changes in food delivery and ease of access. They may be made in-house or bought commercially. It is important to consider the effect of such devices on competition between members of the social group and to provide a sufficient number in order to avoid physical confrontation.
Foraging boards made of wood or plastic and containing holes into which food is hidden can be used to satisfy the searching instincts of extractive foragers (e.g. tamarins, capuchins). The holes can be covered with rice paper that the animals must puncture to obtain the food. Solid boards covered with textured fleece or artificial turf can be mounted on the outside of the enclosure to provide a substrate for foraging and loaded with sticky or small food items (Bayne et al. 1992). Dispensers for example, plastic tubes with holes stuffed with substrate and mealworms (Vignes et al. 2001) can be either passive, in which case the animals must wait for the food to fall out of the holes, or active, in which the holes are of a size appropriate to admit a finger, hand or arm for food retrieval. Some dispensers require operation of a rod to obtain food. This allows the animals to control the frequency of food delivery and duration of feeding and simulates tool use. Balls and toys are available into which food items can be loaded through small holes. As the animals manipulate the device, the food items gradually drop out of holes. Electronic devices (e.g. a moving conveyor) have also been used to alter the frequency of encounter with food. Artificial ‘gum trees’ (e.g. McGrew & Brennan 1986) permit marmosets in the laboratory to perform the natural behaviours of gouging and feeding on gum. Gum arabic (from Acacia senegal) used in frozen desserts and in bakery, confectionery and dairy products, is commercially available for use with artificial ‘gum trees’; it can also be syringed into holes drilled in trees or branches.
Careful consideration of the number, timing and content of feeds can benefit animal welfare both by helping to ensure animals receive adequate nutrition and by increasing habituation and socialization time with humans. Captive primates given a selection of cultivated foods or treats of various nutrient densities do not consistently choose a complete diet (Ullrey 1989, Oftedal & Allen 1996). Intake of highly preferred foods must therefore be controlled, lest overall intake becomes nutritionally unbalanced (Tobin 2003). To prevent feeding selectivity, less preferred foods such as pellets or mash can be fed in the morning and more highly preferred supplementary foods in the afternoon. In this case, animals are fed twice during the day, in addition to other visits to carry out routine husbandry duties, thereby increasing staff–animal contact.
Primates quickly learn timing of meals and they will often become agitated as mealtime approaches. Predictable timing of feeding is thought to be associated with increases in stereotypies and aggression (Krishnamurthy 1994, Carlstead 1996), as well as inactivity and coprophagy (Bloomsmith & Lambert 1995). All these behavioural changes have been interpreted as being indicative of decreased welfare. It has been suggested that for optimal welfare and to avoid food anticipatory behaviour, the time of feeding should be varied throughout the species-typical feeding period but preceded by a reliable signal announcing the arrival of food, such as a buzzer or bell (Gore 1993, Bassett 2003).
Changing dietary programmes
While it is important to regularly review feeding programmes and dietary enrichment, the basic diet should not be changed too often if a colony is in good health. If new primates arrive from another establishment, they should be fed the diet that they are accustomed to, particularly if they are young animals shortly after weaning. Any changes to the diet, within the existing colony or for new animals, should be reviewed and discussed with the relevant personnel.
When making changes to the diet, easing the transition with a gradual, structured change over a two-week period will help to prevent gastrointestinal disturbance, diarrhoea or vomiting. Aside from individual experience, the acceptance of novel foods can vary with age and species. Juveniles are often more willing to try novel foods, and macaques seem to accept novel foods more readily than callitrichids. Observation of other primates consuming food can encourage tasting (Prescott et al. 2005b).
Potential problems with feeding and how to avoid them
Poor dental hygiene is usually a consequence of poor diet and is a particular risk for long-lived species used for breeding or long-term studies. Hard or fibrous items, such as non-toxic browse, dry pellets or synthetic bones, can be provided for Old World monkeys to chew and help clean teeth and stimulate gums. Plants that have been safely used as browse offerings are listed by the National Research Council (2003) and Carroll (2002). Animals should be monitored for dental caries, periodontal disease and abscesses. They can be trained using positive reinforcement techniques to open their mouths for inspection of the mouth and teeth, thereby avoiding capture and restraint (Colahan & Breder 2003, Melfi & Thomas 2005).
Loss of appetite can occur if primates are unwell, when they may refuse to eat the usual diet provided. In such cases, they can be tempted with sweet foods (e.g. fruit juice laced with a protein content). Diet aversion will occur to foods that become associated with negative consequences, for example, a drug dose with unpleasant side-effects. This can affect both the primate(s) and the study outcome. Using a low drug dose may help avoid diet aversion and a pilot study to test the acceptability of the substance, measuring food consumption and body weight, may be necessary.
Older primates are prone to obesity as well as dental problems, so overfeeding should be avoided. They may take longer to eat their food and allowances for this should be made within the feeding regime.
‘Cafeteria style’ presentation of food in bowls can lead to problems as primates normally pick out preferred foods and often throw the rest of the diet out of the bowl. This can lead to individual nutritional imbalance in all group-housed species (Crissey et al. 2001). If food bowls are used for groups of primates, provide numerous bowls in different locations/levels to prevent monopolization by dominant animals. For marmosets and tamarins, position bowls off the floor; these animals prefer to feed away from the ground and vertical clinging and suspension are normal feeding postures (Garber 1993, Buchanan-Smith et al. 2002, Prescott & Buchanan-Smith 1999, 2002).
Social rank and dominance behaviour can result in uneven distribution of food intake within a group. For example, dominant macaques in a social group tend to determine the time that others spend in feeding (Kemnitz 1984, Gore 1993). Food hoarding by high-ranking animals can also occur, particularly in small, restricted environments. Providing low energy food resources may help avoid this, as the animals can be allowed a greater amount. An alternative option is to provide food in small pieces and scatter it to reduce its monopolization. Aggression may occur around feeding, but this can be reduced with training for cooperative feeding (Bloomsmith et al. 1994, Laule 1999). If primates must be separated for feeding, the interval of separation should be as short as possible to allow food to be consumed. Care should be taken to ensure that such separation does not limit the ability to provide enrichment.
5 Refinement of common procedures
There is great scope for refinement of the common husbandry, veterinary and scientific procedures used in primate research, and this section of the report sets out some key principles and recommended texts to help achieve this. Every aspect of all procedures should be carefully considered, and the least aversive study design developed. A useful checklist of questions to consider when planning administration procedures is set out in a previous JWGR report (Morton et al. 2001) and, since many of the points apply equally to other common procedures, a modified version of the checklist is reproduced in Table 7. This, together with the principles set out in Section 2 of this report regarding the need to understand the animals and to interact positively with them at all times, can be used as a basis for all refinements.
Checklist of questions to consider when planning procedures

Checklist of questions to consider when planning procedures

Primates generally do not like to be picked up or restrained, as they perceive this as an aggressive act. Therefore, one of the most effective ways to minimize stress for primates in the laboratory is to ensure that they are handled in a competent and empathetic way. Major improvements in capture, handling and restraint procedures have been achieved in many laboratories in recent years. The aim is for animals to feel unafraid and comfortable whenever they are approached, handled, carried and restrained. Recommended principles for good practice devolve from Section 2 and are summarized below.
Purpose-bred animals of defined health status should be used. The animals should be well-habituated and socialized to humans, preferably early in life and trained to cooperate using positive reinforcement techniques. This will help balance the need to minimize stress for the animals with the need to protect the health and safety of staff. All methods of capture, handling and restraint require considerable experience and skill and should therefore be included as an integral part of staff training and competence assessment programmes. All procedures should be appropriate for the species, individual and the situation. They should be based on an understanding of species-typical behaviour patterns and communication, such that these are interpreted correctly and responded to appropriately. Interaction with the animals starts when they see or hear personnel approaching the room in which their enclosures are situated, not when the enclosure door is opened. Ideally, the confidence of animals should be gained by allowing them to initiate the interaction and approach. Personnel should behave in a passive and non-threatening manner, moving slowly and talking to the animal in a calm, quiet voice. Direct eye contact and potentially threatening body postures should be avoided (see Section 2.2). The need for capture and restraint should be avoided if possible. This can be achieved by carrying out procedures in the home enclosure (see Scott et al. 2003), training animals to enter transport cages and to cooperate in procedures that are relevant to what will be done with them (Prescott et al. 2005a). Handlers need to be aware that primates can be injured as a result of forceful removal from an enclosure, especially when individuals cling to the bars or mesh sides, so where capture and restraint are unavoidable, the least stressful procedures should be used. Situations in which animals escape from their enclosures and need recapture should be avoided. Swift recapture is necessary because the sight of escaped animals can cause others in the room to become agitated. Opportunities for escape should be minimized with good enclosure design, ensuring that outer room doors are closed before entering and leaving the enclosure, using a double door system, or using a secondary enclosure that is portable and fits to the outside of the enclosure. The problem is most easily avoided by training animals for capture and handling using positive reinforcement, and training them to return to their enclosures by offering rewards (see also discussion by Smith et al. 2005). Breeders, suppliers and users of primates should liase closely to ensure that good practice in handling and restraint procedures is standardized between facilities.
There are a number of different methods for catching primates, all of which have advantages and disadvantages and associated welfare implications, including pathological, physiological and behavioural effects. These are described and discussed in recent reviews (Klein & Murray 1995, Reinhardt et al. 1995a, Rennie & Buchanan-Smith 2006b, Sauceda & Schmidt 2000) and therefore only a brief summary is provided below, together with the Working Group's recommendations.
Training to enter a transport container.
The best method for catching and carrying marmosets, tamarins and macaques for routine procedures such as relocation or weighing, is to train them, using positive reinforcement, to move into a catching box (Reinhardt 1992a,b, Klein & Murray 1995, Scott et al. 2003). Reinhardt (1992b) found that rhesus macaques trained by positive reinforcement would enter transport crates in a reliable order (influenced more by age than dominance) each time capture was initiated and that this ensured that specific animals could reliably be caught.
The design of the transport cage should enable the handler to catch the animal safely, without risk of injury. For example, a transport box for marmosets and tamarins should be easily attachable to the front of the enclosure, allowing the caregiver's hands to be free to deliver a reward through a mesh front, and simultaneously close a sliding door. There is debate about whether or not to use a nest box to capture marmosets and tamarins, since they use their nest box as a secure refuge.
A transport box for macaques may be best aligned with the front of the enclosure (or placed within it) and mounted on a trolley, given the weight of these animals (see Smith et al. 2005). The method is not completely stress-free but, if carried out correctly using habituation and positive reinforcement, it can significantly reduce capture stress and is more humane than net-catching or the use of a pole and collar (Rennie & Buchanan-Smith 2006a).
Catching by hand.
Most primates can be caught by hand if well-habituated both to humans and this method of capture. In some cases, bare-handed capture is possible and this offers the greatest protection to the animal, particularly with small species, because the handler can feel the animal well and can exert the correct amount of pressure for safe restraint. If the health status of animals is unknown, or if they are infected by a harmful agent, a protective glove or gauntlet of soft, lightweight leather will give the handler protection from bites or infections. The disadvantage of using a glove is that it can be difficult to determine the amount of pressure needed to restrain the animal safely; thus, lightweight gloves are better than heavy gauntlets in this respect. There are also disadvantages for the animals in that they may bite the glove/gauntlet and damage their teeth and gums. If they find capture and restraint unpleasant, they may associate this with the glove/gauntlet and become agitated and stressed.
Net capture.
Nets are used in conjunction with hand capture, particularly with groups of animals housed in large cages (Sainsbury et al. 1989). However, this method can result in entanglement and injury of the animals when removed from the net, and the resultant stress can cause acute diarrhoea, rectal prolapse and lacerations (Luttrell et al. 1994). It may also frighten other animals in the group. The Working Group does not recommend this method.
Pole and collar capture and restraint.
This method is sometimes used for larger primates including macaques (Klein & Murray 1995). The Working Group considers that it is not good practice and does not recommend this method.
Squeeze-back mechanisms.
This method is used both to catch and restrain primates and is addressed in Section 5.1.2.
Restraint
Restraint for any purpose, whether of short duration for health checks or blood collection, or long duration for elec-trophysiological recording or infusion, can be a highly stressful procedure. It can induce fear and distress responses, such as physical resistance to handling, alarm vocalizations, defensive threatening and aggression, urination and defecation, all of which indicate a serious welfare problem and must be properly addressed. In addition, stress results in a number of physiological responses that will increase unwanted data variability (Suomi et al. 1989, Reinhardt 1991, Brockway et al. 1993, Reinhardt et al. 1995a, Honess & Marin 2006). Justification for restraint procedures should form an integral part of the ethical review of projects, and techniques should be regularly reviewed for further refinement.
It may be possible to avoid the need for physical or chemical restraint through habituation and socialization, together with training of animals to cooperate with procedures using positive reinforcement techniques (Reinhardt et al. 1995a, Sauceda & Schmidt 2000, Reinhardt 2003b, Rennie & Buchanan-Smith 2006b). For example, macaques are quite quickly and easily trained to offer their limbs for injection, which avoids the need to catch them or to use a squeeze-back mechanism. The animals have some control over the situation, by retaining relative freedom of movement, and show reduced stress responses as a result (Reinhardt et al. 1990, Reinhardt 2003b).
Where restraint is necessary, much of the associated stress can be eliminated if the appropriate method is used sympathetically, in conjunction with habituation and positive reinforcement training (Fortman et al. 2002). The method used should provide the least level of restraint, for the minimum frequency and duration necessary. It should protect both primate and personnel from harm, and should avoid causing unnecessary distress or discomfort. Different methods of restraint and their effect on primate physiology and welfare are discussed by Rennie and Buchanan-Smith (2006b); so only a brief summary of the most commonly used methods is presented below as background to the Working Group's recommendations.
Manual restraint.
Macaques that are well-socialized to humans may be restrained in a cradle position using one arm to support the animal's weight and the other to safely immobilize the animal. It is completely unacceptable to hold or carry a macaque with one hand, holding both arms of the animal behind the back, since this is both painful and stressful for the animal and the handler has less control. Wrenching the arms can also dislocate the shoulder or elbow joints (Sainsbury et al. 1989) and excessive force can fracture the humerus (Klein & Murray 1995).
Marmosets and tamarins can be held around the chest between thumb and forefinger. The thumb can be placed under the chin to reduce the risk of the handler being bitten (Sainsbury et al. 1989). Other methods of restraint have been described to facilitate blood sampling and for injections in marmosets where the precision required to sample from these small animals may prevent them from being trained to present a limb (Hearn 1997, Greig et al. 2006).
Squeeze-back cages.
The squeeze-back or crush cage is a commonly used restraint device for larger species of primates (Fortman et al. 2002). It comprises a caging system that incorporates a manual or automatic mechanism to move the back panel of the cage towards the front. The animal is slowly moved to the front of the cage and can be partially or totally immobilized. The advantages are that an animal can be restrained quickly and safely for routine procedures such as injections, the need for manual handling is reduced, and staff can be easily trained to use the method correctly. However, it is not recommended for more complicated techniques, when accuracy is essential (e.g. for cannula placement).
If sensitively used, squeeze-back cages may induce less stress than some other methods. However, there are associated welfare problems and the use of a squeeze-back cage as the animals' home cage is not recommended by the Working Group. This is because such cages are often designed to meet minimum size requirements, and the small size and squeeze mechanism limit the potential for structural complexity and enrichment (Dexter & Bayne 1994). Furthermore, the animal is continually exposed to the threat of capture within the home environment.
As a compromise, a squeeze cage can be incorporated into group-housing systems for macaques and the animals trained, using positive reinforcement training, to move into them when required. They can also be trained to present a specific part of the body to the front of the cage when the squeeze mechanism is started, thus reducing the need for repositioning (Luttrell et al. 1994, Reinhardt 1997).
Restraint chairs.
Restraint chairs are used to support primates in a sitting position when it is deemed necessary to restrain them for prolonged periods (e.g. for electrophysio-logical recordings or chronic infusion). Chair restraint can affect the animal's physiology (Norman & Smith 1992, Norman et al. 1994) and can severely compromise animal welfare (Morton et al. 1987, Klein & Murray 1995). It should only be used where absolutely necessary, and in such circumstances, ways of reducing the effect on the animals should be thoroughly explored and implemented. Some refinement opportunities are listed below.
Select quiet, calm primates. Habituate them to the sensation of being sat in the chair prior to individual studies and reward calm behaviour. Train animals by voice command to enter devices from their home enclosure and place their head through a hole in the top of the chair (as described for macaques by Scott et al. 2003 and Fairhall et al. 2006), or use the chair within the home enclosure. Minimize the duration of restraint. Define the maximum length of chair time in the programme of work after discussion with veterinary and animal care staff. Any time longer than 1 h, even for well-habituated animals, is likely to be a welfare concern and must be carefully weighed against the scientific need. Attend restrained animals all the time. Place compatible animals next to each other where studies allow simultaneous chairing. Numerous psychological studies have demonstrated that the presence of a social partner can ameliorate stress responses to novel and aversive experiences (Gunnar et al. 1980, Coe et al. 1982, Gonzalez et al. 1982, Hennessy 1984). This also applies when using a sling (Coelho et al. 1991). Give glucose drinks by syringe if animals are likely to become thirsty while restrained and are not on food or fluid control protocols. Massage the animal's legs to maintain proper circulation and counter the effects of the abnormal sitting position. Always move occupied chairs carefully and quietly.
Chairs need to be well-designed and suitable in size and operation for the individual animals in order to prevent injury and discomfort including skin abrasions, position-dependent oedema, insiguinal hernias, laryngeal air sacculi-tis and rectal prolapse (Morton et al. 1987). Primates tend to ‘squat’ rather than sit, so chair design needs to reflect this. Useful design features include:
Minimal confinement leaving animals with room to stretch their limbs, in particular leaving the arms free (although, if external catheters are used, the design should ensure these can be taped out of reach). Adjustable sections to accommodate species and individual physical characteristics (Klein & Murray 1995). The position and length of the tail, position of the ischial callosities (where present), crown to rump length, limb length and variations in posture, all need to be taken into account. Use of anti-friction material (e.g. polytetrafluoroethylene [PTFE]) and padding to prevent chafing. Wheels fitted with silent castors and any loose parts constructed such that they do not rattle.
Tethering.
An alternative to chair restraint for larger species and some procedures is the use of the vest and tether system developed by Chatham (1985). This allows continual administration and sampling via surgically-implanted cannulae, which are enclosed in a tether protected by a flexible stainless steel casing held in place in a nylon mesh or leather vest (Klein & Murray 1995). Tethers can also contain lead wires attached to recording devices on the exterior of the cage. Subjects are able to lie down and turn around, thus their movement is not impeded (Reinhardt et al. 1995a). Samples can be obtained remotely so that the animal need not be physically restrained, and the method may therefore be less stressful for the primate and the handler.
The use of vest and tether systems raises several issues of concern with respect to animal welfare. In most cases, the scientific procedures will involve surgical intervention and the use of transcutaneous cannulae with the attendant risk of infection. While wearing the vest, an animal's ability to groom is restricted. In addition, the animal may be singly housed during the instrumentation period because of the potential for cage-mates to interfere with the vest and tether. In chronic studies, this can be for weeks or months (Crockett et al. 1993). The Working Group does not consider this acceptable and ways of overcoming these problems should be found.
The risk of postoperative infection can be minimized using sterile procedures and appropriate antibiotic cover. Localized infection, most frequently around the point of exteriorization, may occur in a small proportion of animals. In such cases veterinary advice on effective remedial therapy should be obtained promptly. Rarely, systemic infection and septicaemia may occur (Morton et al. 2003, Hawkins et al. 2004). In such cases the action taken must recognize appropriate humane endpoints to ensure that individual health and welfare is not unduly compromised, and in some instances this may mean having to euthanize the animal.
Other refinements all of which have been shown to reduce the stress induced by tethering include improving the fit of vests (Morton et al. 2003, Hawkins et al. 2004), improvement of the acclimatization procedure (Crockett et al. 1993) and providing companions in an adjacent cage (Coelho et al. 1991). Reinhardt (1997) has reported that juvenile rhesus macaques were successfully housed in compatible pairs while tethered. The risk and consequences of compromising satisfactory data as a result of damaged cannulae, or other instrumentation, when tethered animals are housed in pairs, must be carefully weighed against the clear animal welfare benefits of social contact. Compatibility of animal pairs, and the robustness of vests and tethers with more than one animal in a cage, should be assessed objectively prior to experimental work, before concluding that single housing is the only practicable approach.
Chemical restraint.
Chemical restraint uses various combinations of different classes of sedatives and tranquilizing drugs. None of these are devoid of side-effects, which must be taken into account according to the procedure to be performed, the species and the level of sedation required.
Examples of the detrimental effects of using chemical agents include pain from intramuscular injection, muscle necrosis, unpleasant psychogenic effects, reduced food and water intake, time taken for recovery and the effect on social interactions within the group (Honess & Finnemore 2005).
Sometimes sedation is used routinely for minor procedures because it seems to be the easier option. However, this is not necessarily true and use of sedatives must always be weighed against the adverse effects on the animal. As with other restraint methods, training animals to cooperate will usually be a better approach where the health status of the animal allows this.
It is good practice for all primates in breeding, supplying or user establishments to be uniquely identifiable, and this is a legal requirement in some countries (e.g. European Community 1986). Some establishments interpret this requirement solely as a tattoo, and animals may receive a number of these if they are marked at both the breeding and the user establishment. However, tattooing is painful and distressing and should not be used without careful consideration of why it is considered necessary, and of alternative methods.
Ideally, the method of identification should not be painful, or cause an adverse reaction. It should not be uncomfortable or likely to cause injury, and should be appropriate for the study. Table 8 lists the advantages and disadvantages of common identification methods. For all of these, staff members need to be well-trained and competent in carrying them out. Non-invasive methods should be the principal method of choice, particularly for pre-weaned animals, or else a combination of highly visible temporary identification with microchipping, as recommended by Rennie and Buchanan-Smith (2006b). Ear notching and other forms of mutilation should never be used.
Advantages and disadvantages of identification methods
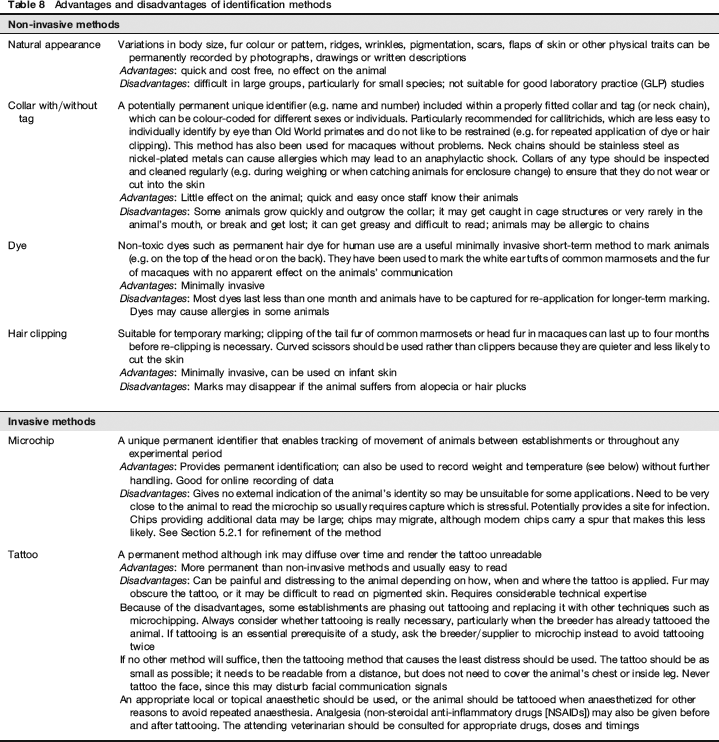
Advantages and disadvantages of identification methods

Primates that are individually recognizable can be given names since this helps to increase recognition of their individuality, facilitate recall of the animal's history, and foster appropriate attitudes to animals (Segal 1989, Reinhardt 2003a). Names are also a useful aid to training, particularly when animals are held in groups. Calling the animal's name gains his or her attention and helps achieve the desired response just as it would with a companion animal.
Site of implantation.
Selecting a site and training the animal to present the part of the body where the microchip is located enables the microchip to be conveniently read without having to catch and/or sedate the animal solely for this purpose. Wolfensohn (1993) found that the use of sites at the elbow or wrist gave the greatest success in re-reading the identification while the animal was conscious, as the microchip could be scanned while the animal held the side of the enclosure or when trained to present a limb.
Use of anaesthesia.
A microchip can easily be applied if the animals are anaesthetized for other purposes. Application of a local or topical anaesthetic should otherwise be considered, particularly for smaller species. Topical anaesthetic cream, although a useful way of reducing pain, takes 30–45 min to be effective. Local anaesthesia involves an intradermal injection, so is not without adverse effects. Always consult the attending veterinarian for appropriate drugs, doses and timings.
Readability of the chip.
Correct use of the reader is important, as is compatibility of microchips and readers. In the UK and Europe, most microchips and readers comply with International Standards Organisation standards (i.e. ISO 11784 and 11785). The USA and Canada have different microchips that necessitate the use of different readers. If animals are to be supplied microchipped to a different world region, the appropriate reader should be supplied with the animals and the location of the microchip specified. Manufacturers of microchips and application techniques should strive towards the production of universally compatible equipment.
Animals should be habituated to the reader, some of which are better tolerated by the animals than others. The reader can be mounted on a disc or pole that the animals can be trained to hold.
Use of microchips to minimize subsequent capture and handling for other procedures.
The need for repeated capture and handling can be reduced by positioning the reader adjacent to a balance and food hopper within the home enclosure of large groups. It can then be used to weigh the animals automatically when they step on the balance to obtain food. Some chips provide additional data, such as temperature, which then avoids the need for rectal temperature recordings.
The World Small Animal Veterinary Association (WSAVA) has made useful recommendations on microchip-ping (see WSAVA at www.wsava.org/site1099.htm for additional suggestions in this respect).
Animal passports
Each animal should have an individual file or ‘passport’ that gives details of their biography and what they have experienced, for example, transport history, social groups, training, husbandry system, type of environmental enrichment, research project history and medical history (see Table 9 for a full list). It should be sent with the animal if he/she is moved between establishments, together with general information on the establishment of origin, such as details of animal care. This will help to achieve continuity (or in some cases improvement) of care and also help to ensure rapid acclimatization to the new facility.
Information to be recorded in an individual history file

Information to be recorded in an individual history file

F1: first generation animals from wild-caught parents; F2: second generation animals whose parents were captive-bred
Information from individual files can be used to form part of a colony database to analyse primate care, use and breeding, and to review the adequacy of systems in order to develop good practice. Electronic databases are particularly useful, since staff can access them across a number of sites. They enable quick and efficient searching for information (e.g. availability, previous use), and can be programmed to automatically alert staff to the need for action (e.g. vaccination and prophylactic treatment schedules, weaning alerts).
General advice on the administration of substances and blood sampling is provided by Wolfensohn and Lloyd (2003). Specific advice on refinement with regard to the administration of substances, together with a checklist of questions to consider when planning administration procedures, is set out in Morton et al. (2001) and a modified version appears as Table 7. Additional questions to ask with respect to the substance administered are set out below.
Will the substance or vehicle have any adverse effects on the animal and are background data available? Will the concentration, dose volume, or nature of the formulation, alter the expected effect and, if so, have the necessary preparations been made? Are there any additional concerns regarding the physio-chemical properties of the substance or associated solvents, e.g. osmolarity? Does the substance need to be freshly prepared? If not, is it stored properly and properly labelled? Is it sterile if it is to be injected?
A European Federation of Pharmaceutical Industries and Associations (EFPIA)/European Centre for the Validation of Alternative Methods (ECVAM) report also provides information on good practice in blood sampling and the administration of substances by various routes (Diehl et al. 2001). Healing and Smith (2000) provide information on intravenous infusion studies, including information on surgical procedures, aseptic techniques, analgesia, choice of catheter, husbandry procedures for each species, vehicles, rates of administration and volumes.
Collection of other body fluids (e.g. saliva, bile, semen, cerebrospinal fluids) often requires invasive procedures and possibly catheterization. Since all of these procedures are likely to cause stress, the need for such collections should be critically reviewed in all cases, and the least invasive, least restrictive, regimes used. For example, saliva can be collected by training animals to chew on a cotton bud and semen can be collected using an artificial vagina.
Samples of urine and faeces can normally be collected without the need for invasive procedures. Welfare issues arise if samples from individual animals, or total collections of excreta (urine, faeces and cage-wash), are required. This may result in primates being housed singly in a restricted space with grid flooring, such as a metabolism cage, in order to determine absorption, distribution, metabolism and excretion values of certain materials. Because of the need for total collection, enrichment items are not usually provided. Typically the period of confinement may be for five to seven days postdose in order to ensure a total collection. Metabolism cages are also sometimes used for shorter periods (up to 24 h) for the collection of small volumes of urine to assess clinical pathology.
Depending on the nature and duration of containment, this type of caging can be very stressful for primates and should be avoided if possible. The need for total collection from individual animals should be critically assessed and the use of less stressful methods carefully considered. For example, it may be acceptable to collect the total excreta from a group and take an average value or use elimination time instead. It may also be possible to train animals to produce a urine sample at a given time (Anzenberger & Gossweiler 1993, McKinley et al. 2003), or to harvest spot samples from clean trays placed underneath home cages during periods when animals are housed singly for other purposes.
If the use of metabolism cages is unavoidable for well-justified scientific reasons, then the details of the animals' confinement should be discussed with the veterinarian, animal care staff and the person charged with advisory duties in relation to animal welfare. Their use should be described and justified in the review of protocols or programmes of work by the licensing authority and/or the ERP or IACUC. Every attempt should be made to refine the cage environment and the way the cages are used. Some key principles are recommended below.
Cage dimensions, materials and structure
The dimensions, design and construction of metabolism cages should be carefully considered so as to reduce their impact on animal welfare. As a basic minimum, the cage dimensions should be large enough to permit freedom of movement and normal postural adjustments. The animals should be able to stand, sit and perch; turn around; rest/ sleep in a natural position; and stretch out comfortably without being restricted by the cage sides or roof. Food and water containers should not interfere with an animal's ability to perform any of the activities stated above. Shelves should be incorporated to allow perching and to provide a comfortable resting place. Non-absorbent materials, such as Trespa™, should be used rather than metal as they may be more comfortable.
The duration of confinement should always be the minimum necessary to achieve the scientific objectives. It is difficult to specify an upper limit because this depends on the individual animal, the nature of the study and the nature of the cage. In most cases sufficient collection of radi-olabel will be achieved in five to seven days (typical target is 90% recovery or greater). Checking the data from samples obtained as sampling proceeds (e.g. daily), should allow a scientific basis for removal from the metabolism cage on a study-by-study basis, which may reduce the confinement period necessary. The ERP or IACUC should be encouraged to set limits on the appropriate maximum acceptable confinement period for any animal, or to approve periods in excess of accepted standards (e.g. >7 days) in each individual case. Current practice is to give primates a minimum recovery period of two weeks, for every week held in a metabolism cage. However, individual animals respond differently and their behaviour should be monitored to determine the ideal period.
For some studies it may be possible to pair animals and accept paired results. Animals in metabolism cages should always be able to see (and hide from) at least one other primate by being housed opposite or alongside other animals, or ideally, by placing the metabolism cage in the home enclosure. Primates in metabolism cages should be provided with positive contact from familiar carers in addition to the contacts needed to conduct the study. This should be an integral part of the establishment's primate socialization programme.
Enrichment, such as chews and toys that have proved attractive for the animals, can and should be provided, although how these are presented needs to be considered with regard to the potential absorption of urine/faeces/ radiolabel. The roof of the metabolism cage can be customized to create a food puzzle to allow foraging and counter understimulation in this type of caging. Reinhardt (1993) found that distributing the standard biscuit ration for caged rhesus macaques on the mesh ceiling of their cages instead of in food-boxes resulted in a marked increase in foraging time (80–290-fold, depending on the shape of the biscuits). Food wastage was minimal and could be avoided altogether by fixing a rim around the top of the cage.
Individual animals should be assessed to ensure that they are likely to be able to cope with the restricted environment of a metabolism cage. Individuals should be calm and confident, with no apparent behavioural problems and little or no apparent anxiety when separated from their cage-mate(s). The animals should be carefully monitored throughout their time in the cage for any adverse physical or behavioural effects such as reduced activity.
Some scientific procedures require the collection and examination of full thickness sections of skin. In order to minimize the potential adverse effects on the animal, as small a piece of tissue as possible should be removed using a new disposable biopsy punch for each sample. Preferably select a site that is relatively hairless (e.g. the medial aspect of the thigh) and not a frequent site for grooming such as the back. A small enough biopsy should be removed such that the wound does not need suturing or stapling, thus avoiding the introduction of foreign materials that the animal may subsequently remove. The small wound can then be readily closed by using tissue adhesives, so a site where there is little tension across the wound should be selected.
Telemetry
Primates are frequently used in telemetry studies, including those carried out to fulfil the requirements of regulatory bodies. Telemetry is widely viewed as benefiting science and animal welfare because it can reduce stress caused to animals (e.g. by restraint), enable reductions in animal numbers and provide indicators of animal wellbeing to help implement humane endpoints. However, telemetry can require invasive procedures such as implantation surgery. It can also involve single housing and the use of jackets, which can cause distress. Thus, although telemetry is described as a refinement, this will not be the case unless the technique itself has been fully refined.
Detailed advice on refinements in telemetry can be found in the JWGR report on telemetry. Part A of the report (Morton et al. 2003) covers experimental design, selection and design of devices, and surgery including analgesia and postoperative care. Part B (Hawkins et al. 2004) details refinements in husbandry and care for animals used in telemetry procedures. This includes behavioural pre-screening to ensure that individuals are suitable for projects and are not implanted unnecessarily, and maintenance of stable group housing. The report explains how to pair or group-house animals implanted with devices that transmit at the same frequency, for example, by using devices that can be turned on and off one at a time. Telemetry companies have developed multi-frequency telemetry devices, which allow telemetered animals to be group-housed. Jackets with sensors for heart rate, electrocardiogram and respiratory parameters are also being developed, which could remove the need for surgery to implant devices. The technology is developing rapidly, so it is important to maintain an up-to-date knowledge of developments that could benefit animal welfare (see www.lal.org.uk/telemetry/index.html).
Food and fluid control protocols
Food or fluid rewards are used as motivators in many studies that require primates to perform conditioned behaviours for extended periods (National Research Council 2003). It has been traditional in such studies to schedule access to the daily food or fluid ration to increase the animals' desire to obtain the reward and ensure they perform the behaviours for the full duration of the daily trial. However, restricting access to food or fluid can elicit behavioural and physiological responses that compromise animal health and welfare and may affect the scientific data being collected. Food or fluid control may further affect animal welfare if the procedures impact on the husbandry of the animals (e.g. where animals on fluid control protocols are individually housed). For these reasons food and fluid control protocols require special authority in some countries.
In recent years, researchers have found that, in some circumstances, animals will work for a reward without the need for food or fluid control, and that performance can be maintained if the reward is sufficiently attractive. For example, common marmosets have been trained to respond to stimuli in order to gain access to a preferred fluid (banana milkshake) that is not encountered elsewhere in the diet. This reward has been used to sustain daily performance on a task for up to 18 months (Williams et al. 2006). It is therefore important to challenge the need for the traditional approach and to explore alternative options.
Clearly, it is better for the welfare of the animals if food and fluid control can be avoided and this would represent a very significant refinement if it could be universally applied to many research projects that use the paradigm. The viability of alternative approaches for motivating macaques to work for extended periods, and the opportunities for refinement of food and fluid control protocols where they are deemed necessary and justified are discussed in Prescott et al. (in preparation).
Recommended principles for refinement include:
The justification for food or fluid control should always be critically examined by the research team and ERP/ IACUC. Such protocols should only be used if it can be conclusively demonstrated that they are the only effective way to accomplish the scientific goals of the study, and this justification should be kept under constant review. Individual animals should be carefully selected with regard to their health and behavioural suitability for learning and performing the tasks required. Rewards should be highly desirable since the desirability of the reward will influence the animal's performance and hence the level of food or fluid control necessary. The protocol should be introduced gradually. The level and duration of food or fluid control for each individual animal should be minimized. Animal health and welfare should be rigorously monitored and easily accessible records maintained. Staff should receive appropriate training not only to carry out the procedures, but also to understand the animal welfare implications of food and fluid control protocols, and to monitor the animals for adverse physical or psychological effects.
6 Long-term maintenance of primates
The lifespan of primates compared with other laboratory animal species is relatively long and some may be housed in the laboratory for many years, either as breeding or experimental animals. This includes telemetered or surgically-prepared primates on long-term studies, and animals authorized for reuse. In such circumstances, there are particular welfare, scientific and ethical issues that must be addressed (see Morton et al. 2003, Hawkins et al. 2004). The welfare concerns stem from inappropriate housing and husbandry and, for experimental animals, their continued use in scientific procedures. Implications for the science include the long-term impact of ageing and health status on the animals' physiology, behaviour and cognition. There may also be technical issues for instrumented animals, for example, in relation to the life of a device or its battery.
Reusing animals in consequential experiments presents a specific ethical problem. Reuse could decrease the numbers of animals used overall. However, the actual or potential harms to the individual animals concerned (e.g. from long-term laboratory housing, effects of procedures) must be weighed against the welfare cost of obtaining and housing (and in some cases surgically preparing) naïve animals. In the UK, reuse is subject to legal constraints (Home Office 2000) and a reduction in overall numbers of primates used is not considered to justify causing a significant increase in harms for individual animals. The difficulty is in deciding what constitutes ‘a significant increase in harms’. This is an issue that needs to be discussed with animal care staff, ideally through the local ERP or IACUC, and kept under regular review. All of the potential harms, throughout the life of each animal, needs to be taken into account, including constraints from laboratory housing.
Deciding how long to keep an animal
Limits need to be set on the length of time that individual animals are maintained, whether in a research or breeding environment. Each laboratory or breeding facility should set out, after discussion between the veterinarian, animal care staff and scientists: (a) the maximum time animals should be kept and/or used; and (b) what is done with them if euthanasia is not required by the experiment. It is not easy to define the criteria to use for these decisions except where there are clear veterinary grounds for euthanasia. It is recommended that the following list of factors be addressed.
Justification for keeping animals long-term.
Contingent suffering (e.g. from long-term laboratory housing) should be factored into the harm/benefit assessment of research projects, and experiments should never be started without a clear idea of how long the animals will be maintained and what will happen to them at the end. This will then need to be kept under review (say at 6 months) to check the continued suitability of the animals and their environment. The view of the Working Group is that it is unacceptable to keep animals that have cranial or other implants for prolonged periods of time without generating scientific data. Experiments should be planned to use the animal in the shortest possible time, maximizing the amount of data collected and minimizing the suffering to the animal. Where animals are not likely to be used further in experiments, reasons for continuing to maintain them should be explored (e.g. to help stabilize a group or provide company for another animal).
The health of the animals.
Good health and welfare are a prerequisite for continuing to keep animals, and the physical and psychological health of all primates held long-term should be regularly reviewed by the attending veterinarian as part of an appropriate clinical and behavioural examination. The temperament of the individual animal also needs to be taken into account.
Husbandry and care system and social environment.
Time limits need to take into account the husbandry system in which the animals are housed. Opportunities for socialization with staff also need to be taken into account. The husbandry system must provide a good quality and quantity of space: small, un-enriched cages are not acceptable for long-term housing. Also important is the length of time the animals have been kept in the laboratory, and the behavioural consequences arising from this. For example, some primates become harder to manage as they get older; they may become more aggressive and then be singly housed as a result. The longer primates are kept in single housing the more likely they are to develop stereotypies or other behavioural problems (Lutz et al. 2003). The Working Group considers single housing to be unacceptable and, therefore, any requirement to singly house animals should be a significant factor against their long-term maintenance or use.
Retirement to non-experimental facilities
The majority of laboratory primates are euthanized following procedures either because their tissues are required as part of the experiment, or because their welfare would be compromised as a result of the procedures that are carried out. In situations where this does not apply, for example, in the case of surplus ex-breeding or stock animals, it may be possible to ‘re-home’ animals as an alternative. This concept is not new and a number of laboratory primate species including common marmosets, squirrel monkeys, capuchins, stump-tailed macaques and chimpanzees have been successfully retired to zoos, sanctuaries, professional collections or breeding colonies.
Retirement and relocation of primates can be beneficial for both animals and staff. However, because of the nature of the animals and the need to find facilities that can properly accommodate and look after them in the long-term, they should only be relocated if it is clear that the process will be in the best interests of the individual animal(s). Each animal's age, state of health, previous life experience and the physical and social conditions in which the animal will be kept, needs to be carefully considered case-by-case. Further information is given by Prescott (2006).
Animals should only be sent to suitably qualified and experienced primate keepers at reputable registered zoos and sanctuaries. Ideally, the new owner should have a recommendation from the relevant committee of a primatological society (e.g. Captive Care Committee of the International Primatological Society) or from an animal welfare organization that already relocates primates (e.g. International Primate Protection League). They need to have the necessary licences (e.g. UK Dangerous Wild Animal Licence for most primate species other than marmosets), have access to veterinary advice, and be able to catch and handle animals if they require treatment. Their premises should be inspected before considering the release of any animals to them. The housing and husbandry requirements set out in this report should be met in full and, in particular, animals should never be relocated to an establishment where they will be kept on their own.
Any establishment that intends to relocate its primates outside a laboratory environment should develop a clearly defined and documented procedure that enables all issues relating to animal welfare (including the suitability of each individual animal's character), health and safety, the prospective owner and any participating organizations to be addressed. Careful and timely planning of the whole process is important as is the follow-up of relocated animals to monitor success and deal with any problems (Seelig & Truitt 1999, Brent 2004, Prescott 2006a). In addition, it is beneficial to regularly review in-house socialization, habituation and training programmes, using input from follow-up studies, to ensure that they facilitate safe and humane relocation.
For re-homing to continue to be successful, receiving establishments and any intermediaries, need to participate enthusiastically, with full knowledge of the reasons for the original use of the animals. To avoid the process being misrepresented, any statements that will be made about the process need to be agreed by all parties prior to receipt of the animals (Kerwin 2006).
7 Euthanasia
Principles and methods of euthanasia for primates are set out in the following publications.
Recommendations for Euthanasia of Experimental Animals (Close et al. 1996, 1997) http://www.lal.org.uk/pdffiles/LA1.pdf, http://www.lal.org.uk/pdffiles/LA2.pdf
Report of the American Veterinary Medical Association (2001) http://www.avma.org/issues/animal_welfare/euthanasia.pdf
Euthanasia of Animals Used for Scientific Purposes (Australian and New Zealand Council for the Care of Animals in Research and Teaching 2001) www.adelaide.edu.au/ANZCCART/news/Euthanasia.pdf
All of these discuss the animal welfare concerns with regard to euthanasia. The ANZCCART document notes that: ‘When animals are killed, both the method of euthanasia, particularly the time taken to produce unconsciousness, and how the technique is performed can result in animals experiencing pain, distress, fear and anxiety. Further, the animal's psychological response to the environment in which it is killed, including interactions with other animals and humans and how it is handled, can result in emotional distress.’
The AVMA report states that ‘It is our responsibility as veterinarians and human beings to ensure that if an animal's life is taken it is done with the highest degree of respect, and with an emphasis on making the death as painless and distress free as possible.’
All three documents emphasize the importance of a painless and rapid induction of unconsciousness, followed by cardiac or respiratory arrest and the ultimate loss of brain function. The animal must be killed humanely and efficiently. Staff members who are charged with carrying out the procedure should be competent in the selected method and be willing and mentally prepared for the task. Good training and supervision are therefore essential, with emotional support, if needed.
Skilful and empathetic handling, and selection and preparation of the environment for euthanasia can help to minimize any potential distress. Euthanasia should be conducted in an area separate from other primates, not least because in the event of a problem, an animal may become stressed which will affect other animals in the room. Guidance on humane killing under the UK Animal Scientific Procedures Act (ASPA) states that, as a general rule, animals of any species to be killed should be removed from the immediate presence of others (Home Office 1997).
The most acceptable method of euthanasia for primates is intravenous injection of an overdose of anaesthetic, such as sodium pentobarbitone, but always refer to veterinary staff for advice. Sedation should be considered if it is likely to reduce animal stress or help with staff safety.
When animals are euthanized, use the opportunity to conduct a postmortem as part of a continued health screen. Morphometric measures (e.g. adrenal weights, bone density) can also be made at postmortem for comparison between healthy and diseased states, or of the effects of different housing regimes.
Whenever animals are euthanized, full use of their tissues and blood should be made, or these should be frozen for future use. Setting up tissue banks and data-exchange networks (both in-house and between user and breeding establishments) with good communication systems, which help match supply and demand, is an effective means of coordinating, optimizing, reducing and refining primate use. A number of EU-funded projects involve the establishment and maintenance of banks of primate tissues and body fluids (e.g. www.eupeah.org, www.euprim-net.eu and www.inprimat.org). The NIH National Institute of Ageing (NIA) also has a repository of primate tissues for research on ageing.
8 Source and supply of primates
There is considerable potential to apply reduction and refinement to the sourcing and supply of primates (Prescott & Jennings 2004). Many establishments importing and using primates already recognize this, and devote considerable resources to promoting refinement down the supply chain. Factors to consider include the health status of the animals (and whether they are wild-caught or captive-bred), the suitability of individuals for the study procedures, husbandry and care at breeding and supplying centres, quarantine, transport and acclimatization (Animal Procedures Committee 2006a). All of these can affect both animal welfare and scientific data. Thus, the availability and financial cost of animals should not be the only concerns.
As a basic principle, animals should never be ordered until experiments have been adequately planned and justified, the anticipated requirements (e.g. number of animals, sex and age) are realistic and accurately predicted, and funding is secure. Good coordination between users, breeders and suppliers, and between and within multidisciplinary research teams, is essential to ensure that animals are never wasted, either because they are surplus to requirements, or inappropriate for the research. Matching the individual characteristics of the animals to the specific demands of the research programme, and providing feedback to the supplier on how animals coped with experimental programmes, is also very important. For example, some animals may be more suited to some behavioural tasks than others by virtue of their temperament or intelligence. This sort of information can be entered on to the animal's passport and incorporated into future breeding plans. The need for a single sex or specific age of animals should also always be examined.
Selection of breeding centres
Primates can be bred in-house (in the laboratory where they are used) or bought in. There are a number of advantages to in-house breeding; there is greater control over housing and husbandry, the primates are acclimatized to their environment, socialization and training specific to their purpose can begin at an early age, and there is no need for transport and quarantine. However, some facilities may be too small or lack the necessary experience to breed primates, and balancing supply with demand can be a problem. Where primates are acquired from elsewhere, it is important to recognize that standards of husbandry and care vary enormously in breeding centres around the world. Some offer high-quality accommodation with creative enrichment and husbandry practices thoughtfully designed to reduce adverse effects, and these should be used in preference. At others, however, animals are maintained in barren enclosures with little or no enrichment, young macaques are weaned and removed from their mothers very early (e.g. at 6 months), and animals are ‘conditioned’ by housing them singly in very small cages for long periods prior to export (Prescott 2001, Animal Procedures Committee 2006a). Primates from some centres have to undergo lengthy, multi-staged transport from the country of origin to the laboratory where they are used and where they may undergo additional quarantine procedures (Wolfensohn 1997, Prescott 2001, Scientific Committee on Animal Health and Welfare 2002, Honess et al. 2004b).
Users and regulatory authorities can have a significant influence on conditions at breeding and supplying centres by visiting and assessing standards and choosing their supplier accordingly (Table 10). For example, some users insist on increasing use of F1 animals for breeding, innovative environmental enrichment programmes, pair or group housing for ‘conditioning’, shipment of compatible animals in pairs and efficient transport procedures that reduce journey times. Visits provide the opportunity to observe the facilities and the animals, to encourage improvements in animal welfare, and to offer support and advice in this regard. Closer communication between breeding/supplying and user establishments helps to ensure continuity of husbandry and care (e.g. to help with rapid acclimatization to conditions at the user establishment); facilitates socialization, habituation and training programmes; and helps balance supply and demand. The breeder/supplier may view users and regulators differently so both should visit, and regular feedback should be encouraged among all concerned. It is easier to do this when breeding/supplying establishments are in the country of use and/or if there is an authorization process involving licensing and inspection that identifies the best sources. Since not all customers or authorities may be able to visit their suppliers regularly, the Working Group considers that it would be helpful to set up a network of communication, to provide up-to-date information on individual centres.
Factors to consider when assessing the suitability of breeding or supplying establishments

Factors to consider when assessing the suitability of breeding or supplying establishments

Capturing wild primates and maintaining them in captivity is known to be stressful to the animals (Johnson et al. 1973, Laudenslager et al. 1999, Suleman et al. 2004) and can often result in high morbidity and mortality (Suleman et al. 1995, 1999, 2000, Tarara et al. 1995, Uno et al. 1989). Use of wild-caught primates, either to replenish breeding stock or as experimental animals, has therefore been identified as a serious animal welfare concern (Prescott 2001, Scientific Committee on Animal Health and Welfare 2002, Prescott & Jennings 2004, International Primatological Society 2007) and, in 1997, the Council of Europe published a ‘Declaration of Intent’ to limit the use of animals to those which are purpose-bred, and to encourage initiatives and measures to end the use of wild-caught primates (Council of Europe 1997). There are also issues relating to the quality and health status of the animals that are likely to impact on the validity of research results (Table 11). The capture of wild primates also presents a threat to the survival of some species and local populations, as the demography of a population can be severely altered with repeated removal of animals of specific age and sex (Cyranoski 2000, Institute for Laboratory Animal Research 2003). Notwithstanding this, in countries such as Mauritius where non-indigenous primate species are seen as ‘pests’, breeders may be required to capture a requisite number per year as a condition of their licence to export.
Comparison of purpose-bred and wild-caught primates in relation to use in scientific procedures
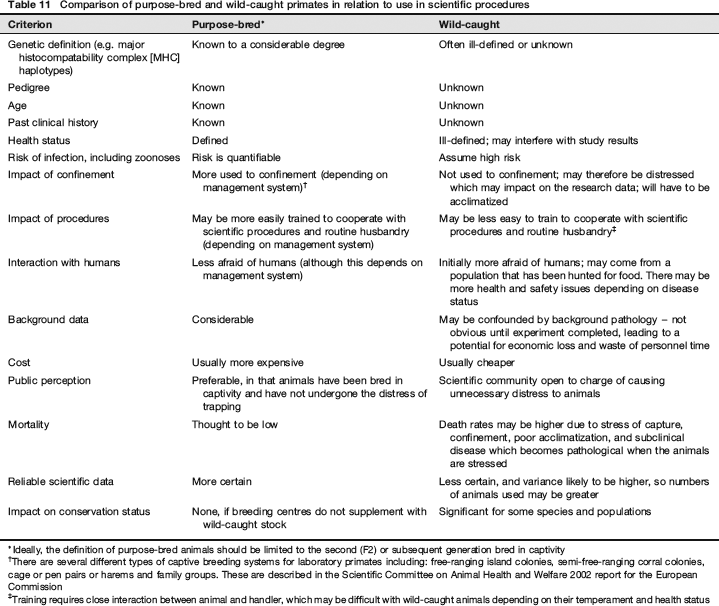
Comparison of purpose-bred and wild-caught primates in relation to use in scientific procedures

Ideally, the definition of purpose-bred animals should be limited to the second (F2) or subsequent generation bred in captivity
There are several different types of captive breeding systems for laboratory primates including: free-ranging island colonies, semi-free-ranging corral colonies, cage or pen pairs or harems and family groups. These are described in the Scientific Committee on Animal Health and Welfare 2002 report for the European Commission
Training requires close interaction between animal and handler, which may be difficult with wild-caught animals depending on their temperament and health status
Significant numbers of wild macaques are captured to supplement captive breeding colonies. Thus, 70–80% of the long-tailed macaques imported into the UK in the years 2001–2004 were the first-generation offspring of wild-caught parents (Hansards 2004). Breeding colonies should aim to be self-sustaining and such a move has to be linked to a clearly defined strategy to decrease the reliance of breeding and supplying establishments on wild populations. This requires trapping quotas to be reduced, and significant and increasing proportions of first-generation offspring retained for breeding second-generation stock (Animal Procedures Committee 2006a).
While capture of wild animals continues, centres that trap should ensure that any persons trapping primates are adequately trained and competent in humane methods of capture (see American Society of Mammalogists 1998 and Jolly et al. 2003 for guidance). National authorities in the country of origin should encourage proper handling procedures in both the field and holding facilities, and institute a system of licensing of trappers and exporters, with provisions for inspection of procedures and facilities, taking the Guidelines of the International Primatological Society (2007) as a minimum standard. Establishments using primates can help drive these changes by ordering only purpose-bred animals (ideally F2) and explaining the reasons to breeding and supply centres.
Use of wild-caught primates in procedures
In some countries such as the UK, the use of wild-caught primates is prohibited except where exceptional and specific scientific justification can be established and the majority of primates used in the rest of the EU are also now purpose-bred (Scientific Committee on Animal Health and Welfare 2002). The European Primate Research Network (EUPREN), for example, states ‘For ethical and scientific reasons, it is generally recognised that wild-caught primates are inappropriate to use…’. However, wild-caught primates are still used in scientific procedures in countries such as Japan, USA and South Africa. Moving to the use of purpose-bred primates for all scientific procedures would have a significant positive impact on both animal welfare and science (see Table 11), and there has been considerable progress with this in recent years. Not only would this solve the animal welfare issues associated with capture and confinement of wild animals, but it would also reduce the need for the additional health screening, quarantine and remote handling practices that add to the animals' stress.
For all of the reasons stated above, the Working Group endorses the UK Home Office position that wild-caught primates should not be used in experiments. If a species is not available from a purpose-bred colony, then the potential for using an alternative, purpose-bred species should be explored. Other national authorities that regulate primate breeding, supply, import and use should also endorse this approach.
Quarantine
During their lifetime, laboratory primates may be held in a variety of situations that serve to quarantine and/or isolate them for various lengths of time. Most forms of quarantine are intended to protect public health through health screening and treatment of the animals, or to detect disease in the animals, but the process can have a considerable impact on animal welfare. The nature and magnitude of the impact is largely dependent on the nature of the quarantine required, and on the way that animals are housed and handled and the facilities managed.
Some exporting centres and facilities that are set up to deal specifically with large batches of imported primates, usually house the animals singly in small cages with little or no enrichment. On arrival, imported primates are likely to be disoriented and fearful of their new surroundings, and stressed by isolation from former social companions, coupled with fear of unfamiliar animals and humans in close proximity. Primates from the wild or outdoor settings also appear to be more sensitive to acoustic stress. The use of metal cages (which are inherently noisy) during these quarantine periods may be a particular source of stress, and every effort should be made to reduce cage-related noise. For recently imported primates, human attention may be an additional stressor and there may be insufficient time for staff to develop appropriate and non-threatening interactions with the animals. Negative experiences and behavioural pathologies that develop during quarantine periods can have long-term effects and may counter the value of refinement and enrichment efforts applied subsequently.
These are very serious welfare issues for the animals and need to be addressed by the relevant competent national authorities and those exporting, importing and using animals. As yet, the opportunities to refine quarantine and associated procedures have not been properly explored, perhaps because the period is considered to be short and for a defined purpose, and/or because of practical problems associated with disease control. Nevertheless, although the quarantine period may seem short to humans, an animal will not interpret its experience in this way. Furthermore, in some countries such as the USA, quarantine may be repeated for individual states and/or research establishments, thereby compounding its effects. 10 Some shipments of primates may be subjected to as many as four separate quarantine periods, i.e. pre-export, mandatory import, state and establishment.
The US Center for Disease Control (CDC) requires that imported primates be held under quarantine for at least 31 days, at any one of about 30 registered facilities. Animals are held behind physical pathogen barriers; there are special procedures for handling or disposing of potentially contaminated material; animals undergo testing for Mycobacterium tuberculosis and filovirus; and are monitored for clinical signs of zoonotic diseases of major public health significance. If test results are questionable, the required quarantine period is extended, until additional testing clarifies the health status. Buyers may ask the quarantine facility to conduct additional disease testing or treatment, beyond what is required by law, during the 31-day post-entry quarantine period. Most recipient facilities additionally quarantine the primates at their own facility, often in similar conditions to the mandated quarantine, for some variable period (3-30 days), regardless of where the animals are from; this is not required by law. Some individual states, such as California, also require quarantine prior to the primates crossing into the state.
It is recommended that the necessity of each quarantine episode, and for the number and type of tests requested, be reviewed and reduced if possible. It may be that importers of primates, as in the UK, can maintain their own registered quarantine facility for direct import, as this avoids at least one journey and single housing/regrouping experience. Regulators and importers need to be more explicit about their disease control requirements in order to avoid holding animals unnecessarily under quarantine with the associated constraints on their husbandry and care.
Pairing or group-housing animals in quarantine is a major refinement and some users and quarantine centres have already begun to challenge traditional requirements and have moved away from caging animals singly. One reason given for single housing is concern that the spread of disease within an animal room cannot be controlled if animals are paired or grouped. However, in the event of individual clinical or subclinical disease cases, it is common practice to treat near neighbours or the entire room, so this reason should be challenged if treatment or other actions would be the same whether or not animals were singly-housed. A second reason given is that there is lack of time to establish compatible pairs or groups. This too should be challenged, since where animals have come from the same breeding colony, it should be possible to form new pairs among juveniles or even infants fairly quickly, or better still, arrange for animals to be pre-paired or pre-grouped at the colony of origin.
If single housing is unavoidable for justifiable veterinary or human health reasons, then this should be for as short a time as possible, and animals need to be able to see and hear their conspecifics (see Section 3.3). Since the duration of individual housing generally depends on the time taken for the required health tests, ways of expediting test results should be explored. Resources should be targeted to the welfare and care of animals to ensure that their behavioural, psychological and physical needs are satisfied as far as possible.
Rooms in many primate import stations were designed and built to accommodate an entire shipment with the smallest legally permissible cages occupying all of the available space within rooms. This makes it difficult to provide newly imported animals with greater freedom of movement, or to provide more space to facilitate other forms of environmental enrichment. Furthermore, staff may believe that keeping animals in a large enclosure, where they can avoid a squeeze-back mechanism, will result in handling difficulties. Capturing animals in larger cages should not be a problem if animals are adequately habituated and socialized to humans early in life (see Section 2.4), although the stress of shipment may make them less cooperative.
Some quarantine facilities are reluctant to provide much environmental enrichment believing that enrichment items will be too difficult or too time-consuming to sanitize and replace. This concern is far outweighed by the benefit for the animals' welfare and subsequently the quality of the science. Therefore, enrichments should be devised to overcome any such problems. One way of doing this is to focus on a ‘softer’ environment and disposable materials such as hay, wood, cardboard and paper (avoiding printed paper). For animals who have been living under natural conditions, such enrichments may afford more familiarity and comfort-value than artificial objects or devices. (Note that the response of wild-caught animals to environmental enrichment may differ from that of purpose-bred animals and they may take longer to interact with enrichment items.)
New cages/enclosures need to be designed, and old ones modified, with the specific needs of newly imported animals in mind (see Section 3). For example, a panel or strip of muslin, burlap or opaque plastic has been used successfully as a visual barrier on the outside of the enclosure to calm frightened primates. It is especially important during this time to monitor reactions toward other animals in facing enclosures.
If a prolonged period of quarantine is required in the importing country, and the health status of animals has been defined, it may be possible to commence positive reinforcement training before the end of the quarantine period. This will be easier if the animals have been exposed to habituation/socialization and positive reinforcement techniques (e.g. training them to accept food from the hand) at the exporting centre.
Pre-export ‘conditioning’
Pre-export conditioning is a particular form of quarantine that covers the processes used in source countries to assess the animals' physical health before transport and relocation, and to habituate group-housed animals to transport and other forms of caging. The manner in which conditioning is carried out, and the duration of the conditioning period, depends on a number of factors. These include the husbandry and other management practices at the centre; the health status of the colony; the diseases endemic in the country of origin; the real or presumed needs of the customers; and the import/export regulations of the importing/ exporting country.
Some centres maintain animals in their groups prior to supply, and carry out all routine health check procedures in the home enclosure. However, in many cases, animals are housed individually in very small, barren cages for up to 45 days or more and this is a serious concern. None of the reasons for the pre-export conditioning period actually require animals to be maintained either in single housing or in a barren environment, and the Working Group considers such housing to be unacceptable. In some cases, there seems to be confusion over what the regulatory and customer demands are. Greater clarity on these points is therefore essential.
If cage conditioning is unavoidable for a justifiable veterinary reason, then animals should be housed in compatible pairs or groups with appropriate environmental enrichment to allow and encourage a wide range of different behaviours (see Sections 2 and 3). The animals can then be transported with their companion and be kept with the same animal on arrival, which will help reduce stress during relocation (Schaffner & Smith 2005).
Health status: Herpesvirus simiae (B virus) free colonies
The implementation of a number of the refinements described in this document depend on the animals being of high health status, such that human workers are not put at risk by close contact with the primates in their care. Using only captive-bred rather than wild-caught animals has clear benefits in this respect. There is particular concern over the potential transmission of H. simiae to humans and not all colonies are H. simiae-free. Although many laboratories test their macaques frequently for antibodies, a negative antibody test result is not viewed as entirely reliable due to the possibility of delayed seroconversion. In addition, there are differences in sensitivity and specificity of the various available serological tests leading to variations in interpretation. This has had a major impact on the way Old World primates are housed and handled in some countries. For example, in the USA, all macaques are treated as though they are potential H. simiae carriers, whatever their origin or prior test results.
The Working Group recognizes the problems caused by the zoonotic potential of H. simiae and the seriousness of the infection in humans. In order to facilitate refined husbandry practices and safeguard human health, a priority in the long term is for all colonies to be H. simiae-free. However, there is a serious dilemma in that the development of such colonies has associated welfare concerns. The testing and segregation scheme used to produce and maintain them is analogous to quarantine in principle and effects. There is an additional concern that infants are weaned at 6–12 months (sometimes earlier), and housed individually for a period of initial screening/testing. To mitigate the effects of early weaning in these circumstances, animals should be weaned into small groups rather than individually. It is also recommended that methods of diagnosis and interpretation of H. simiae screening results are regularly reviewed in order to ensure that results are as accurate as possible (Schapiro 2000, Wolfensohn & Gopal 2001, Ward & Hilliard 2002).
9 Transport and acclimatization
Most primates will undergo transportation at some time in their life, either from one country to another, between establishments or buildings, or from room to room. Transport can be a stressful experience for primates (e.g. Wolfensohn & Honess 2004); even cage change can cause prolonged disturbances in physiological parameters, such as heart rate (Line et al. 1990). Transport also involves changes to the environment into which the animals are relocated and changes in staff, which compounds the transport stress. For primates travelling to the USA or EU from Asia, journeys can be as long as 70 h, with loading/unloading between stages which can cause stress and presents opportunities for problems to occur (Prescott 2001). In all circumstances, the objective should be to move the animals in a manner that does not jeopardize their well-being and ensures their safe arrival at their destination in good health, with minimal distress (Claassen 1994, Reilly 1998).
There are national and international legislative requirements and guidelines relating to animal transport, but these usually reflect minimum standards and it is important to try to further reduce the stress and discomfort involved. A Working Group of the Laboratory Animal Science Association (Swallow et al. 2005) has produced detailed guidance on transporting laboratory animals, including primates, as has a committee of the National Research Council (2006). The Working Group endorses the recommendations in both these guidance documents. In summary, journeys between establishments should be planned in advance to minimize disruption to the transported animals' welfare. This is especially important for the long, multi-stage journeys experienced by some primates. A journey plan should be produced detailing the responsibilities of various individuals involved in the transport. This should be agreed with the intended recipient and should give accurate details of events at each stage of the journey. It should also provide instructions for contingencies in the event of delays and adverse events, and include contact details for all those involved in the journey. Communicating clearly and often with all parties involved will help ensure a smoother journey for the animals.
Transport should be by the most direct means possible, with the minimum number of stages between departure and arrival at the final destination. For transport by air, use of private charter jets as opposed to commercial air carriers can reduce the number of stops or plane changes before the final destination and hence opportunities for delays or mishaps. Time in transit and waiting times should be kept to a minimum; however, the quality of the journey is just as important as the duration. Primates should be supervised until they are shipped, and properly attended by competent persons while awaiting transfer.
Transport containers should be of an appropriate size and construction for the journey length (see Swallow et al. (2005) for recommended container dimensions and designs, including a new design of container for macaques that has been adopted by the International Air Transport Association (2005)). Animals should, where possible, be transported in compatible pairs or family groups since this may reduce stress (Schaffner & Smith 2005, Swallow et al. 2005). Young macaques are usually transported at an age when they are unlikely to fight, so pairing should not be difficult.
Prior to transportation, animals should be habituated to transport containers for short periods of time to minimize levels of fear and apprehension, particularly prior to longer journeys. This can be achieved by first placing the container in the primates' enclosure and then confining the animals for short periods. Such activities can be included in the general habituation and training programmes.
Bedding, browse or fruit can be provided during longer journeys including international transport, but it is essential to research whether there are any relevant import restrictions. The US Department of Agriculture prohibits the entry of many plant materials from primate source countries, but tamarind, any type of onion, coconut fibre and wood-wool are acceptable from nearly any source, as are tinned fruits and vegetables.
An individual history file (see Table 9) should accompany each animal. It is also recommended that information on the animals' husbandry and care (e.g. diet, enclosure type and furnishings, established pairing or groupings) is shared between sender and recipient well in advance of shipment, so that arrangements can be made for animals to become quickly acclimatized on arrival at the new premises. On arrival, a suitable acclimatization period is essential to allow animals to overcome any stress imposed by transport. It should also allow sufficient time and opportunity for them to become accustomed to a new physical and social environment, and allow a period of adjustment for behavioural and physiological variables to return to normal before scientific investigations are carried out (Clarke et al. 1988a,b, Wolfensohn 1997, Scientific Committee on Animal Health and Welfare 2002, Honess et al. 2004a). This will also allow time for positive reinforcement training.
There are insufficient scientific data available to define ideal acclimatization periods and more information on this is required. Animals should be monitored post-relocation, with particular attention paid to abnormal and self-injurious behaviour, activity patterns, changes in the dominance hierarchy and alopecia (Honess et al. 2004b). Whatever the acclimatization period, it should at the very least be sufficient such that food and water consumption is normal for the species, and individual animals are interacting with each other, their environment and care staff.
10 Summary principles for refinements in husbandry, care and common procedures for primates in research
The document provides specific recommendations for improving the welfare of primates used in research. Many of these are encompassed within more general principles which are provided below. Consultation with a primate behaviour specialist can provide valuable help with implementation of these principles and is therefore recommended.
Understanding the animal and nature of suffering as a basis for refinement
Understanding the biology and behaviour of primates and recognizing the signs of suffering and good and poor animal welfare, is crucial for designing and implementing refinement (and some reduction) strategies. This understanding should be used to examine every aspect of the animals' lifetime experience to identify ways of reducing potential suffering and improving welfare. Procedures that cause pain and/or distress in humans may have similar effects in other primates; conversely some procedures might cause more or different distress in primates than they would in humans. Staff should be able to recognize when an animal's physical health or psychological wellbeing is compromised (including the subtle signs) and know what to do about this. Each primate should be treated as an individual with needs that will vary throughout life and which should be responded to appropriately. There should be regular meetings between research teams and animal care staff to discuss refinements, review progress and ensure uptake of recent developments. The implementation of refinement should be monitored and documented, and an individual identified to have overall responsibility for the refinement strategy.
Harm/benefit analysis
The justification for primate use through the harm/ benefit analysis should include a cumulative assessment of all potential adverse effects during the lifetime experience of the animal. This should encompass those due to transport, housing, husbandry and procedures such as handling and restraint, as well as suffering that results directly from experimental procedures and their effects.
Training animals
Positive reinforcement training, combined with an ongoing socialization and habituation programme, can provide both animal welfare and scientific benefits. Thus, it is important to make positive reinforcement training an everyday part of the laboratory routine for husbandry, veterinary and scientific procedures. Positive reinforcement is always preferable to negative reinforcement, or reinforcement with previous deprivation.
Staff
There should be sufficient staff to care for and monitor each individual animal effectively. All staff working with primates should be well-trained and competent to interact with the animals in a positive manner and to carry out the procedures required of them. There should be a programme of continuing professional development for all staff who plan or review projects involving primates, and/or care for or carry out procedures on them (including animal care staff, veterinarians, scientists and members of ERPs and IACUCs). The programme should ensure that all relevant staff members are up-to-date with refinements in husbandry and experimental protocols and with recent developments in understanding primate behaviour.
Housing and care
Primates should be housed in compatible social groups, unless there are compelling animal health or welfare reasons for single housing, and disruption to established groups should be minimized. Managers of primate units should establish husbandry routines that allow the adequate detection and monitoring of aggression within social groups and enable staff to forestall potential problems. Scientific and/or procedural reasons for single housing should always be rigorously challenged. Primates should be provided with sufficient quantity and quality of space to enable them to carry out an extensive range of species-specific behaviours, taking into account the natural social structure and environment of the species. It is important to recognize that minimum enclosure dimensions in regulatory guide lines and codes of practice are minima not optima, and some existing standards may not reflect current best practice. Existing housing should be reviewed for opportunities to provide additional usable space of an appropriate design and the enclosures adapted accordingly. Refinement of housing, like refinement of procedures should be monitored, assessed and documented. Records of successful (and unsuccessful) enrichments and refinements should be kept and this information shared with other users and breeders of primates. Diet should be varied and balanced. Food presentation should be managed so that it occupies the animals and stimulates them mentally and physically. Consultation with an expert nutritionist is recommended.
Procedures
Studies should be designed to have the mildest possible adverse impact on the animals taking into account all aspects of all procedures whether scientific, veterinary or husbandry. This should include technical elements, identification methods, any requirement for single housing, the duration of the study, adverse effects and humane endpoints. Handling and restraint procedures should be refined to avoid welfare problems and confounding variables. For example, many species of primates can be trained by positive reinforcement to enter a restraint chair or transport box so that physical capture and restraint, or use of a crush back cage, is unnecessary. Net capture and the pole and collar method are not recommended.
Long-term use, re-homing
Primates should not be ordered, nor experiments started, without a defined plan regarding the length of time that the animals will be maintained in the laboratory and the fate of animals at the end of the experiment. Animals (particularly if instrumented) should not be kept long-term in anticipation of being used at some time in the future. The potential for re-homing animals to a well-managed zoo or private collection with appropriate experience in husbandry and care of primates should always be seriously considered. The sharing of tissues within and between establishments should be maximized as a means of reducing animal use. A Pan-European database should be established to facilitate tissue/data sharing.
Sourcing, transport and quarantine
In the interests of quality of science, animal welfare and human health and safety, primates should be of defined and high health status (in particular H. simiae-free). This facilitates many refinements because risks to human health and safety are significantly reduced. Wild-caught primates should only ever be used under exceptional and highly specific circumstances, for example, in procedures closely directed at the benefit and conservation of an endangered species. The responsibility for animal welfare begins with the sourcing of animals, not just with their arrival at the user establishment. Sources of animals should be chosen carefully. It is important to insist on appropriate weaning age, the setting-up of stable social groups post-weaning, socialization/habituation to humans and social housing for pre-transport conditioning and quarantine procedures, working together with breeders and suppliers to challenge any real or perceived requirement for single housing. Visits to breeders and suppliers should be made as an integral part of this process. Transport and quarantine can be significant stressors for primates, so it is essential to assess the potential for improving transport, quarantine and acclimatization periods to reduce stress, and for refining all associated husbandry and procedures. Requirements for repeated quarantine should be challenged.
Funding and publication of research into primate needs and 3Rs strategies
Research funders and the industries that use primates should increase their support for research into reduction, refinement and replacement of primate use. Information on such issues should be more widely published and disseminated.
Footnotes
Acknowledgements
Thanks to Graham Tobin and Jim Lawson.
