Abstract
Chronic pain and subfertility are the main symptoms of concern in women with endometriosis. In order to find new therapeutic options to suppress the pain, translational animal models are indispensable. We have developed a new automated, experimental setup, with full consideration for animal wellbeing, to determine whether operant behaviour can reveal abdominal hyperalgesia in rats with surgically-induced endometriosis, in order to assess whether abdominal hyperalgesia affect behavioural parameters. Endometriosis was induced by transplantation of uterine fragments in the abdominal cavity. Control groups consisted of sham-operated rats and non-operated rats. We have developed an operant chamber (Skinnerbox) which includes a barrier. The rat can climb the barrier in order to reach the food pellet, increasing in this way the pressure to the abdomen. We show that endometriosis rats collect significantly less sugar pellets when compared with the control rats after the introduction of the barrier. In the Skinnerbox experiment, we showed that in a positive operant setting, the introduction of a barrier results in a contrast of operant behaviour of endometriosis rats and control groups, perchance as a result of abdominal discomfort/hyperalgesia due to surgically-induced endometriosis. This is a promising start for the further development of a refined animal model to monitor abdominal discomfort/hyperalgesia in rats with surgically-induced endometriosis.
Endometriosis is a common, chronic gynaecological condition, defined as the extra-uterine presence of functional endometrial-like tissue.1,2 Studies have shown that this ectopic tissue develops its own autonomic and sensory innervation and that upon mechanical stimulation of these lesions, an increase in sensory nerve response has been observed.3–5 This indicates that mechanical stimuli, as for example physical activity or pressure (e.g. defaecation, coitus) can trigger pain.3–5 The pain symptoms in endometriosis are diverse and have a considerable impact on the quality of life.68 Current therapies are mainly aimed at providing pain relief. Since it is an oestrogen-dependent disease, most treatment modalities are aimed at creation of a hypo-oestrogenic state.2,9,10 Apart from the disabling side effects, treatment success-rates are often disappointing.1,2 Therefore, there is a pressing need for more effective treatment options.
Animal models are valuable in endometriosis research in order to develop these novel therapies.11–15 One of the translational challenges is that animals cannot report pain or discomfort.16,17 Therefore traditionally, animal responses to inflicted noxious stimuli are studied. 12 Still, this behaviour can be subtle and easily be missed during its normal explorative behaviour. Besides, the behavioural response of an animal to a noxious stimulus is highly variable.17,18 Another important aspect to consider, is that objective measures such as withdrawal response time to a noxious stimulus measure hypersensitivity rather than the pain itself. 17 Hypersensitivity to a noxious stimulus is often seen in chronic pain patients, although it is not as often reported as continuous or spontaneous pain.19,20
Unfortunately, monitoring spontaneous pain is considered challenging in animal pain studies and can be addressed by studying spontaneous behaviour.11,15,17,18,21–25 After irritation of the peritoneum, chemical inflammation of the uterus, artificial ureteral calculosis or abdominal surgery, changes in spontaneous behaviours have been observed including for instance hunching, licking of the abdomen, stretching and arching of the back, loss of balance during grooming or rearing and pushing the abdomen to the floor.12,18,22–25 Also, the absence of behavioural aspects can be indicative of pain/discomfort. Exploration of the environment is typical baseline behaviour that is lost after administration of painful stimuli.12,15 Feeding and weight gain are diminished, grooming is reduced and social interactions are altered.12,15 However, care should be taken, as recognizing and interpreting pain induced behaviour is considered strenuous and subjective, especially when it comes to mild to moderate pain or stress-related symptoms.16,18 This is illustrated for instance by Giamberardino and coworkers, who have analysed spontaneous pain behaviours from continuous videotape recordings and were not able to show changes in pain behaviours after the induction of either endometriosis or sham-endometriosis and control rats. 21 These findings were corroborated by Lopopolo et al., who also did not observe spontaneous pain behaviour or muscle hyperalgesia in a rat model of endometriosis. 23 The fact that rats with surgically-induced endometriosis act ‘normally’ has prompted us to design an open field test which allows the demonstration of abdominal hyperalgesia, while guarding the wellbeing of the animals. We have studied behavioural changes during positive operant conditioning using a Skinnerbox paradigm, designed to reveal signs of abdominal hyperalgesia by means of introducing a barrier the animals have to overcome while collecting sugar pellets. We hypothesized that endometriosis rats, compared to control rats, collect significantly less sugar pellets in a Skinnerbox after the introduction of a barrier due to abdominal hyperalgesia.
Materials and methods
Animals
The experiment was conducted with adult, female WAG/Rij rats (150–200 g; n = 40) obtained from Charles River, The Netherlands. This WAG/Rij strain is an inbred Wistar strain with genetically identical rats.
All animals were randomly housed pairwise in Macrolon cages (type 3; Komeco, Alkmaar, The Netherlands) in a temperature-controlled room (21–25℃) with a reversed 12 hour dark/light cycle (light off phase between 08:00 and 20:00 h) and free access to sterile water and food. Shelters and cage enrichment (EnvitoDry®) were provided. Rats were allowed to acclimatize for at least one week before surgery. After surgery, rats were housed individually for one day, before they were reunited again.
Our study was approved by the ethical committee of the Radboud University's Animal Experiment Committee (approval number RU-DEC 2012-061). This committee is recognized by the Central Authority for Scientific Procedures on Animals. All experiments were carried out in the Radboud Research Facility in which an animal welfare body is stated to oversee the performance of animal experiments and animal welfare in accordance with the Animal Experimentation Act. Animals and their housing and husbandry were observed daily and their wellbeing was recorded in standardized notebooks.
Surgical procedure
Cyclicity of the female rats was monitored daily through vaginal swabs and microscopical evaluation of the smears. Rats received daily subcutaneous injections with oestradiol (3 µg/kg subcutaneously) for three days prior to the day of surgery, to increase endometrial thickness to facilitate the transplantation of the uterine explants. In a pilot study we established that the oestrous cycle stage of the female rats did not affect the collection of sugar pellets at the different barrier heights (data not shown).
Surgical procedure was performed according to the well-accepted and studied surgical method to induce endometriosis by Vernon and Wilson (1985). 26 The day prior to surgery, the surgical area was prepared by removing the hair using clippers. On the day of surgery, prophylactic Buprecare (0.05 mg/kg subcutaneously) analgesia was administered. Rats were anaesthetized with inhalation anaesthesia (a mixture of N2/O2 (66/33%) and isoflurane (2.5%)) and were placed on heating pads. The surgical procedure was performed aseptically, and the surgical area was sterilized using a Betadine solution in 70% alcohol. An incision was made along the Linea Alba, through the skin and muscle layer. The left uterine horn was exposed and a 2–3 cm section was tied off and removed. In the autotransplantation group, this uterine horn was then opened lengthwise and divided into five pieces of 4 × 4 mm2 which were sutured to the inner abdominal wall (n = 2) and the mesentery (n = 3).
In the allotransplantation procedure, both uterine horns were kept intact and five pieces of exogenous uterine tissue from a donor rat were grafted. Further, a sham-operated control group was included. In these rats, the left uterine horn was removed without re-introducing uterine explants into the abdomen. Instead, five stitches were placed on the same locations as the uterine explants.
The wound was closed by suturing the abdominal wall and skin separately. After surgery, the rats received Buprecare (0.05 mg/kg subcutaneously) daily for an additional two days. No abnormal behaviour of the rats was observed as a result of the administration of Buprecare and prior to the initiation of the Skinnerbox experiment. Lesions were allowed to develop for two weeks until the initiation of the Skinnerbox evaluations.
At the end of the experiment, rats were euthanized by exsanguination under anesthesia, and the presence of endometriosis lesions was confirmed.
Experimental design
Animals were randomized per study group. All animals were similarly treated and assessed. Sample size was estimated based on literature and experience. The experimental design in the Skinnerbox experiment:
Group 1 (n = 10): control group with no surgical procedure. Group 2 (n = 10): sham-operated group with removal of one uterine horn without uterine tissue transplantation. Group 3 (n = 10): endometriosis group with endometriosis induction by allotransplantation. Group 4 (n = 10): endometriosis group with endometriosis induction by autotransplantation.
Skinnerbox experiment
We compared operant behaviour of WAG/Rij rats with surgically-induced endometriosis to non-operated control and sham-operated rats, in order to assess the most distinguishing settings of the operant paradigm.
The rats were trained and tested in 15-minute sessions on 18 consecutive working days in a standard operant chamber with two levers and two signal lights (Figure 1). Rats were not fasting beforehand and no external noxious stimuli were applied. All experiments were carried out in the dark phase of the light–dark cycle. In the centre of the chamber, a barrier was placed. This barrier had an opening with a variable size (2 × 2, 2.5 × 2.5, 3 × 3, 4 × 4 cm2) which was situated at different heights (9, 16 and 20 cm) above ground level. During the testing period, a green light on one side of the box was switched on after which a sugar pellet was released. At the moment the rat consumed the pellet, the light on the other side of the box switched on and another sugar pellet was presented on the opposite side. A computer counted the collected number of pellets; the experiments were therefore not blinded.
Schematic representation of the operant chamber model: (a) top view; (b) side-view of the separator.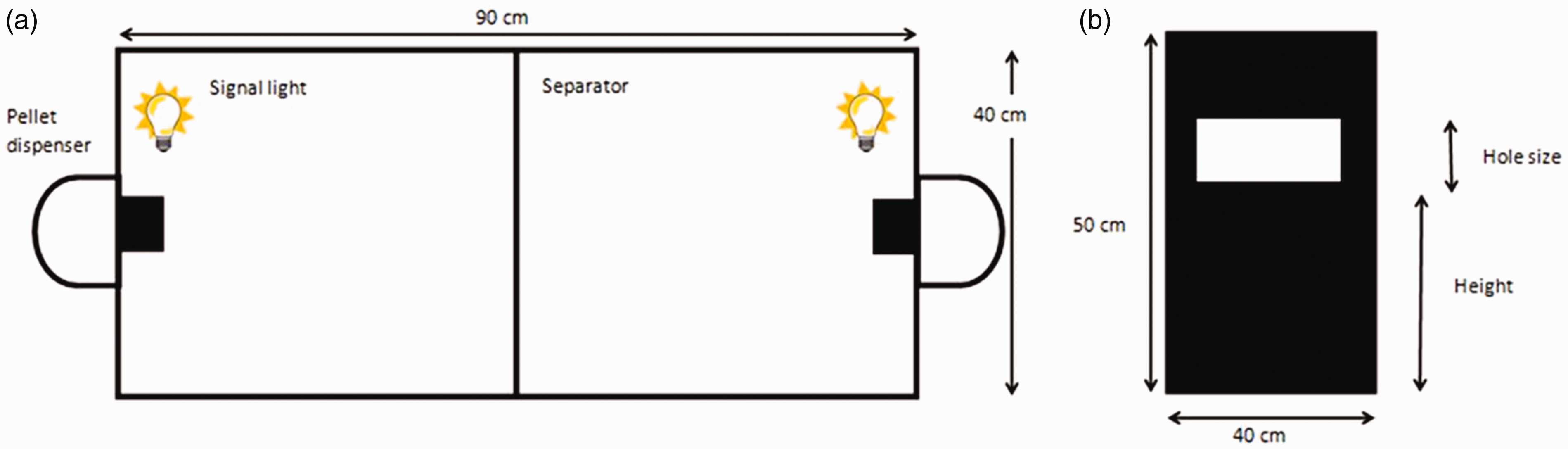
The rats started the experiment with two training days in which they had to cross the chamber to collect pellets, without the barrier present. In the following 16 days, the barrier was put in place. The height and opening size were varied throughout this testing period. Only one parameter was varied at a time.
Initially, the height was set at 9 cm with an opening size of 4 × 4 cm2, after which the opening size was reduced to 3 × 3 cm2 (one day) and 2 × 2 cm2 (one day) and subsequently increased again to 2.5 × 2.5 cm2 (two days) and 4 × 4 cm2 (five days). Finally, in the presence of a 4 × 4 cm2 opening size, the height was increased from 9 to 16 cm (two days) and 20 cm (two days). The design is schematically presented in Supplemental Figure 1.
Statistical methods
Statistical analysis was performed using SPSS version 22.0; 27 p values of < 0.05 were considered significant.
We used a two-way repeated measures ANOVA using training day as a within factor and study group as between factor to measure group differences during the training days. Two-way ANOVA was used to test for group differences for the height/opening size combinations used in the barrier. The number of sugar pellets collected was set as a dependent variable with the study group and barrier characteristics (hole size or height) as fixed factors. Post-hoc Tukey's multiple comparisons tests were conducted when significant group differences were seen.
Results
The operant behaviour of WAG/Rij rats with endometriotic lesions was monitored in the operant chamber experiment for signs of behavioural changes. Analysis of the data revealed no interaction between the groups and hole size or barrier height (p = 0.336 resp. p = 0.516), meaning that all groups follow the same operant behaviour pattern (Supplemental Figure 1).
During the two training days, in absence of the barrier, no significant differences in number of pellets between the groups (F (3, 36) = 1.301; p = 0.29) were observed. On day two, the rats collected significantly more pellets than on day one (F (1, 36) = 122.6; p < 0.0001), which is illustrative of the ‘learning behaviour’ in which the rats were conditioned to cross the chamber after the light signal (Supplemental Figure 2). We found no interaction between the experimental condition and training days, indicating that all groups had a similar learning curve (F (3, 36) = 0.328; p = 0.805).
The presence of the barrier, barrier height and opening size had a significant impact on the amount of collected pellets (Figure 2). When analysing differences between the endometriosis state and the different hole sizes, we saw significant differences between the endometriosis and control groups when comparing pellet collection after passing the barrier at a height of 9 cm and opening size of 4 × 4 cm2 (F (3. 316) = 6.282; p < 0.001). Rats from the autotransplantation and the allotransplantation collected significantly less pellets than the sham-operated group (resp. p = 0.029 and p = .001). When compared to the non-operated control group, pellet collection was significantly different only in the autotransplantation group (p = 0.011). There was no difference between the sham- and non-operated groups (p = 0.899), nor between the autotransplantation and allotransplantation groups (p = 0.739).
Hole size settings. The number of collected sugar pellets (mean ± SD) is shown for the four different study groups on the different hole size settings of the barrier. Allo = allotransplantation. Auto = autotransplantation. *p < 0.05; ***p < 0.001.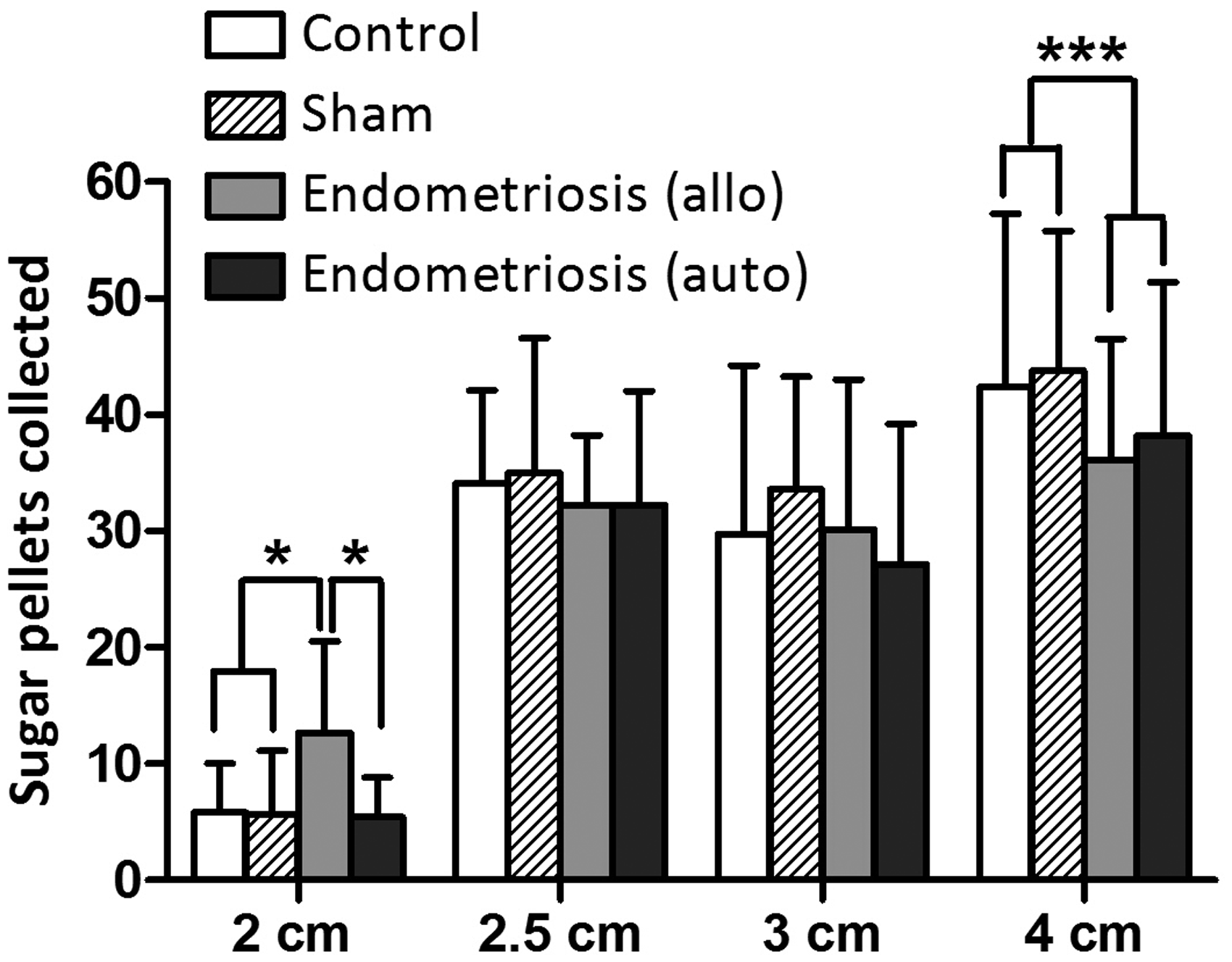
At the smaller opening sizes 3 × 3, 2.5 × 2.5 and 2 × 2 cm2, the frequency of pellet collection was further reduced (Figure 2). When comparing the pellet collection frequency between the groups at these opening sizes, no significant differences were observed at 3 × 3 cm2 (F (3, 36) = 0.443; p = 0.724) and 2.5 × 2.5 cm2 (F (3, 36) = 5.559; p = 0.644) opening sizes. In contrast, we found a significant difference when the opening size in the barrier was set at 2 × 2 cm2 (F (3, 36) = 4.4147; p = 0.013).
At a barrier height of 16 cm, and an opening size of 4 × 4 cm2, the difference in effects on operant behaviour was still observed between the test groups (F (3,76) = 3.519; p = 0.019) (Figure 3). Post-hoc test showed that the sham-operated and non-operated control groups did not differ from each other (p = 0.395). However, the allo- and autotransplantation group collected significantly less pellets compared with the sham operated group (resp. p = 0.046 and p = 0.024) but not with the non-operated control group (resp. p = 0.702 and p = 0.543). At a barrier height of 20 cm, the number of pellets collected in all groups was lower and the difference in the number of pellets was no longer statistically significant between groups. This appears to be mostly due to the fact that at this height, control rats also had increasingly difficulty to get across the barrier (F (3, 76) = 0.627; p = 0.600) (Figure 3).
Height settings. The number of collected sugar pellets (mean ± SD) is shown for the four different study groups on the different height settings of the barrier. Allo = allotransplantation. Auto = autotransplantation. *p < 0.05; ***p < 0.001.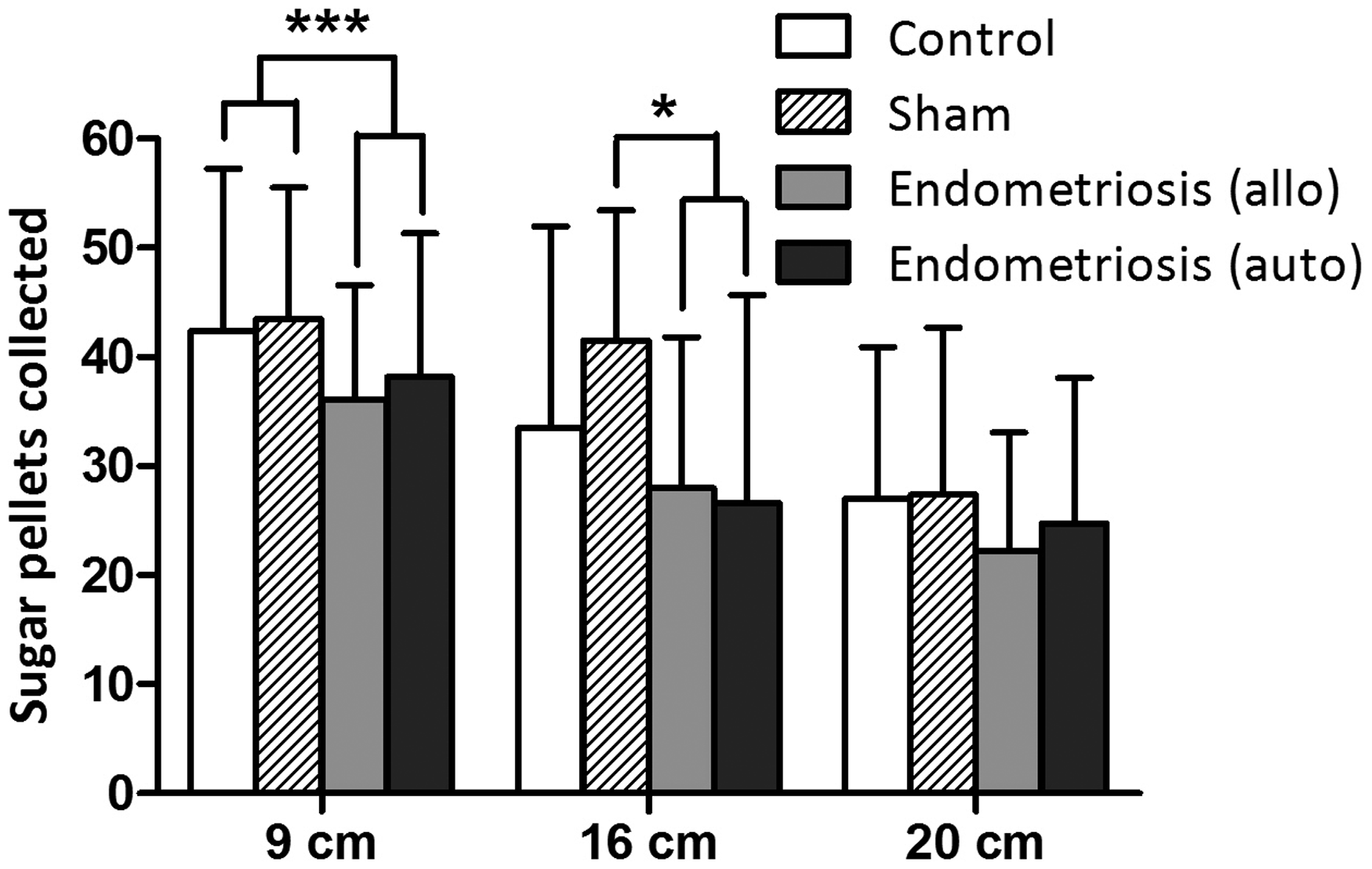
No correlation was observed between the total lesion burden and the number of sugar pellets collected.
Discussion
In this study, we aimed to find behavioural indicators of endometriosis-related abdominal hyperalgesia. To this end, we studied operant-induced behaviour in rats with and without surgically-induced endometriosis.
The results of our Skinnerbox experiment suggest that we observed an ‘endometriosis effect’ on operant behaviour. Likely, as a result of the presence of abdominal lesions in the endometriosis groups, an impact on behaviour was measured in a cage in which the animal had to climb over a barrier to reach food; when the barrier was left out of the operant chamber, no significant differences were observed between the groups. This indicates that the recovery time after surgery was sufficient and the surgical procedure and Buprecare did not affect behaviour and appetite. This is in line with earlier reports demonstrating that behavioural changes are only apparent within the first few days after laparotomy.19,28
After introducing the barrier in the Skinnerbox, both the barrier height and opening size significantly inhibited the operant stimulated collection of pellets by the endometriosis rats. Endometriosis rats consumed fewer pellets due to less frequent crossing of the chamber through the hole compared to the control groups. The hole size of 4 × 4 cm2 was the optimal size to detect the effect of endometriosis state on operant behaviour. When the opening was reduced to 3 × 3, 2.5 × 2.5 or 2 × 2 cm2, no group differences were observed anymore. Apparently, openings smaller than 4 × 4 cm2 resulted in fewer crossings due to physical limitations instead of abdominal hyperalgesia. Similarly, when the height of the opening in the barrier was increased to 16 or 20 cm, rats seemed to be physically limited in crossing the barrier independent of endometriosis state. Our findings indicate that a barrier with a 4 × 4 cm2 hole at 9 cm high is the most discriminatory setting to detect a difference in operant behaviour as a result of abdominal hyperalgesia in WAG/Rij rats.
We did not observe a significant correlation between the lesion burden of the rats and the number of sugar pellets they collected. Moreover, Lopopolo et al. also showed that there is no significant correlation between the number and size of cysts and the spontaneous pain behaviour as well as muscle sensitivity in endometriosis rats after ureteral stone implantation. 23 This is in line with the observation in women in which endometriosis-associated pain also does not correlate with disease severity, lesion size or location.29,30
In this study, we furthermore evaluated whether the operant conditioning behaviour was influenced in a different manner due to the surgical induction technique, that is through auto- (uterine horn removed, uterine explants grafted) or allotransplantation (no horn removed, uterine explants grafted) of endometrial tissue. In both, surgical induction techniques are used to create ectopic lesions, but one method involves the removal of one of the two uterine horns which is replaced by donor rodent endometrial tissue or even human endometrial tissue. 11 In our experiment, we did not observe behavioural differences between the two endometriosis groups. Therefore, we conclude that for endometriosis research, the operant chamber is applicable to both types of rat models.
A limitation of the operant chamber is that more factors might influence the outcome. For example: to press a lever to receive sugar pellets, the rat has to have an intact cognitive function, a good appetitive motivation, a stamina and good coordination of movements. 28 If the rate of pressing the lever is affected, does it mean that the appetite or activity level of the rat has been affected or that it is suffering more from its endometriosis?17,28
The relatively small and specific effects observed in our study are illustrative for the challenges encountered when studying pain-associated behaviour. Recognizing and interpreting pain behaviour can be very difficult and subjective, especially when it comes to mild to moderate pain symptoms.16,18 Most women report only pain that is induced by physical activity (e.g. defaecation or coitus), predominantly during menstruation. 21 Berkley and coworkers have used this phenomenon to design a rat model in which vaginal hyperalgesia is tested by using balloon distention during different phases of the oestrous cycle.3,12,31,32 Another technique for testing sensitivity is placing animals on a hot plate to test heat allodynia.12,15 We wanted to avoid the use of external stimuli, but studying rat behaviour is cumbersome, subjective, time consuming and labour intensive. The advantage of our operant chamber in comparison with these other techniques is that, in contrast to the subjective evaluation of secondary behavioural indicators such as for example licking, stretching, shaking or scratching, the Skinnerbox provides objective information regarding behavioural changes presumably as a result of abdominal hyperalgesia, namely the collection of sugar pellets as counted by a computer.12,16 Moreover, the Skinnerbox is an automated system, which does not require the continuous monitoring of the animals for prolonged periods of time, animals were tested for only 15 minutes per day, and fasting was not needed.
In this study, we aimed to find objective indicators for endometriosis-related abdominal hyperalgesia in operant behaviour in endometriosis rats. In our Skinnerbox experiment, we showed that in a positive operant setting, the introduction of a barrier (4 × 4 cm2 opening size; 9 cm height) results in a contrast of operant behaviour of endometriosis rats and control groups, perchance as a result of abdominal hyperalgesia due to surgically-induced endometriosis. This is a promising start for the further development of a refined animal model to monitor abdominal hyperalgesia in rats with surgically-induced endometriosis, in an objective and automated fashion, without having to compromise on animal wellbeing or comfort. Such a model could be valuable in studies aimed at developing and/or testing novel treatment approaches for endometriosis.
Supplemental Material
Supplemental material for An objective and automated method for evaluating abdominal hyperalgesia in a rat model for endometriosis
Supplemental Material for An objective and automated method for evaluating abdominal hyperalgesia in a rat model for endometriosis by Mieke AW van Aken, Patrick G Groothuis, Maria Panagiotou, Marcel van Duin, Annemiek W Nap, Tineke CM van Rijn, Tamas Kozicz, Didi DM Braat and Ard BWMM Peeters in Laboratory Animals
Footnotes
Acknowledgements
The authors would like to thank S Mulder for her contribution and support on the Skinnerbox experiments, TL Emmerzaal for his contribution to the data analysis and Prof. Dr M Ritskes-Hoitinga for her support on the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article DB and AN receive research support from Merck Serono and Ferring. DB further receives research support from Goodlife. These research grants were not related to the research described in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
