Abstract
Murine aggression has profound implications on animal welfare and husbandry. This report examines how three distinct combinations of environmental enrichment – wheel, igloo and tunnel; wheel, igloo, and tunnel with nesting; and tunnel with nesting – affect aggressive behaviour in CD-1 male mice. We found that combining wheel/igloo/tunnel enrichment with nesting or replacing the wheel/igloo with two tunnels while maintaining the nesting enrichment reduced aggression. These findings not only suggest how enrichment can improve the welfare of aggressive male mice but also emphasise the need for further research to determine the optimal combination of enrichment.
Introduction
Intermale aggression between cage-mates is a natural behaviour in mice essential for establishing hierarchy. 1 However, aggression can cause mild signs, such as ruffled fur or minor wounds, to severe injuries requiring euthanasia. Standard husbandry practices, including removing the aggressor or single-housing all cage-mates, can mitigate harm but also raise welfare concerns owing to social isolation and increased stress. 2
Aggression is influenced by multiple factors,1,3–9 including environmental enrichment10,11 and genetic background, 1 yet the specific impact of enrichment remains unclear. Given the well-documented high aggression levels in CD-1 males 1 which were housed in our facility, we report on the effects of three different enrichment conditions on their behaviour, that is, wheel/igloo/tunnel (W + TbE), wheel/igloo/tunnel plus nesting (W + T + NbE) and tunnel plus nesting (T + NbE). Less aggression was observed under W + T + NbE and T + NbE conditions. These results suggest that incorporating nesting materials plays a crucial role in mitigating conflict. Our findings highlight the importance of refining enrichment strategies to improve animal welfare aiming to preserve social housing.
Animals, materials and methods
Animals
CD-1/Crl (referred to as CD-1) and C57BL/6J male mice were imported from Charles River (UK) and kept in our AAALAC-accredited, UK Home Office-compliant, barrier-protected, specific-pathogen-free animal facility. Animal management was performed using PyRAT software (Scionics).
Cage environmental enrichment
Mice were housed in controlled conditions (20–24°C, 45–65% humidity, 12 h light/dark cycle with 1h twilight) with ad libitum food (2016 Teklad, Envigo, UK) and reverse osmosis water in ventilated cages (Green/Emerald line, Tecniplast, UK). Bedding (Safe Bedding Asp, Aston Pharma, UK) and enrichment (Datesand, UK) included clear handling tubes, Fast Trac (wheel/igloo), aspen bricks, nestlet, tunnel clips, cardboard tunnels, and sizzle nest. Mice were housed in one of three conditions: W + TbE (aspen brick, wheel/igloo, clear tunnel), W + T + NbE (aspen brick, wheel/igloo, cardboard tunnel, nestlet, 10 g sizzle nest) or T + NbE (aspen brick, two cardboard tunnels, tunnel clip, nestlet, 10 g sizzle nest). Animals were monitored (visually) daily, with the animals removed from the cage and inspected weekly. Since the mice were not removed from the cage daily, we cannot rule out the possibility that the weekly inspections captured more severe aggressive encounters. Per UK Code of Practice, group-housed mice had ≥330 cm2 floor space, 45–60 cm2 per 20–30 g mouse, with a maximum of five per cage.
Data extraction and analysis
CD-1 male aggression was analysed over 10 months using imported three-week-old CD-1 males for experiments. Data were collected during three periods: 2020/2021 (June to March, W + TbE: 569 C57BL/6J, 321 CD-1 mice), 2022/2023 (June to March, W + T + NbE: 371 CD-1 mice) and 2023/2024 (June to March, T + NbE: 314 CD-1 mice). Given that data were collected at different timepoints, which might imply different experimental conditions, no statistical test was performed; instead, the data collected are presented as absolute numbers.
Fight wounds, typically found on the back or tail and often accompanied by bleeding, were documented in PyRAT. To avoid duplicate entries, wounds were recorded only upon their initial discovery, with the experimental unit being the individual animal. The average age of the mice upon the initial discovery was 10.9 ± 5.53, 12.14 ± 5.35 and 14.6 ± 3.55 weeks for W + TbE, W + T + NbE and T + NbE, respectively. Mice were housed in groups of two to five, with average group sizes of 3.06 ± 0.99, 2.5 ± 1 and 4.33 ± 0.5 for the W + TbE, W + T + NbE and T + NbE conditions, respectively. The methodology did not account for the potential variation in incidence due to cage density effects. A scoring system for the severity of the wounds was not used. Known aggressors were isolated; otherwise, all males were singly housed. Superficial wounds were untreated, while deeper or clustered wounds received pain relief per veterinary advice. Males with exposed muscle were euthanized.
Results
Between two and five CD-1 males were housed in W + TbE cages (Figure 1(a)). Over 10 months, 77 of 321 CD-1 males in W + TbE cages showed aggression-related wounds (Figure 1(di)), consistent with prior studies. 1 In contrast, signs of aggression were absent in 569 C57Bl/6J males also housed in W + TbE cages (Figure 1(di)). We modified W + TbE cages by adding nesting material and replacing the plastic tunnel with cardboard (W + T + NbE; Figure 1(b)) or by adding nesting material, removing the wheel/igloo linked to increased aggression in CD-1 males 8 and increasing the numbers of cardboard tunnels (T + NbE; Figure 1(c)). Among 371 CD-1 males in W + T + NbE cages and 314 in T + NbE, only seven and six males displayed fight wounds, respectively. The number of males with fight wounds was lower when compared with males housed in W + TbE cages (Figure 1(di) to (diii)).
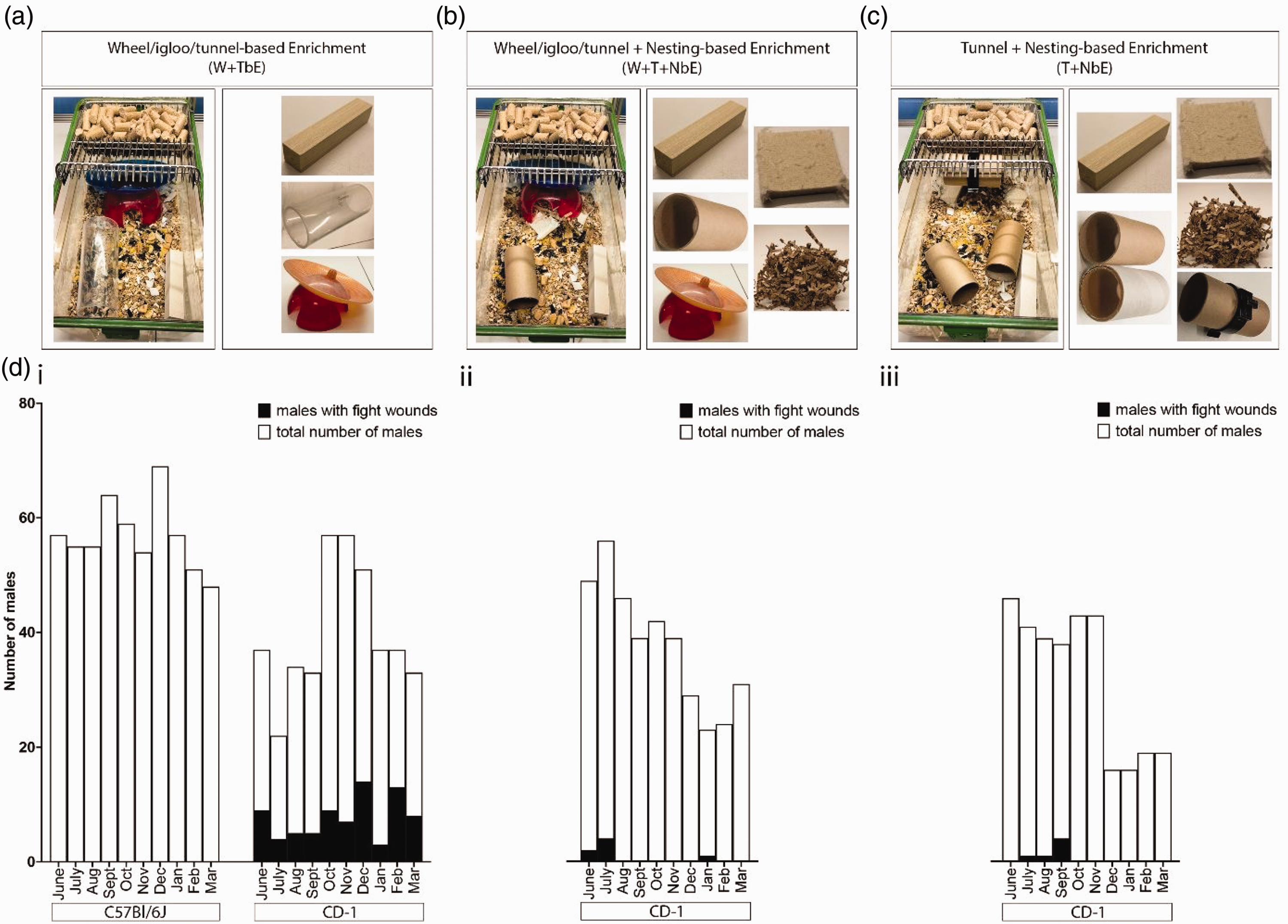
Environmental enrichment and male aggression. (a) W + TbE: wheel/igloo, plastic tunnel, aspen brick. (b) W + T + NbE: wheel/igloo, cardboard tunnel, aspen brick, 10g sizzle nest, nestlet. (c) T + NbE: two cardboard tunnels, tunnel clip, aspen brick, 10g sizzle nest, nestlet and (d) Fight wound data: total males (white) versus males with fight wounds (black) over 10 months. (di) C57BL/6J and CD-1 in W + TbE; (dii) CD-1 in W + T + NbE; (diii) CD-1 in T + NbE.
Discussion and conclusion
Aggression among co-housed male mice is a major concern in laboratory settings, resulting in stress, injuries and euthanasia. Current management strategies, such as removing aggressive mice or single-housing, are costly and increase staff workload. Therefore, optimising co-housing conditions is essential. CD-1 mice are particularly susceptible to aggression, and our report suggests that incorporating nesting materials, with or without a wheel or igloo, lowers this aggressive behaviour (Figure 2). Materials such as nestlets, sizzle nests and cardboard tunnels promote shredding, which may help redirect aggression. Future research could explore whether increasing access to these materials further encourages nesting behaviour and, hence, reduces aggression, as is the case for BALB/cAnNCRLBr. 12
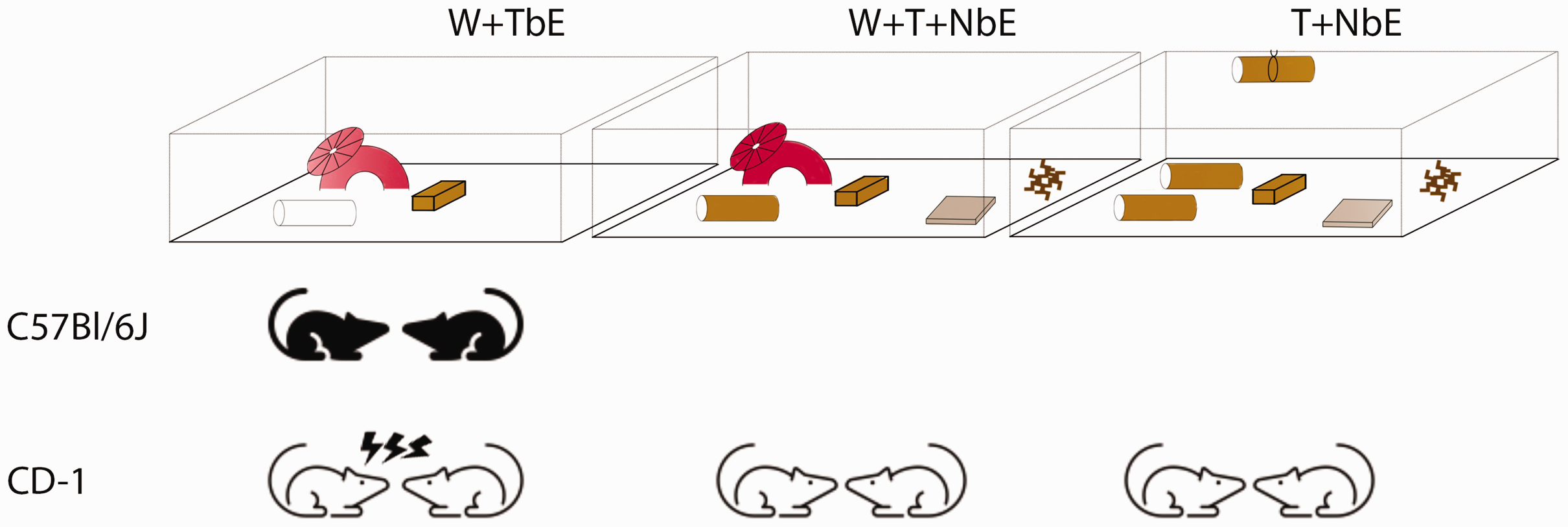
Schematic summary. Illustration of C57BL/6J and CD-1 male aggression in W + TbE (wheel/igloo, plastic tunnel, aspen brick), W + T + NbE (wheel/igloo, cardboard tunnel, aspen brick, 10g sizzle nest, nestlet) and T + NbE (two cardboard tunnels, tunnel clip, aspen brick, 10g sizzle nest, nestlet) housing conditions.
The enrichment conditions presented in this report were not tested concurrently. W + TbE condition was used during 2020–2021, coinciding with the reduced research activity during the COVID-19 pandemic. W + T + NbE and T + NbE were tested in subsequent years, potentially introducing confounding factors. Further studies with stress indicators and continuous cage monitoring to observe aggressive encounters directly are needed to determine whether transitioning to W + T + NbE or T + NbE effectively mitigates aggression, reducing the need for single-housing. This report has significant implications for managing aggression in co-housed male mice.
Footnotes
Acknowledgments
We thank the Isogai lab members for providing mice for this study, the NRF members for discussions, and Mateo Velez-Fort, Troy W Margrie and Tom Otis for reviewing the manuscript.
Author contributions
Conceptualisation: YI and EMA. Methodology: AV, CG, SM and TO’M. Data curation: AV. Analysis: AV and CG. Writing: CG and EMA. Writing – review: YI, SM and TO’M.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Data availability
To access data, interested parties can contact the corresponding author (
Ethical considerations
All experiments carried out were reviewed by the SWC Animal Welfare and Ethical Review Body.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sainsbury Wellcome Centre Core Grant from the Gatsby Charitable Foundation and Wellcome (grant numbers GAT3755 and 219627/Z/19/Z).
