Abstract
Jugular catheterization is a common procedure used under experimental conditions. However, there is considerable variation in the reported techniques, particularly for grower pigs (>40 kg and <60 kg) when larger volumes of blood per sample (>10 mL) are required. This paper provides a complete methodology including the use of current equipment and anaesthetic regimen for grower pigs. This surgical jugular catheterization method was carried out in 30 large white grower pigs. Firstly, the pigs were habituated to human handling for at least two weeks prior to surgery. Animals were sedated and anesthetized. Following intubation, an incision was made in the jugular fossa, and the jugular vein was located. A catheter was then inserted and fixated. The wound was stapled and the catheter line secured to the back of the neck. The pigs recovered fully from the surgery and the catheters remained patent for the duration of the blood sampling period (min 72 h). Twenty millilitres of blood were collected every 15 min, taking approximately 2 min per pig. No haemolysis was detected in any samples. Jugular catheterization of pigs using this procedure proved successful both in terms of animal recovery and quality of samples. Catheters remained patent and pigs remained calm during sampling.
Pigs are increasingly being used as models for human studies, 1 due to their anatomical and physiological similarities. 2 For studies requiring frequent and accurately timed blood samples, venous catheterization is essential, as venepuncture at frequent intervals is known to be stressful to the animals, potentially leading to misleading results. In addition, blood sampling in pigs has long been considered to be a difficult and uncertain task due to the lack of suitable superficial blood vessels. 3 Different approaches have been used for different age groups (live weights) of pigs. For example, in young piglets (up to 4.8 kg), catheterization of the percutaneous external jugular vein is common, particularly for a single sample. 4 Piglets of a similar weight (1.5–5.75 kg) have also been subject to non-surgical jugular vein cannulation to collect 3 mL per sample, with minimal anaesthetics. 5 Similarly, a rapid and non-surgical procedure was used for piglets weighing around 6.2 kg using the Vacutainer system with wire guide-sampling approximately every 30 min. 3 For larger pigs (lactating sows) venous cannulation of the mammary gland has enabled collections of 10 mL per sample three times daily. 6 Similarly, Phillips et al. 7 used home-made catheters made out of medical-grade microbore tubing in ear-vein catheterized sows to collect 20 mL per sample once daily. In adult/grower pigs there are significant amounts of subcutaneous adipose tissue beneath the skin in the jugular groove. Hence, the jugular vein may be 3–6 cm below the skin. Typically the diameter of the jugular vein in pigs of this size might be 5–6 mm. Thus while transcutaneous access to the jugular vein is possible there is significant risk of laceration to the vein and uncontrolled haemorrhage. Blood collection is routinely performed by transcutaneous venepuncture of the jugular vein when a single blood sample is needed. However when multiple samples are needed over a period of hours or days then surgical catheter placement as we describe is desirable.
From an anatomical perspective, pig blood samples have been taken from the following vessels, including the ear vein, 7 saphenous artery,8,9 mammary vein 6 and jugular vein.3–5,10–12 However, the mammary vein and ear vein methods do not typically allow for frequent large quantities (∼20–30 mL) of blood to be sampled within a short time.
For a single sample, puncture of the jugular vein using the Vacutainer® system (BD Diagnostics, Franklin Lakes, NJ, USA) has become the most commonly used technique which requires restraint such as the snout snare. However, in situations where repeated blood sampling is required, this technique is inappropriate. Restraining pigs for any duration of time has been shown to result in increased stress hormones, such as cortisol, which may induce variations in biochemical, haematological and hormonal metabolites. 13 Catheterization procedures using physical restraint without anaesthesia exist, 3 though these are more appropriate for piglets and smaller pigs. Therefore, to reduce restraint-associated stress and to allow for repeated blood sampling, catheterization of a vein becomes essential.
The aim of this work was to develop a technique for jugular vein catheterization in grower pigs, which would allow multiple blood samplings of 20–30 mL per sample within a short time period – 4 h in this case.
Materials and methods
All procedures were approved by the University of Queensland Animal Ethics Committee (AEC Approval No. NFS/364/13/ARC), and were conducted under veterinary supervision.
Anatomical localization
The location of the external jugular vein was approximated as shown in Figure 1.
4
The triangulation technique was initially used, however due to extensive biological variation between animals in terms of the depth of the external jugular vein, the percutaneous external jugular vein catheterization triangulation technique was not sufficient to accurately locate the jugular vein.
Photograph of a dissected grower pig’s neck in dorsal recumbency showing landmarks used to identify the external jugular vein.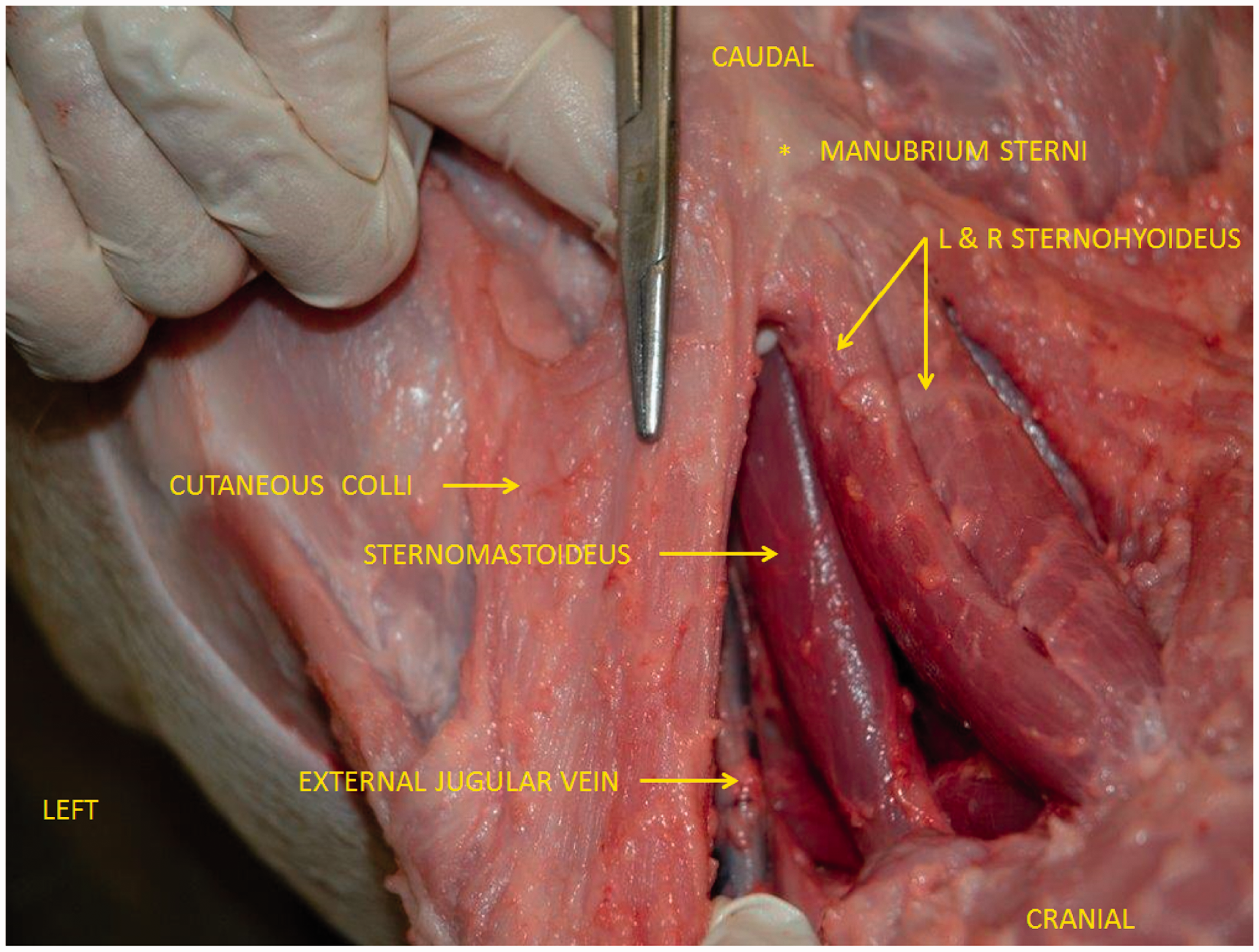
Animals and housing
Thirty large white male pigs (University of Queensland Piggery, Gatton, QLD, Australia) aged 17 weeks (body weight [BW] 60 ± 2 kg) were used. The pigs were housed individually on raised floors (commercial purpose built pig floor tile, G.S.C. Engineering, Goomeri, QLD, Australia) in 1.8 m2 pens. The pens were designed by The Queensland Animal Science Precinct, and built by Stark Engineering, Gatton, QLD, Australia. The walls of the pens were covered in plastic sheeting resulting in smooth walls (to reduce the risk of pigs dislodging the catheters), with an external feeding trough at one end. Water was available ad libitum. Pigs could see and hear each other, but touch was restricted by the pen walls. Pigs had room to turn around. Lights were on from 06:00 to 20:00 h. The shed was automatically ventilated.
Surgery was carried out weekly, with 10 pigs being handled per week over a three-week period. The catheterization procedure for 10 pigs was carried out in one day, which was made possible by organizing two teams with a veterinary surgeon, an anaesthetist, and an assistant per team. Ancillary staff were involved in tasks working between the two teams, such as preparation of equipment for each catheterization (sterile surgical instruments, etc.), and observation of pigs immediately post-surgery.
Habituation
In order to reduce the stress that would have been associated with blood sampling every 15 min for a minimum of 2 h, the pigs were started on a programme of ‘habitation’ to human contact. For approximately two weeks prior to surgery, the pigs were habituated to human contact with handling for a minimum of 10–15 min daily, by a variety of co-workers (a minimum of three per day, from a pool of 12). This involved patting, rubbing, and scratching the body of the pig all over, while simultaneously talking to them quietly. Pigs were fed within an approximately 2 h framework morning and afternoon, specifically not in relation to initial human entry to the barn, to ensure that the pigs would not associate human entry with a signal for being fed.
Anaesthesia
Sedation was achieved by intramuscular administration of 0.15 mg/kg butorphanol (Butorgesic®; Ilium Veterinary Products, Smithfield, NSW, Australia) and 3 mg/kg of Zoletil® (1.5 mg/kg zolazepam and 1.5 mg/kg tiletamine) Zolazepam and tiletamine were obtained combined in equal quantities in the formulation Zoletil® (Virbac Animal Health, Peakhurst, NSW, Australia). Isoflurane (IsoFlo®; Abbott Laboratories, Melbourne, VIC, Australia) in oxygen was then delivered by mask while a 20 G catheter was secured in the lateral ear vein. Some pigs were sufficiently anaesthetized without further drug administration, but most required a small dose of intravenous thiopentione sodium (2.5 mg/kg, 0–4.8 mg/kg) (Ilium Thiopentone®; Troy Laboratories, Smithfield, NSW, Australia). The trachea was intubated with a 7.5 mm endotracheal tube and anaesthesia was maintained with isofluorane delivered in oxygen for the duration of surgery. Each pig received 15 mg xylazine intramuscularly (Xylazil-20®; Ilium Veterinary Products) as the isoflurane was turned off to diminish excitement on recovery. Antibiotic cover was achieved with intravenous oxytetracycline 386 mg at the commencement of surgery (Alamycin 10®; Norbrook, Tullamarine, VIC, Australia). Oxytetracycline was continued intramuscularly for two days post-surgery. Each pig received a single dose of meloxicam 20 mg intravenously before recovery (Metacam 20®; Boehringer Ingelheim, Macquarie Park, NSW, Australia).
Surgical procedure
Surgeons prepared for the aseptic surgery in a standard manner. Following sedation, each pig was placed in the dorsal recumbency position on an operating table. After complete anaesthesia induction, a skin incision was made in the jugular fossa on one side of the neck in a caudomedial to craniolateral direction. The incision was approximately 3–4 cm long. Blunt dissection was used to go between the sternomastoideus and the sternohyoideus muscles of the neck and into the deeper tissues until the jugular vein was located, usually at a depth of 3–4 cm below the skin. Connective tissue was gently removed from around the jugular vein and blunt dissection was used to free a section of the jugular vein approximately 2–3 cm in length (Figure 2).
Photograph demonstrating the external jugular vein prior to catheterization.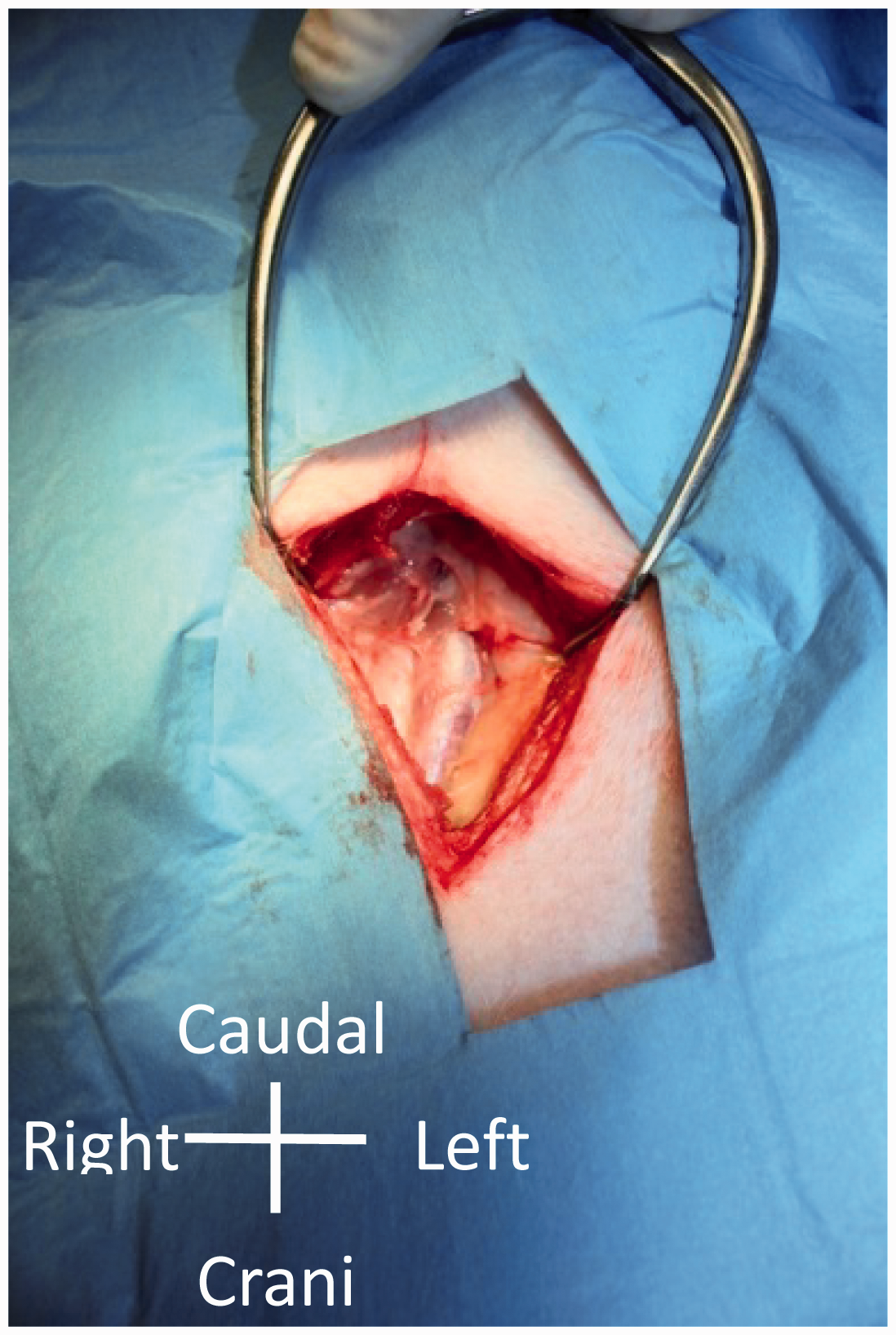
A complete central venous catheterization set (Arrow® CV-04306; Arrow International Inc, Reading, PA, USA) was used to position a catheter into the vein. This involved the following: a sterile introducer needle from the kit was used to penetrate the vein. In some instances a sterile blunt-ended hook (spey hook) was gently placed under the vein and elevated slightly in order to better immobilize the vein before the introducer needle was inserted. Once the introducer needle was in place, a spring-wire guide with a flexible curved tip was then passed through the introducer needle. The introducer needle was removed and a tissue dilator was passed over the spring-wire guide into the vein (Figure 3). The tissue dilator was removed while leaving the spring-wire guide in place. The catheter was then passed over the spring-wire guide and into the vein to its full extent (10 cm). The spring-wire guide was removed and an extension set which had been preloaded with sterile saline and with an injection port attached, was secured to the end of the catheter. Once the catheter had been checked for patency by the withdrawal of 2–3 mL of blood, the catheter and extension set were then flushed with 3 mL of heparinized saline (4 IU). The end of the catheter was sutured into place adjacent to the jugular vein with Ethicon 3/0 chromic catgut (Johnson & Johnson Medical Pty Ltd, Sydney, Australia). Subcutaneous tissues were also sutured with 3/0 catgut in order to minimize dead space. The extension set was positioned at the cranial end of the skin incision and the wound closed with surgical staples and a sterile dressing placed over the wound.
From left to right: introducer needle 18 Ga × 2.5″ (6.35 cm); spring-wire guide marked 0.032″ (0.81 mm) diameter × 17 and 13/16″ (45 cm); tissue dilator; indwelling catheter 16 Ga × 6″ (16 cm) Radiopaque with Blue FlexTip® with integral extension line, extension line clamp and integral suture wing.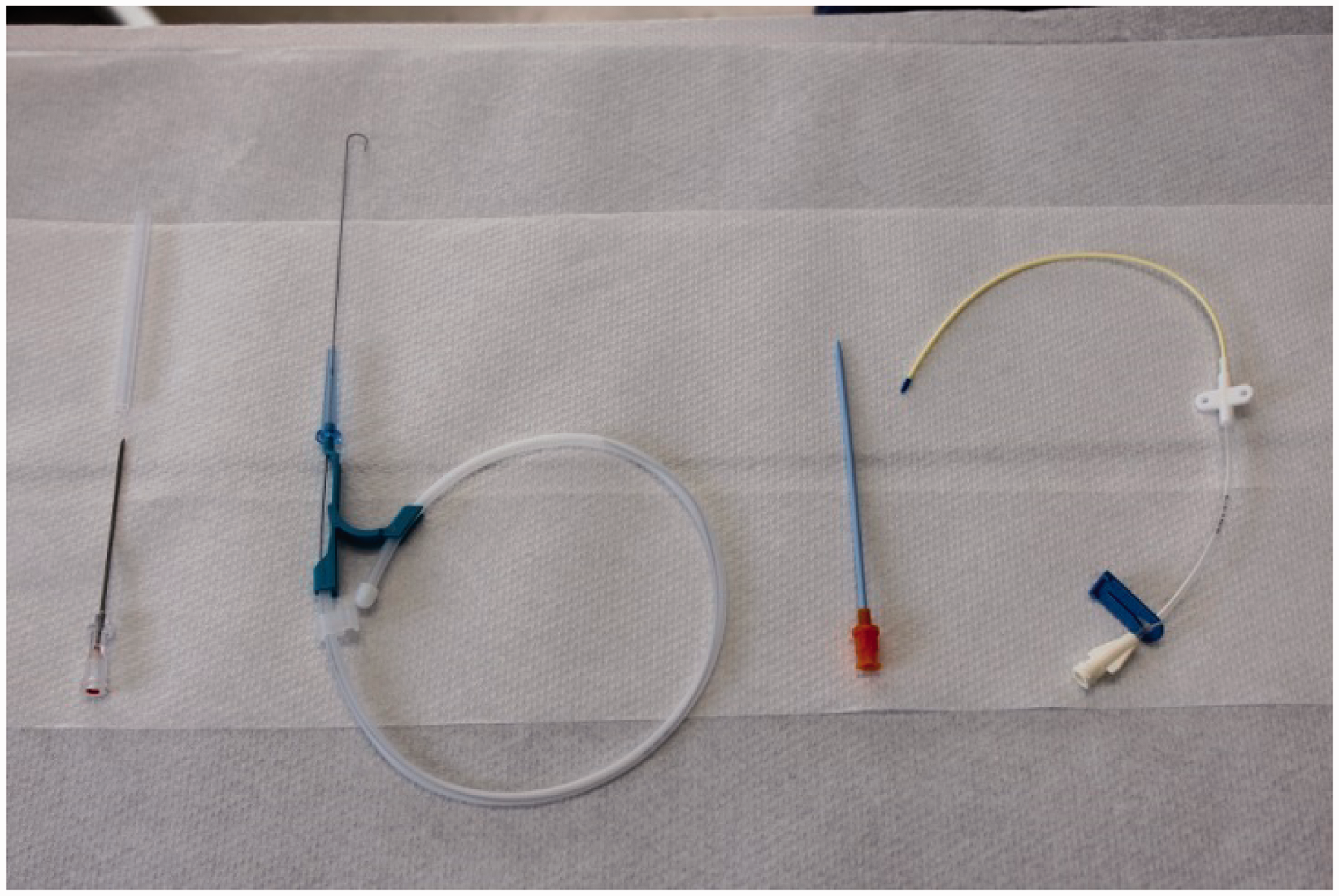
Securing the catheter externally
The catheter and extension tube were secured by means of tape (Elastoplast), superglue (Selleys Pty Ltd, Padstow, NSW, Australia) and staples onto the back of the neck. Once sutured into place, and the incision stapled, the incision was sprayed with a moisture vapour permeable spray and covered with transparent adhesive waterproof film, which was in turn covered with Sleek (Smith & Nephew Australia, North Ryde, NSW, Australia). These procedures were to ensure that the wound remained dry, as when drinking, water tended to run down the neck.
The B Braun Heidelberg 22 cm Luer Lock extension tube (ZebraVet, Brisbane, Australia) was secured to the catheter, and a ‘butterfly-wing’ piece of Elastoplast was then used to trap the end of the tubing at the top of the neck, and secured using both superglue to the skin and either staples or sutures. Elastoplast was then used to secure the extension tubing and catheter further, by winding loosely at least 2–3 times around the entire neck.
Recovery and catheter patency
Following surgery, the pigs recovered from the procedure for 48 h prior to the blood sampling procedure.
During this recovery period catheters were flushed twice daily, first with normal saline (0.9% HCl), followed by heparinized saline containing 100 IU of heparin (heparin lock). The volume of heparinized saline was 3 mL, which was the volume of the catheter and the two extension tubes. However, in between blood samples, while still flushed with normal saline, 3 mL of heparinized saline was added at a concentration of 4 IU prior to the next sample being collected.
The oral glucose tolerance test (OGTT) and blood sampling
Following an overnight fast, a 1.5 g/kg BW D-glucose solution was fed to the pigs using 50 mL syringes (D-Glucose anhydrous; Chem-Supply Pty Ltd, Gillman, SA, Australia). A silicon tube was attached to the end of each 50 mL syringe and the tube was placed onto the bottom lips of the pigs, which allowed them to drink the glucose solution with a minimal loss of the solution. The syringe was pushed in slowly to allow time for the pigs to swallow, but they all drank it readily, probably due to the sweet taste. All pigs consumed the glucose solution within 2 ± 1 min.
On the day of sampling, an extra extension tube (as before) was attached to the fixed extension tube outlet, and was either allowed to move about freely, or, if the pig seemed to respond to its touch on the skin, it was then stuck to the Elastoplast bandage with another short piece of bandage. This remained in place for the duration of the blood sampling per day. Ten-millilitre syringes were used to remove blood at regular intervals. For example, to remove 30 mL, three × 10 mL syringes were used. This was done by direct connection to the extension tube (no needles), and the appropriate volumes were then added to various vacutainers as required, though with the lids removed, before being re-sealed. Approximately 20–25 s was required to remove each 10 mL blood sample. This minimized any incidence of haemolysis of the red blood cells (RBC). When the required amount of blood was removed, the tubing and catheter were flushed with 10 mL of 0.9% saline solution, followed by 3 mL of 4 IU heparinized saline. Before the next sample was taken, approximately 10 mL of blood was removed and discarded before taking the actual samples. In this way, one team of three people could sample four pigs in 15 min. One person was inside the pen taking the samples and flushing the catheter, one person was outside the pen handing that person syringes as required, and also taking the filled syringes away, and one person added the blood to the appropriate vacutainers. As a result of the habituation process, the pigs were used to people getting into and out of their pens, as well as being handled.
Analytical procedures
Blood was collected in heparinized Vacutainers™ for insulin analysis and in Vacutainers™ coated with K2EDTA anticoagulant for enteroendocrine hormone analysis (study not reported here). Blood was collected one hour prior to feeding (–60 min) and then every 15 min from feeding time (T0 min) to 2 h (120 min) postprandial and then at 3 (180 min) and 4 (240 min) h post-glucose dose. Blood was centrifuged at 3000 g for 10 min and the plasma frozen at –20℃. Plasma samples were analysed for glucose according to a standard procedure using glucose hexokinase II and enzymatic colorimetric determination (GAHK20; Sigma-Aldrich, St Louis, MO, USA).
Results and discussion
Catheters were placed into the jugular vein of grower pigs of roughly 60 kg (n = 30) by experienced veterinarians. The mean time required for the surgical procedure was 65 min, though this did not include pre-medication, or affixing of the extension tubes using bandaging. In addition, variable amounts of time elapsed per pig during the removal of the endotracheal tube. However, pigs were under constant supervision for this procedure.
Selection of appropriate vein for sampling
Considerations to be taken into account were the comparatively large volume to be removed per sample (∼20–30 mL), the frequency of sampling (every 15 min for 2 h, and then hourly to 4 h), and ease of access without disturbing the pig excessively. The ear vein was rejected as being unlikely to allow removal of 30 mL of blood per sampling time. Both the saphenous and mammary veins in these pigs were too small for the volume of blood required. Due to the volume of blood required, ease of access, location of vein and previous works as guidance, the jugular vein was decided to be the most appropriate for our requirements.
Catheterization methodology
Overall, this jugular catheterization method had a high success rate. It has been recommended that catheterization should be carried out at least one to two days in advance
14
to minimize any changes in blood parameters resulting from surgery. Thirty large white male grower pigs were catheterized. No pigs were excluded from the experiment due to complications of the procedure itself. All 30 catheters remained in place and were patent during the two consecutive days of sampling. Pigs remained calm during the blood sampling (Figure 4). Prior to blood sampling, one intravenous extension was found to have been dislodged, however, this was easily replaced.
Handling stress was eliminated by prior exposure to handling and using extended cannulation tubing which allowed for ‘remote’ blood collection. This picture illustrates blood collection from a grower pig while it remains calm. Some pigs slept during blood collection.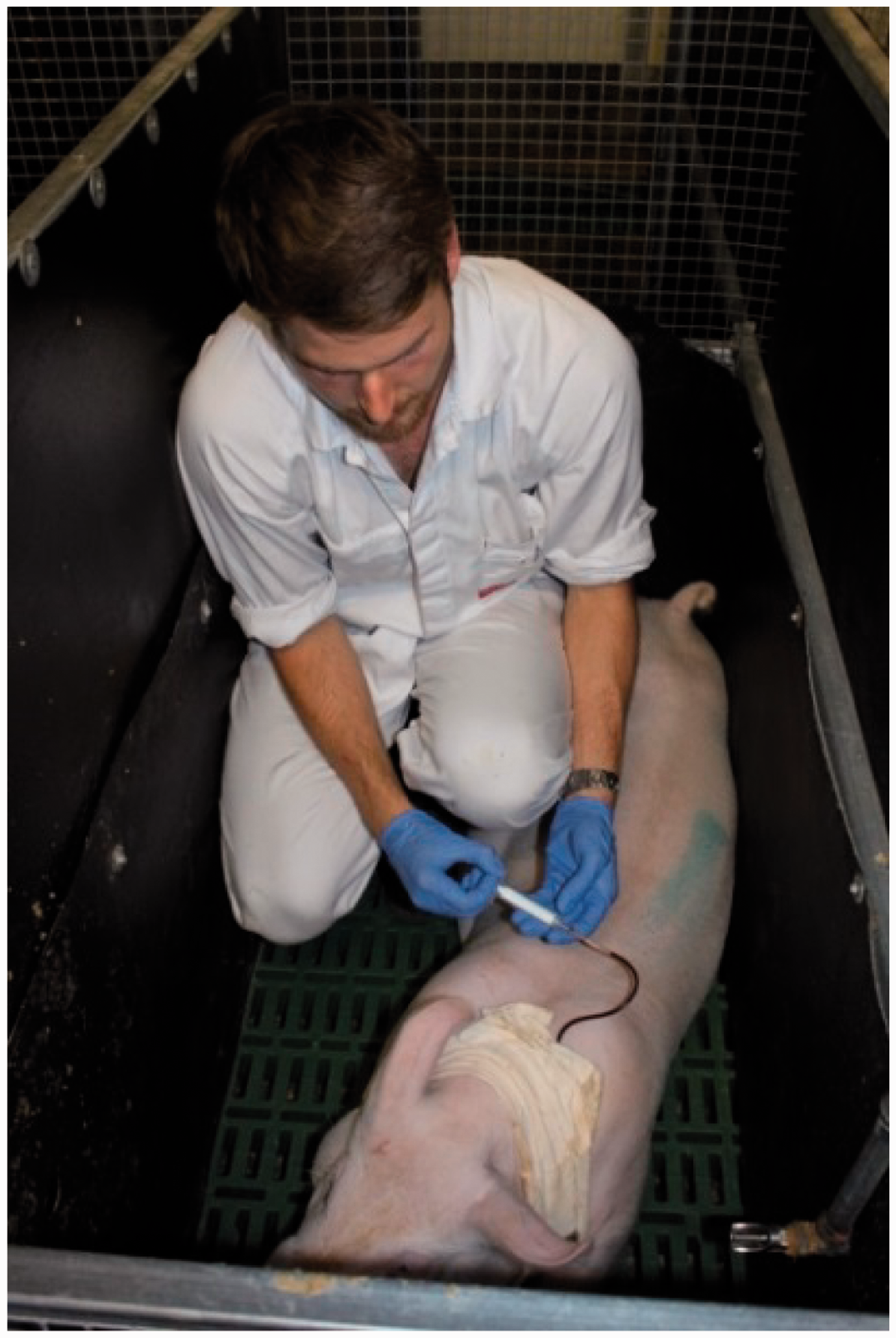
The glycaemic response following an OGTT from six pigs is presented in Figure 5. The glucose response curve following the OGTT was biphasic.
15
Fasting blood glucose was 5.0 mmol/L and peaked at 6.6 mmol/L at 30 min after the glucose dose. Pigs became hypoglycaemic at 90 min (4.8 mmol/L) and then experienced a secondary rise in blood glucose until 180 min when they became euglycaemic.
Blood glucose (mmol/L ± SE) of six pigs following an oral glucose tolerance test (OGTT) given 1.5 g/kg body weight of D-glucose.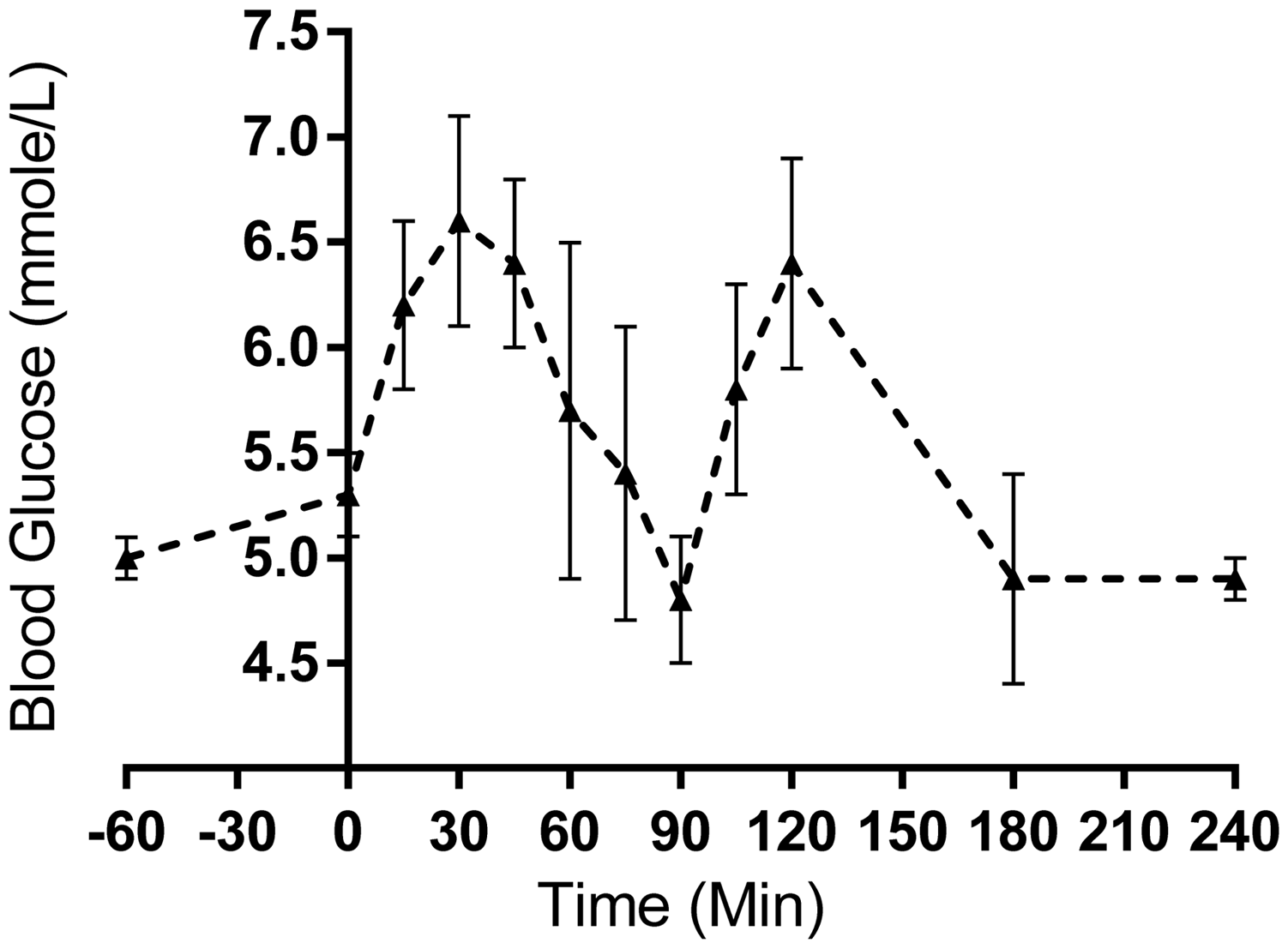
Humans demonstrating a biphasic glucose curve in response to the OGTT are thought to have higher insulin sensitivity, lower fasting plasma glucose, insulin levels and glucose and insulin area under the curve in the OGTT, 15 and hence are thought to be more glucose-tolerant. The first phase following the OGTT was between 0 to 90 min. This initial increase in plasma glucose was associated with the consumption of the glucose solution and its subsequent absorption into the systemic circulation. After the initial peak in plasma glucose, these pigs were very efficient in reducing their plasma glucose concentrations and developed hypoglycaemia (90 min).
Conclusion
Given the volume of blood required and the frequency of collection, the jugular vein was the most appropriate vein for blood collection from this age and size of pig. This technique while not rapid, and requiring full anaesthesia, resulted in a minimum amount of stress to the animal. In terms of the frequency of blood sampling required, it is strongly recommended that a habituation procedure be included in the process.
Footnotes
Acknowledgements
Our thanks to Debra Laws for her role in the pig anaesthesia, and to Dr Cora Lau for her veterinary expertise and care of the pigs. We would also like to thank the staff of the Queensland Animal Science Precinct (Gatton campus), and the University of Queensland for organizing the pig housing, and providing the equipment. Xinyu (Zoe) Zhang assisted in all aspects of pig husbandry. Finally our thanks also to the CNAFS staff and students who spent so much time habituating the pigs to human touch and presence.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research has been funded by the Australian Research Council Centre of Excellence in Plant Cell Walls.
