Abstract
The postprandial hypertriglyceridaemia (PHT) rabbit, developed as a new animal model of metabolic syndrome, is characterized by PHT, central obesity and glucose intolerance. For detailed investigation of lipid metabolism characteristics in PHT rabbit, the plasma levels of apolipoproteins A-I, B, C-II, C-III and E were measured. Movements of apolipoproteins B100 and B48 were investigated using sodium dodecyl sulphate–polyacrylamide gel electrophoresis to determine whether postprandially increased triglyceride is exogenous or endogenous. The level of apolipoproteins A-I, B, C-II and E were increased in PHT rabbit after feeding. Apolipoproteins B100 and B48 were detected in the plasma fraction of d < 1.006 g/mL of the PHT rabbit. The postprandial increase in apolipoprotein B in the PHT rabbit reflects a numerical increase in lipoprotein particles in the blood; the increase in apolipoproteins C-II and E suggests some disturbance in lipoprotein catabolism. Apolipoprotein B48 was detected postprandially in PHT rabbits. These results suggest that delayed catabolism of exogenous lipids caused the retention of chylomicron remnants in the blood. Results also suggest that activities of the lipolytic enzyme lipoprotein lipase and hepatic triglyceride lipase were deficient and that the hepatic uptake of exogenous lipoproteins was delayed in the PHT rabbit. Especially, for examining remnant hyperlipoproteinaemia in humans, PHT rabbit is an excellent animal model for hypertriglyceridaemia research.
Keywords
Ischaemic heart disease is closely related to the plasma cholesterol (CHO) level. However, results of recent studies have demonstrated that not all patients developing ischaemic heart disease have hypercholesterolaemia 1 and that elevated levels of serum triglycerides (TG) increase the risk of ischaemic heart disease. 2,3 Furthermore, metabolic syndrome, a condition in which central obesity is complicated by multiple risk factors of atherosclerosis (hypertriglyceridaemia, glucose intolerance and hypertension), has recently been suggested to increase the risk of ischaemic heart disease. From the Watanabe heritable hyperlipidaemic (WHHL) rabbit, Tomoike et al. of the Yamagata University Faculty of Medicine selectively bred the WHHL-TGH (triglyceride-high) rabbit, in which serum CHO and TG levels were both high, and the WHHL-TGL (triglyceride-low) rabbit, in which the CHO level was high but the TG level was normal. 4 Tomoike et al. also developed the postprandial hypertriglyceridaemic (PHT) rabbit model, which showed normal serum TG and CHO levels during fasting, but an abnormally high level of TG alone postprandially. 4 The PHT rabbit, which showed a high postprandial TG level, was derived from the mutant F2 hybrid by crossbreeding between the Japanese White (JW) rabbit and the WHHL-TGH rabbit.
The PHT rabbit was derived from the WHHL-TGH rabbit by eliminating the factor of hypercholesterolaemia and selecting individuals who showed PHT. Actually, the PHT rabbit strain is considered to be polygenic. 5 Expected to be a novel animal model of metabolic syndrome, it is characterized by central obesity, glucose intolerance and hypertriglyceridaemia. 5,6
The kinetics of various apolipoproteins of the PHT rabbit, especially postprandial changes in lipoproteins, were analysed to elucidate the detailed characteristics of lipid metabolism in the PHT rabbit.
This study was designed to examine whether the postprandial increase in TG in the PHT rabbit resulted solely from an increase in VLDL released from the liver, or reflected the increase in exogenous (diet-derived) lipoproteins, from the kinetics of apolipoprotein B100 and B48 which are present, respectively, in endogenous and exogenous lipoproteins.
Materials and methods
Animals and feeding conditions
The PHT rabbits used in this study had been bred and kept at the Laboratory Animal Center, Yamagata University Faculty of Medicine. They were weaned at one month after birth, and maintained in individual cages (480 mm wide, 610 mm deep, 370 mm high). As plasma levels of lipid components in PHT rabbits, plasma TG and CHO levels were evaluated before feeding (during fasting) and 24 h after the start of feeding in six-month-old PHT rabbits that had been subjected to more than 24 h of fasting followed by feeding to satiation. After the measurement of plasma lipid components, 3–5 male and 10–15 female PHT rabbits showing the highest postprandial TG levels were selected, randomly crossed to prevent inbreeding degeneration, and propagated for generations.
Postprandial plasma TG levels in PHT rabbits peaked at 15–21 h after the start of feeding. 5 Therefore, plasma samples collected at 15–21 h after the start of feeding were used in this study.
The PHT rabbits and control JW rabbits were kept on a 12 h light–dark cycle (with lights-off at 18:00 h) at 22 ± 2°C and 40–60% humidity, fed 120 g of chow diet (Labo R Grower; Nosan Co, Yokohama, Japan) at noon every day; they were provided access to water ad libitum.
All experiments were performed in accordance with the Guidelines for Animal Experiments of Yamagata University Faculty of Medicine.
Lipoprotein fractionation
Two seven-month-old male PHT rabbits and two six-month-old male JW rabbits were bled from the auricular artery after 24 h of fasting and at 21 h after the start of feeding. Blood was collected in a tube containing lithium heparin; it was immediately cold-centrifuged at 850
Postprandial apolipoproteins
Plasma was obtained from six seven-month-old male PHT rabbits and six 6–9-month-old male JW rabbits after 24 h of fasting and at 21 h after the start of feeding, as described above. Sodium azide (0.05%) and gentamycin (0.005%) were added to the plasma to prevent protein degeneration.
TG, CHO and total protein (T-Pro) levels were measured using the enzymatic method with the instruments described above.
Apolipoproteins A-I, B, C-II, C-III and E were measured using immunoturbidimetric methods with antihuman apolipoprotein antibodies (HA Test; Wako Pure Chemical Industries Ltd, Osaka, Japan).
Plasma was delipidated with ethanol and ether
7
to purify proteins. Air was discharged from a 15 mL centrifuge tube by sealing nitrogen gas; then 500 μL of plasma and 9 mL of chilled anhydrous ethanol were added and mixed well. The mixture was then allowed to stand for 30 min. Subsequently, 6 mL of diethylether was added and mixed for 30 min, and the mixture was centrifuged at 850
The delipidated sample was then dried at a low temperature; a small volume of pure water was added for protein determination using a protein assay reagent kit (BCA; Pierce Biotechnology Inc, Rockford, IL, USA). To solubilize the sample, an equal volume of buffer (EzApply; ATTO Corp, Tokyo, Japan) consisting of 100 mmol/L Tris–HCl (pH 8.8), 2% SDS (w/v), 20% sucrose (w/v), 0.06% bromphenol blue (w/v) and 100 mmol/L dithiothreitol was added; the mixture was heated at 95°C for 5 min, and cooled to room temperature.
As a marker to identify apolipoprotein B48, lymph-derived purified proteins were used and analysed by sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS–PAGE). Lymph was collected from the mesenteric lymph duct of 16-month-old female JW rabbits under anaesthesia with sodium pentobarbital (40 mg/kg, i.v.) at 8.5 h after the start of feeding, and used for the analysis after protein purification and solubilization, as described above for the plasma sample.
According to the method described by Laemmli, 8 SDS–PAGE was performed for about 150 min at a constant current of 20 mA using a precast 3–10% gradient gel (PAGEL and PageRun system; ATTO Corp). To each well, a sample (adjusted to a protein content of 40 μg), apolipoprotein B48 marker, positive control apolipoprotein B100 (Apolipoprotein B; Athens Research and Technology, Inc, Athens, GA, USA), or molecular weight marker (High Molecular Weight SDS Calibration Kit for Electrophoresis; GE Healthcare UK Ltd, Little Chalfont, UK) was added for electrophoresis.
After electrophoresis, the gel was shaken for 30 min in a fixation solution consisting of 20 mL of methanol, 7.5 mL of acetic acid and 72.5 mL of ultrapure water; then it was soaked in a staining solution containing 0.25% Coomassie brilliant blue R-250 in a 10:9:1 mixture of methanol, ultrapure water and acetic acid. The stained gel was placed in a destaining solution (88:7:5 mixture of ultrapure water, acetic acid and methanol) and shaken on a shaker until the background became clear. The destained gel was sandwiched between cellophane sheets and air-dried completely.
Apolipoprotein B kinetics in the plasma fraction of d < 1.006 g/mL
Two 12-month-old male PHT rabbits and two 14–15-month-old male JW rabbits kept under normal feeding conditions (120 g of chow diet daily at noon) were bled from the auricular artery immediately before and 15 h after the start of feeding. The obtained plasma was immediately kept chilled on ice.
The plasma fraction with a density of less than that (d < 1.006 g/mL) of VLDL was collected by ultracentrifugation 9 using a preparative ultracentrifuge (Optima XL-90, 50.2Ti rotor; Beckman Coulter Inc, Fullerton, CA, USA) to exclude the influence of LDL-derived apolipoprotein B100. After solubilization, the sample was fractionated using SDS–PAGE; the electrophoretic images were analysed for B100 and B48 apolipoproteins.
Statistical analysis
The measurement results obtained in each experiment were tested, respectively, for a normal distribution and for significance using Student's t-test, and using Welch's t-test in cases of homoskedasticity and heteroscedasticity. For this study, P values of <0.05 were inferred to be statistically significant.
Results
Lipid levels in plasma and lipoprotein fraction
No difference was found between the fasting TG levels in the PHT rabbits (48.5 mg/dL) and JW rabbits (44.0 mg/dL). However, the postprandial TG level in the PHT rabbits (878.5 mg/dL) was eight-fold higher than that in the JW rabbits (105.5 mg/dL). The fasting CHO level in the PHT rabbits (25.0 mg/dL) did not differ from that in the JW rabbits (22.5 mg/dL), but the postprandial CHO level in the PHT rabbits (76.0 mg/dL) was four-fold higher than that in the JW rabbits (18.5 mg/dL) (Table 1).
Plasma lipid levels
Plasma lipid levels are expressed as means (range of minimum–maximum). TG: triglyceride; CHO: total cholesterol; fasting: fasting for 24 h; postprandial: 21 h after the start of feeding; PHT: postprandial hypertriglyceridaemia; JW: Japanese White
Regarding the fasting lipoprotein TG levels, no difference was found between VLDL levels in the PHT rabbits (11.5 mg/dL) and in the JW rabbits (7.8 mg/dL). However, the LDL level in the PHT rabbits (8.4 mg/dL) was one-third of that in the JW rabbits (25.4 mg/dL). Furthermore, the HDL level in the PHT rabbits (28.5 mg/dL) was three-fold higher than that in the JW rabbits (10.7 mg/dL). In the postprandial VLDL, LDL and HDL TG levels in the PHT rabbits (498.6 mg/mL in VLDL, 284.5 mg/dL in LDL and 92.7 mg/mL in HDL) were 7–11-fold higher than those in the JW rabbits (68.7 mg/mL in VLDL, 28.4 mg/dL in LDL and 8.4 mg/mL in HDL). No significant difference was found between the fasting lipoprotein CHO levels in the PHT rabbits (0.0 mg/dL in chylomicron [CM], 0.5 mg/mL in VLDL, 4.8 mg/dL in LDL and 19.7 mg/mL in HDL) and JW rabbits (0.0 mg/dL in CM, 1.7 mg/mL in VLDL, 9.2 mg/dL in LDL and 11.7 mg/dL in HDL). However, the postprandial VLDL, LDL and HDL-CHO levels in the PHT rabbits (27.3 mg/mL in VLDL, 34.0 mg/dL in LDL and 14.7 mg/mL in HDL) were 3–5-fold higher than those in the JW rabbits (7.1 mg/mL in VLDL, 7.5 mg/dL in LDL and 3.9 mg/mL in HDL) (Figure 1).
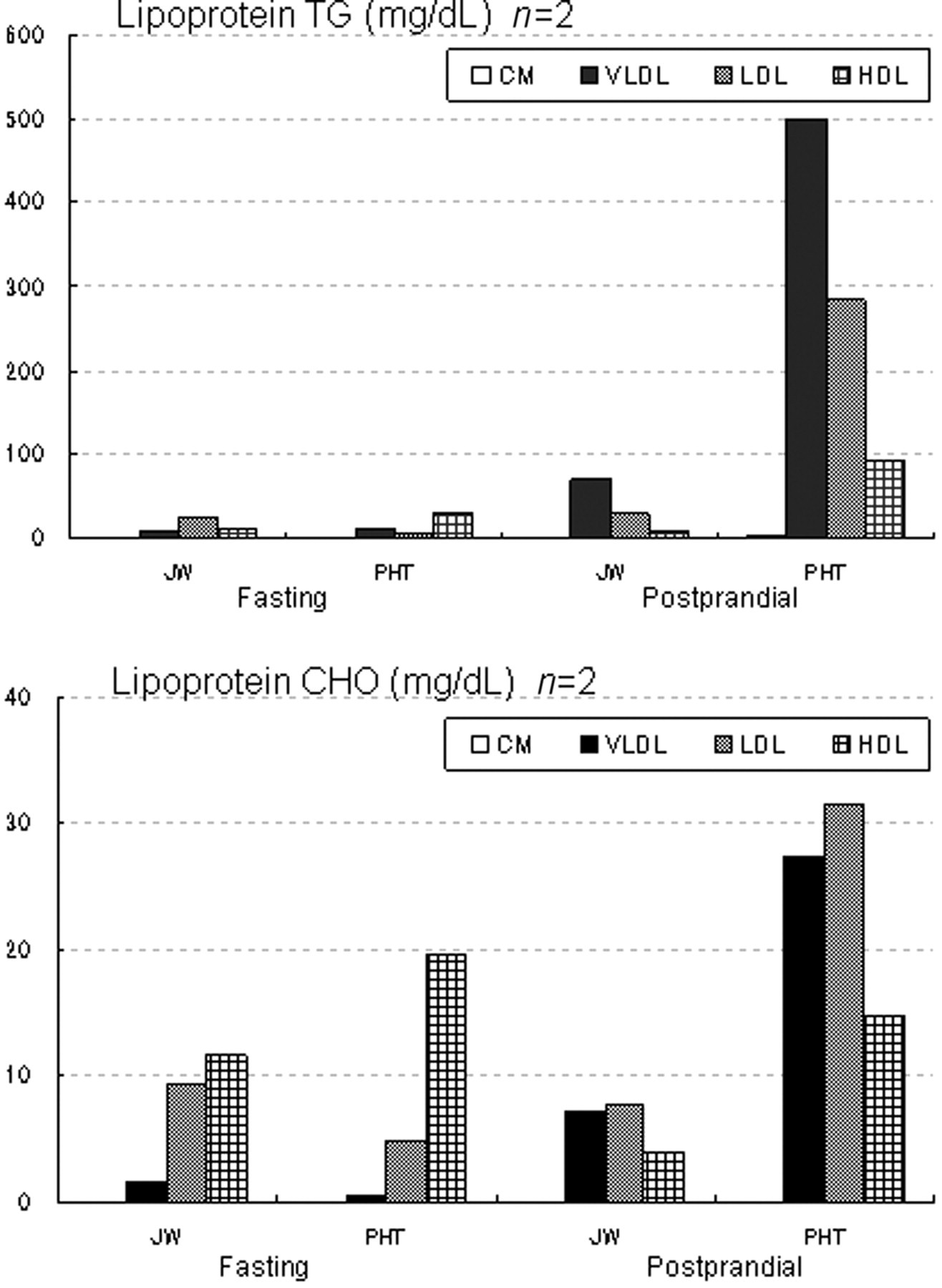
Lipid levels in lipoprotein fractions. From lipoprotein fractions on stained electrophoretograms, the lipid levels in the fractions were obtained and are expressed as means. TG: triglyceride; CHO: total cholesterol; fasting: fasting for 24 h; postprandial: 21 h after the start of feeding; CM: chylomicron; VLDL: very low-density lipoprotein; LDL: low-density lipoprotein; HDL: high-density lipoprotein; PHT: postprandial hypertriglyceridaemia; JW: Japanese White
The fraction images obtained by the analysis of electrophoretic images with image analysis software are presented in Figure 2. During fasting, the fraction images of lipoprotein TG were similar in the PHT and JW rabbits (Figure 2 T-A, T-B, T-C and T-D). However, the postprandial VLDL TG and LDL TG levels in the PHT rabbits were markedly higher than those in the JW rabbits (Figure 2 T-E and T-F). On the other hand, during fasting, lipoprotein CHO levels in the PHT and JW rabbits were high in the HDL fraction (Figure 2 C-A, C-B, C-C and C-D), whereas the postprandial CHO levels in the PHT rabbits were low in the HDL fraction, and higher in the VLDL and LDL fractions (Figure 2 C-E and C-F). In the JW rabbits, the postprandial CHO level was decreased in the HDL fraction, but the postprandial CHO levels in the VLDL and LDL fractions were similar to their fasting levels (Figure 2 C-G and C-H).
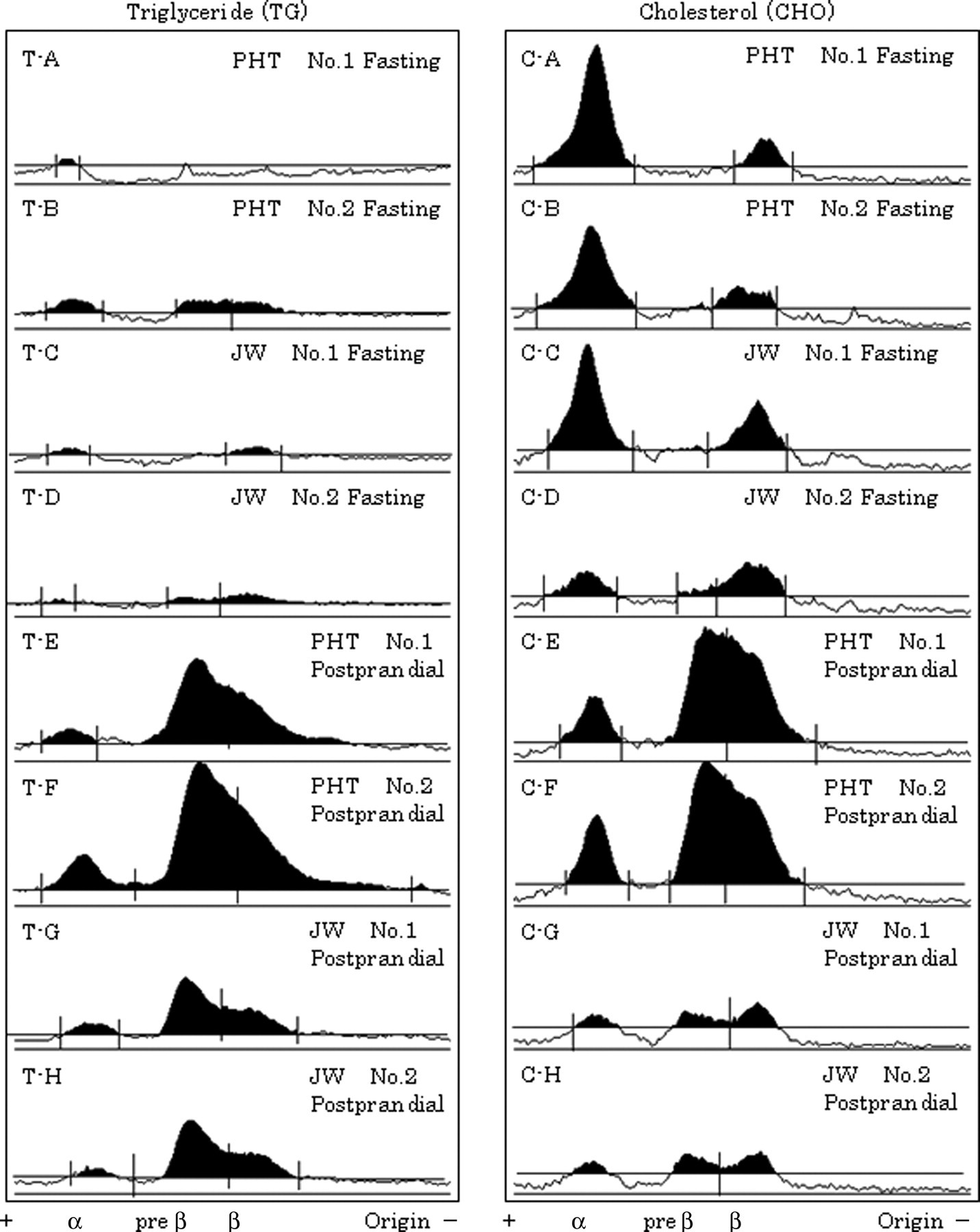
Fractionation of lipoproteins. Lipoproteins in plasma, collected after 24 h of fasting and at 21 h after the start of feeding, were fractionated using agarose gel electrophoresis and stained for triglyceride (TG) and cholesterol (CHO). The resulting electrophoretic patterns were analysed using image analysis software. TG: T-A and T-B, for fasting postprandial hypertriglyceridaemia (PHT); T-C and T-D, for fasting Japanese White (JW); T-E and T-F, for postprandial PHT; T-G and T-H, for postprandial JW. CHO: C-A and C-B, for fasting PHT; C-C and C-D, for fasting JW; C-E and C-F, for postprandial PHT; C-G and C-H, for postprandial JW
Postprandial apolipoproteins
The fasting and 21 h postprandial TG levels in the JW rabbits were, respectively, 38.2 ± 3.1 mg/dL and 104.2 ± 13.7 mg/dL; those in the PHT rabbits were 102.7 ± 27.0 mg/dL and 1876.7 ± 242.5 mg/dL, indicating a marked postprandial increase in the TG level. The fasting and 21 h postprandial CHO levels in the JW rabbits were, respectively, 28.5 ± 4.9 mg/dL and 37.8 ± 6.3 mg/dL; those in the PHT rabbits were 29.0 ± 6.7 mg/dL and 98.3 ± 16.2 mg/dL. The fasting and postprandial T-Pro levels in the JW rabbits were 6.0 ± 0.4 mg/dL and 6.3 ± 0.3 mg/dL, respectively, which were similar to the measured levels of 6.4 ± 0.2 mg/dL and 6.3 ± 0.2 mg/dL in the PHT rabbits (Table 2).
Lipid and total protein levels in the samples used for apolipoprotein measurement
Values are expressed as means ± SE. TG: triglyceride; CHO: total cholesterol; T-Pro: total protein; fasting: fasting for 24 h; postprandial: 21 h after the start of feeding; PHT: postprandial hypertriglyceridaemia; JW: Japanese White
*P < 0.01 (vs. fasting value)
† P < 0.01 (vs. JW-postprandial value)
‡ P < 0.05 (vs. fasting value)
The levels of several apolipoproteins are shown in Table 3. The fasting and postprandial apolipoprotein A-I levels were, respectively, 17.6 ± 1.7 mg/dL and 33.3 ± 5.6 mg/dL in the PHT rabbits, and 18.2 ± 2.3 mg/dL and 21.9 ± 1.2 mg/dL, respectively, in the JW rabbits, indicating a significant postprandial increase in the PHT rabbits, but not in the JW rabbits.
Measurements of apolipoprotein levels
Values are expressed as means ± SE. Fasting: fasting for 24 h; postprandial: 21 h after the start of feeding; PHT: postprandial hypertriglyceridaemia; JW: Japanese White
*P < 0.05 (vs. fasting value)
† P < 0.01 (vs. fasting value)
‡ P < 0.01 (vs. JW-postprandial value)
Similarly, the fasting and postprandial apolipoprotein B levels were, respectively, 18.5 ± 1.4 mg/dL and 44.9 ± 8.1 mg/dL in the PHT rabbits, and 20.9 ± 7.5 mg/dL and 28.1 ± 7.8 mg/dL, respectively, in the JW rabbits, indicating significant postprandial increases, particularly in the PHT rabbits.
The fasting and postprandial apolipoprotein C-II levels were 1.3 ± 0.0 mg/dL and 7.3 ± 2.7 mg/dL, respectively, in the PHT rabbits, and 4.1 ± 2.5 mg/dL and 1.8 ± 0.8 mg/dL, respectively, in the JW rabbits, indicating a significant postprandial increase in the PHT rabbits and a significant postprandial decrease in the JW rabbits.
The fasting and postprandial apolipoprotein C-III levels were 0.9 ± 0.0 mg/dL and 2.1 ± 0.8 mg/dL, respectively, in the PHT rabbits; and 3.0 ± 1.8 mg/dL and 1.4 ± 0.7 mg/dL, respectively, in the JW rabbits, indicating a postprandial increase and decrease in the PHT and JW rabbits, similar to the apolipoprotein C-II levels.
The fasting and postprandial apolipoprotein E levels were, respectively, 2.2 ± 0.1 mg/dL and 7.5 ± 1.7 mg/dL in the PHT rabbits, and 4.8 ± 2.2 mg/dL and 2.5 ± 0.6 mg/dL, respectively, in the JW rabbits, indicating a significant postprandial increase in the PHT rabbits.
Figure 3 presents SDS–PAGE patterns of apolipoprotein B. In the PHT rabbits, apolipoprotein B100 was detected during fasting (Figure 3D); both apolipoproteins B100 and B48 were detected after feeding (Figure 3E). On the other hand, in the JW rabbits, apolipoprotein B100 was detected during fasting (Figure 3B) and after feeding (Figure 3C), but apolipoprotein B48 was not detected (Figures 3B and C).
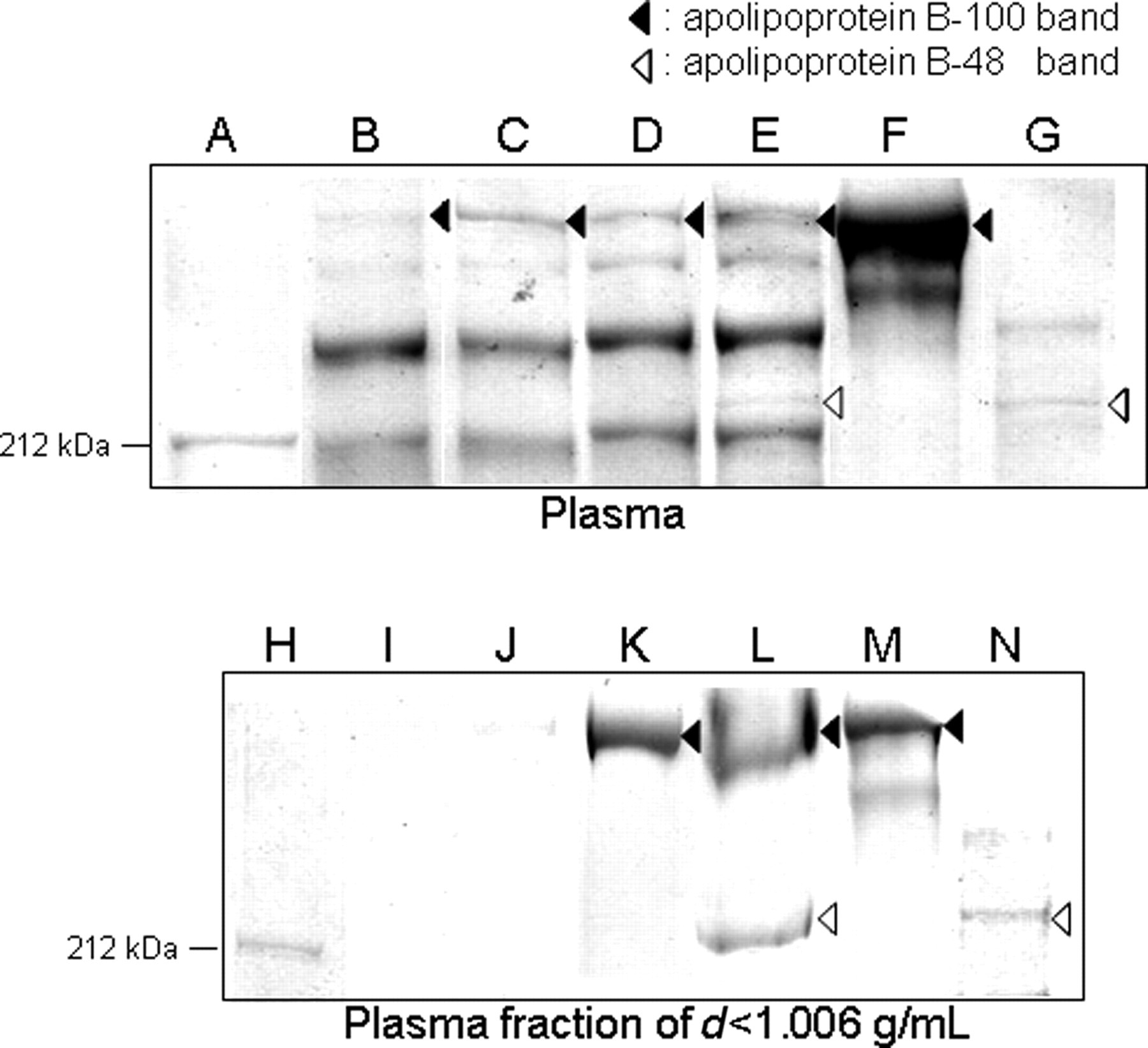
Sodium dodecyl sulphate–polyacrylamide gel electrophoresis analysis of apolipoprotein B. Upper panel: Analysis of apolipoprotein B using plasma. Lower panel: Analysis of apolipoprotein B using the fraction of d < 1.006 g/mL after ultracentrifugal fractionation of plasma. A and H, molecular weight marker (212 kDa); B and I, fasting (preprandial) Japanese White (JW); C and J, postprandial JW; D and K, fasting (preprandial) postprandial hypertriglyceridaemia (PHT); E and L postprandial PHT; F and M, apolipoprotein B100 marker; G and N, apolipoprotein B48 marker (lymph-derived)
Kinetics of apolipoproteins B100 and B48 in the plasma fraction of d < 1.006 g/mL
In the PHT rabbits, apolipoprotein B100 was detected before feeding (Figure 3K); apolipoproteins B100 and B48 were both detected 15 h after feeding (Figure 3L). In contrast, in the JW rabbits, neither apolipoprotein B100 nor apolipoprotein B48 was detected before or after feeding (Figures 3I and J).
Discussion
Figure 4 portrays a model of the separate transport pathways for lipoproteins carrying exogenous and endogenous lipids. Goldstein et al. reported that the lipoprotein emerges from the liver as VLDL and from the intestine as CM. 10 Because CM and VLDL contain many TG in lipoproteins, they are called TG-rich lipoproteins. The causes of hypertriglyceridaemia are considered to be deficient hydrolysis of TG in TG-rich CM and VLDL lipoproteins, increased hepatic production of VLDL and deficient metabolism of metabolic remnants of triglyceride-rich CM and VLDL lipoproteins. 11 The marked postprandial TG and CHO increase in the PHT rabbit. The results of lipoprotein fractionation suggest that this increase mainly reflects an increase in lipoprotein TG and CHO in the VLDL and LDL fraction migrating to the pre-β to β position (Table 1 and Figure 1). After feeding, although plasma apolipoprotein B100 was detected in both strains (Figure 2), apolipoprotein B100 in the d < 1.006 g/mL fraction was detected only in PHT (Figure 3). Therefore, it was considered that LDL remained unmetabolized in blood after feeding in JW, as did VLDL in PHT. In addition, results showed that exogenous lipoproteins remained unmetabolized after feeding because apolipoprotein B48 was detected only in PHT after feeding (Figures 2 and 3). Consequently, the increase in lipids in PHT after feeding was presumed to be the result of both remaining endogenous VLDL and exogenous lipoproteins in blood. Moreover, it can be presumed that the remaining exogenous lipoproteins are CM remnants because apolipoprotein B48 was detected (Figure 3), although there was only a slight increase in CM (Figures 1 and 2) in PHT after feeding.
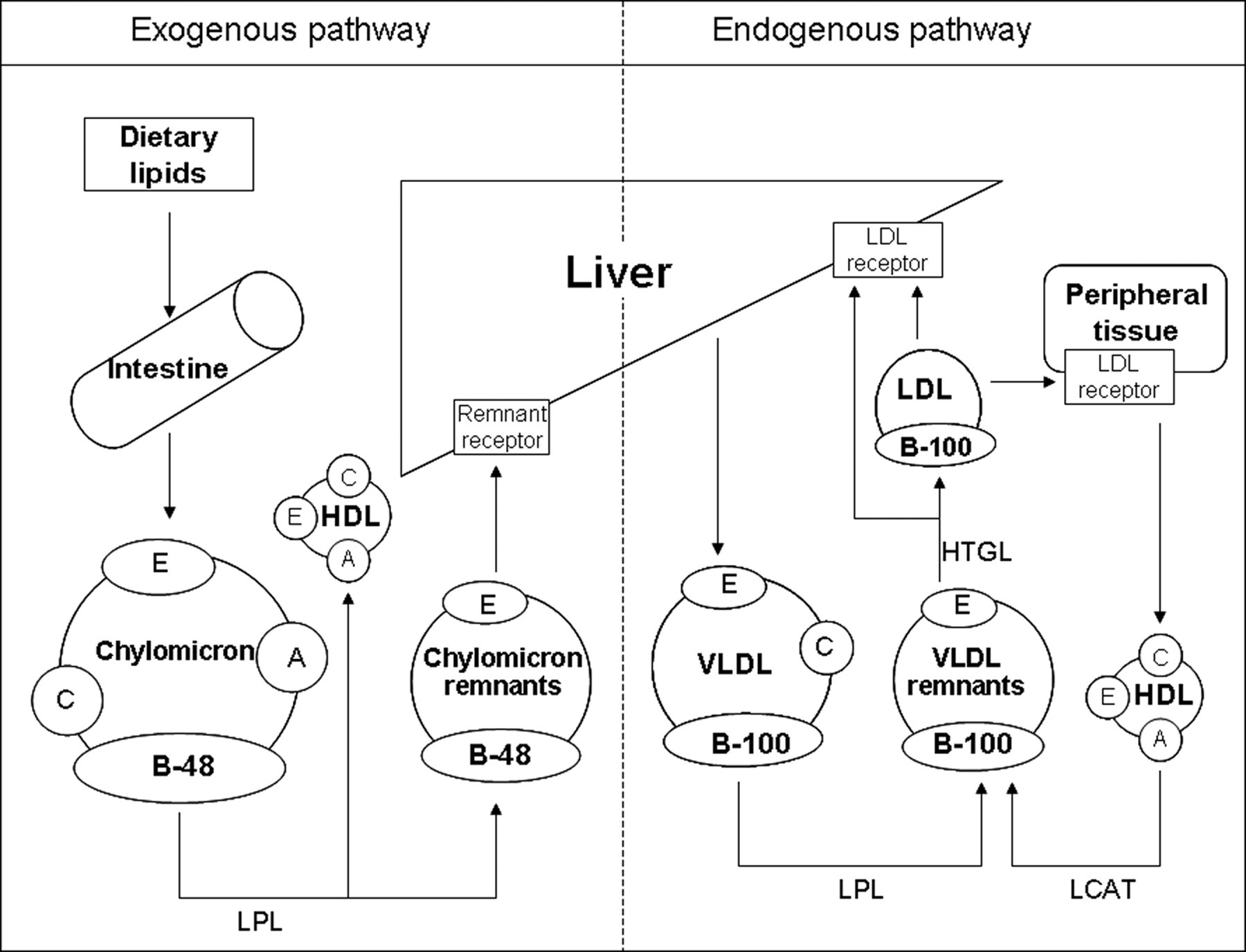
Metabolic pathway of lipoproteins. Metabolic pathway of endogenous and exogenous lipoproteins (modified from the figure by Goldstein et al. 10 ): VLDL: very low-density lipoprotein; LDL: low-density lipoprotein; HDL: high-density lipoprotein; LPL: lipoprotein lipase; HTGL: hepatic triglyceride lipase; LCAT: lecithin-cholesterol acyltransferase
Each apolipoprotein plays an important role in lipoprotein metabolism. 12 Because apolipoprotein A-I is a structural protein of HDL and has the function of activating lecithin-cholesterol acyltransferase (LCAT), which is a free-CHO esterification enzyme, the postprandial increase in apolipoprotein A-I in the PHT rabbit is related closely to the increase in HDL-TG (Figure 1). Apolipoprotein C-II is a lipoprotein lipase (LPL)-activating factor. Furthermore, apolipoprotein C-III reportedly acts antagonistically against apolipoprotein C-II and suppresses LPL activity. 13,14 Therefore, apolipoprotein C-II is considered to have increased postprandially in the PHT rabbit, because LPL must be activated to decompose the VLDL-like lipoprotein, which increased postprandially in the PHT rabbit. Apolipoprotein C-III is considered to have increased in response to this excessive increase in apolipoprotein C-II. A postprandial increase in apolipoprotein E was observed in this study. It is a remnant receptor-binding protein that is necessary for the uptake of CM remnants by the liver. This finding concurs with the increases in CM remnant in the circulation of the PHT rabbit.
Generally, an increase in TG is attributable to a lipoprotein metabolism abnormality caused by an insufficiency of LPL or hepatic triglyceride lipase (HTGL) activity, the functional impairment of hepatic lipoprotein receptors, or defect or structural abnormality of apolipoproteins. 15 In humans, endogenous and exogenous lipoproteins reportedly increase postprandially; they consume LPL and bring about insufficiency in the amount or activity of LPL and a decrease in catabolic activities by LPL. 16 In the postprandial PHT rabbit, the amount and activity of LPL became deficient because of the presence of CM released from the small intestine and VLDL released, in large amounts, from the liver.
In the postprandial PHT rabbit, a decrease in the CM remnant uptake by the liver, because of an abnormality of remnant receptors of the liver (defect, numerical decrease or decrease in activity of receptors), might be a cause of the postprandial retention of CM remnants in the PHT rabbit. In addition, evaluation of the properties of apolipoproteins in the PHT rabbit is necessary, as suggested by a report that apolipoprotein E2, among apolipoprotein E isomers, scarcely binds to receptors, causing the retention of remnants in the circulation. 17
Taken together, these observations suggest that the abnormally high postprandial TG levels in the PHT rabbit arise not only from VLDL; they partially reflect the TG derived from exogenous lipoproteins (CM remnants). We speculate that postprandial hypertriglyceridaemia in the PHT rabbit resulted from the release of large amounts of VLDL and exogenous lipoproteins into the blood, thereby causing a transient deficiency of TG degradation. An increase in remnant lipoproteins reportedly promotes atherosclerosis. 18 For that reason, we consider that the PHT rabbit is useful as an animal model to study the relationship between hypertriglyceridaemia and atherosclerosis.
For elucidation of the mechanism of abnormal lipid metabolism it is important that further studies be conducted on the activity of LPL involved in the degradation of VLDL and CM, and on the causes of the increase in CM remnants in blood.
Footnotes
ACKNOWLEDGMENTS
We are very grateful to all employees of the Laboratory Animal Center of Yamagata University Faculty of Medicine for their helpful assistance. We would also like to thank Dr T Kawai of the Central Pharmaceutical Research Institute of Japan Tobacco Inc, for his advice on the study of apolipoprotein B kinetics.
