Abstract
Pharmacokinetics of drugs may differ between small and large mammals (including humans); therefore, drug testing in animal models must be carefully designed. Sprague–Dawley rats were used in cardiac experiments, during which the lopinavir concentration in serum had to match human therapeutic levels (4–10 μg/mL). Lopinavir was administered as a co-formulated drug of lopinavir and ritonavir. It was found that after a single administration of a standard human peroral dose (lopinavir 13.3 mg/kg of body weight), the serum concentration of lopinavir was only one-tenth of the target level. It remained below the minimum target level even after 10-fold the standard dose was administered. After initial pilot tests, a dose escalation study was conducted with oral doses 10- and 15-fold the standard clinical dose of lopinavir (i.e. 133 and 200 mg/kg, respectively). A second administration 2 h later effectively increased and maintained higher concentrations during the experimental ischaemia and reperfusion periods. A dose-dependent increase in serum concentration of the drug was observed. Thus, the target therapeutic serum level of lopinavir in the rats was achieved by administrating 10- to 15-fold the standard human dose twice, separated by a 2 h interval.
Apoptosis is an indispensable treatment target in cardiology because it markedly contributes to myocardial cell death early in the course of ischaemia and is further induced by reperfusion.1,2 Human immunodeficiency virus (HIV) protease inhibitors are potentially valuable as antiapoptotic agents, in addition to and independent of their primary antiviral effect. Particularly, the beneficial effect of HIV protease inhibitors on CD4+ T-cell count in HIV-infected patients is thought to be due in part to inhibition of T-cell apoptosis. 3 Lopinavir is a potent protease inhibitor 4 that is commercially available and is used clinically as an anti-HIV medication.
Antiapoptotic effects of the HIV protease inhibitors nelfinavir and ritonavir are well documented in different models of non-viral disease associated with excessive apoptosis; these include experimental sepsis in mice, 5 mice with experimental-induced fatal hepatitis, staphylococcal enterotoxin B-induced shock, middle cerebral artery occlusion-induced stroke 6 and experimental mice pancreatitis. 7 Compared with nelfinavir, lopinavir can (in some cases) greatly increase CD4+ cell count in HIV-infected patients, a property likely attributable to its antiapoptotic properties. The primary purpose was to study the effect of lopinavir on cardiomyocyte apoptosis using a well-established rat model of ischaemia–reperfusion. However, the dose rate required to achieve an effective serum concentration of lopinavir in rats (i.e. drug serum levels similar to target therapeutic levels for humans)5–7 had not been described to date and required prior investigation.
Indeed, potential differences between species in drug pharmacokinetics need to be considered carefully when conducting animal studies. Pharmacokinetics data have been published on lopinavir in rats, dogs and humans. 8 The pharmacokinetic properties of lopinavir are known to differ between humans and rats; i.e. the drug has a shorter half-life and reaches lower maximum plasma concentration in rats.8,9 The aim was to develop and test the optimal dose rate and regimen of lopinavir in rats so that the predetermined target therapeutic serum level (i.e. 4–10 μg/mL, similar to that observed in patients with HIV treated with lopinavir 10 ) could be reproducibly achieved.
Material and methods
The Institutional Animal Care and Use Committee at Mayo Clinic approved all animal procedures.
Animals
Twenty-two male Sprague–Dawley rats (Rattus norvegicus; Harlan Laboratories, Indianapolis, IN, USA) were obtained and acclimatized for at least 48 h. Vendor health reports indicated that the rats were free of known viral, bacterial and parasitic pathogens. Mean (SD) weight was 483 (14) g. Age was between 100 and 120 days.
Rats were housed singly in ventilated, solid-bottom, polycarbonate cages with wire lids and cellulose bedding (Cell-Sorb Plus; Fangman Specialties, Inc., Cincinnati, OH, USA) in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. PicoLab Rodent Diet 20 (PMI Nutrition International, Henderson, CO, USA) and reverse-osmosis water were provided ad libitum. The room temperature was maintained at 20–22°C, and the relative humidity was 30–70%. The automatically controlled photoperiod was 12 h of light (06:00–18:00) and 12 h of dark (18:00–06:00). Autoclaved cotton material (Nestlets; Anacare, Inc., Bellmore, NY, USA) and plastic bones (Nylabone Products, Neptune City, NJ, USA) were provided as enrichment to all rats.
Study design
The study comprised two parts: pilot dose tests in five animals and a dose escalation study in 17 animals. Pilot dose tests were preliminary evaluation of lopinavir pharmacokinetics by measuring the serum concentration after a single administration. A dose escalation study was conducted in order to find the appropriate dose rate and regimen in the setting of myocardial ischaemia and reperfusion.
During pilot dose tests, animals received a single oral gavage (mean administered volume, 0.8 mL/dose) of drugs at the standard human oral dose (13.3 and 3.3 mg/kg of body weight of lopinavir and ritonavir, respectively, 4 2 animals) or the 10-fold dose (3 animals). Blood samples were taken during time intervals from 1–6 h after drug administration: at one hour after the standard dose administration in the first animal, at 2 h in the second animal, and at 2, 4 and 6 h after the 10-fold dose administration in each of the final three animals.
During the dose escalation study, two groups of animals received two equal drug administrations 2 h apart. Animals in group 1 (n = 7) received lopinavir equivalent to 10-fold the standard human dose (133 mg/kg lopinavir and 33 mg/kg ritonavir). Animals in group 2 (n = 10) received lopinavir equivalent to 15-fold the standard human dose (200 mg/kg lopinavir and 50 mg/kg ritonavir with mean administrated volume 1.2 mL/dose).
Figure 1 summarizes the timing and dosages of lopinavir, timing of blood sampling, and periods of ischaemia and reperfusion in the dose escalation study.
Schematic of the ischaemia–reperfusion model in rats shows the drug administration regimen in the dose escalation study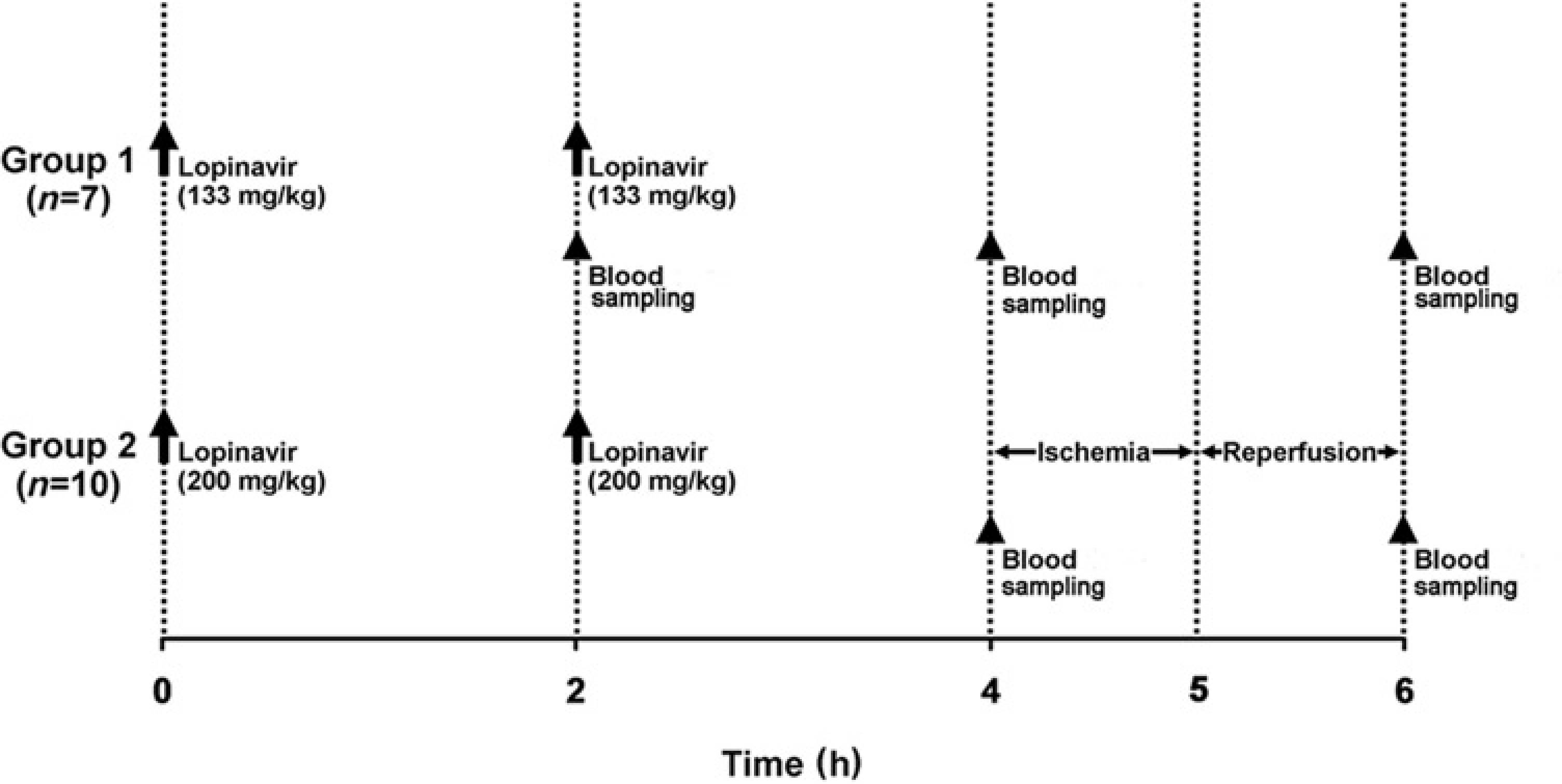
Medication
Drugs
Kaletra (Abbott Laboratories, Abbott park, IL, USA) is a coformulation of lopinavir and ritonavir (4:1 ratio). The latter component inhibits P450-mediated metabolism of lopinavir, thereby supporting the therapeutically desired plasma level of lopinavir. 9 Kaletra was available only in a peroral form. Kaletra tablets containing 200 mg of lopinavir and 50 mg of ritonavir were manually ground and suspended in 2% ethanol in distilled water. 5 A liquid preparation was also available containing 80 mg of lopinavir and 20 mg of ritonavir per millilitre.
Kaletra tablets were used only in the first two rats in the pilot dose tests. The oral liquid was used in the rest of the study because the liquid formula was more concentrated than the ground-tablet preparation; this allowed maintenance of the same administered volume (mean, 0.8 mL/dose) while delivering 10-fold the standard dose.
Dosing procedure
Medications were administered by gavage. The first administration was performed with animals under short-term anaesthesia with isoflurane. The animal was held vertically. A gavage needle was inserted along the tongue, and the drug was administered slowly, which made the animal involuntarily swallow. The gavage needle was smoothly withdrawn after administration of the drug and the animal was observed carefully until it was fully conscious.
The second administration during respirator control used the same gavage technique, only it was performed on the surgical table, with animals under anaesthesia and after tracheal intubation. It was difficult to keep the animal in a vertical position after tracheal intubation; although all animals in group 1 received the second dose without complication after intubation, this procedure allowed partial regurgitation of the drug in five animals from group 2 (these data were excluded from the analysis). Based on this experience, for the remaining five animals in group 2, the second dose was administered before tracheal intubation while holding the animal in a vertical position. This successfully prevented regurgitation of the drug.
Blood sampling and analysis
Blood samples (0.5 mL each) were collected from a femoral vein using a 22-gauge catheter. After blood centrifugation, the serum was stored at –70°C until analysis. Lopinavir levels in the serum samples were measured using highperformance liquid chromatography (Advanced Diagnostic Laboratories, National Jewish Health, Denver, CO, USA).
Anaesthesia and surgery
Anaesthesia was induced by intraperitoneal injection of pentobarbital (40 mg/kg of body weight) (Nembutal; Lundbeck, Inc., Deerfield, IL, USA). The animals were intubated and ventilated through tracheotomy, and anaesthesia was maintained by isoflurane inhalation (1–2%). A left lateral thoracotomy was performed to expose the heart. The anaesthetized animals were maintained under ventilation control with an open chest for approximately 120 min. In the group 2 animals of the dose escalation study, a 5-0 polypropylene ligature was placed around a proximal portion of the left anterior descending coronary artery and tied by a slip knot. Coronary occlusion was maintained for 60 min; the slip knot was then released and reperfusion followed for 60 min. After completion of the pilot dose tests and dose escalation study protocols, the animals were euthanized.
Results
Pilot dose tests
In the first two animals receiving the standard human dose, the serum level of lopinavir was 0.399 μg/mL at one hour after administration in one animal and 0.538 μg/mL at 2 h after administration in the other, i.e. approximately one-tenth of the minimum target serum concentration (Figure 2, empty circles).
Serum concentration of lopinavir in the pilot dose tests over time. Animals received a single dose of lopinavir. (○ 13.3 mg/kg of body weight; • 133 mg/kg of body weight) The dotted line represents the minimum target for serum concentration level. Time starts at 0 h with the administration of the first dose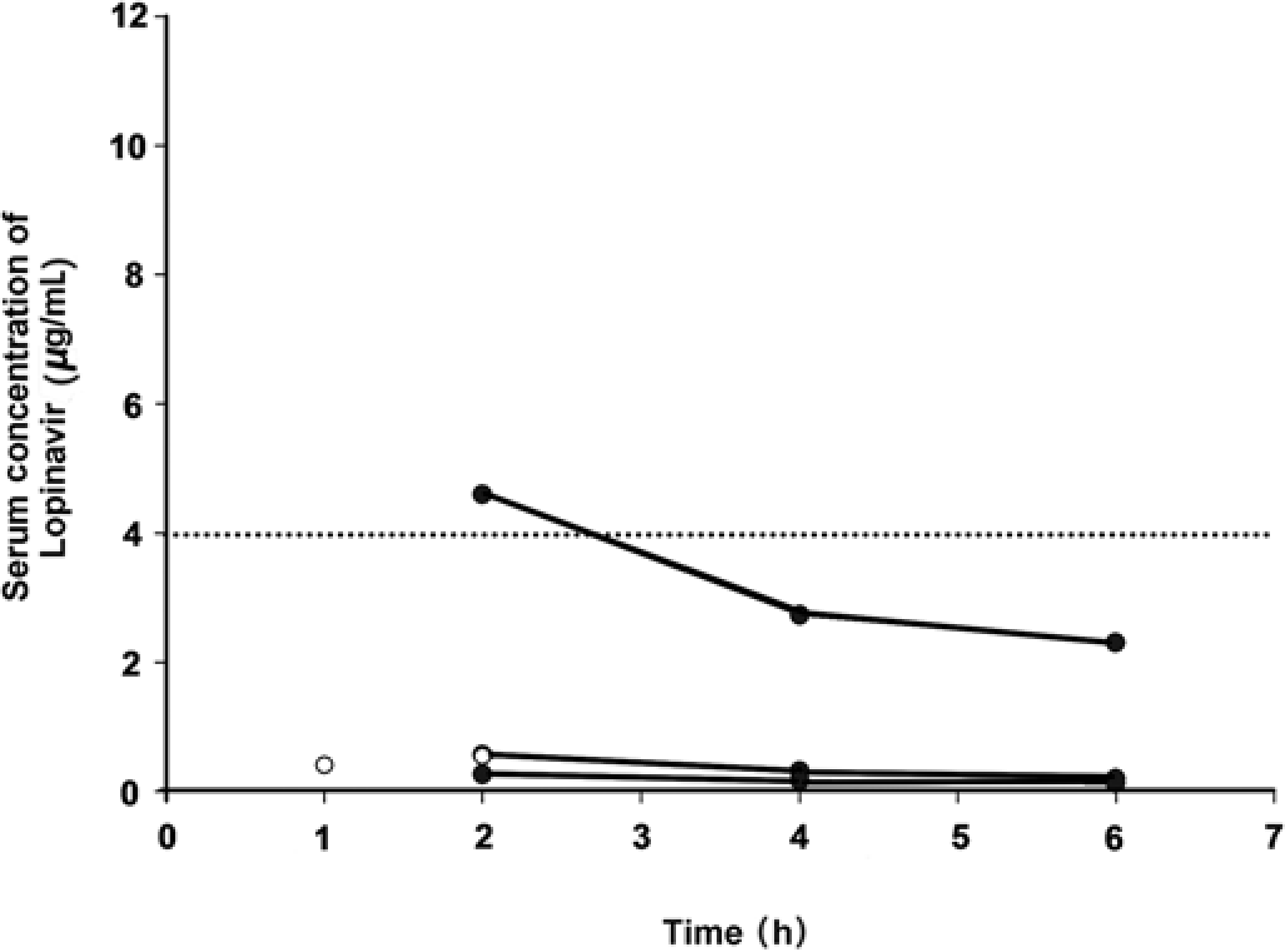
The results from the latter three animals receiving a larger dose of liquid Kaletra showed that the maximum measured serum concentration (mean ± SD, 1.8 ± 2.4 μg/mL) was achieved 2 h after administration and then decreased (at 4 h, 1.1 ± 1.5 μg/mL; at 6 h, 0.9 ± 1.2 μg/mL). The minimum target concentration was only reached at one time point in one animal out of five in the overall pilot dose tests (Figure 2, filled circles).
Dose escalation study
Considering the preliminary results, it was decided to administer a second dose of lopinavir and further increase the peroral dose to achieve the desired serum concentration of lopinavir (Figure 1). In group 1, mean ± SD serum concentration of lopinavir was 5.5 ± 1.9 μg/mL at 4 h and 5.3 ± 2.1 μg/mL at 6 h after the initial administration. These levels, which were considerably higher than that reported in the pilot tests, achieved the predetermined minimum target serum level in four of five animals at 4 h and in five of seven animals at 6 h after the initial administration. The second administration appeared to support maintenance of the target level throughout the mock 2 h ischaemia–reperfusion period (Figure 3a). In group 2, serum concentration of lopinavir remained high throughout the whole ischaemia–reperfusion period (7.4 ± 0.9 μg/mL at 4 h and 6.5 ± 2.2 μg/mL at 6 h after initial administration) (Figure 3b).
Serum concentration of lopinavir in the dose escalation study over time. Mean (SD) concentrations are shown for several time points. (a) Group 1 (n = 7) received two doses of lopinavir (133 mg/kg of body weight; 10 times the human oral dose). (b) Group 2 (n = 5, data from five animals were excluded because of drug regurgitation) received two doses of lopinavir (200 mg/kg of body weight; 15 times the human oral dose). The dotted line represents the minimum target for serum concentration level. Time starts at 0 h with the administration of the first dose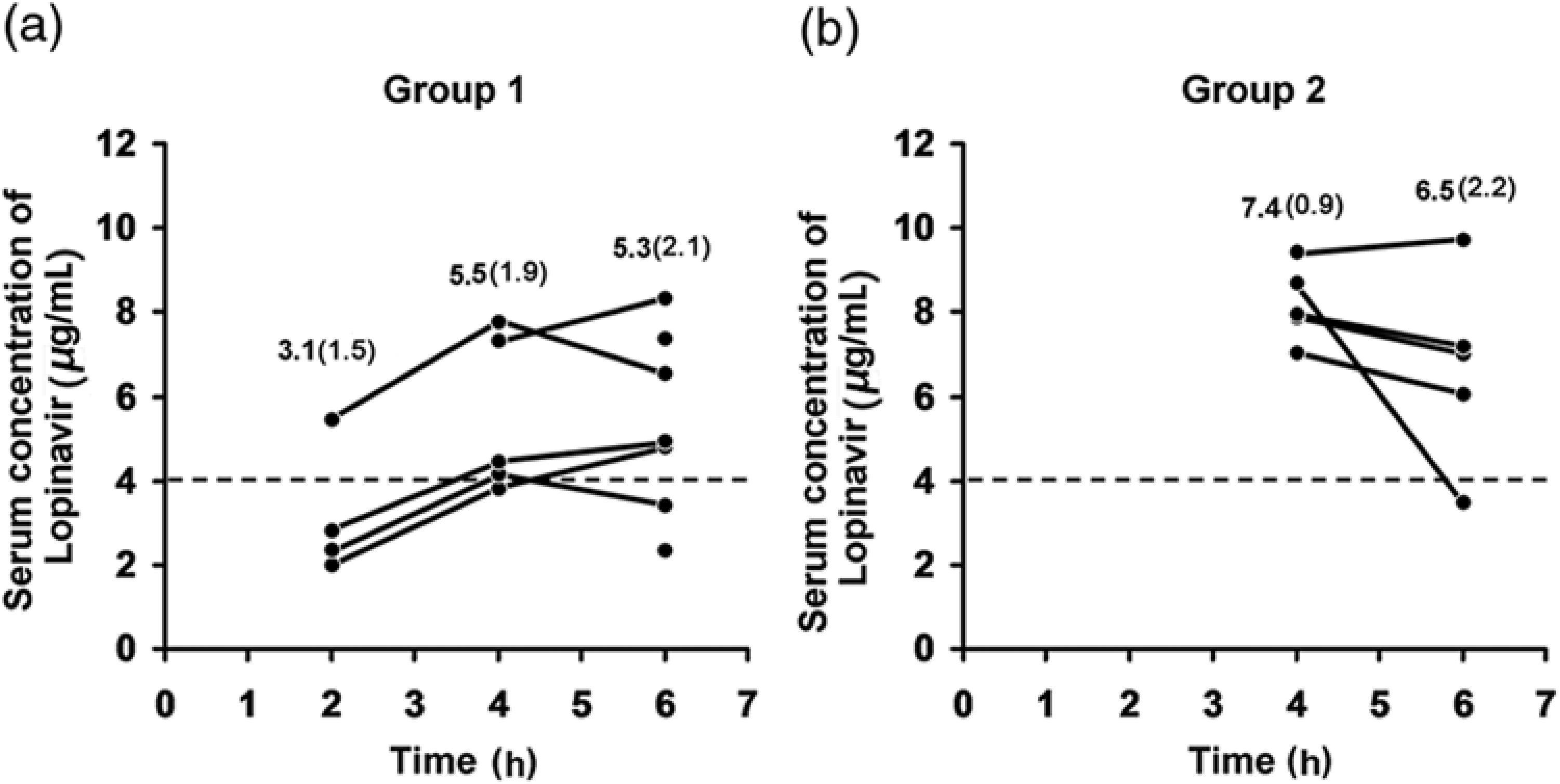
Discussion
Multiple factors may affect the actual serum level of the investigated drug in rat blood circulation. Determination of the dose to be administered in preclinical animal studies simply by adjusting the recommended human dose for body weight can be considerably misleading. Results showed that in a rat experimental model, the serum lopinavir levels that correspond to human therapeutic levels could not be achieved by using a body weightadjusted standard human peroral dose. Only increasing the dose to at least 10-fold the standard dose and repeating that dose after a 2 h interval achieved the target drug serum level, although considerable variability among individuals was found. A dose which was 15-fold the standard lopinavir dose led to a further increase of the drug serum level, although marked inter-individual variability remained.
Interestingly, in a mouse model, in order to obtain therapeutic levels of nelfinavir (another antiapoptotic protease inhibitor) similar to those achieved in humans, the nelfinavir dose had to be increased three-fold and the drug had to be combined with ritonavir, 5 which decreased elimination of nelfinavir. 9 The data presented here are supportive for conclusion that careful attention should be paid to drug dosage optimization in rodents due to possible differences in pharmacokinetics of a drug as compared with that in humans.
There were drug- and procedure-related difficulties that had to be overcome to achieve the target (i.e. antiapoptotic) serum concentration of lopinavir. Firstly, although the metabolic profile of lopinavir in different species is qualitatively similar, the peak concentration of the drug is lower in rats compared with humans.8,9 It is difficult to estimate the role of a number of factors (bioavailability, metabolic rate, etc.) in the species-related difference in lopinavir serum concentration. Nevertheless, it can be noted that serum concentration of lopinavir in humans increases by concurrent ingestion with dietary fat. 4 Possibly, absence of fat in rat diet is one of the factors underlying the discussed differences.
Secondly, half-life for lopinavir in male rats is approximately 2 h. 8 Therefore, two administrations within a 2 h interval were implemented to achieve a sustained serum level, as the results presented here confirmed, during the period of experimental ischaemia and reperfusion.
Thirdly, another problem to overcome was that the administered amount of the drug was relatively large for laboratory rats, and regurgitation or aspiration by gavage feeding was a potential complication. 11 Regurgitation was combated by slowing the rate of drug administration by gavage and by holding the animal in a vertical position. This problem was especially important when 15-fold the human dose (mean volume, 1.2 mL/dose) was administered, resulting in excluding the first five animals in group 2 of the dose escalation study. This issue was successfully overcome in the latter five animals in group 2 of the dose escalation study by giving the second administration before tracheal intubation. An alternative approach to reach the target serum concentration with increasing a single dose was considered, but the consensus was not to use this strategy because it would require at least a two times increase in the volume of the single drug administration, which would be impractical, as discussed above.
Lastly, the experimental setting in group 2 was based on an ischaemia–reperfusion model. Such an experimental setting inevitably is associated with various abnormalities in cardiac performance, including left ventricular dysfunction, arrhythmias or heart failure. Consequently, unstable haemodynamic conditions can occur and may negatively affect organ perfusion and, ultimately, drug absorption, distribution, metabolism and excretion. Results document, however, that all the aforementioned difficulties can be overcome by a careful initial optimization of the drug dose rate and regimen. The current dose rate and regimen of lopinavir assured its target serum concentration in the 2 h myocardial ischaemia and reperfusion model involving male rats.
Administering lopinavir at a dose 10- to 15-fold the standard human dose was necessary to reach the target serum concentration. The increase in serum concentration of the drug was dose-dependent within the range studied (i.e. up to 15 times the human dose). Similarly, high doses of lopinavir have been tested in rats to examine the possible adverse effects and toxicity of long-term use. 12 Long-term daily administration of this dosage did not cause critical adverse effects. Therefore, this dosage should be acceptable in the acute experimental setting.
This study had limitations. Firstly, the fixed commercial ratio of lopinavir:ritonavir of the drug formula used in this study is optimized for maximum clinical effect in humans, so a different ratio might have been more effective to increase the serum concentration in rats. Secondly, technical reasons such as difficulties of blood collection because of animal hypotension or hypovolaemia reduced the number of time points when the serum concentration of lopinavir could be measured. The serum analysis was skipped at some points to minimize hypovolaemia from blood loss.
In conclusion, it was demonstrated that despite the differences in pharmacokinetics between small and large mammals (i.e. rats versus humans), and despite various confounding effects associated with the experimental setting, the target therapeutic serum level of lopinavir could be achieved by optimizing the dose rate and regimen.
Footnotes
Acknowledgements
This study was supported by the American Heart Association Grant-in-Aid 2060631 (to MB). We are indebted to Dr Andrew Badley for sparking our interest in the antiapoptotic effects of protease inhibitors and for many helpful discussions, and we thank his research associate, Gary D Bren, for practical advice and help. We thank Jacquelyn Lombari for her assistance with experiments during her internship and Gillian Murphy for secretarial help.
