Abstract
Summary
Transplantation of epithelial cells into cleared fat pads is a widely used technique in the study of mammary gland biology. It was first described in 1959 and has remained a valuable technique, most recently in conjunction with the analysis of mammary anlagen from knockout mice with an embryonic lethal phenotype or reproductive defect, and for mammary epithelial stem-cell assays or analysis of precancerous cells. Mammary glands, unlike most other organs, mainly develop postnatally. When the small amount of endogenous epithelium present in the fat pad of a prepubertal mouse is removed, this clearance leaves a natural microenvironment that can be repopulated with exogenously supplied epithelial cells. Cells with the appropriate developmental potential (stem cells or progenitor cells) can regenerate the epithelial portion of the mammary gland after puberty and pregnancy. The conventional clearance of the fat pad is an involved surgical procedure. We have improved the technique and minimized surgery and recovery time, while maintaining an efficient removal of endogenous epithelium from the mammary fat pad.
Transplantation of cells into cleared fat pads of the mouse is often used to study mammary epithelial stem-cell behaviour or to analyse the effects of genetic alterations on mammary gland growth, differentiation and apoptosis. The easily accessible structure of the mammary gland and the fact that the gland is not a vital organ makes these experiments possible. The technique was first described by DeOme et al. (1959). The experimental course requires a removal of the endogenous epithelium from the fat pad of young mice; tissue fragments or cell suspensions can then be introduced into the cleared fat pad. These transplanted cells can grow and differentiate and form a ductal tree nearly indistinguishable from the endogenous one.
Although the technique of mammary gland clearing and transplantation was developed in the late 1950s, it is still most useful to answer many of the modern experimental questions. Transplantation of cells into the fat pad and consequent analysis of the resultant structures have been used to assess their stem-cell properties and their regenerative capacity (see Smith & Boulanger 2003, Visvader 2006 and for references therein). Cells that have been altered by retroviral infections have been analysed to assess the consequences of oncogene expression on the mammary gland (Edwards 2000) and have provided insights into mammary tumorigenesis (Daniel et al. 1968, Medina 1976, Edwards et al. 1996).
Transplantation of mammary tissue fragments from transgenic or knockout mice into cleared fat pads of wild-type mice has been used when the genetic manipulation resulted in a sterile animal or embryonic lethal phenotype. The fat pad environment from wild-type mice provides normal developmental conditions and thus phenotypic effects emanating from genetic alterations in the epithelial or the stromal compartments can thus be distinguished (Klinowska & Streuli 2000, Parmar & Cunha 2004).
Precancerous mouse mammary cells can be maintained and their cancerous progression observed in the cleared fat pad (Cardiff et al. 2006). It is also possible to humanize the fat pad in order to support normal and cancerous human breast epithelial cell growth (Kuperwasser et al. 2004, Liu et al. 2006).
Mice have 10 mammary glands, five on each side, located ventro-laterally. They can be distinguished as the thoracic glands, numbers 1–3, and the inguinal glands, numbers 4 and 5. The inguinal gland number 4 is most easily accessible and, therefore, commonly used for clearing and transplantation experiments.
A small mammary gland anlage can be observed in the developing mouse embryo. The glandular tree grows out during puberty and functional differentiation occurs in the adult gland during pregnancy and lactation. Proliferation of epithelial cells and the extension of epithelial ducts fill the pad during puberty. Terminal end buds are essential for the extension of the ducts during puberty. At pregnancy, alveolar cells are specified, which differentiate into secretory cells synthesizing milk proteins. After lactation ceases, the mammary gland undergoes programmed cell death and remodelling, known as involution, returning to a state comparable with the virgin gland (Hovey & Trott 2004).
Before cells can be transplanted, it is necessary to completely remove the endogenous epithelial tissue from the pad. This is most practically most feasible during the first three weeks after birth. During this time, the ductal structure is restricted to the proximity of the nipple. After puberty, the ducts extend to the margins of the mammary fat pad and they cannot be totally removed without destroying the fat pad. After removal of the epithelial cells, the remaining mammary fat pad is a microenvironment conducive for epithelial proliferation and differentiation. If epithelial cells remain after clearance, they can grow out together with transplanted cells and complicate the interpretation of the histological analysis.
The current surgical method involves the removal of the anlage using a dissecting microscope (Figures 1a–c). The abdominal skin is opened by a large Y-shaped incision. The mammary gland anlage is pulled out with the part of the fat pad closest to the nipple. The nutritive blood vessels and the area of the nipple are cauterized as well as the connection to the fifth inguinal gland, and the wound is closed. Ductal cells immediately underneath the nipple remain and may contaminate the transplant, if the nipple is left in the skin. The incision is a few centimetres in length and a large amount of tissue is disconnected from the body.
Comparison of the old and new methods of mammary fat pad clearance. Old method: (a) Lines of incision for the classical way to clear the mammary fat fad. (b) After incision, the skin is folded back to expose the entire mammary gland. The same procedure would be repeated on the contralateral side. (c) The wound is closed using six Michel clips. New method: (d) circles around the nipple mark the incision to resect the mammary anlage. (e) The skin around the nipple is excised and the nipple and only a portion of the mammary fat pad is exposed. (f) The wound is closed using two Michel clips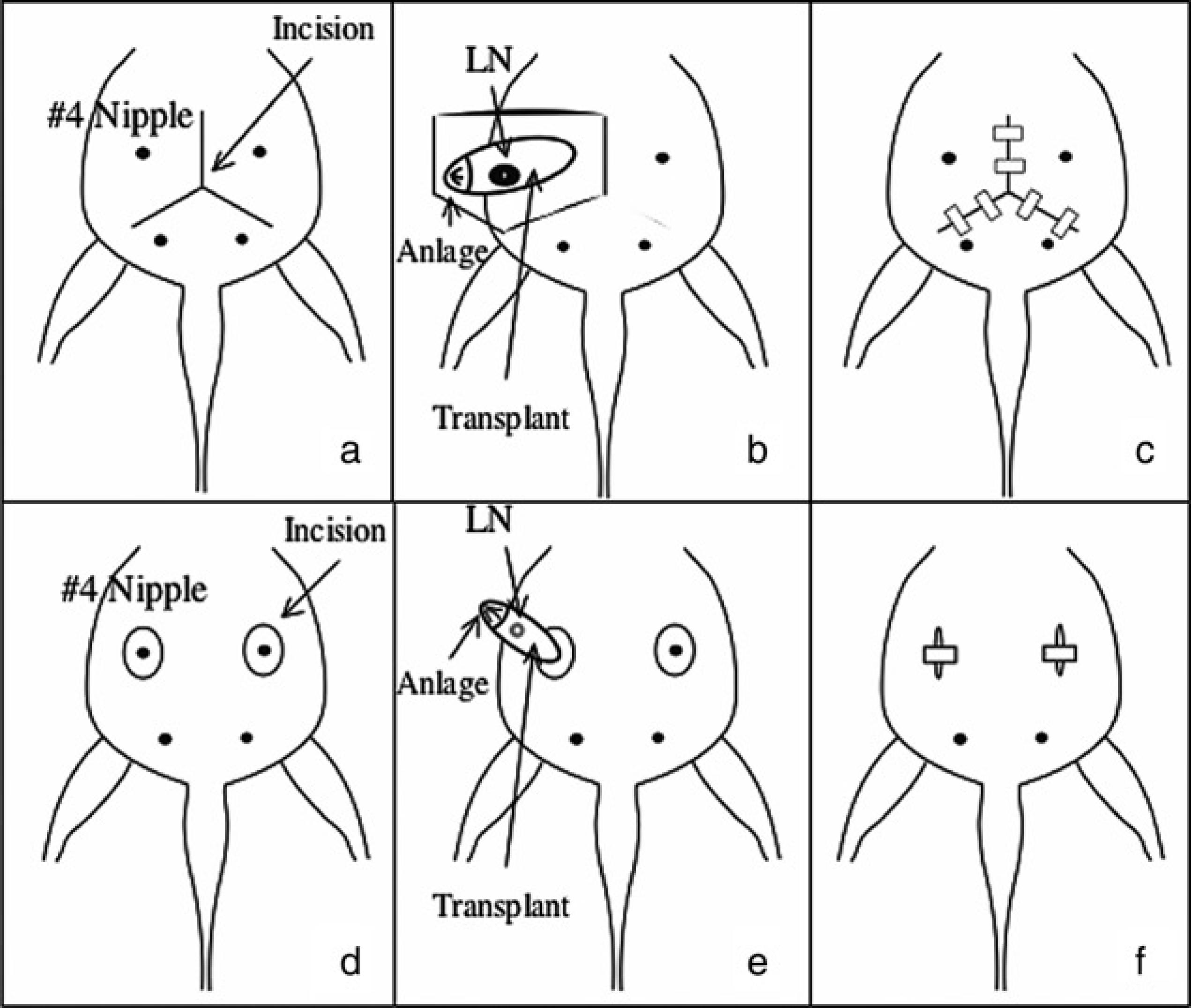
We altered the procedure to minimize the wound from the surgery and, at the same time, achieve a high success rate for fat pad clearance. We present a variation of the original method described by DeOme et al. (1959), which can be considered less harmful for the animals and an improvement of the technique.
Materials and Methods
Animals
Female C57BL/6NCrl, BALB/cAnNCrl mice (Charles River, Sulzfeld, Germany) and enhanced green fluorescent protein (EGFP) transgenic mice (C57BL/6Tg(ACTB-EGFP) Osb, Jackson Labs, Bar Habor, ME, USA) were housed conforming to the German animal welfare laws and the guidelines for animal housing from the Federation of European Laboratory Animal Science Associations (FELASA). Food and water were given ad libitum. Animals were kept in open cages or individually ventilated cages. The animal experiments were reviewed by the committee for animal welfare in Regierungspräsidium Darmstadt.
Clearing of the fat pad
Female C57BL/6 or BALB/c mice at 3–4 weeks of age were used for clearance. Mice were anaesthetized with ketamine (Pharmacia & Upjohn, Erlangen, Germany) 100 mg/kg BW and xylazine (Rompun, Bayer Vital, Leverkusen, Germany) 10 mg/kg BW i.p. and attached dorsally on a heating pad at 37°C. Alternatively, mice were anaesthetized by inhalation of enfluran (Baxter, Unterschleißheim, Germany) and analgesia using metamizol (Novalgin, Ratiopharm, Ulm, Germany) 200 mg/kg BW i.p. After disinfection of the skin, an incision around the fourth nipple was made by small scissors. The skin and the nipple were removed together with a region containing the closest part of the fat pad and the mammary anlage. The incision was closed using a Michel clip, which was removed two weeks after the surgery.
Isolation of primary mammary epithelial cells
Primary mammary epithelial cells were isolated by excision of the mammary glands from 8–12-week-old virgin mice. The whole mammary fat pad containing the epithelial cells was cut into small pieces and digested using DMEM/F12 medium 1:1 with collagenase (Roche, Basel, Switzerland) and hyaluronidase (Sigma, München, Germany). After 3 h of digestion, the tissue was washed several times in phosphate buffer saline (PBS) containing 1% fetal calf serum (FCS), the cells were collected by sedimentation and plated on collagen coated cell culture plates in DMEM/F12 medium containing 5% FCS, 1 % glutamine, epidermal growth factor and insulin (Sigma). Cells were cultured for three days before they were trypsinized and used for transplantation.
Implantation
The implant of either mammary tissue or cultured cells was inserted or injected into the remaining fat pad. Either primary mammary tissue or single cells (1 × 104 cells in 10 μL PBS) from female BALB/c or C57BL/6 mice were used for implantation into the cleared recipient. To distinguish the mammary epithelial cells derived from the host from those derived from the transplanted cell population, mammary epithelial cells from transgenic animals carrying the EGFP gene (C57BL/6Tg(ACTB-EGFP)Osb) (The Jackson Laboratories) (Okabe et al. 1997) were used as donors.
Whole mounts
Morphological analysis of mammary gland tissue was carried out. Whole mammary glands including the fat pads were fixed with 4% formaldehyde in PBS on glass slides and stained with haematoxylin for 8 h. After staining, the slides were rinsed in water for 15 min. The whole mounts were dehydrated in decreasing concentrations of ethanol before clearance in xylene and mounted with Euckit (Merck, Darmstadt, Germany).
Tissue from animals implanted with EGFP positive cells was native and analysed under a fluorescence microscope. The EGFP expressing tissue was detected at a wavelength of 488 nm for excitation and 509 nm emission.
Results
Overview of the method
Our method minimizes the invasiveness of the mammary fat pad clearance procedure (Figures 1d–f). First, the nipple of the number 4 mammary gland is identified and marked (Figure 2a). An incision is made around the nipple (Figure 1d) and part of the fat pad is pulled up through the incision (Figures 1e and 2b). The nipple and the portion of the gland extending to the lymph node is excised. This excision is important for two reasons. The first is that the contaminating epithelial cells near the nipple and the elongating ductal tree are removed (Figures 3a, b). The second reason is that the connection between the fourth and fifth glands is disrupted by the excision. At this point during the surgery, material can be transplanted into the remaining fat pad (Figure 1e). The small incision can be easily closed (Figure 1f).
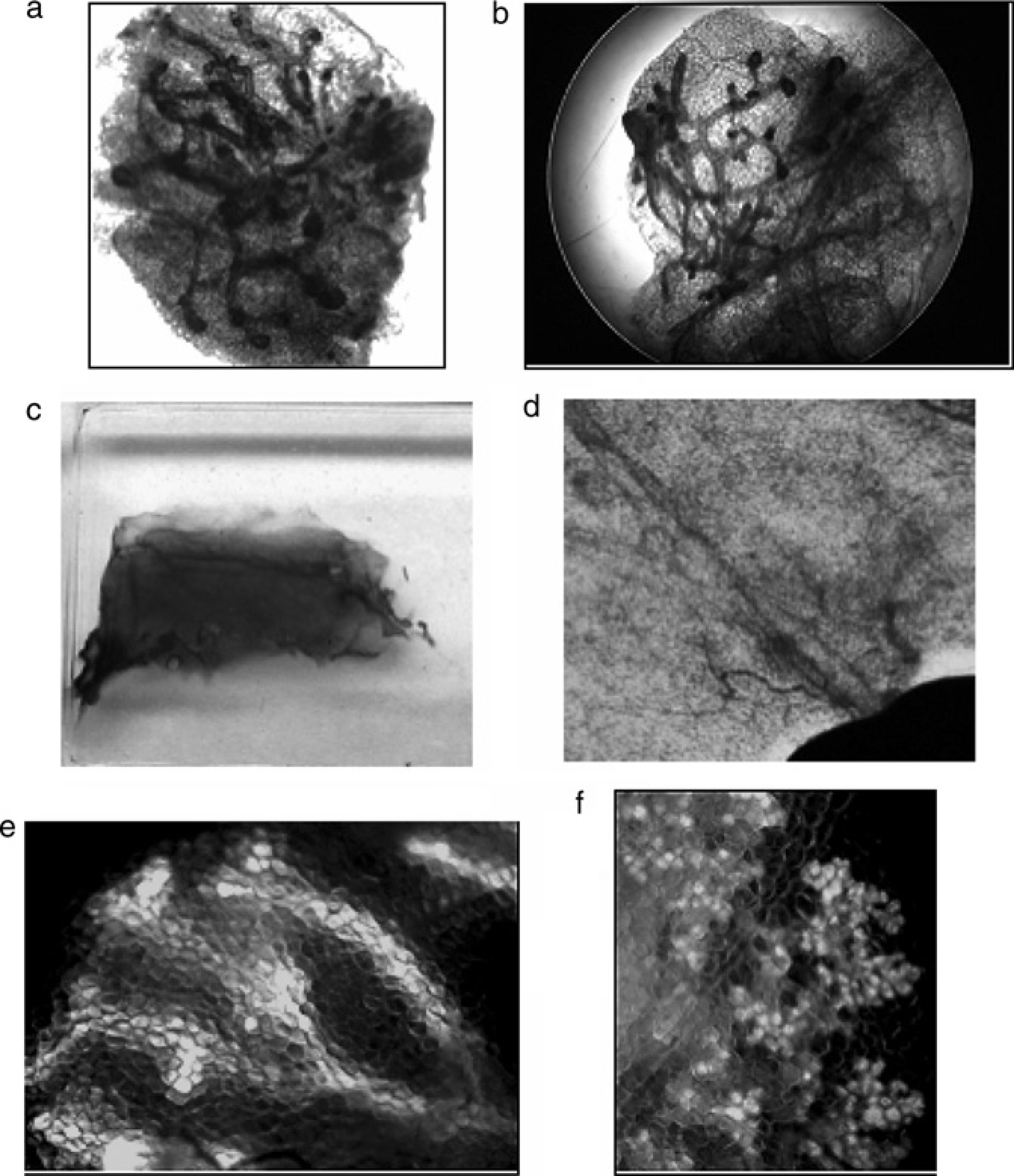
Removal of the mammary anlage using the new method. (a) The 4th mammary nipple marked in black in preparation for the surgery. (b) Resection of the skin containing the nipple and fixation of the fat pad with the mammary anlage. The exposed portion of the mammary gland will be excised in order to remove the epithelial portion of the gland. Note the very small incision and that the skin does not need to be fixed in place to expose the gland as is necessary in the classical procedure Histological examination of the mammary gland mounts. (a, b) Explants (mammary anlage) removed during surgery and stained with haematoxylin. The initial outgrowth of the ductal tree of the early pubertal gland is detectable. (c) Whole mount of cleared fat pad six weeks after clearance. (d) Magnified view of cleared fat pad. (e) Primary cells of enhanced green fluorescent protein (EGFP) transgenic mice were implanted into the cleared fat pad of wild-type mice, and the resulting regenerated mammary gland of the juvenile shows fluorescent ductal structures. (f) Pregnancy induces the transplanted EGFP tissue to form alveoli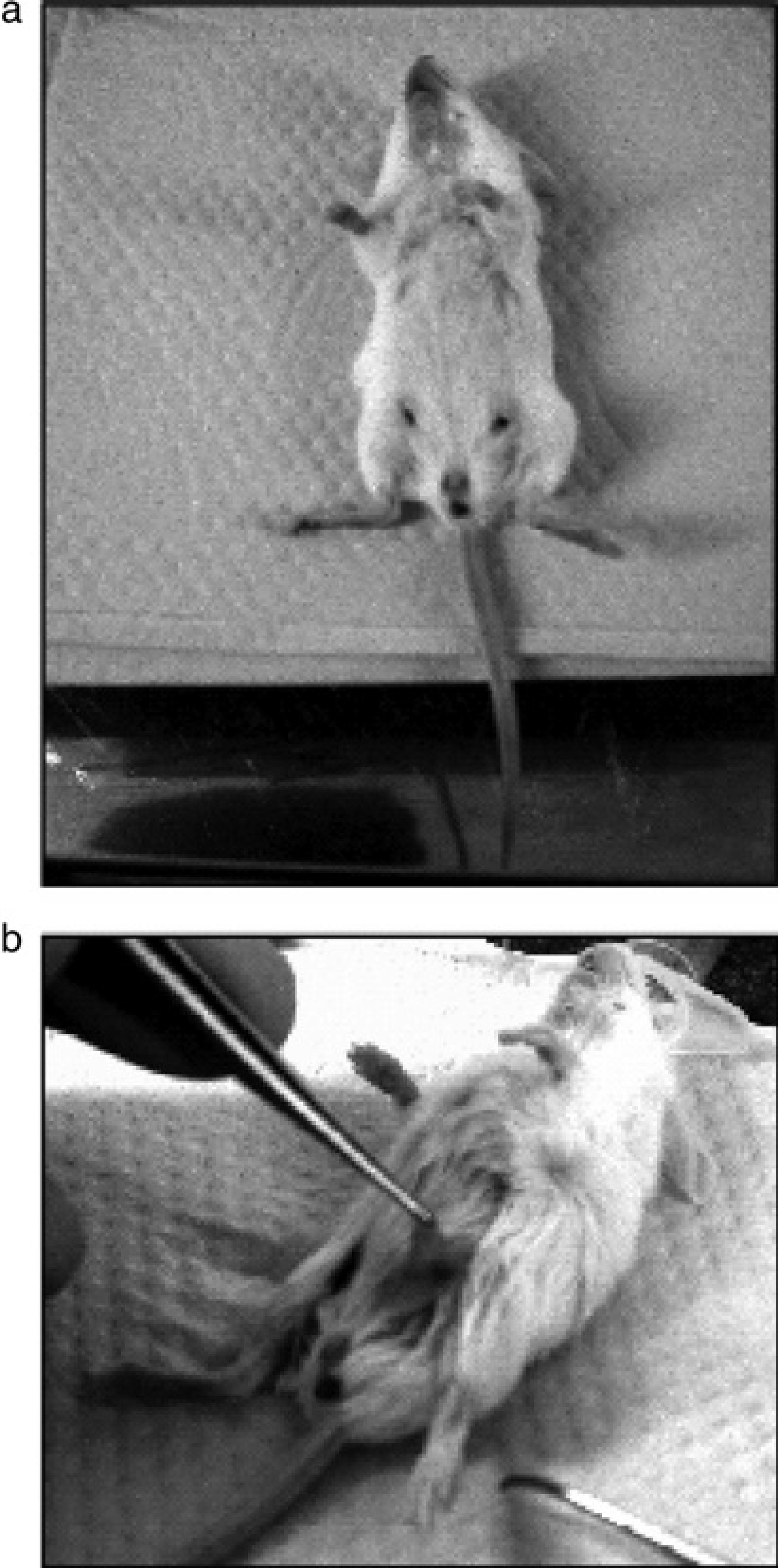
Success of the transplantation method
Our modification of the fat pad clearance-method successfully produced mammary fat pads cleared of endogenous epithelial tissue (Figures 3c, d). The subsequent development of the donor mammary epithelial tissue in the host mammary fat pad was very similar to the results described by DeOme et al. (1959). When cleared mammary glands were analysed six weeks to three months after implantation, contaminating endogenous mammary epithelial tissue was not detected in 125 of 130 cleared animals (96%), indicating a very good rate of clearance.
To distinguish endogenous and transplanted mammary epithelial cells beyond doubt, we used primary cells from EGFP transgenic animals for the implantation into cleared mammary fat pads of wild-type mice. We analysed the ability of the EGFP expressing mammary epithelial cells to repopulate the fat pad using fluorescence microscopy. Transplantation of EGFP-expressing cells into a cleared fat pad showed the successful repopulation of the fat pad including ductal structures in the non-pregnant gland (Figure 3e) and pregnancy-induced alveoli (Figure 3f).
Animal welfare
Our procedure is less harmful to the welfare of the animals. We did not observe complications during wound healing after the surgery. In order to determine the extent of tissue adhesion, we performed dissection after surgery. We observed that adhesions related to scarring after surgery were not found using our protocol (data not shown), but could be seen with the classical method.
Owing to the minimal invasiveness of our procedure and the short time period required for surgery, approximately 5 min once the anaesthesia takes effect, versus 25 min for the classical procedure, inhalation anaesthesia could be applied. Consequently, the young animals had a shorter recovery period after surgery. The recovery time by using injected analgesia and inhalation anaesthesia was 2–3 minutes. Under these conditions, no deaths due to complications of anaesthesia occurred. Intraperitoneal-injected anaesthesia requires a long recovery time until the drugs are eliminated and is accompanied by reduced acceptance of the pups by the mother. We noted fewer problems with acceptance of the pups using shorter exposure to the drugs and smaller cutaneous incisions.
Discussion
Mammary gland transplantation is a widely established method for testing the developmental capacity of mammary cells and tissue in vivo. In order to obtain completely cleared mammary fat pads, it is necessary to work with animals of 2.5 to 3.5 weeks of age, depending upon the mouse strain, in which the outgrowth of the ductal tree is still very limited. This young age adds to the complication of surgery. The original procedure requires a large incision and longer treatment time with anaesthesia. The subcutaneous position of the mammary fat pad naturally allows a modified approach that reduces the size of the incision while maintaining successful clearance and ease of transplantation. This modified procedure is also highly practical and can be performed quickly, which becomes important when dealing with large numbers of mice.
Both methods prepare the mammary fat pad as a microenvironment for mammary epithelial cells, and both surgeries still result in the destruction of the mean excretory duct. The increasing pressure produced in the tissue during lactation leads to apoptosis of the secretory cells and involution of the regenerated mammary gland.
The classic procedure for clearing the fat pads has been relatively unmodified since its first description in 1959. Our modification ensures a faster procedure, reduces the size of the incision and provides for a faster recovery time of the mice. It makes it possible to use mice at two weeks of age, if necessary, and return them successfully to the mother. Surgery that results in a large incision closed by three or four clips or with a suture, often results in rejection or ingestion of the pup by the mother. This leads to the use of older mice with an age of more than three weeks and a lower complete clearance rate, due to ductal elongation occurring in older pups. The incomplete clearance is higher in mice 21–28 days of age than in animals 14–21 days old.
We established a modification to the fat pad clearance procedure that reduces the effects of intensive surgery. The method is time-efficient, enables the use of short-term anaesthesia and allows the use of very young mice that can be successfully returned to the nursing mothers.
Footnotes
Acknowledgement
The Natural Sciences and Engineering Research Council of Canada funded part of this work.
