Abstract
Administering monosodium glutamate (MSG) to neonatal rodents induces obesity and type 2 diabetes. In addition, several studies have shown that MSG administered to pregnant animals can cross the placenta and reach the foetus. The present study was performed to investigate the effects of administering MSG to pregnant ICR mice on dam and neonatal growth. Pregnant mice were treated with 60 or 120 mg MSG once daily from day 5 of pregnancy to one day before parturition by subcutaneous injection. In addition, the body weights of the neonates were determined until nine weeks of age. The birth weights of neonates were not different between the control and MSG-treated groups. However, MSG treatment resulted in a lower body weight gain of neonates during lactation. In addition, this underweight of the MSG-treated group at weaning returned to normal compared with the control group at five weeks of age. Cross-fostering experiments indicated that the lower body weight gain of neonates in the MSG-treated group during lactation was due to its effects on the dam. Serum prolactin levels and mammary gland development of the mice were examined next to determine the reasons for this lactation problem. Although there were no differences in prolactin levels, morphological analyses of the mammary glands revealed apparent differences, including low numbers and altered phenotype of alveoli, between the control and MSG-treated groups. Taken together, our results show that treating pregnant mice with excess MSG induced lower neonate body weight gain during lactation.
Monosodium glutamate (MSG) has been widely used since its identification as a flavour enhancer in foods in 1908. The safety of MSG has been affirmed by the governments of most countries under conditions of normal use.1,2 However, there is still a controversy regarding its safety among officials and scientists.3,4 Olney first raised questions regarding the safety of MSG, i.e. the repeated injection of large doses of MSG into neonatal rodents could produce visible brain damage, notably in the hypothalamus, and increased body weight. 5 Many subsequent studies in various animal models have shown that exposure to excessive amounts of MSG leads to obesity, type 2 diabetes, macular degeneration, neuronal diseases, liver inflammation, and dysplasia.6–11 The pathophysiology of MSG administration to neonates involve the passage of MSG across the immature blood–brain barrier (BBB), allowing it to reach the brain and thus induce neuronal cell death in specific regions of the central nervous system, such as the arcuate nucleus, area postrema, and retina.12,13
Several studies have also shown that MSG can penetrate the placental barrier and cause developmental brain damage in the foetus after maternal administration14,15 using rodents as models. Hence, pregnant mice were used in this study.
Based on the results from several studies indicating that MSG in neonates induced obesity because of brain damage and that MSG can be transferred from the dam to the foetus via the placenta, it is postulated that the administration of a high dose of MSG to pregnant mice may induce obesity in their neonates. To examine this hypothesis, an experiment was done to ascertain the changes in body weight of offspring from dams that received MSG until nine weeks old. In addition, in this study, an assay was done on serum prolactin levels of the dams and their mammary gland development was analysed because the neonates of dams in the MSG-treated group had lower body weight gain during lactation.
Materials and methods
Animals and MSG treatment
ICR female mice aged 7–8 weeks were used in this study. The mice were provided with drinking water ad libitum and with a normal diet. They were kept under a 12 h light–dark cycle at 24 ± 1℃ and 50% humidity. Female mice were mated with ICR males >10 weeks old. The presence of a vaginal plug was taken to indicate day 1 of pregnancy (P1). Pregnant female mice were divided into three groups (n = 4 each). All pregnant mice included in the study were primiparous. MSG was administered by subcutaneous injection on the back from P5 to one day before parturition. MSG (Sigma-Aldrich, St Louis, MO, USA) was dissolved in distilled water (DW) at a concentration of 60 or 120 mg/200 µL. The control mice were treated with DW in the same manner. Four pregnant mice per group were used.
Neonatal administration of MSG was carried out by subcutaneous injection of 2 mg/g body weight into newborn mice from normal pregnant females on consecutive days until four days old.
The body weight of each offspring was measured within one day after birth and litter size was standardized to eight pups with equal numbers of males and females where possible. At three weeks of age, the offspring were weaned, separated according to sex, and kept in separate cages. All mice were ear punch marked for identification. The body weight of each offspring was monitored until nine weeks of age. The mean body weight at the indicated weeks was calculated for 28 offspring, including equal numbers of males and females in each group (Figure 1). To minimize bias, the highest and lowest body weights in each sex were excluded from the calculation.
The experimental design. DW: distilled water.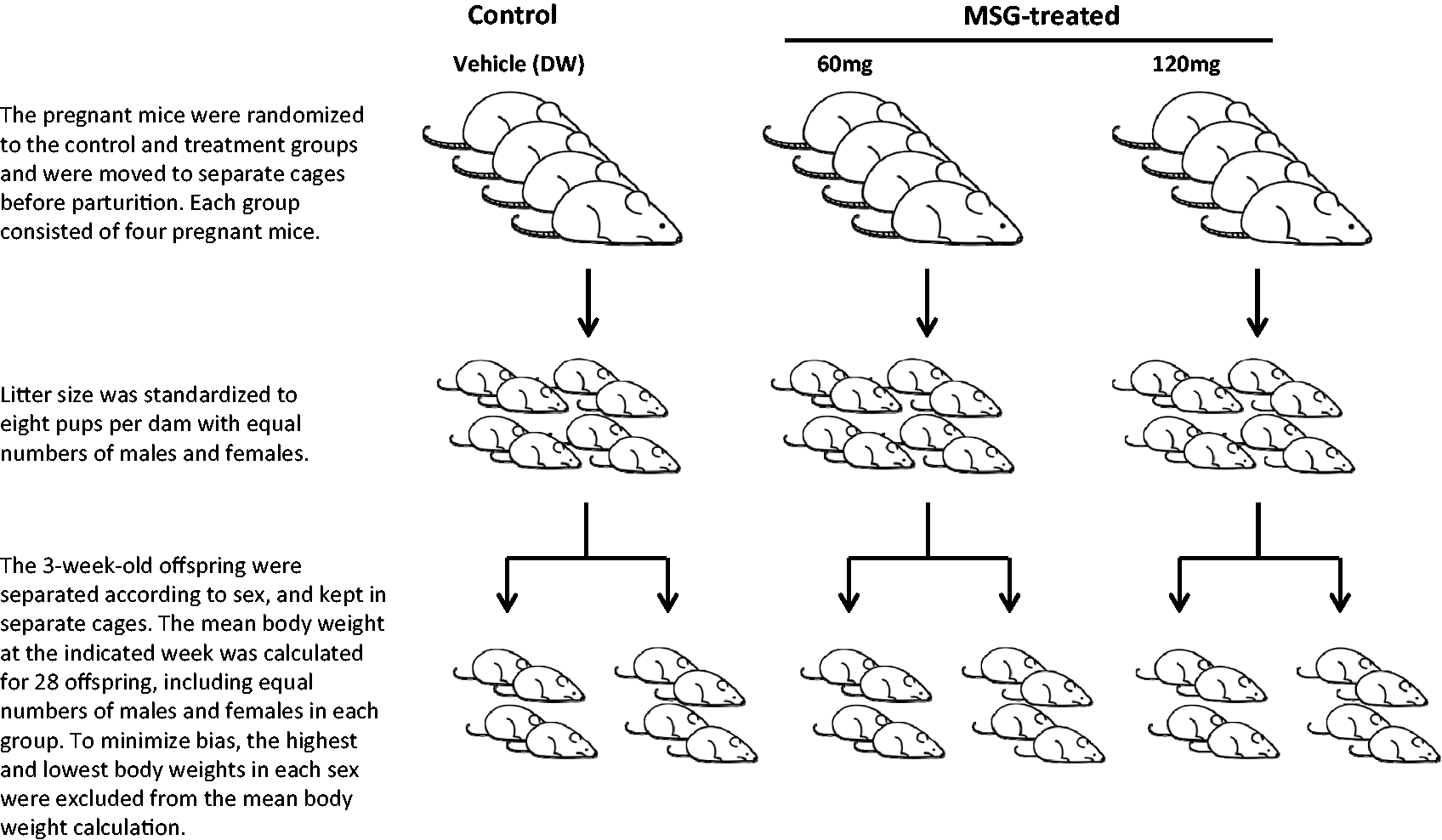
To show the differences in body weight gain between each group, the birth weight of a control individual was defined as 100%.
Cross-fostering was performed between control and MSG-treated dams to allow the exchange of their newborn mice within 24 h of parturition. Eight pups from the control dams or the MSG-treated dams were fostered to a corresponding dam from the other group.
All animal studies were conducted in compliance with the guidelines for the Care and Use of Research Animals established by the Animal Studies Committee of Dankook University. This animal care and use protocol was reviewed and approved (DKU-12-04) by the institutional animal care and use committee at Dankook University.
Prolactin assay
Serum samples were collected by tail incision from pregnant (at P12, P17) or lactating (at day 2 of lactation [L2]) mice treated with MSG or the vehicle control. Measurement of serum prolactin level was performed using a mouse prolactin enzyme-linked immunosorbent assay (ELISA) kit (Abcam, Cambridge, MA, USA) according to the manufacturer's instructions. To minimize daily variation in prolactin levels, all serum samples were collected between 17:00 and 18:00 h. The serum was aliquoted and frozen at −80℃ until use.
H&E staining of mammary glands
Mammary glands were obtained from the mice killed by cervical dislocation on day 10 of lactation and fixed overnight in 10% formalin. The fixed tissues were embedded in paraffin blocks using automated processing and embedding equipment. Tissue sections of 5 µm thick were cut and mounted onto glass slides, which were stained with Harris' haematoxylin and eosin (H&E).
Statistical analyses
The significance of differences among groups was analysed by one-way analysis of variance. Data are shown as the means ± standard error of the mean, and P < 0.05 was taken to indicate significance.
Results
Effects of MSG treatment on changes in body weight of offspring from birth to nine weeks old
There were no differences in birth weights between control and MSG-treated groups (n = 20/group) (Figure 2), while the weight gain of pups from the MSG-treated group over three weeks of lactation was decreased compared with that of the controls, and the difference in the 120 mg MSG-treated group was significant. The lower body weights in both the 60 and 120 mg MSG-treated groups recovered to the same levels as that in the control group at five weeks. In addition, body weights of all offspring from the MSG-treated groups were similar to those of the control group until nine weeks of age. On the other hand, the offspring treated with MSG (neonatal administration of MSG) as a positive control had higher body weights at five and nine weeks of age, although they were similar in body weight to the control group until three weeks of age.
Effects of monosodium glutamate (MSG) on body weight in mice. There were no differences in the mean birth weights of pups between the MSG-treated (MSG 60 or 120 mg) and control groups. The body weight gains over three weeks (during lactation) of MSG-treated groups were decreased compared with the control group. This lower body weight recovered by five weeks of age and then increased at the same rate as the control group. In the group with foetal administration of MSG (MSG foetal ad.), significant weight gain was observed after three weeks of age. Data represent the means ± SEM of 28 mice per group with equal numbers of females and males in each group. *P < 0.05 for control group versus MSG 120 group at three weeks.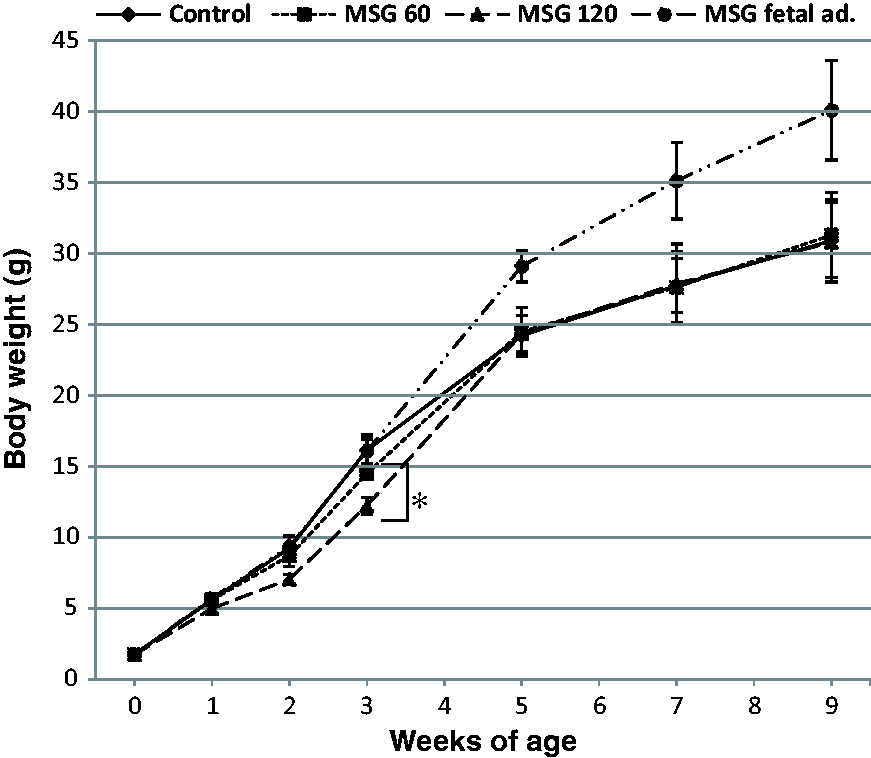
Effects of cross-fostering nursing on weight gain during lactation
Cross-fostering studies were performed next to investigate whether the lower body weight gain in neonates during lactation was due to the effects of MSG on the dam and/or offspring. Figure 3 shows that the control pups fed by dams treated with MSG had lower body weight gain than pups from dams treated with MSG fed by control dams. In particular, a significant difference (P < 0.05) in body weight was observed between the two groups at three weeks. These observations suggested that the effects of MSG on the dams are responsible for the observed lower body weight gain in pups during lactation.
Effects of cross-fostering on body weight in pups . The control pups fed by monosodium glutamate (MSG)-treated dams (msg-mom/con-pup) showed lower body weight than pups from MSG-treated dams fed by control dams (con-mom/msg-pup). The number at the top of each inside column indicates the body weight. Data represent the means ± SEM of 28 mice per group with equal numbers of females and males in each group. *P < 0.05 for con-mom/msg-pup group versus msg-mom/con-pup group at three weeks.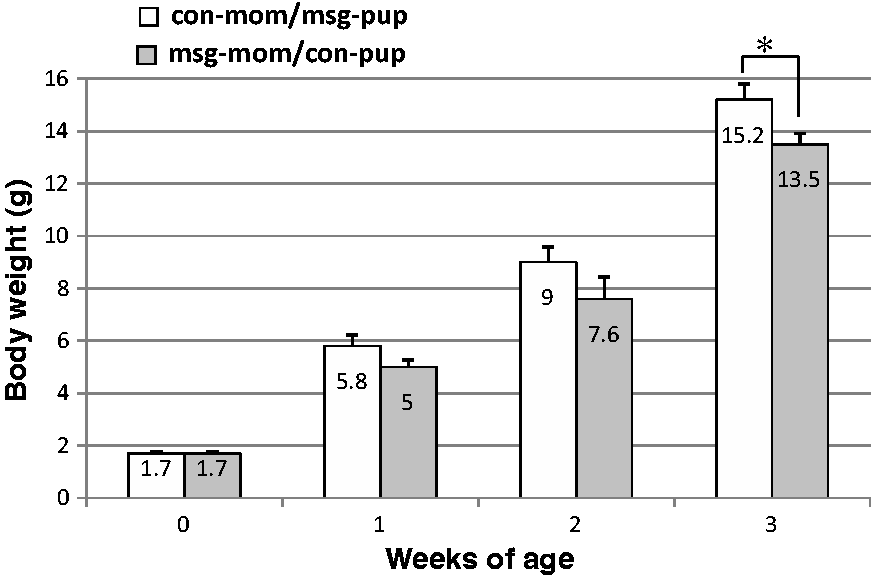
Serum prolactin levels of MSG-treated mice
Prolactin is a hormone known to promote mammary gland development and milk production. The prolactin levels were measured in serum samples collected at P12, P17, and L2 to examine the reasons for the lactation problems in MSG-treated mice. Consistent with the results reported previously,
16
low levels of prolactin were detected at P12 and P17, while high levels were detected at L2 in mice from all three groups (Figure 4). There were no significant differences in prolactin levels between the control and MSG-treated groups, although the prolactin levels at L2 in the MSG-treated groups tended to be low.
Serum prolactin levels in control and monosodium glutamate (MSG)-treated mice. Serum samples from mice for each group were collected at P12, P17, and L2 and examined for prolactin levels by enzyme-linked immunosorbent assay (ELISA). Data represent the means ± SEM.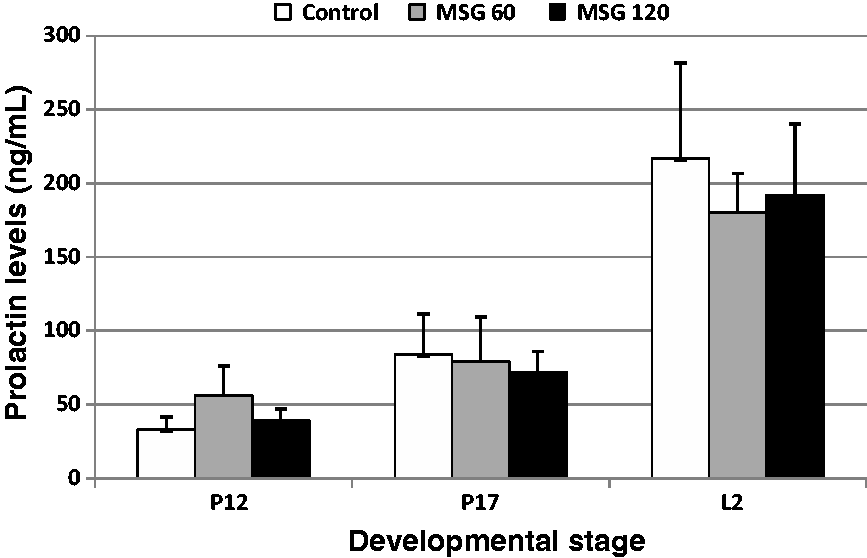
H&E staining of lactating mammary glands from control and MSG-treated mice
Histological analyses of 10-day lactating mammary glands from control and MSG-treated mice were performed by H&E staining. In the control group, the mammary glands were well developed and multiple alveoli were seen (Figure 5). However, the mammary glands of MSG-treated mice showed an abnormally low number of dilated alveoli and each individual alveolus had an altered morphology.
Histological analysis of mammary tissue on day 10 of lactation. Paraffin-embedded sections of control (a and c) and monosodium glutamate (MSG)-treated mouse (b and d) mammary glands on day 10 of lactation were stained with haematoxylin and eosin. Mammary glands from MSG-treated mice (b and d) showed altered development compared with controls (a and c). Arrow indicates presence of alveoli. Images are from one representative of groups of three mice at magnifications of 40 × (a and b) and 200 × (c and d). Scale bar = 100 µm.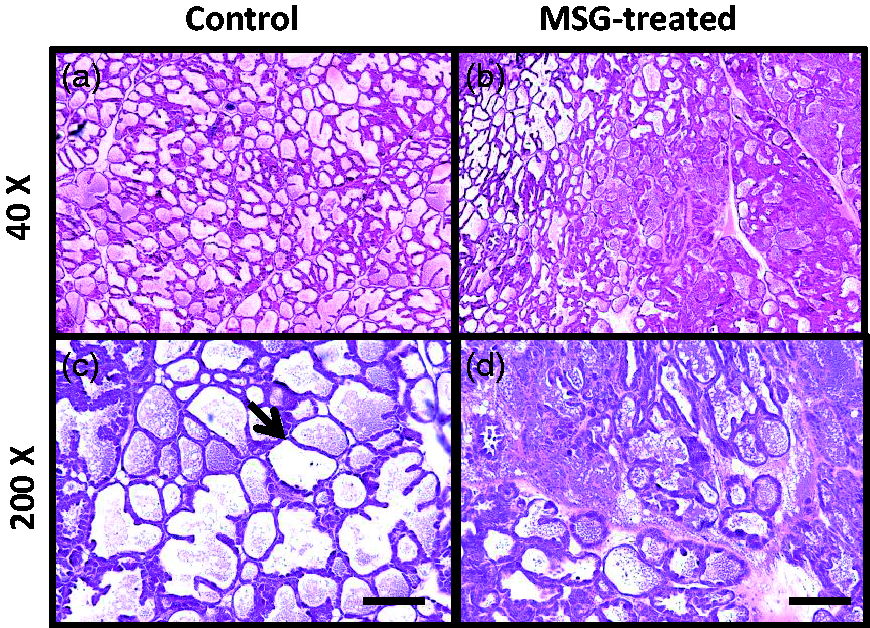
Discussion
Neonatal administration of a high dose of MSG caused destruction of the ventromedial nucleus and arcuate nucleus in the hypothalamus, which controls food intake and energy expenditure, 17 and has resulted in obesity and type 2 diabetes.5,8,9 Several studies have also shown that MSG in pregnant animals could be transported across the placenta into foetal tissues, including the brain and kidney. 14 Based on these previous observations, we initially postulated that obesity would be induced in offspring of dams treated with MSG. However, the results of the present study indicated that MSG treatment of dams had no influence on the body weight of their offspring. Instead, we found that MSG treatment resulted in a lower body weight gain of neonates during lactation.
The concentrations of MSG (60 mg or 120 mg/mouse/daily) used in this study were similar to those used for neonatal administration of MSG (2 mg or 4 mg/g/mouse), taking into consideration that the weights of mice used in this study were between 30 g and 40 g. These concentrations were about 10–50-fold greater than the human daily intake. 18 However, administration of MSG into pregnant mice for two weeks in this study might not represent an excessively high dose compared with the total lifetime human intake.
In this study, no signs of obesity were found in the pups of the MSG-treated groups based on the evaluation of their body weights until nine weeks of age. However, previous studies have indicated that oral administration of MSG to pregnant mice induced obesity of offspring with increasing age.5,8 Although we cannot explain the discrepancy in the results, there are several possible differences in the conditions, including dose used, administration route, and mouse strain. 3
MSG treatment of pregnant mice was found to have resulted in a lower body weight gain of their neonates during lactation in this study. Although similar results have been reported in pregnant rats with oral MSG administration, the causes were not discussed. 19 The results of cross-fostering experiments indicated that the effect of MSG on the dam was the primary cause of this decrease in body weight gain in the pups. In general, factors influencing weight gain during lactation can be thought of as problems affecting milk production and secretion. Histological analyses of 10-day lactating mammary glands of the vehicle control and MSG-treated mice showed both qualitative and quantitative differences in the alveoli. The mammary glands of MSG-treated mice showed an abnormally low number of dilated alveoli and each individual alveolus showed altered morphology. We could not exclude the possibility that the incomplete mammary gland development was due to the local effects of injury by the repeated subcutaneous injection of MSG. However, the adverse effects of MSG on lactation were probably systemic rather than local, as previous studies have reported similar results following oral administration. 19
Prolactin plays a crucial role in lactation, inducing mammary gland development and milk secretion. Therefore, prolactin levels were measured to examine the relation to the incomplete mammary gland development seen in the MSG-treated group. However, there were no significant differences in prolactin levels between the control and MSG-treated groups. A recent study using Src-knockout mice has indicated that low levels of prolactin receptor lead to lactation failure. 20 Therefore, further studies are required to examine the effects of MSG treatment on prolactin receptor expression.
In conclusion, treatment of pregnant mice with excessive amounts of MSG by subcutaneous injection did not induce obesity in their offspring, but induced lower body weight gain of neonates during the lactation period. This underweight during lactation was probably due to abnormal mammary gland development in the dam. Further studies are needed to determine the direct causes of incomplete mammary gland development induced by MSG treatment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by research funds provided by Dankook University in 2013.
