Abstract
Summary
To determine if administration of the anaesthetic cocktail, telazol–ketamine–xylazine (TKX) and pentothal (PEN) decreases serum concentrations of luteinizing hormone (LH) in pigs, the following experiment was performed. On day 1, eight gilts and six barrows of similar weight (75 kg) were anaesthetized with TKX (1 mL/22.5 kg body weight [BW] intramuscularly) and indwelling jugular catheters were inserted. On days 2, 6 and 8 blood samples were taken every 20 min, for 4 h before pigs were administered saline (day 2) or anaesthetized with TKX (day 6) and PEN (8.9 mg/kg BW intravenously, day 8). Blood samples were taken every 20 min for 4 h following administration of saline and anaesthetics. Mean serum concentrations of LH and cortisol did not differ (P > 0.05) within barrows or gilts from before administration of saline (day 2) to following saline administration. Mean serum concentrations of LH and cortisol were not different (P > 0.05) within barrows and gilts before administration of TKX (pre-TKX) or PEN compared with day 2 samples. Following administration of TKX (post-TKX), mean serum concentrations of LH decreased (P < 0.05) and remained decreased for 140 min, while mean serum concentrations of cortisol increased (P < 0.05) post-TKX and remained elevated for 140 min. In gilts, mean serum concentrations of LH did not differ (P > 0.05) from pre- to post-TKX. However, mean serum concentrations of cortisol increased (P < 0.05) post-TKX in gilts and remained elevated for 240 min. Following administration of PEN, mean serum concentrations of LH and cortisol within barrows and gilts were not different (P > 0.05) from concentrations before administration of PEN. Administration of TKX to barrows and gilts increased serum concentrations of Cortisol, but transient decreases in serum concentrations of LH were observed only in barrows, indicating gonadal status and/or sex may influence the ability of TKX to alter circulating concentrations of LH in pigs.
Stress can have profound negative effects on reproduction, and swine are excellent models for experiments that investigate the effects of stress on gonadotropin secretion because they respond with decreased serum concentrations of luteinizing hormone (LH) coincident with increased serum concentrations of cortisol (Estienne et al. 1991, Popwell et al. 1996, Turner et al. 1999a,b). Stress is often manifested by a decrease in serum concentrations of gonadotropins caused by an elevation in serum concentrations of Cortisol. This response has been observed in the monkey (Norman 1994), rat (Suter et al. 1988) and pig (Popwell et al. 1996). Furthermore, elevated serum concentrations of Cortisol, the predominant glucocorticoid in swine, are considered a valid indicator of stress (Stott 1981).
Accurate measurement of serum concentrations of gonadotropins requires that blood samples be collected at frequent intervals because these hormones are released from the pituitary gland in an episodic fashion (Dierschke et al. 1970, Brinkley 1981). This is usually accomplished by obtaining blood samples from indwelling jugular catheters, which are inserted either surgically or non-surgically. Surgical placement of catheters allows visualization of the vein and allows the catheter to be secured in the vein. Disadvantages of the surgical procedure are the inherent surgical complications such as those associated with infection and reactions to specific drugs used in the procedure, especially the anaesthetic, which itself may induce a stress response.
Barbiturates and their derivatives can decrease the activity of the gonadotropin releasing hormone (GnRH) pulse generator and therefore decrease pulsatile secretion of gonadotropins as has been observed in rats (Legan & Karsch 1975). Dissociative agents such as ketamine HCl have been the anaesthetic of choice for surgical procedures such as venous catheterizations in a number of species, however, its effects on gonadotropin secretion are species-specific and/or depend on the animal model used. Ketamine anaesthesia of cynomolgus monkeys has been shown to have no effect on pulsatile secretion of cortisol or bioactive LH (Malaivijitnond et al. 1998). Other researchers (Goubillon et al. 1999) have observed no disruption in pulsatile LH release in gonadectomized rats anaesthetized with ketamine. However, ketamine anaesthesia was found to inhibit pulsatile LH release in ovariectomized steroid-primed rats (Yilmaz & Gilmore 1999). Administration of ketamine to humans has been shown to cause a marked stimulation of adrenocortical function and an increase in plasma concentrations of LH (Oyama et al. 1977). In the pig, administration of ketamine has been shown to decrease serum concentrations of LH and increase serum concentrations of cortisol in gilts (Popwell et al. 1996).
An alternative to the use of ketamine alone to anaesthetize pigs has been the use of ketamine in combination with other dissociative agents and tranquillizers. An anaesthetic cocktail termed telazol–ketamine–xylazine (TKX) consists of 50 mg/mL each of tiletamine HCl, ketamine HCl (both dissociative agents), zolazepam HCl (a benzodiazepine tranquillizer) and xylazine (a non-narcotic tranquillizer). The TKX cocktail has been shown to have a rapid onset of action, and to provide excellent sedation, analgesia and muscle relaxation (Ko et al. 1994). The effects of TKX on pulsatile release of LH in the pig are unknown. A better understanding of the effects of TKX is necessary if it is to be used for surgical placement of catheters for blood sample collection to study the secretory release of LH in swine. Therefore, the objectives of this experiment were: (1) to determine if serum concentrations of LH are reduced in barrows and gilts due to activation of the hypothalamic-pituitary-adrenal axis in response to administration of TKX or the barbiturate, pentothal (PEN) and (2) to determine the duration of the suppression in serum concentrations of LH.
Materials and Methods
Animals
Crossbred (Duroc × Yorkshire × Landrace) barrows (n = 6) and gilts (n = 8) of similar age (144.6 ± 0.7 days) and weight (75.0 ± 3 kg) were stratified by litter and used in this experiment. On day 1 of the experiment, gilts and barrows were anaesthetized with TKX (1 mL/22.5 kg body weight [BW] intramuscularly [i.m.]) and an indwelling jugular catheter was surgically inserted. The anaesthetic cocktail TKX consists of 50 mg/mL each of tiletamine HCl, ketamine HCl, zolazepam HCl and xylazine. Following recovery from anaesthesia, pigs were moved to individual pens and were fed 3 kg of a 16% crude protein corn-soybean meal diet containing 0.9% lysine and 3.4 mcal metabolizable energy/kg daily and given ad libitum access to water. On day 2, blood samples (10 mL) were collected from all pigs every 20 min for 4 h beginning at 08:00 h, then 5 mL of 0.9% sterile saline was injected intravenously (i.v.) and blood samples were collected for an additional 4 h at 20 min intervals. Each sample was immediately placed into chilled tubes on ice. Blood samples were allowed to clot overnight at 4°C and then serum was obtained by centrifugation (1500 ×
Measurement of luteinizing hormone
Serum concentrations of LH were determined in duplicate by radioimmunoassay (RIA) in all blood samples (Niswender et al. 1970, Diekman & Hoagland 1983, Clapper et al. 1993). Porcine LH (AFP11043B, National Hormone and Pituitary Programme, NIDDK) was used as the radio-iodinated antigen and standard. LH antiserum (AFP15103194, National Hormone and Pituitary Programme) was used at an initial dilution of 1:300,000. Sensitivity of the assay was 0.14 ng/mL. Intra-assay coefficient of variation (CV) was 6.6% and inter-assay CV was 11.3% (n = 3 assays).
Measurement of cortisol
Serum concentrations of cortisol were determined in duplicate by RIA in blood samples obtained 1 h prior to and in all samples obtained after administration of saline, TKX and PEN using a coated tube assay as per the directions from the manufacturer (DSL-2100, Diagnostic Systems Laboratories, Webster, TX, USA). Recovery of 2, 4 and 8 ng of Cortisol added to porcine sera was 85.0 ± 5.1%, 88.3 ± 4.3% and 86.2 ± 4.4%, respectively. Inhibition curves of sera ranging from 10–25 μL were parallel to standard curves. Sensitivity of the assay was 1.24 ng/mL. Intra-assay and inter-assay CV were 12.3% and 10.8%, respectively (n = 3 assays).
Measurement of oestradiol-17β
Serum concentrations of oestradiol-17β were determined in duplicate by RIA (Kesler et al. 1977, Rempel & Clapper 2002) in the first sample taken from all gilts on days 2, 6 and 8. Oestradiol-17β (E-8875, Sigma Chemical Company, St Louis, MO, USA) was used as the standard, and tritiated oestradiol-17β (NET 517, NEN, Boston, MA, USA) was used as the tracer. Antisera (GDN no. 244 anti-oestradiol-17β-6-BSA, Gordon Niswender, Ft Collins, CO, USA) was used at an initial dilution of 1:30,000. Sera (500 μL) were extracted twice with a 3 mL volume of diethyl ether. Recovery of [3H]oestradiol-17β added to porcine sera before extraction averaged 100 ± 3%. Sensitivity of the assay was 6.8 pg/mL. All samples were analysed in a single assay with an intra-assay CV of 9.0%.
Measurement of progesterone
Serum concentrations of progesterone were determined in duplicate by RIA (Niswender 1973, Diekman & Hoagland 1983, Clapper et al. 1993) in the first sample taken from all gilts on days 2, 6 and 8. Progesterone (P-0130, Sigma Chemical Company) was used as the standard, and tritiated progesterone (TRK 413, Amersham Biosciences, Piscataway, NJ, USA) was used as the tracer. Antisera (GDN no. 337 anti-progesterone-11-BSA, Gordon Niswender) was used at an initial dilution of 1:700. Sensitivity of the assay was 0.15 ng/mL. All samples were analysed in a single assay with an intra-assay CV of 7.7%.
Statistical analyses
Based on the standard deviation of baseline concentrations of cortisol and LH, a sample size of six animals was sufficient to detect treatment differences at a significance level of 5% with a power of 85%. Statistical analyses were conducted separately for barrows and gilts because mean serum concentrations of LH differed (P < 0.05) due to sex. Data were found to be normally distributed as determined by the Shaprio-Wilk test. To correct for multiple pair-wise comparisons, a Dunnett's test was used. The effect of the anaesthetics on mean serum concentrations of LH was determined by comparing the mean of values obtained before administration of saline, TKX and PEN with those obtained after administration of the anaesthetics (Statisical Analysis System 1985). The effect of the anaesthetics on mean serum concentrations of cortisol was determined by comparing the mean of values obtained 1 h prior to administration of saline, TKX and PEN, with those obtained after administration of the anaesthetics. Differences in mean serum concentrations of oestradiol and progesterone obtained in the first sample taken on days 2, 6 and 8 (gilts) were tested by one-way analysis of variance.
Results
Anaesthesia was induced in all pigs on days 1 and 6 within 2 min of administering TKX. Pigs remained in a surgical plane of anaesthesia throughout the installation of the catheters. Mean time to install the catheters was 37.8 ± 3.5 min. Pigs regained ventral recumbency within 2.5 h after administration of TKX on days 1 and 6. Anaesthesia was induced in all pigs within 1–2 min post-administration of PEN on day 8. Pigs regained ventral recumbency within 1 h.
Mean serum concentrations of LH before administration of saline did not differ (P > 0.05) from those after administration of saline on day 2 in barrows (Figure 1a). Similarly, mean serum concentrations of cortisol in samples taken 1 h prior to administration of saline did not differ (P > 0.05) from those in the remaining 4 h period on day 2 in barrows (Figure 1a). After administration of TKX on day 6, mean serum concentrations of LH in barrows decreased (P < 0.05) within 40 min compared with pre-TKX concentrations and remained decreased for 140 min (Figure 1b). After administration of TKX on day 6, mean serum concentrations of cortisol increased (P < 0.05) in barrows within 20 min compared with 1 h prior to administration of TKX and remained elevated for 140 min (Figure 1b). Mean serum concentrations of LH prior to administration of PEN did not differ (P > 0.05) from those in the remaining 4 h period on day 8 in barrows. Similarly, mean serum concentrations of cortisol in samples taken 1 h prior to administration of PEN did not differ (P > 0.05) from those in the remaining 4 h period on day 8 in barrows (Figure 1c).
Mean serum concentrations of luteinizing hormone (LH) and cortisol in barrows (n = 6) before and after administration of saline on day 2 (a), telazol-ketamine-xylazine (TKX) on day 6 (b) and pentothal (PEN) on day 8 (c). Means are least squares means ± SD. *Means with a symbol represent mean serum concentrations of LH that differ by time (P < 0.05). +Means with a symbol represent mean serum concentrations of cortisol that differ by time (P < 0.05). Arrows indicate the time of administration of saline, TKX and PEN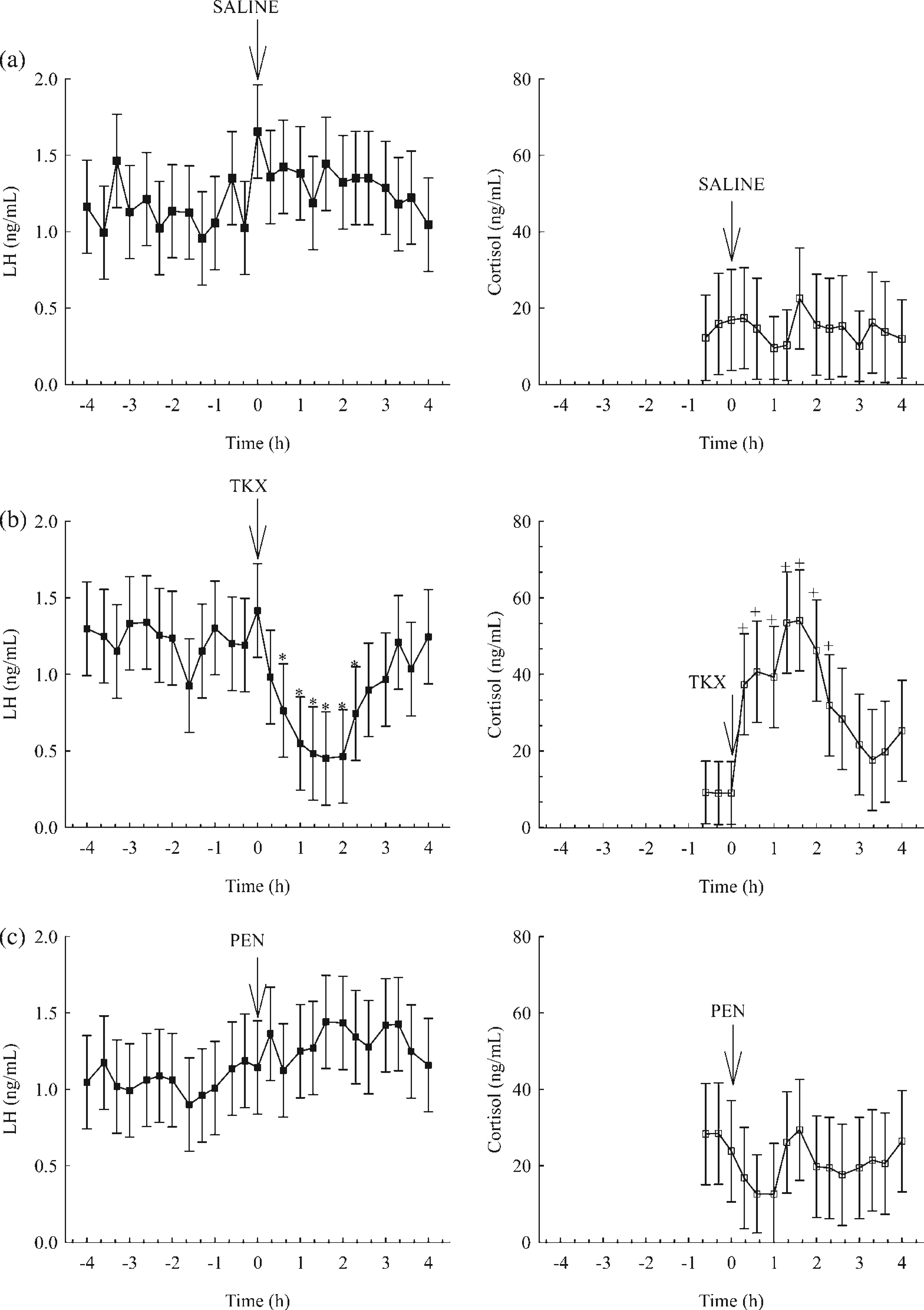
Mean serum concentrations of LH prior to administration of saline did not differ (P > 0.05) from those after administration of saline on day 2 in gilts (Figure 2a). Similarly, mean serum concentrations of cortisol in samples taken 1 h prior to administration of saline on day 2 in gilts did not differ (P > 0.05) from those in samples obtained in the remaining 4 h (Figure 2a). Mean serum concentrations of LH were greater (P < 0.05) in gilts from 220 to 260 min than at all other times throughout the blood collection period on day 6. Mean serum concentrations of cortisol increased (P < 0.05) in gilts within 20 min of administration of TKX on day 6 compared with the samples taken 1 h prior to TKX and remained elevated for 220 min (Figure 2b). Neither mean serum concentrations of LH nor cortisol differed (P > 0.05) before or after administration of PEN on day 8 in gilts (Figure 2c).
Mean serum concentrations of luteinizing hormone (LH) and cortisol in gilts (n = 8) before and after administration of saline on day 2 (a), telazol-ketamine-xylazine (TKX) on day 6 (b) and pentothal (PEN) on day 8 (c). Means are least squares means ± SD. *Means with a symbol represent mean serum concentrations of LH that differ by time (P < 0.05). +Means with a symbol represent mean serum concentrations of cortisol that differ by time (P < 0.05). Arrows indicate the time of administration of saline, TKX and PEN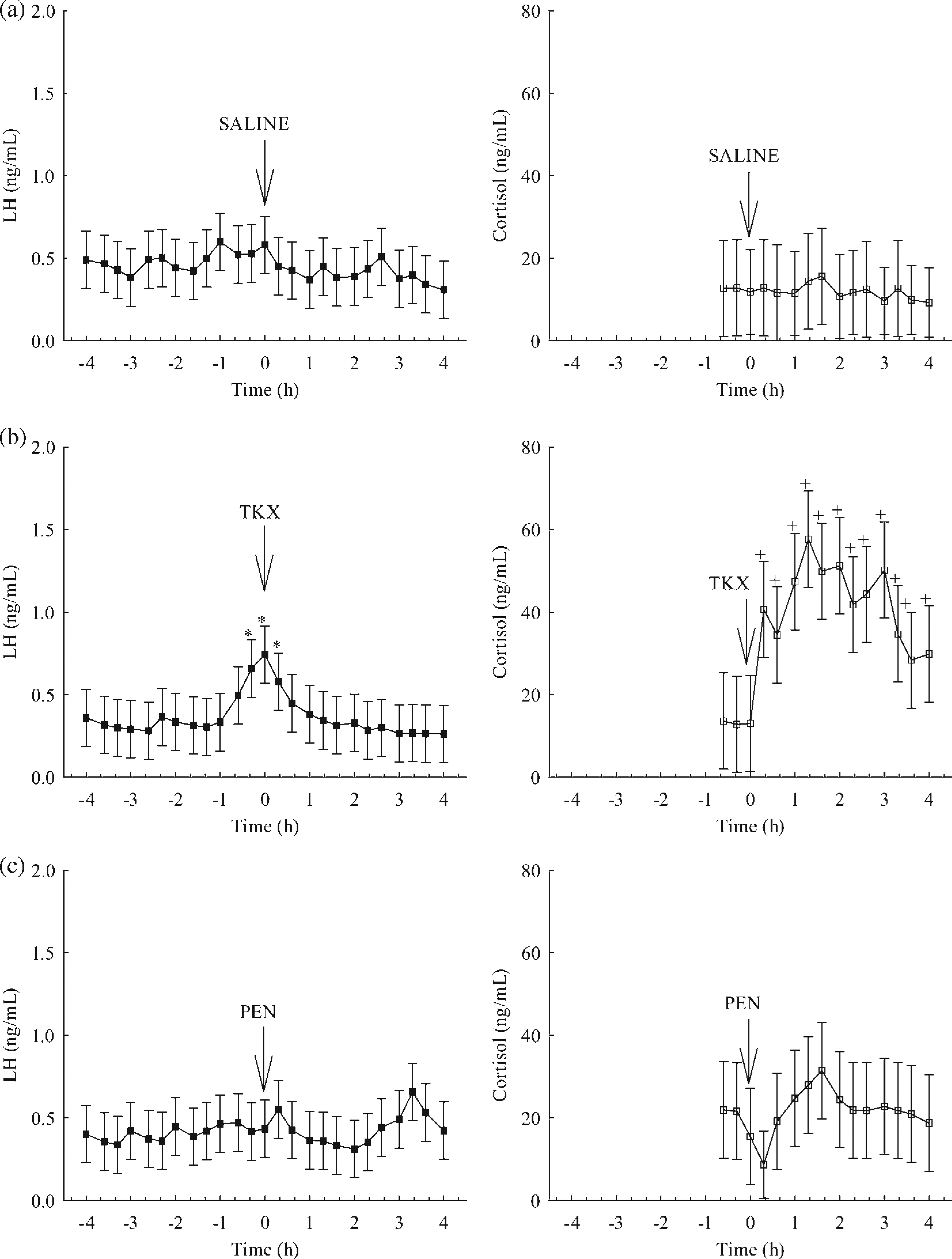
Mean serum concentrations of progesterone in gilts were not different (P > 0.05) on day 2 (0.22 ± 0.11 ng/mL), day 6 (0.15 ± 0.11 ng/mL) and day 8 (0.24 ± 0.11 ng/mL). Mean serum concentrations of oestradiol-17β in gilts did not differ (P > 0.05) on days 2, 6 and 8 and all samples were at or below the detection limit of the assay (6.8 pg/mL).
Discussion
Ketamine anaesthesia has been used in a number of species and its effects on serum concentrations of LH are equivocal. Ketamine anaesthesia of cynomolgus monkeys did not affect pulsatile secretion of bioactive LH (Malaivijitnond et al. 1998) and did not disrupt pulsatile secretion of LH in gonadectomized rats (Goubillon et al. 1999), but decreased pulsatile LH release in ovariectomized steroid-treated rats (Yilmaz & Gilmore 1999). In man, administration of ketamine caused an increase in plasma concentrations of LH (Oyama et al. 1977). It has been shown that anaesthesia of ovariectomized gilts with ketamine resulted in a decrease in serum concentrations of LH while increasing serum concentrations of cortisol (Popwell et al. 1996). However, reports of the effects of the anaesthetic cocktail TKX on gonadotropin secretion in the pig are unknown.
In the present study, administration of TKX to barrows decreased serum concentrations of LH. This is similar to the findings of Popwell et al. (1996) where serum concentrations of LH decreased in ovariectomized gilts after administration of ketamine. In contrast, serum concentrations of LH did not decrease in the prepubertal gilts in the present study after administration of TKX. Because serum concentrations of LH in gilts are naturally less than those in barrows, some may argue that it is more difficult to detect changes in LH. However, mean serum concentrations of LH in gilts in this study were above the detection limit of the LH assay, therefore, should the anaesthetics have caused a decrease in LH it would have been detected. The fact that serum concentrations of LH were elevated in gilts before administration of TKX was likely due to six gilts having coincident episodic releases of LH. It is unlikely that TKX decreased LH in gilts because serum concentrations of LH decreased in a manner indicative of the normal disappearance of LH in the blood following an episodic release (Diekman et al. 1991) and did not decrease to levels less than those measured prior to administration of TKX.
One difference which may account for the discordant response to TKX between barrows and gilts in the present study is the fact that gilts were not ovariectomized. In the present experiment, gilts were gonadally intact but mean serum concentrations of progesterone and oestradiol-17β were not different among the treatments and neither progesterone nor oestradiol-17β were at levels indicative of mid-luteal or follicular phase concentrations, respectively (Van de Wiel et al. 1981, Fonda et al. 1984). Other researchers have shown that the effect of ketamine on the gonadotropin system is dependent upon the presence of steroids and/or gonads.
Gouibillon et al. (1999) found no disruption of pulsatile LH release in gonadectomized rats anaesthetized with ketamine, but this same anaesthetic has been shown to inhibit LH release in ovariectomized steroid-primed rats (Yilmaz & Gilmore 1999). It is not certain if low concentrations of steroids or other ovarian factors may have contributed to the inability of TKX to decrease serum concentrations of LH in the gilts in the present study. Species differences may exist with respect to the role that progesterone and oestradiol-17β have in modulating the effect of TKX on LH secretion. Certainly, components of the TKX cocktail may function differently when used together, but identification of those mechanisms was not the purpose of this experiment.
Administration of TKX was accompanied by activation of the hypothalamo–pituitary–adrenal axis in barrows and gilts as evidenced by an increase in serum concentrations of Cortisol. Increases in serum concentrations of cortisol in pigs after administration of ketamine have also been documented (Popwell et al. 1996). Increased serum concentrations of cortisol may have been responsible for the decrease in serum concentrations of LH in the barrows in the present study as has been observed in ovariectomized gilts treated with adrenocorticotropic hormone (ACTH) (Estienne et al. 1991) or hydrocortisone acetate (Turner et al. 1999b). The fact that serum concentrations of LH did not decrease in gilts in this study after administration of TKX may have been due to the presence of ovarian steroids and/or other ovarian factors. Administration of ACTH or hydrocortisone acetate decreased serum concentrations of LH in the ovariectomized prepubertal gilt, but not in the gonadally intact prepubertal gilt (Fonda et al. 1984). Further evidence to support this notion is that repeated acute elevations of serum concentrations of cortisol did not disrupt the preovulatory LH surge, oestrus or ovulation in gilts (Turner et al. 1999b) nor did it inhibit the secretion of LH in ovariectomized gilts treated with oestradiol (Turner et al. 1999a).
While administration of TKX caused an increase in serum concentrations of cortisol in barrows and gilts and a decrease in serum concentrations of LH in barrows, administration of PEN had no effect on either LH or cortisol in barrows and gilts. Administration of PEN has been shown to block the preovulatory LH surge in the rat (Legan & Karsch 1975). The inability of PEN to decrease serum concentrations of LH in pigs in this study may have been due to the dosage used. The recommended dosage for pigs of this size was used and has been reported to provide adequate anaesthesia for surgical procedures such as hernia repair, tumour removal, tusk removal and castration (Muhrer 1950). Approximately, four-fold more PEN per kilogram body weight was used in the rat (Legan & Karsch 1975) than in the pigs in this study. However, response to PEN may also be due to species differences.
Administration of TKX to barrows caused a decrease in serum concentrations of LH, however, serum concentrations of LH had recovered within 140 min. Serum concentrations of cortisol increased after administration of TKX but returned to pre-TKX levels by 140 min. Even though administration of TKX did not decrease serum concentrations of LH in gilts, serum concentrations of cortisol did increase within 20 min and remained elevated for the remainder of the blood collection period. The effects of TKX on gonoadotropin and cortisol secretion in the barrow are transient. Therefore, TKX may prove to be a useful anaesthetic for surgical procedures provided approximately 2.5 h elapses before blood samples are collected for accurate quantification of gonadotropins and glucocorticoids. Serum concentrations of cortisol may require longer than 4 h before they return to pre-TKX levels in gilts.
In summary, the anaesthetic cocktail TKX may be used as an effective anaesthetic in swine for procedures lasting approximately 40 min. Caution should be exercised when it is used in studies designed to quantify serum concentrations of LH or components of the hypothalamo-hypophyseal-adrenal axis because it causes a transient increase in serum concentrations of cortisol in barrows and gilts and a concomitant decrease in serum concentrations of LH in barrows. PEN anaesthesia would be useful for surgical procedures requiring less time because it does not affect gonadotropins or cortisol in gilts or barrows.
Footnotes
Acknowledgements
This article was presented in part at the Midwestern Section of the American Society of Animal Science 2003 Meeting, Des Moines, IA. Journal Paper No. 3375, South Dakota State University Agricultural Experiment Station, Brookings, SD.
