Abstract
Summary
The effect of a commonly used anaesthetic, ketamine/xylazine and/or carbon dioxide (CO2) on plasma luteinizing hormone releasing hormone (LHRH) and testosterone concentrations was determined in male Sprague-Dawley rats. These values were compared with values obtained from pre-anaesthetic control samples. Ketamine/xylazine treatment did not significantly affect testosterone concentrations. In contrast, LHRH started to decrease one hour after ketamine/xylazine administration and continued to significantly decrease after 24 h. In addition, in the CO2 euthanasia-only group, LHRH concentrations were also significantly decreased. These results suggest that ketamine/xylazine anaesthesia followed by CO2 euthanasia 24 h later is exerting a significant effect on LHRH concentrations 24 h after anaesthetizing, while only having a slight effect on testosterone, and that CO2 is exerting an immediate significant effect on LHRH. In conclusion, LHRH analysis should be avoided after ketamine/xylazine anaesthesia and CO2 euthanasia.
Keywords
Ketamine is a commonly used anaesthetic in medicine, veterinary practice and animal research. In scientific research, it is important to verify what other effects, besides anaesthesia or sedation, an anaesthetic will have in relation to the systems to be measured, for example hormone analysis. In addition to the anaesthetic used throughout the experiment, carbon dioxide (CO2) is also often used to euthanize animals. Thus, one must be cogniscent of the effects of the first anaesthetics on hormonal changes subsequent to its application, and of the effects of CO2 used to euthanize animals.
Ketamine has been found to increase serum prolactin and cortisol concentrations in men (Hergovich et al. 2001) and increase prolactin in adult male rhesus monkeys (Rizvi et al. 2001). However, serum thyroxine and triiodothyronine were slightly lower in rats treated with ketamine plus xylazine compared with control rats (Alfonso et al. 1998). In contrast, testosterone was neither affected in the male rhesus monkey (Zaidi et al. 1982) nor were testosterone, cortisol, immunoreactive luteinizing hormone (LH) and bioactive LH affected in the male cynomolgus monkeys when ketamine was administered either as an acute or chronic dose (Malaivijitnond et al. 1998). When ketamine was given with atropine and diphenylhydantoin to rats, there was no effect on oxytocin (Zierer 1991). Goubillon et al. (1999) found that ketamine anaesthesia did not alter mediobasal hypothalamic multiunit volleys that immediately preceded LH pulses and that LH was not affected in gonadectomized rats.
Another anaesthetic, halothane, has been found not to have any effect on rat serum testosterone, LH and follicle stimulating hormone (FSH) (Cameron et al. 1983). However, a significant increase in rat adrenocorticotropic hormone-like immunoreactivities immediately after exposure and corticosterone-like immunoreactivities up to 24 h after exposure was observed, indicating the activation of the hypothalamic-pituitary-adrenal axis over a 24 h period (Karuri et al. 1998).
We have recently identified a problem when measuring testosterone, progesterone and luteinizing hormone releasing hormone (LHRH) concentrations in rat plasma up to 24 h after ketamine/xylazine sedation (unpublished results). The present study was performed in order to confirm and report the effect of ketamine/xylazine application and then the additional effects of CO2 on testosterone and LHRH concentrations in male rat plasma.
Materials and Methods
Animals and housing conditions
All work was carried out with prior approval from the Ruakura Animal Ethics Committee (no. 10412). Twenty-two Ruakura Animal Colony-bred Sprague-Dawley sexually naïve male rats (weight range 300-400 g, minimum age 12 weeks) were used in the experiment. The rats were housed in groups of four to five in single sex cages on sawdust bedding and kept at 22 ± 1.5°C, 50-70% humidity on a 12:12 h light/dark cycle (07:00:19:00). All rats had ad libitum access to food (Sharps Grain and Seed, Diet 86, Carterton, NZ) and tap water. No microbiological screening of these rats was performed in this facility as these rats are held under conventional housing conditions.
Experimental procedures
The rats were assigned to one of the three treatment groups: KX1 + CO2 (ketamine + xylazine one hour plus CO2 euthanasia) (n = 9); KX24 (ketamine + xylazine 24 h plus CO2 euthanasia) (n = 9) and CO2 only (non-anaesthetized but CO2 euthanized) (n = 4). The preanaesthetic blood samples taken from all rats, including the CO2 only group were used as the control group. All sampling was performed during the light phase of the day (09:00 h-11:00 h).
Each rat was transferred singly on the day of sampling from the home room to the surgery in a transfer cage, containing bedding, food and water as normal. Each rat was restrained by hand for approximately 5 min while the preanaesthetic blood sample was taken. All rats had preanaesthetic blood samples taken by venepuncture from the saphenous vein, collected in heparinized capillary tubes (BD VACUTAINER™; Becton Dickinson, Hamilton, NZ) and the plasma extracted and stored at -20°C until required. Plasma from the preanaesthetic control samples were pooled by treatment group because of the small volume collected and the need for large volumes to be analysed.
The ketamine-treated groups were given an intraperitoneal dose of ketamine (100 mg/mL)/xylazine (20 mg/mL)/sterile MilliQ water (3:2:3) at 0.1 mL/100 g body weight. Thus, each rat received 3.75 mg ketamine and 0.5 mg xylazine per 100 g of body weight, enough to cause sedation (approximately one hour). Following the ketamine/xylazine anaesthesia, the KX1 + CO2 group was then euthanized by CO2 one hour after anaesthesia and the KX24 + CO2 group 24 h after anaesthesia. After anaesthesia in the KX24 + CO2 group, the rat was allowed to recover in the transfer cage for 6 h before being replaced with its littermates. After 24 h, the KX24 + CO2 group were euthanized by CO2 and a blood sample was taken. The CO2 group did not receive any ketamine/xylazine anaesthesia, but were euthanized by CO2. Blood was extracted after CO2 euthanasia by cardiac puncture, collected in heparinized tubes, and the plasma was extracted and stored at -20°C until required.
Hormone measurement
Total testosterone concentrations were measured by enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, MN, USA, Cat. No. DE2300) and LHRH by radio immunoassay (Phoenix Pharmaceuticals, Inc, Belmont, CA, USA, Cat. No. RK-040-02).
Statistical analysis
The hormone data were analysed following logarithmic transformation of the data points by a t-dist test in Microsoft® Office Excel 2003 (Microsoft Corporation, USA), with all groups being compared with each other.
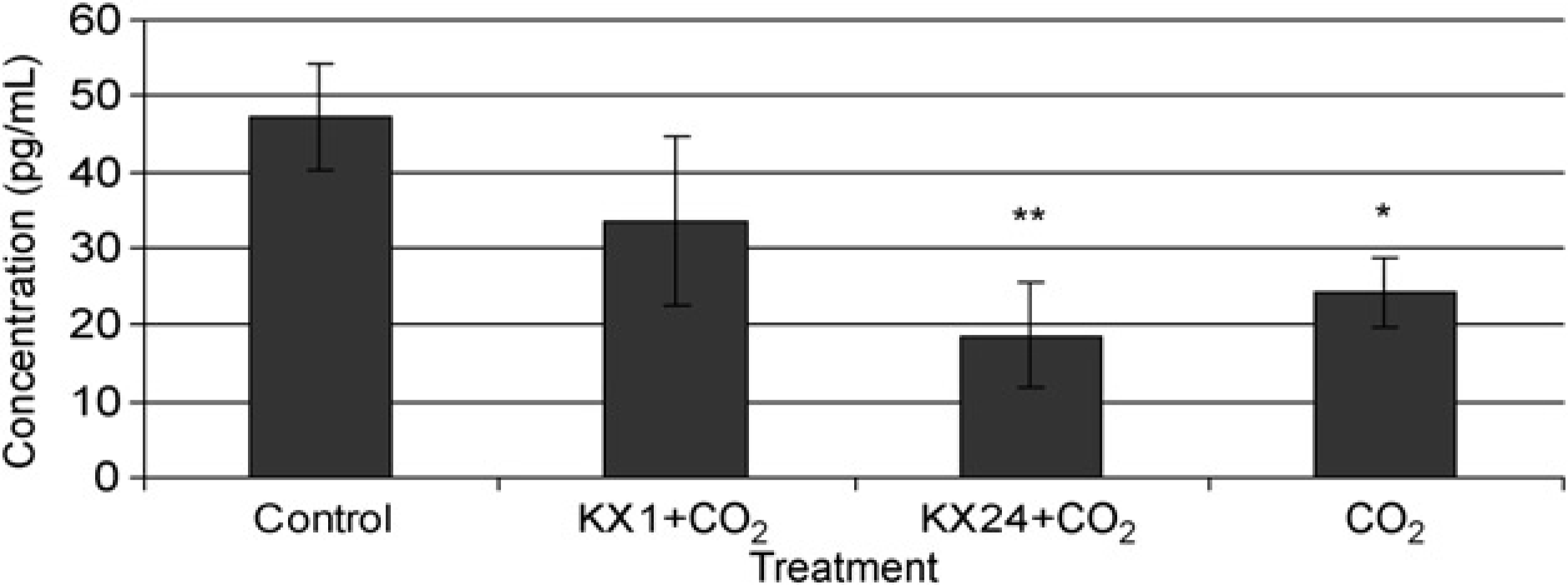
Testosterone concentration in male rat plasma pre-ketamine/xylazine anaesthesia, one hour (n = 9) and 24 h (n = 9) later and after euthanasia by CO2 (n = 4). Values are presented as the means of the treatment groups ± standard error of mean. Control is the pooled concentration of the preanaesthetic blood samples; one hour and 24 h are the concentrations of testosterone one and 24 h after anaesthesia, respectively, CO2 is the concentration of testosterone after CO2 euthanasia only Luteinizing hormone releasing hormone (LHRH) concentration in male rat plasma pre-ketamine/xylazine anaesthesia, one hour (n = 9) and 24 h (n = 9) later and after euthanasia by CO2 (n = 4). Values are presented as the means of the treatment groups ± standard error of mean. Control is the pooled concentration of the preanaesthetic blood samples; one hour and 24 h are the concentrations of LHRH one hour and 24 h after anaesthesia, respectively, CO2 is the concentration of LHRH after CO2 euthanasia only. *Denotes a significant difference between CO2 and control (P < 0.05); **denotes a significant difference between KX24 + CO2 and control (P < 0.01)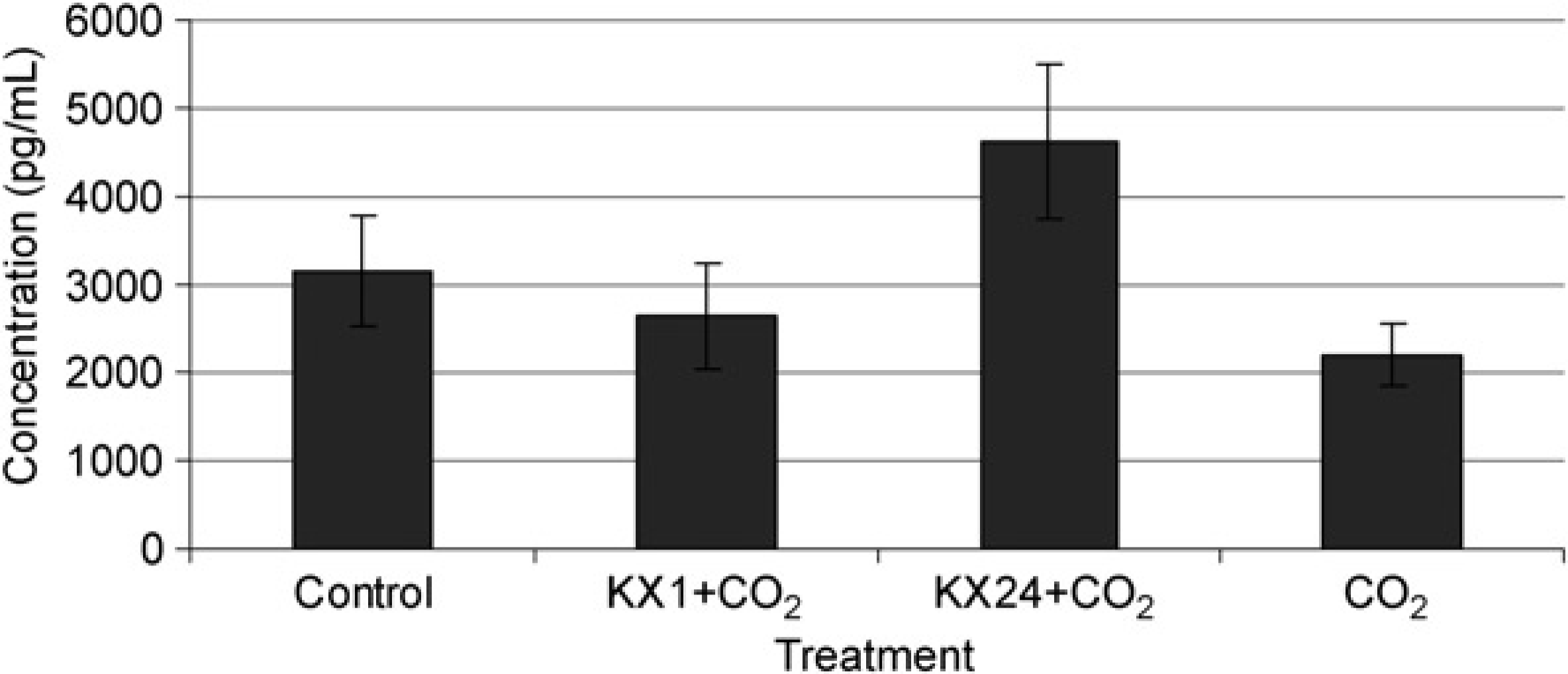
Results
Figures 1 and 2 show the effects of ketamine/xylazine, one hour and 24 h after sedation and CO2 euthanasia, as well as CO2 euthanasia alone compared with preanaesthetic control samples, on testosterone and LHRH, respectively.
There was no significant change in testosterone concentration one hour or 24 h after ketamine/xylazine anaesthesia plus CO2 euthanasia, and after CO2 only euthanasia compared with the control samples. In addition, there was no significant difference in testosterone concentration between KX1 + CO2 and KX24 + CO2, KX1 + CO2 and CO2 only and KX24 + CO2 and CO2 only groups (P > 0.05 for all comparisons).
LHRH concentration started to decrease one hour after ketamine/xylazine sedation (P > 0.05) and continued to decrease up to 24 h after sedation, becoming significantly lower than preanaesthetic control concentrations after 24 h (P ≤ 0.01). LHRH concentration in the CO2 only group was also significantly decreased compared with preanaesthetic control concentrations (P ≤ 0.05). There was no significant difference in LHRH concentration between KX1 + CO2 and KX24 + CO2, KX1 + CO2 and CO2 only and KX24 + CO2 and CO2 only groups (P ≥ 0.05 for all comparisons). The CO2 used to euthanize the animals was not neutral but had an additional effect for LHRH (Figure 2).
Discussion
The present experiment showed that LHRH concentrations after one hour were below preanaesthetic concentrations in all rats and had decreased further by 24 h. The CO2 euthanized only group also had significantly lower LHRH concentrations, thus it is possible that CO2 was exerting an immediate effect on LHRH, either synergistically or competitively. Since the CO2 only group had significantly lower LHRH concentration compared with the control group, and there was no significant difference in LHRH between the three treatment groups, there is the possibility that the decreases in LHRH were solely because of the effect of the CO2 euthanasia and not because of the ketamine/xylazine anaesthesia. However, the KX1 + CO2 group did not have significantly lower LHRH concentrations compared with the control group, whereas the KX24 + CO2 group did. This raises the possibility that one of the following is occurring. The first suggestion is that the ketamine/xylazine anaesthesia does not exert an immediate effect on LHRH, but after a period of time (24 h), the anaesthetic or its metabolites, do exert an effect, at a similar level to CO2 alone. This would also suggest that the combined effect of the ketamine/xylazine anaesthesia and CO2 euthanasia are not additive after 24 h. Another suggestion would be that the CO2 is exerting an immediate effect, as it is a highly penetrable gas, but at one hour, the anaesthesia is counteracting that effect. After 24 h, however, the anaesthesia can no longer counteract the effect, probably because it has been metabolized from its original form. A further study looking at the effect of the ketamine/xylazine anaesthesia on LHRH after 24 h without CO2 euthanasia would clarify this point.
In the present study, testosterone concentrations were not significantly affected by the anaesthetic mix. In addition, testosterone decreased slightly but not significantly in the CO2 group compared with control concentration, suggesting that CO2 does not exert any effect on testosterone.
It has been observed before in this laboratory that if a group of anaesthetized male rats are returned to their littermates before recovering from the anaesthetic, the rats that recover more quickly can attack and kill the remaining sedated littermates. Thus, the KX24 + CO2 group were kept in individual cages for 6 h after anaesthesia before being returned to their home cage and littermates. It could be argued that the separation could induce a separational stress and thus alter hormone levels. However, the testosterone levels did not change significantly. These data are supported by studies on hamsters where separation from a female did not lead to a change in testosterone levels (Castro & Matt 1997); and guineapigs where separation from littermates did not alter testosterone concentrations, but testosterone significantly decreased if the male had been housed with a single female (Hennessy 1999).
There are few publications on the effects of CO2 on animal hormones. Krystal et al. (1989) found that there were significant increases in cortisol, growth hormone and prolactin in rhesus monkeys (Macaca mulatta), but only after prolonged exposure to doses of 5, 7.5 and 10% CO2.
In humans, there have been some reports of decreases in testosterone following general anaesthesia, possibly because of surgical trauma, but more often an increase occurred one hour after surgery (Cartensen et al. 1973). In neotropical and domestic cats, ketamine/xylazine sedation did not seem to cause stress, as cortisol concentrations were reduced 30 min after sedation compared with before sedation. In addition, sedation did not affect testosterone concentrations or plasma LH concentrations (Genaro et al. 2007). These data are similar to the data in this experiment and suggest that sedation did not affect the stress levels of the rats and thus affect the hormone levels. An interesting point to note is that while LHRH showed a decreasing trend, total testosterone increased, indicating an increase in secretion. López-Calderón and colleagues suggested that the decrease in LHRH because of chronic stress is a result of decreasing LHRH secretion (López-Calderón et al. 1990, 1991).
Stress has been shown to have an effect on hormonal changes. The response depends upon the stressor (Retana-Márquez et al. 2003), the type of stress and the animal's social status (Sapolsky 1992). Acute stress, such as short-term restraint, was found to decrease hypothalamic LHRH content with a subsequent increase in plasma LH and FSH in rats (López-Calderón et al. 1990). In contrast, chronic stress decreases LHRH, LH, FSH and testosterone concentrations (López-Calderón et al. 1991). In the short term, stress may increase testosterone concentrations by increasing circulation to the testis by catecholamines and lactate. There is also evidence of neural pathways producing testosterone release (Lee et al. 2002). Stress owing to the handling of the rats (couple of minutes of hand-held restraint) and the procedures (blood sampling and anaesthesia) would be the same for all the rats used in this experiment. Therefore, the differences in hormonal responses between the different treatment groups cannot be attributed to stress in this case.
There is a certain degree of individual variation in both the hormones, although the trends seen after anaesthesia and CO2 discount this variation being the sole effect.
Since LHRH and testosterone operate in a feedback mechanism, it is difficult to ascertain whether ketamine/xylazine is having a direct effect on either LHRH or testosterone or both. LHRH is required to increase the pituitary content and circulatory levels of LH (Cattanach et al. 1977), which in hamsters, in turn increases the levels of testosterone approximately 45 min after the initial LH surge (Richardson et al. 2004a). In turn, LHRH is also sensitive to the negative feedback control by gonadal steroids, but the extent of this control is dependent on season in the seasonal breeders (Lehman et al. 1997) and on stages of puberty in other species (Richardson et al. 2004b). Since in this experiment testosterone was slightly increased at 24 h after ketamine anaesthesia and LHRH was decreased, it could be argued that testosterone was exerting a negative feedback control on LHRH. Sherwood et al. (1980) noted a loss of the LHRH proestrus surge in rats given a dose of 100 mg/kg body weight of ketamine. In contrast, Goubillon et al. (1999) did not observe any change in the LHRH pulses in rats given the same dose. Ketamine has also been observed not to have an effect on testosterone in monkeys (Zaidi et al. 1982, Malaivijitnond et al. 1998), and ketamine/xylazine and xylazine alone did not affect FSH and LH up to 24 h after administration in pony mares (Thompson et al. 1988).
This experiment has shown that up to 24 h after ketamine/xylazine anaesthesia, any measurements of plasma testosterone concentration, and after anaesthesia and CO2 euthanasia, LHRH concentration, should be treated with caution, as the real effect of the experiment or treatment could be hidden by the anaesthesia and euthanasia. In future studies, it may be advisable for the researcher to study the effect of an anaesthetic or euthanizing agent on hormones using a small group of animals, prior to embarking upon a full study.
Footnotes
Acknowledgements
The author wishes to thank Dr Christian Cook for his support with this trial. This work was approved by the Ruakura Animal Ethics Committee.
