Abstract
The Desert hamster, Phodopus roborovskii, is one of the lesser-known laboratory animal models and therefore knowledge of its reproductive system and physiology is limited. This study investigated the time course of vaginal and behavioural oestrus cycles by means of vaginal smear cytology, serum luteinizing hormone (LH) levels, wheel-running activity patterns, and pairing tests. The oestrus cycle lasts between four and six days and follows a pattern rather similar to that described for other rodent species. An important finding of this study is the existence of a fifth cycle stage between pre-oestrus and oestrus, the so-called early oestrus. Early oestrus differs from pre-oestrus by a complete lack of leucocytes in the vaginal cytology and a dramatic increase of serum LH levels just before activity onset. The early oestrus stage lasts 4–6 h, but was not observed in every female. When present it affects the length of the whole oestrus cycle. With early oestrus the pre-oestrus stage lasts only 14–18 h and the total length of the oestrus cycle length is four days. Without early oestrus, the pre-oestrus is prolonged to 18–36 h and the oestrus cycle length varies between four and six days. Desert hamsters showed only subtle oestrus-correlated changes in wheel-running activity, i.e. they failed to show the characteristic scalloping of activity onset, but showed prolonged activity during early oestrus. Pairing tests revealed characteristic changes in the relative frequencies of socio-positive, neutral, aggressive and sexual behaviour during the course of the oestrus cycle, with an elevated level of sexual behaviour during oestrus.
Hormonal changes accompanying the oestrus cycle have been investigated in great detail in rodent laboratory animals because they affect the behaviour and physiology of female animals in a complex manner.1–3 Vaginal smear histology, a well-established technique for determining oestrus stages, 4 was used to characterize the oestrus cycle in various laboratory species and revealed species-specific differences especially regarding the length of oestrus stages (rat,4–6 Golden hamster, 7 Mongolian gerbil, 8 Siberian hamster 9 ). Using an alternative, more elaborate technique, serum luteinizing hormone (LH) levels have also been analysed, because a nocturnal increase of LH triggers ovulation every 4–5 days, thereby characterizing the night of oestrus.6,10,11
The aim of the present study was to characterize the oestrus cycle in the Desert hamster, Phodopus roborovskii, a species which is of interest for chronobiological studies, especially if associated with the sociobiological aspect as this species combines well-defined locomotor activity, 12 similar to the Golden hamster, with a social way of life and with intense paternal care. Besides studies concerning physiological adaptations to desert life, 13 this species has recently been used for the analysis of chemosignals,14,15 obesity16,17 and community structure in a changing environment.18,19
Desert hamsters are the smallest hamsters of the Cricetinae family with a weight of 12–20 g and a body length of 6–8 cm. They prefer semi-desert habitats on sandy soil in Mongolia, Northern China and parts of Russia where they eat seeds and insects. The semi-fossorial species uses burrows with a single nesting chamber and a food storing chamber in order to avoid the harsh weather conditions in the desert; but hibernation or daily torpor has not been observed up to now. Different adaptations to life in the desert have been described, such as white fur colour on the ventral side, hairy foots, highly concentrated urine, and sand-coloured fur color on the back side.
It is well known from chronobiological studies in other rodent species that the oestrus cycle affects circadian rhythm parameters. For example, activity levels increase on the days of pre-oestrus and oestrus in Golden hamsters20–22 and rats. 23 In addition, activity onset occurs earlier in the oestrus cycle in rats23,24 and Golden hamsters 25 resulting in a so-called ‘scalloping’ of activity onset. Oestrus-correlated changes in locomotor activity rhythms have been interpreted as a strategy for increasing the probability for physical contact between males and females and thus increasing mating success. This hypothesis is further supported by behavioural studies demonstrating an increase in socio-positive or sexual behaviours on the day of oestrus (Golden hamster, 26 Djungarian hamster, 27 Mongolian gerbil,28,29 review8,30,31).
For an extensive study on the oestrus cycle of the Desert hamster, Phodopus roborovskii, four approaches were employed: (i) vaginal smears were used to characterize the length of the oestrus stages especially in comparison with other rodent species, such as Golden hamsters, rats and Mongolian gerbils; (ii) serum LH levels were analysed to further verify the timing of ovulation; (iii) running-wheel activity recordings were used to determine oestrus-correlated effects on circadian rhythms; and (iv) pairing behaviour in the course of the oestrus cycle was investigated.
Materials and methods
All the experiments were performed in accordance with the guidelines for the use of animals in research, the legal requirements of Germany, and all institutional guidelines. The experiments were reported to the Regional Council of Stuttgart (AZ: 35–9185.82/0236 and AZ: 35–9185.82/0246).
Animals and housing
Desert hamsters were kept in our laboratory under controlled environmental conditions (light–dark [LD] 14:10, namely 14 h light alternating with 10 h darkness with lights on at 07:00 h, regulated room temperature 22 ± 1℃, regulated relative air humidity 50 ± 10% and 15 air changes per hour), with food (breeding and maintenance diet for rats and mice in a 1:2 ratio; Altromin, Lage, Germany) and untreated mains water provided ad libitum. The animals were housed in solid bottom Makrolon® polycarbonate plastic cages with wire grid lids (Type IV, Tecniplast, Hohenpeissenberg, Germany) embedded with wood shavings (Altromin) and enriched with nesting material, houses and sand baths. Until the beginning of the experiments, the animals were housed in groups of 3–5 littermates of the same gender. Cages and bedding were renewed every two weeks, water bottles once a week. All the animals were bred from our own laboratory breeding stock, which were based on breeding pairs provided by the Institute of Zoology of Martin-Luther University Halle-Wittenberg, Germany in 2006 (three years between the start of breeding in Stuttgart and the beginning of the study with at least three generations raised up in our laboratory). After these measurements, six animals were paired and were used in our breeding stock; all the other animals were sacrificed using carbon dioxide. 32
An overall total of 59 females and 28 males were used in the experiments: 12 females and 12 males for the analysis of vaginal smears and the registration of running-wheel activity; 36 females for the follow-up analysis of vaginal smears and the final determination of LH, and 11 females and 16 males for the analysis of behaviour. The males were placed next to females in order to maintain the females’ cycles.
Histological analysis
For the experiments, 12 female and 12 male hamsters (age: 32.6 ± 2.2 weeks) were separated in single cages (Makrolon Type II) equipped with a running wheel (diameter: 26.0 cm) and enrichment (nesting material, sand bath and houses). The animals were kept in an inverse LD regime with lights on at 17:00 h and lights off at 07:00 h. Vaginal smears were taken twice a day, namely within the first hour of the dark phase, i.e. between 07:00 and 08:00 h, with the use of red lights (λ 630–700 nm), and within the first hour of the light phase, i.e. between 17:00 and 18:00 h. Two samples were taken because pre-trials had shown that the length of the di-oestrus might be less than 12 h. Vaginal smears were analysed with a light microscope (AX70, Olympus, Hamburg, Germany) and CellR software (Olympus Soft Imaging Solutions, Hamburg, Germany). Qualification and quantification of cell types were performed in 40–400fold magnification. Four components were distinguished: epithelial cells, cornified cells, leucocytes, and mucus (the latter not being quantified for analysis, Figure 1). Vaginal smears were analysed over a total of 50 days. In a follow-up experiment with 36 females (27.1 ± 1.6 weeks), the length of each oestrus stage was investigated by vaginal smears taken every 2 h for a duration of 24 h.
Vaginal smears of Desert hamsters showing characteristic impression of cell types and ratios. (a): Pre-oestrus includes leucocytes (neutrophils, basophils, lymphocytes) recognizable by large nuclei with a comparably small cell-lumen. Epithelial cells have a nucleus which is surrounded by a larger cell-lumen with a square shape. Size of these cells varies, but size of nuclei remains stable. Cornified epithelial cells do not include a nucleus; these components appear to be as large as epithelial cells and show a square shape. (b): Early oestrus smears include only epithelial cells and cornified epithelial cells. Occasionally, cornified epithelial cells seem to have nuclei or shadows of former nuclei. (c): Oestrus is characterized by a lack of leucocytes and epithelial cells, only cornified epithelial cells are recognizable. (d): Met-oestrus can be described as pre-oestrus, both stages are not distinguishable only on the basis of smears, as ratios and numbers of leucocytes vary between animals. (e): Di-oestrus includes the most obvious component – the mucus, which cannot be quantified but which can usually be found at the edge of the smear, besides leucocytes and epithelial cells occur with a low number of cornified epithelial cells. (f): Sperms of Desert hamsters found after copulation.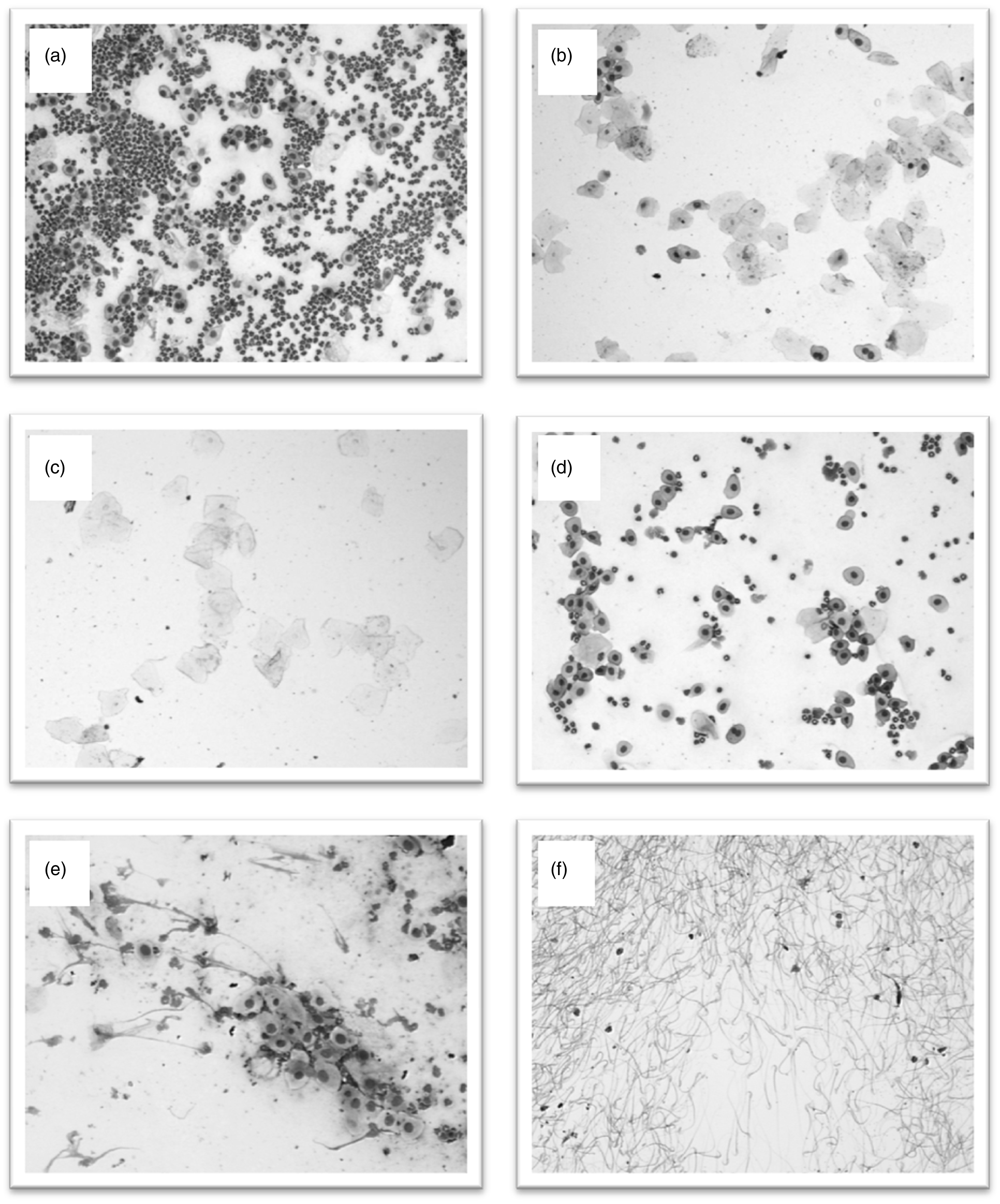
LH determination
Measurements of serum LH levels were taken twice, and different animals (age 30.7 ± 1.6 weeks) were used for the first (n = 18 females) and second (n = 18 females) determinations of LH. Vaginal smears were analysed at least three weeks before taking the first blood sample. The first measurements of LH were performed for each of the oestrus stages with blood samples taken by decapitation during the light period, i.e. 3.5 h before lights off. When after extensive analyses of vaginal smears the existence of an additional stage, the early oestrus, became apparent, LH measurements were repeated in a more detailed manner for that early oestrus stage by taking blood samples 3.5, 2.0, 0.5 h before and 1.0 h after lights off. In this case blood samples were taken from retrobulbar blood flow under short inhalation anaesthesia (isoflurane, Henry Schein Vet, Hamburg, Germany) with a microhaematocrit tube (No. 563, Na-heparinized, DIN ISO 12772, VWR, Darmstadt, Germany) and two blood samples (70 µL) at least two weeks apart were taken from each animal. Serum LH levels were analysed with standard test kits (Rat LH ELISA, IBL Hamburg, Germany).
Activity recordings
At the age of 32.6 ± 2.2 weeks, 12 female and 12 male hamsters were transferred to individual cages (Makrolon Type II) containing nesting material and running wheels (diameter 26.0 cm) for the recording of locomotor activity. Running-wheel activity of all animals was registered using a ClockLab system (Actimetrics, Wilmette, IL, USA); calculations and statistical analyses were performed with MatLab and SPSS 17.0, respectively. Onsets and offsets of running-wheel activity were calculated using a template-matching algorithm that searched for 6 h of time ‘bins’ without activity followed by an activity level of at least 5% of the activity measured over the whole day. The inverse algorithm was used to define activity offset, i.e. 6 h of activity followed by an activity level of less than 5% of the whole day activity. Activity itself was measured as the number of wheel revolutions per bin as exported by the time series function of ClockLab. Only bins exceeding a threshold of 20% of all non-zero counts were defined as bins showing activity.
Behavioural tests
A total of 11 females (28.6 ± 3.3 weeks) and 16 non-related, adult males (age 18.3 ± 0.9 weeks) were used for the behavioural pairing tests. Vaginal smears were taken at least three weeks before the tests, and then pairing experiments were performed 3 h after lights on for 10 consecutive days. Each test lasted 30 min per day and per animal. The tests were terminated earlier if serious biting behaviour was observed. Each day another male was presented in order to prevent any influence by individual preferences. A total of 92 pairing tests were conducted, i.e. 9.2 ± 0.6 tests per female. The frequency of oestrus stages varied: pre-oestrus (n = 22), oestrus (n = 22), met-oestrus (n = 40) and di-oestrus (n = 8). The early oestrus stage was not included in the behavioural tests because of the short duration of this stage. Behaviour was characterized in four categories: sexual behaviour (approach from a distance, olfactory investigation of the anogenital region, lordosis, mounting, and copulation), socio-positive behaviour (sniffing, contact-sitting, and allogrooming), neutral behaviour (all behaviours without interaction), and aggressive behaviour (threatening, submission, biting, and chasing).
Statistics
Statistical analyses were carried out with SPSS 17.0. First, all data were tested for normality against a standard normal distribution using the Shapiro–Wilk test (n < 50) or the Kolmogorov–Smirnov test (n > 50). Chi-square test of independence was used for the analysis of cellular components of vaginal smears and for the analysis of behavioural differences during the oestrus cycle, because these data were equally distributed and continuous measurements were available for each female. Analysis of variance (ANOVA) was performed for the analysis of cell types within cycle stages. Since activity and endocrine data were not equally distributed, non-parametric tests were performed. The Kruskal–Wallis test was applied for the analysis of LH data because more than two independent groups were needed for comparison, followed by the Mann–Whitney U-test as a post hoc test in case of significance. Activity data were investigated with Friedman tests followed by a Wilcoxon post hoc test, because in this case data were related and more than two groups were needed for comparison. Differences were accepted at P < 0.05.
Results
Histology
Five oestrus stages were determined from vaginal smears of the 12 females according to different cell types and their relative quantities. The chi-square test of independence revealed a highly significant effect of the oestrus stage on the relative quantities of the different cell types (χ2 = 179.1, DF = 8, P < 0.001). As shown in Figure 2, pre-oestrus was characterized by similar quantities of leucocytes (36.5 ± 2.1%), epithelial cells (27.1 ± 0.8%) and cornified epithelial cells (36.4 ± 1.5%). Cell composition of the vaginal smear changed during early oestrus. Here leucocytes were almost absent (0.9 ± 0.3%), while epithelial cells made up 33.5 ± 1.6% and cornified cells 65.5 ± 1.8%. The ratio of cornified cells (94.1 ± 0.6%) increased even more during oestrus, when only a few epithelial cells (5.8 ± 0.6%) and leucocytes (0.07 ± 0.07%) were observed. During met-oestrus leucocytes (37.9 ± 1.3%), epithelial cells (30.9 ± 0.8%) and cornified cells (31.2 ± 1.1%) occurred again in similar quantities. The most obvious feature of the di-oestrus was the high amount of mucus, which could not be quantified.
Relative quantities of cell types in vaginal smears of Phodopus roborovskii – females during five oestrus stages. Black and white bars at the x-axis indicate the light–dark (LD) regime but do not represent the actual length of the light and dark phase (LD 14:10). Instead, they are included in the diagram to emphasize the fact that the early oestrus stage was rather short and occurred only during the dark period, while the di-oestrus stage was observed only during the light phase.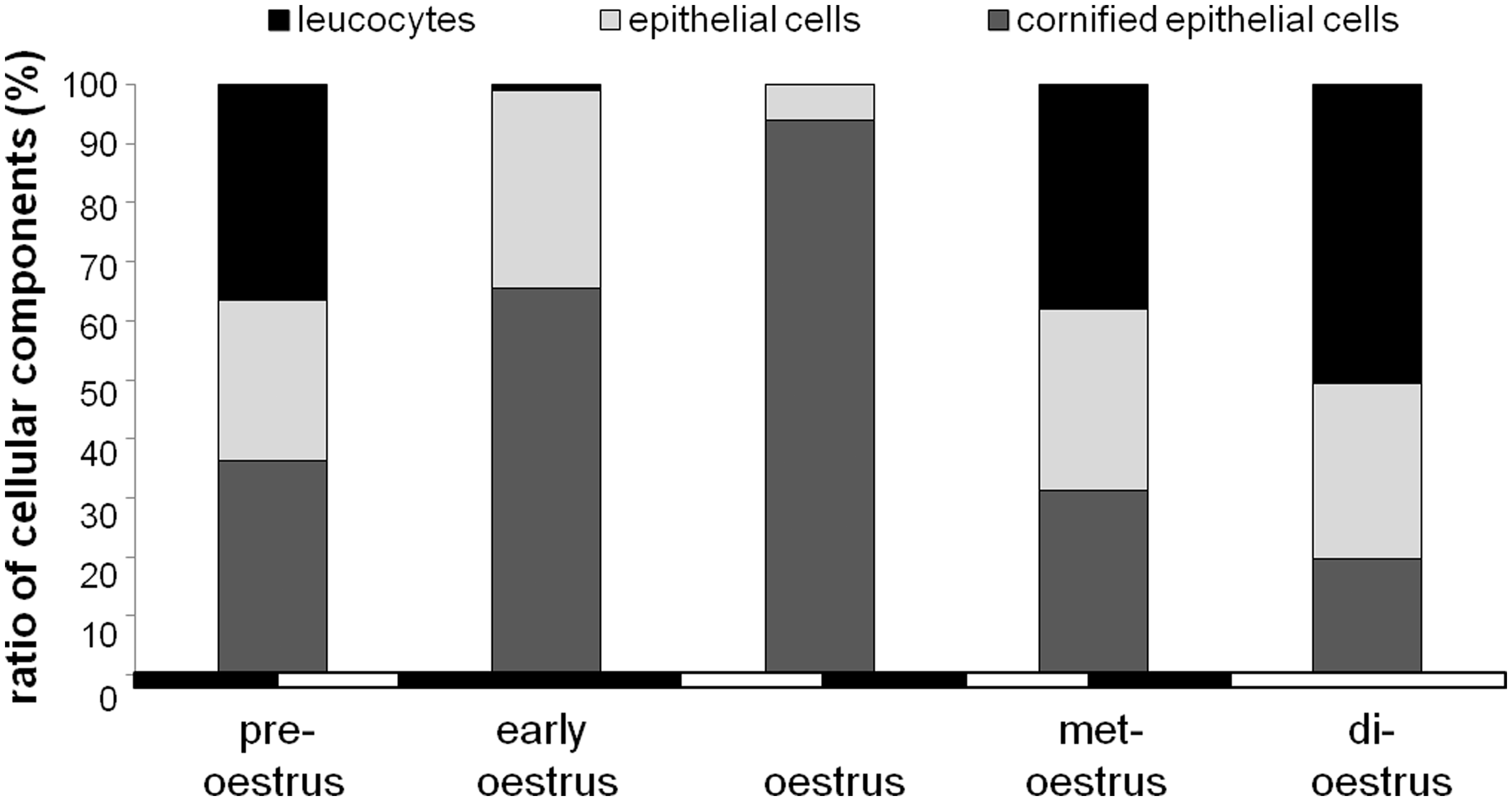
Further statistical analyses revealed significant differences in the relative quantities of cell types during the oestrus cycle. The quantities of cornified (ANOVA F = 466.17, P < 0.001) and epithelial cells (ANOVA F = 90.33, P < 0.001) varied systematically during the oestrus cycle. The quantities of cornified cells were highest during oestrus and lowest during di-oestrus, while the quantities of epithelial cells were lowest during oestrus and highest during early oestrus.
The length of each oestrus stage was determined in more detail, with vaginal smears taken in 2 h intervals over a 24 h period (Figure 3). Duration of pre-oestrus varied considerably: when followed by an early oestrus stage, pre-oestrus lasted 14–18 h. However, when no early oestrus stage was observed, the length of pre-oestrus was prolonged to 18–22 h or to 32–36 h. As already mentioned, the early oestrus stage was not observed in each female. When it occurred, however, it lasted 4–6 h (total range 2 to 12 h). The oestrus stage ended after 32–36 h during the next resting period. The following met-oestrus stage was the longest stage with a period of 38–42 h. Both the beginning and the end of this stage occurred during a resting period. The di-oestrus stage lasted only 4–5 h and was restricted to one resting period. In summary, the whole oestrus cycle lasted between four and six days. Stages of early oestrus and di-oestrus were rather short.
Schematic pattern of the oestrus cycle of the Desert hamster, Phodopus roborovskii. Different cycle stages are depicted as follows: pre-oestrus (white), early oestrus (grey), oestrus (bright grey), met-oestrus (dark grey), di-oestrus (black). Transition periods are indicated by bright grey squares in the background. Black and white bars at the top indicate the light–dark cycle with 14 h of light and 10 h of darkness. Parts (a) and (b) represent oestrus cycles of four days with (a) and without (b) the early oestrus stage. Part (c) represents an oestrus cycle of six days with characteristic prolongation of pre- and met-oestruses.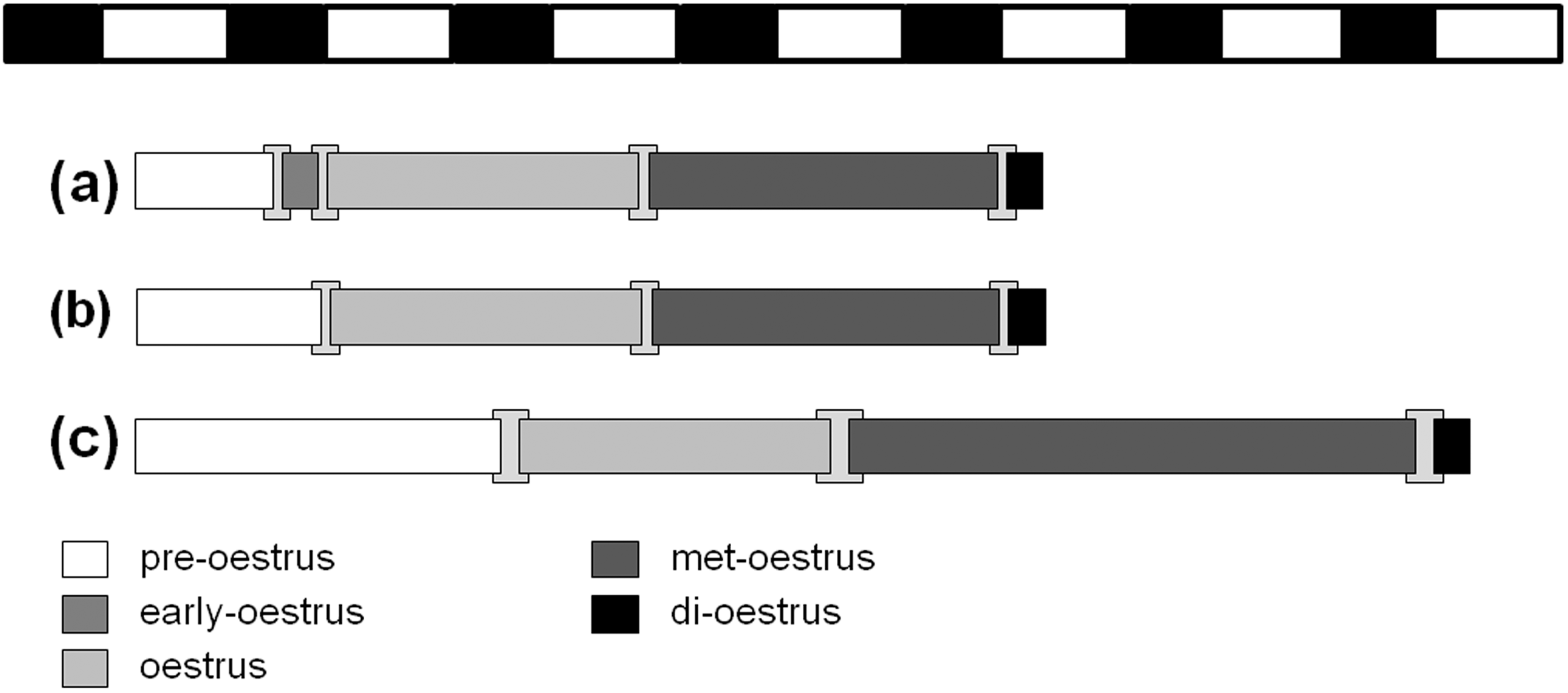
Luteinizing hormone
In a first series of measurements, serum LH levels were analysed for each oestrus stage. Statistical analysis failed to reveal any significant differences (Kruskal–Wallis: χ2 = 7.584, P = 0.1). Serum LH levels showed only small variations ranging from 1.6 ± 0.04 ng/mL (n = 4) during oestrus to 2.5 ± 0.1 ng/mL (n = 4) in pre-oestrus. Furthermore, LH concentrations were 2.1 ± 0.3 ng/mL (n = 5) in the newly described stage of early oestrus (3.5 h before lights off), 1.8 ± 0.2 ng/mL (n = 5) in met-oestrus and 2.3 ± 0.2 ng/mL (n = 5) in di-oestrus.
In a second series of measurements, serum LH levels were analysed in more detail for the early oestrus stage. As seen in Figure 4, serum LH levels increased significantly just prior to activity onset (Kruskal–Wallis: χ2 = 7.971, P = 0.047). At 3.5 h before lights off, LH levels of 2.1 ± 0.3 ng/mL (n = 5) were determined. These levels differed significantly from concentrations of 8.2 ± 2.7 ng/mL (n = 5, U = 3.0, P = 0.047) determined at 2 h before darkness and concentrations of 6.1 ± 1.8 ng/mL (n = 5, U = 3.0, P = 0.047) determined at one hour after darkness. The highest concentrations of 15.9 ± 4.0 ng/mL (n = 5, U = 3.0, P = 0.047) were measured at 0.5 h before the dark phase.
Luteinizing hormone (LH) concentration in female Desert hamsters during different stages of the oestrus cycle and in detail during early oestrus. Black and white bars at the x-axis indicate the light–dark cycle (LD 14:10, lights on at 07:00 h) and the light phase during blood sampling. An additional box marks deviation from the sampling schedule during early oestrus. Asterisks (*) indicate significance level of P < 0.05.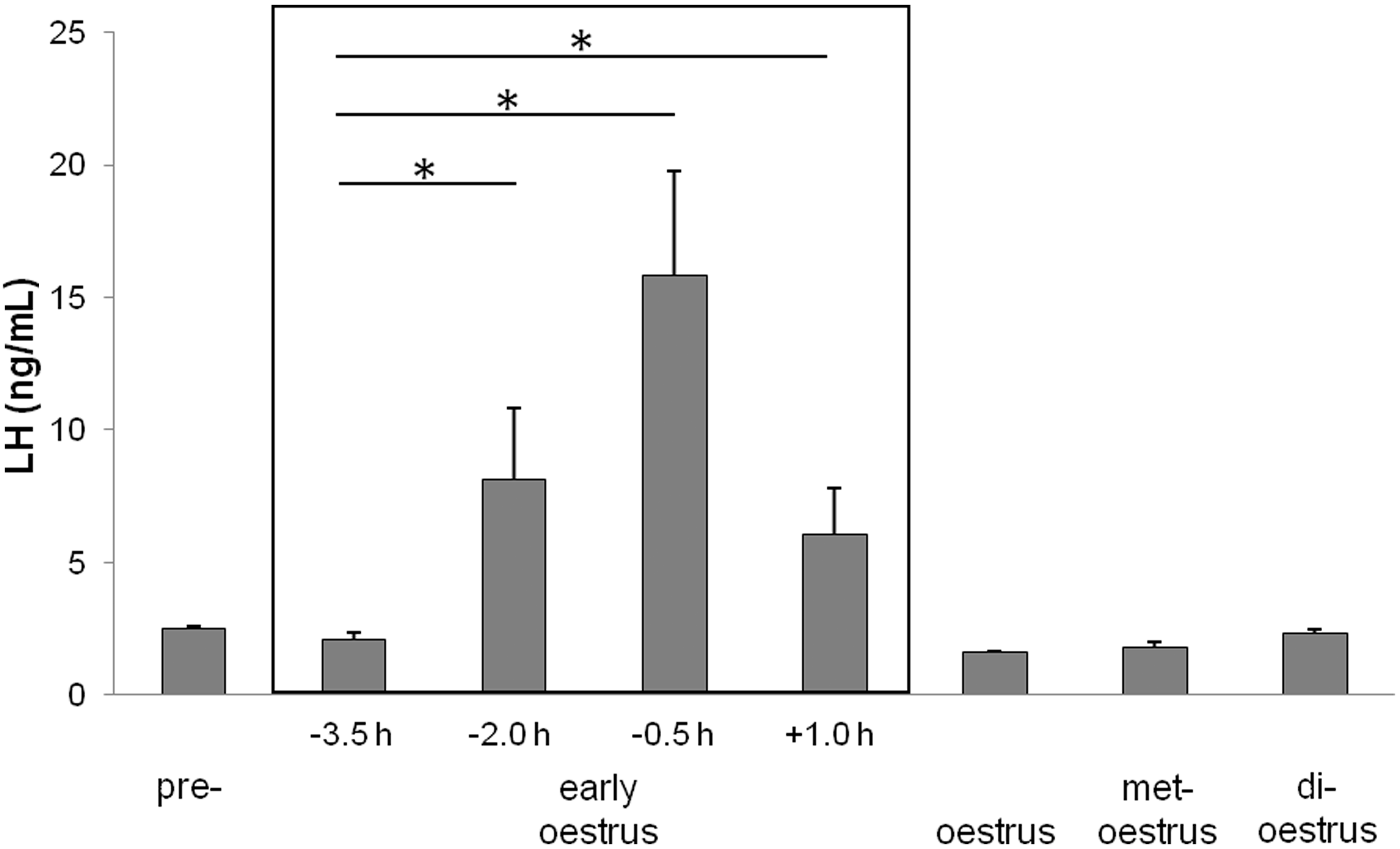
Activity
Oestrus-correlated effects on locomotor activity rhythms were investigated in 12 females over a period of 50 days. Visual inspection of activity plots failed to show any evidence for a so-called scalloping of activity onset, i.e. an earlier onset of activity during oestrus. As shown in Figure 5, activity onset was rather stable and did not differ between males and females. For a more detailed analysis, parameters of locomotor activity were determined on a daily basis and correlated with vaginal smears taken within the first hour of the dark period, i.e. at the beginning of the activity period. Statistical analyses failed to reveal any oestrus-correlated effects on activity onset (Friedman: χ2 = 3.588, P = 0.5). Activity onset was almost identical on the days of pre-oestrus (0.28 ± 0.05 h after lights off), early oestrus (0.27 ± 0.07 h), oestrus (0.26 ± 0.05 h), met-oestrus (0.28 ± 0.09 h) and di-oestrus (0.28 ± 0.9 h). Similar negative results were found for the amount of activity. Although wheel-running activity was highest during the stage of oestrus (12,275 ± 2025 revolutions per day) and lowest during met-oestrus (10,887 ± 1821 revolutions per day), this difference did not prove significant regardless of whether calculations were based on the total amount of activity during the dark phase (Friedman: χ2 = 7.491, P = 0.1) or on the amount of activity without the sampling period for taking vaginal smears (χ2 = 5.382, P = 0.3). By contrast, significant oestrus-correlated effects were found for activity offset (Friedman: χ2 = 14.6, P = 0.005) and the duration of the activity period (Friedman: χ2 = 13.4, P = 0.009). Females were active longer during early oestrus (8.21 ± 0.11 h) than during met-oestrus (7.87 ± 0.15 h, Wilcoxon: Z = –2.136, P = 0.03), di-oestrus (7.87 ± 0.17 h, Z = –2.758, P = 0.006) and pre-oestrus (7.87 ± 0.20 h, Z = –2.401, P = 0.016), while no significant difference was found between early oestrus and oestrus (8.17 ± 0.15 h, Z = −1.718, P = 0.09). Along the same line, activity ended later during early oestrus (1.37 ± 0.19 h before lights on) compared with pre-oestrus (1.83 ± 0.15 h, Z = −2.312, P = 0.02), met-oestrus (1.89 ± 0.12 h, Z = −2.268, P = 0.02) and di-oestrus (1.85 ± 0.11 h, Z = −2.713, P = 0.007), while no significant difference was found between early oestrus and oestrus (1.57 ± 0.11 h, Z = −1.785, P = 0.07).
Actograms of running-wheel activity of a female (left side) and a male (right side) Desert hamster kept under light–dark cycles (LD 14:10) marked by the white and black bar at the top of each actogram. Each row represents one day with activity shown as black columns. Time in days progresses from top to bottom. Arrows identify the days of oestrus as determined by vaginal smears one hour after lights off.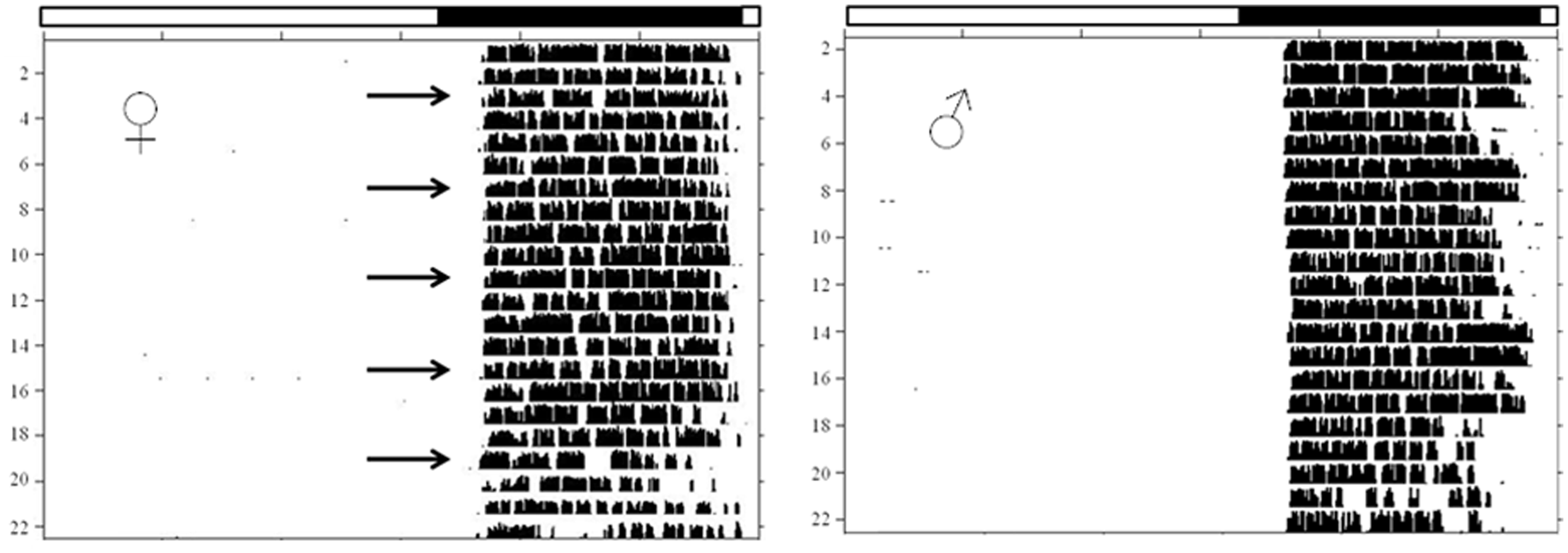
Behavioural tests
For behavioural testing, fertile males were paired for 30 min with females (n = 11) during different oestrus stages. Only the early oestrus stage was not tested due to its short duration. Four types of behaviour were differentiated: socio-positive, neutral, aggressive and sexual behaviours. The relative frequencies of these four types of behaviour during the observation period are summarized in Figure 6. Statistical analysis by the chi-square test of independence revealed oestrus-correlated differences in the relative frequencies of all four types of behaviour (χ2 = 19.2, DF = 9, P = 0.024). The most obvious differences were found in the relative frequencies of sexual behaviour, which was most pronounced during oestrus (34.4 ± 4.8%) compared with met-oestrus (25.4 ± 4.4%), pre-oestrus (25.6 ± 3.7%) and di-oestrus (13.8 ± 4.0%).
Relative frequencies of aggressive (black), neutral (white), socio-positive (bright grey) and sexual (dark grey) behaviours in the course of the oestrus cycle in females of Phodopus roborovskii (n = 11).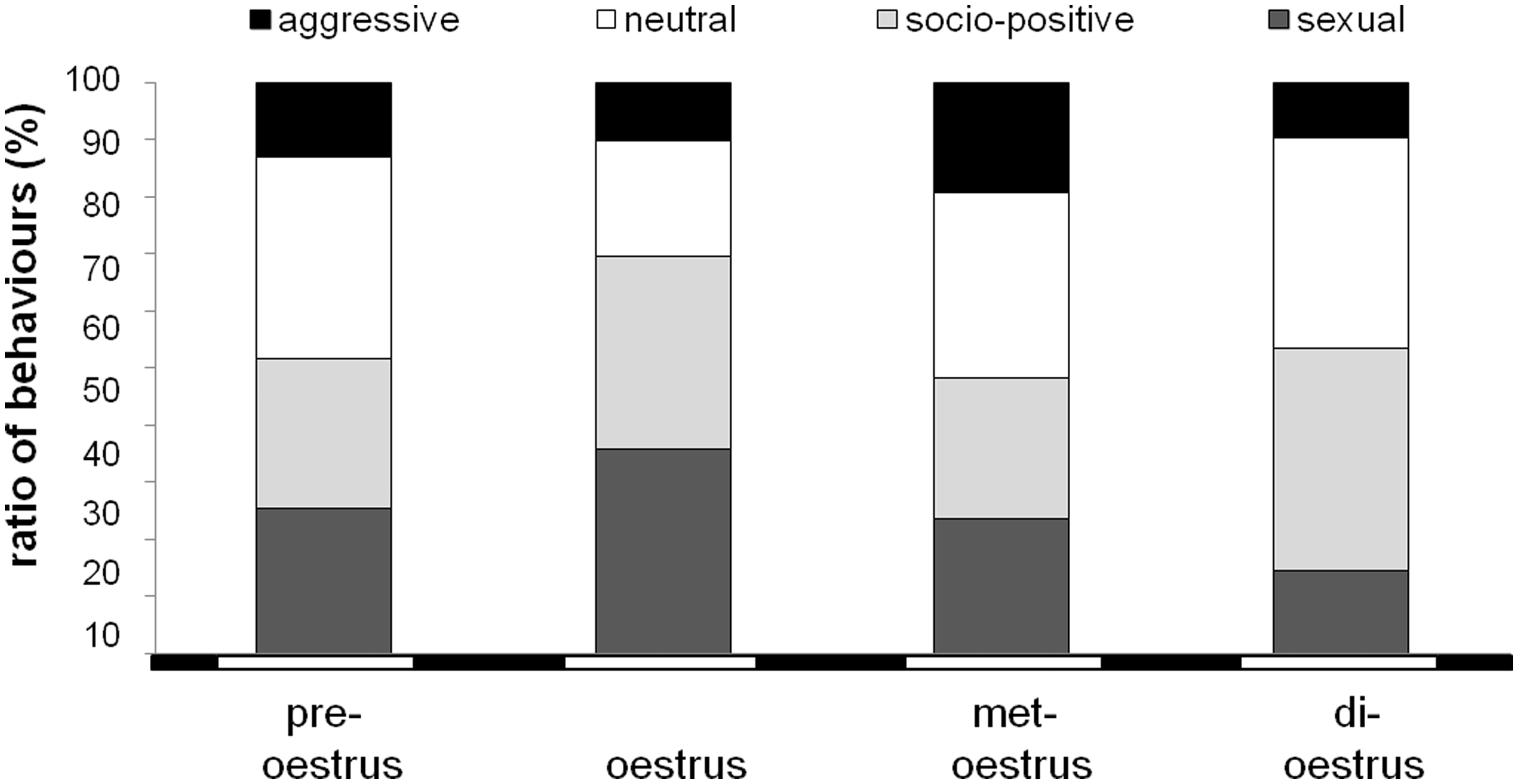
Different types of behaviour were more pronounced or reduced for single oestrus stages. For example, during oestrus, sexual (34.4 ± 4.8%) and social (32.8 ± 3.8%) behaviours were more frequent than neutral (22.9 ± 5.6%) or agonistic (9.9 ± 4.3%) behaviours. In met-oestrus all types of behaviour occurred with a similar frequency. The subsequent di-oestrus stage was dominated by social (42.7 ± 10.1%) and neutral (34.7 ± 9.6%) behaviours rather than sexual (13.8 ± 4.0%) or agonistic (8.8 ± 3.6%) behaviours. Finally, the stage of pre-oestrus was dominated by neutral behaviour (36.9 ± 8.2%) rather than social (25.9 ± 5.6%), sexual (25.6 ± 3.7%) or agonistic (11.5 ± 3.4%) behaviours.
Discussion
The present study produced some fundamental information on the oestrus cycle of the Desert hamster, the smallest species of the hamster family. Histological analyses of vaginal smears were used to verify characteristic cell compositions and the length of each oestrus stage. In general, the oestrus cycle of the Desert hamster lasted between four and six days and followed a pattern rather similar to that described for other hamster and rodent species. 30 However, an important finding of our study is the existence of a fifth cycle stage, the so-called early oestrus. The appearance of this stage seems to indicate an oestrus cycle of increased fertility, because regular oestrus cycles with a length of four days and an increase from 3 to more than 15 ng/mL of LH were observed only in female hamsters that showed the early oestrus stage. It has to be mentioned here that females either showed early oestrus or failed to, but a switch cannot be excluded indicating a changing fertility level. Females without the early oestrus stage showed the LH peak in pre-oestrus, even though a detailed analysis in 2 h intervals was not revealed and the highest values measured were only 3.5 ng/mL. A detailed analysis of wheel-running activity revealed no oestrus-correlated changes at activity onset, but a phase delay of activity offset and thus a longer activity period during early oestrus and oestrus. The present study also included behavioural testing during the oestrus cycle. We found an increase in sexual behaviour during oestrus and an elevation of aggressive or agonistic behaviour in met-oestrus whereas neutral or socio-positive behaviour remained unaffected. The histological analysis of vaginal smears is a well-established method for the determination of oestrus cycle stages.4,6 However, species-specific differences and peculiarities are almost as equally important as the stages themselves. A significant finding of the present study is the documentation of a fifth cycle stage, the early oestrus. The early oestrus stage was characterized by vaginal smears dominated by epithelial and cornified cells and a complete lack of leucocytes. To our knowledge, such a stage has neither been described for the Golden hamster, 7 the rat,5,6 the Mongolian gerbil, 8 nor for any other species of the genus Phodopus.33–35 Due to the rather short duration of the early oestrus stage (4–6 h) it might have been missed in previous studies or only considered to be a transitional stage. Alternatively, the early oestrus stage could be evaluated as unique to the Desert hamster, Phodopus roborovskii. This was further supported by the dramatic rise of serum LH levels during early oestrus, because the rise is known to indicate ovulation in hamsters and other mammals 36 and rats. 6
In general, the vaginal cytology of the Desert hamster showed more similarity with that of gerbils (Meriones unguiculatus 8 ) and rats5,6 than with that of any other hamster species. For example, we did not find any elongated squamous cells as described for the Golden hamster (Mesocricetus auratus 7 ). Furthermore, mucus has not been described in vaginal smears of Golden hamsters, whereas mucus was a characteristic feature of the di-oestrus stage in Desert hamsters and gerbils.
Previous studies on the oestrus cycle in the Phodopus species reported difficulties in determining oestrus cycle stages based on vaginal cytology in Djungarian (Phodopus campbelli 27 ) and Siberian (Phodopus sungorus9,11) hamsters. Unfortunately, the sampling procedures were not well described in these studies, but it is rather likely that vaginal smears were taken only once a day, most likely during the light phase. By contrast, vaginal smears were taken twice a day in the present study, i.e. during the first hour after lights on and during the first hour after lights off. Due to this sampling procedure, it was possible to determine two rather short phases of the oestrus cycle, namely the early oestrus, which lasted only 4–6 h and was restricted to the dark phase, and the di-oestrus, which lasted only 4–5 h and was restricted to the light phase. Taking samples of vaginal smears only once a day would explain why Dodge et al. 9 failed to determine the oestrus cycle in Siberian hamsters, Phodopus sungorus. Another difference to previous studies in the Phodopus species concerns the presence of large quantities of mucus during di-oestrus in the Desert hamster. This has not been described for the other Phodopus species. Since there is no obvious difference between vaginal smears taken during pre-oestrus and met-oestrus, verification of the other two oestrus stages, namely oestrus and di-oestrus, is absolutely necessary to determine a complete oestrus cycle. Previous studies have probably failed to determine the oestrus cycle in the other Phodopus species, because they could not differentiate the di-oestrus from other oestrus stages. This may have been due to the sampling procedure of vaginal smears (only once a day) or the lack of mucus during di-oestrus.
In order to further verify the timing of ovulation in the Desert hamster, LH serum levels were analysed during the oestrus cycle with special focus on the newly described early oestrus stage. It is well known from other rodent species that a dramatic increase of LH at night triggers ovulation.6,37,38 However, detailed analyses of LH serum levels during the oestrus cycle have been published only for Golden hamsters37,39 and rats. 4 In all these studies oestrus stages were determined by vaginal smears and the pre-ovulatory LH surge occurred during pre-oestrus. In contrast to these findings, the present study on the Desert hamster revealed a new oestrus stage characterized on the one hand by vaginal smears with only epithelial and cornified cells and no leucocytes, and on the other hand by a dramatic increase of serum LH levels. We therefore suggest that this stage is a separate oestrus cycle stage specific to the Desert hamster rather than a transitional stage between pre-oestrus and oestrus.
Another aspect of the present study was the analysis of oestrus-correlated changes in wheel-running activity. It is well known that the oestrus cycle modulates the amplitude, phase and period of circadian rhythms in rodent species like the Golden hamster4,21 and the rat.23,24 The Desert hamster differs from these species in that oestrus-correlated changes in wheel-running activity are only subtle. A detailed analysis revealed no significant effects on activity onset, but a significant lengthening of activity duration during early oestrus and oestrus. The more dramatic changes of wheel-running activity reported for Golden hamsters or rats have been interpreted as a strategy to increase the probability for physical contact between males and females and thus increase mating success. The lack of such oestrus-correlated changes in the Desert hamster may be due to the fact that as social animals female Desert hamsters have regular contact with males anyway.
The final aspect of the present study was the behavioural tests demonstrating an increase in sexual behaviour on the day of oestrus. In this part of the study the early oestrus stage was omitted from testing because of its short duration and the requirement to perform all behavioural tests at the same time of day. As expected, oestrus-correlated changes were observed for sexual behaviour, which was most pronounced during oestrus. This is in good agreement with a previous study in the Golden hamster. 40 By contrast, aggressive behaviour was observed during each stage of the oestrus cycle with only slightly lower ratios during the oestrus and the met-oestrus stages (9%) compared with pre-oestrus (11.5%) and di-oestrus (10%). Previous studies in Golden hamsters reported divergent results. While Takahashi and Lisk 26 observed rather constant levels of aggressive behaviour during the oestrus cycle, a more recent study demonstrated that the number of aggressive attacks was highest during met- and di-oestrus. 22 In general, females should reduce aggressive behaviour in order to mate, but a review 30 showed that this is not the case for all rodent species. While Norway rats (Rattus norvegicus) and short-tailed shrews (Blarina brevicauda) reduced aggressiveness against male opponents during oestrus, collared lemmings (Dicrostonyx groenlandicus) and grey opossums (Monodelphis domestica) showed no oestrus-correlated changes in their levels of aggression, i.e. they were always aggressive irrespective of their oestrus cycle stage. 30 The rather constant level of agonistic behaviour observed during the oestrus cycle is thus a feature that the Desert hamster has in common with other rodent species.
Our findings show that it is possible to determine the oestrus stage of Desert hamsters on the basis of vaginal smears. However, it may be necessary to take vaginal smears twice a day in order to identify two rather short phases of the oestrus cycle, namely the early oestrus and the di-oestrus.
Footnotes
Acknowledgements
All experiments could not have been conducted without the support of numerous colleagues and students. Special thanks go to Miriam Ziegler, Kristin Schmiech, Sandra Böker and Harald Feuchter. The authors also wish to thank Hans-Peter Piepho from the University of Hohenheim for his kind advice on statistical matters and Andreas Herrmann for a linguistic review of the manuscript.
Funding
Elke Scheibler was funded by the Federal State of Baden–Württemberg and the European Social Fund with the Margarete–von-Wrangell scholarship.
