Abstract
Objective:
This study was designed to assess the severity of lung tissue changes due to COVID-19 in a pediatric cohort that still complained of post-COVID-19 symptoms.
Materials and Methods:
Parents and adolescents, 13 to 22 years old and have had a confirmatory positive polymerase chain reaction (+PCR) and/or rapid antigen test for COVID-19, consented to have a series of diagnostic tests to determine the level of lung tissue changes due to COVID-19. Sixteen adolescents completed the intake form and lung sonography. The lung sonogram was completed using the bedside lung ultrasound in emergency (BLUE) point method and a GE Logiq I laptop ultrasound system, as well as a linear 7.5-MHz or 3.0-MHz convex-array transducers. Thirteen adolescents completed a chest magnetic resonance imaging (MRI) examination on a 0.55T Siemens MAGNETOM Free.Max scanner. The examination included the heart and lungs of each participant and took about 90 minutes. A radiologist, sonographer, and computed tomography (CT) technologist reviewed and scored the images. Those positive lung sonogram scores were matched anatomically to the same slice within the chest MRI examination.
Results:
Based on the 13 participants, the acoustic mapping process demonstrated no diagnostic matches for positively scored sonograms at the pleural line or pulmonary parenchyma with the corresponding MRI image slices. There were diagnostic matches for negatively scored sonograms at the pleural line or pulmonary parenchyma with the corresponding MRI image slices.
Conclusion:
With lingering post-COVID-19 symptomology, this adolescent cohort demonstrated that acoustic mapping of their lung sonograms and chest MRI images had important combined diagnostic value. Lung sonography demonstrated diagnostic effectiveness for evaluating the periphery of the lung tissue, in this cohort but limited in viewing deeper structures. Conversely, MRI provided definitive views of the entire chest cavity and lung tissue, which is important for properly evaluating deeper pulmonary parenchyma.
At the onset of the COVID-19 pandemic, the health care system was overwhelmed with the number of adults presenting with acute pathology from this zoonic virus. The guidance provided for diagnosis and treatment was geared toward adults suffering from the SARS-CoV-2 viral infection, which was in part due to the belief that children had less severe presentations. 1 This early information on a possible pediatric lessening of viral symptoms and severity was believed to be related to children’s age-related immunity and vascular endothelial damage, as well as their clotting function. 1 This was also supported by the results of extensively reported epidemiologic studies that suggested that pediatric patients, suffering from the SARS-CoV-2 viral infection, made only 1% to 2% of reported cases.2 –4 This focus on adults as opposed to children was also due to those children testing positive for the virus having less observable presenting symptoms.5 –7 Owing to the apparent lesser severity and limited testing for children, those infected likely either were asymptomatic carriers or quickly recovered. This was reported based on the many of the “lock down” mandates that were instituted globally, which placed asymptomatic children at home while recovering from the virus.8,9 An additional factor believed to assist pediatric patients to recover more quickly than adults was the hypothesis that children were infected with secondary and weaker viral strains that originated with parents, teachers, and so on.10,11 Regardless of the mechanism or the innate resistance to the severity of the virus, children were infected and many are now showing long-term deficits. These pediatric deficits are now coming to the forefront of medical treatment as children report difficulties with breathing, upon exertion, headaches, fatigue, sleep disturbance, and inability to focus on cognitive tasks. 12 Given these perplexing presentations, pediatricians continue to look for a combination of diagnostic tests that might help to elucidate the presence of lingering disease, because of the SARS-CoV-2 viral infection. Obviously, blood tests and diagnostic imaging are common tools used for adults; however. children present challenges for utilizing invasive procedures and ionizing radiation. During the pandemic, computed tomography (CT) was commonly used to obtain diagnostic images of adult lungs for both SARS-CoV-2 viral infiltrates and possible blood clots; 13 however, the radiation dose made this not translatable for children. Therefore, the objective of this research project was to determine the diagnostic value of using non-ionizing imaging to ascertain the presence of pediatric lung tissue damage due to surviving the COVID-19 pandemic. The research question posed was as follows:
Materials and Methods
Study Protocol
A prospective cohort study was designed and approved for execution (OSU IRB: 2021H0448). Pediatric participants and their parents/guardians were primarily recruited through the children’s hospital’s pulmonary clinic. The clinic routinely evaluated and provided surveillance for patients with self-reported residual COVID-19 symptoms. The inclusion criteria were that participants must be within the age range being seen in the clinic (13-22 years of age), have had a confirmatory positive polymerase chain reaction (+PCR), and/or rapid antigen test for COVID-19. The exclusion criteria were based on participants having had any prior history of cardiac or renal disease. The pediatric pulmonologists informed the patients and parent/guardian about the approved study. The parents were directed to complete a Qualtrics scheduling form that they could access from the study flyer’s QR code. Once the Qualtrics scheduling form was submitted, research lab staff contacted the family to make appointments for a lung and heart sonogram, blood draw, and survey data collection. As part of the Qualtrics scheduling form and at the first appointment, the pediatric participant was screened for MRI safety. A subsequent appointment was made with the family to complete the MRI appointment at a local outpatient facility. All pediatric participants and their parents/guardians signed a written consent form to participate in all aspects of the study. Incentive $15 gift cards were provided at the first appointment, and an additional one was presented at the conclusion of the MRI examination. The research staff made several phone calls to remind parents about the appointments and ensure participants complied with MRI safety. Parents were also told that any positive diagnoses resulting from the diagnostic testing would be communicated to them for further testing and continuity of the participant’s care.
Diagnostic Imaging Protocol
For this portion of the study, the consented participants were examined with sonography for their hearts and lungs. The sonogram was performed on a GE Logiq I laptop ultrasound equipment system (GE Medical, Waukesha, Wisconsin). A set of 7.5-MHz linear and 3.0-MHz convex-array transducers were used to evaluate each patient’s heart and lungs. Owing to the participant’s shortness of breath and to screen for cardiac abnormalities, a modified cardiac sonogram was performed. The cardiac sonogram consisted of two main views, subcostal and apical four chambers. Once the modified cardiac exam was completed, the lungs were imaged based on the lung ultrasonography protocol established by Lichtenstein. 14 Dr Lichtenstein’s bedside lung ultrasound in emergency (BLUE) protocol was used to examine six zones of the right and left chest (e.g., upper anterior, lower anterior, upper axillary, lower axillary, upper posterior, and lower posterior chest; See Figure 1). A total of 12 images were taken of the participant’s lungs, bilaterally, and real-time clips were also captured to assess breathing and lung sliding. All the DICOM images and clips for each patient were saved securely on the research lab’s cloud-based storage system.
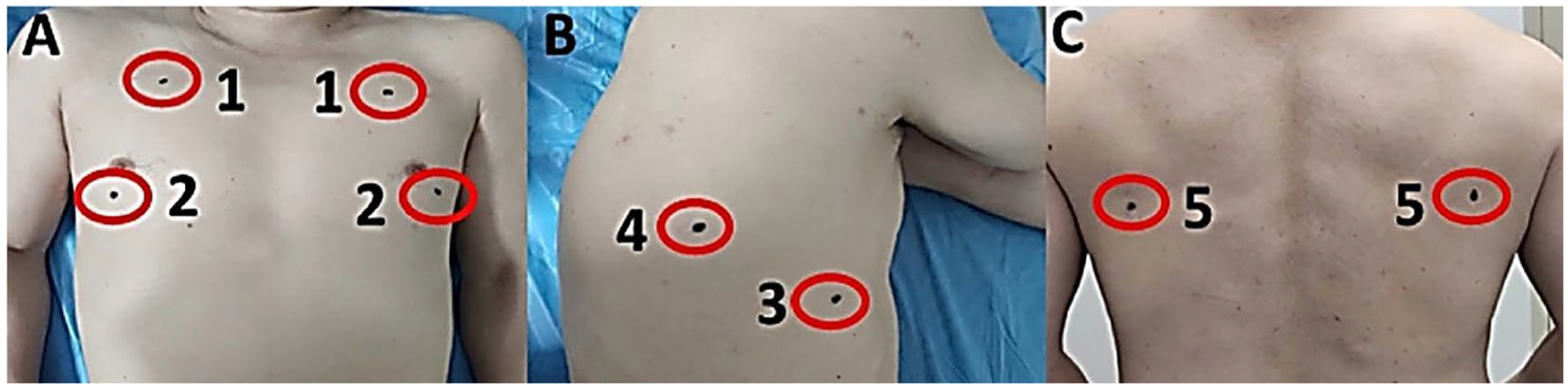
Tan et al’s bedside lung sonography in emergency (BLUE) protocol: 5 points. A lower posterior lung point was added to this study (6) to make sure that the lower lung base was imaged anteriorly and posteriorly (reprinted permission).
After completing the MRI screening form, an appointment was scheduled for the participant and their parent/guardian to proceed with the MRI examination. The MRI images were acquired using a 0.55T system (MAGNETOM Free.Max, Siemens Healthineers AG, Erlangen, Germany). The MRI included a comprehensive multi-planar examination of the heart and lungs for each participant and took about 90 minutes. The completed MRI images and clips were securely uploaded to Precession (Heart IT, Durham, North Carolina) and processed using validated software (suiteHEART, NeoSoft LLC, Pewaukee, Wisconsin).
Image Analysis
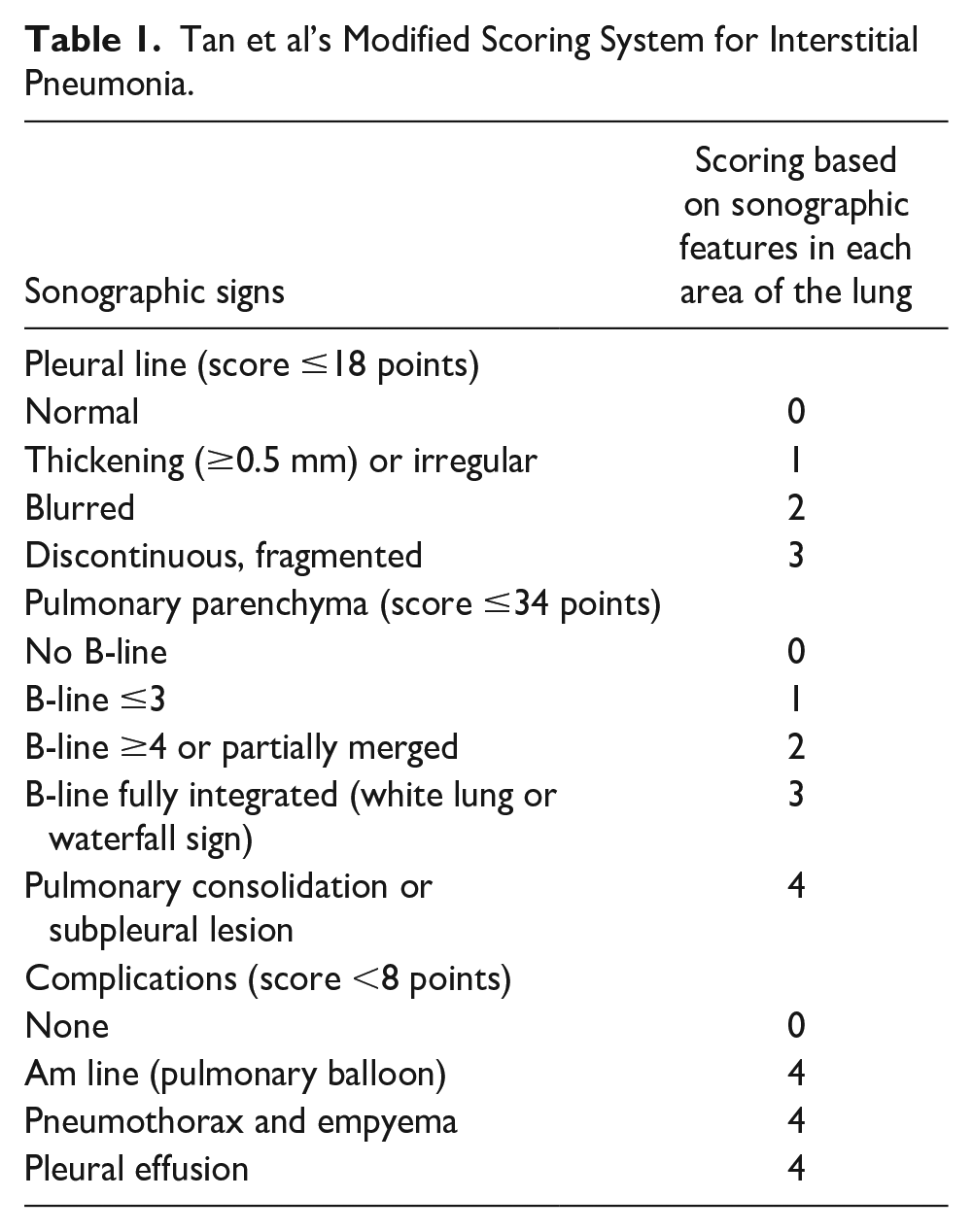
The lung sonographic, BLUE protocol, images were separately scored using Tan et al’s 15 modified scoring system for interstitial pneumonia. The scoring is focused on the pleural line involvement and the lung parenchyma. The scoring values are based on the sonographic features seen in each of these sections and across all 12 BLUE points. The values assigned were normal/no artifacts = 0, pleural irregularity/≤3 B-line artifacts = 1, blurred pleura/≥4 B-line artifacts =2, and a fragmented pleura/white lung sonographic appearance =3 (See Table 1). 15 The overall diagnostic summary from the scoring system was based on a normal score of zero or an abnormal score of ≥1.
Tan et al’s Modified Scoring System for Interstitial Pneumonia.
The analysis of the lung sonogram and MRI images was completed using the recently published method of acoustic mapping. Smargiassi et al 16 discussed the use of sonography to describe pleural interstitial syndrome. They further extended their use of a multi-modality approach to assessing fibrotic interstitial lung disease by creating an acoustic pulmonary map, which was designed to correlate the sonography and CT findings (See Figure 2). 16 For this study, the MRI lung images, specifically the T2-weighted axial views of the chest, were used and correlated with the six BLUE points to create acoustic maps. Correlations were made with the superficial anatomical structures that could be easily identified on the MRI chest image.
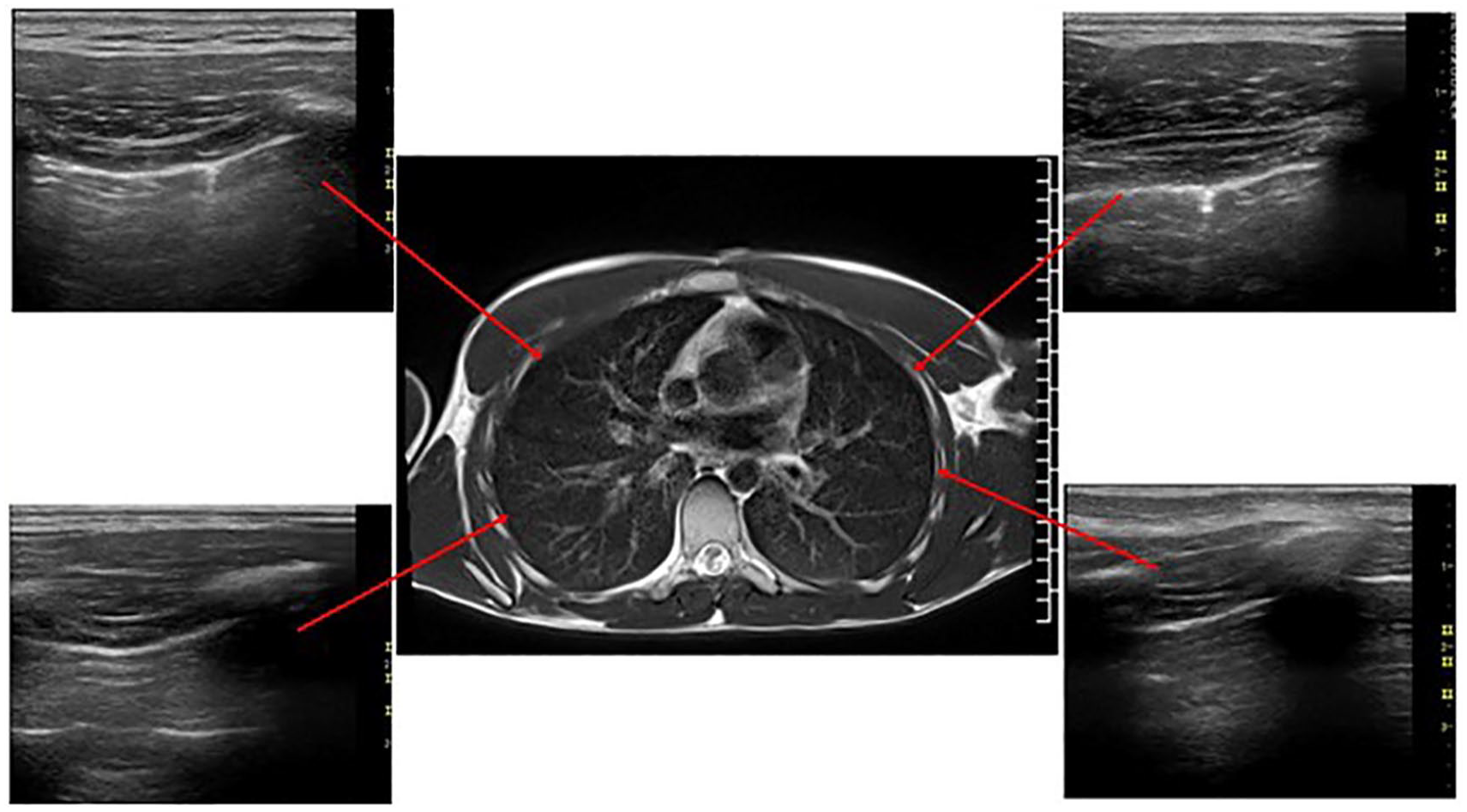
Acoustic map of an MRI image from a 21-year-old male with lingering COVID-19 symptomatology. The anterior upper, axillary, and posterior lung sonograms are matched to their anatomical locations on the axial MRI T2 chest image.
An example is that the anterior (right and left) upper BLUE points were located using the clavicle, the anterior (right and left) lower BLUE points were located using the nipple, the upper posterior and axillary BLUE points were located using the paravertebral line at the spine of the shoulder blade, and the lower posterior and axillary BLUE points were located using the edge of the inferior border of the scapula. The identified MRI slice was chosen and evaluated as well as the slice above and below the reference slice.
The radiologist worked with the sonographer and a CT technologist to review all the images and score the 12 images across the corresponding MRI axial chest images for diagnostic matches and no matches.
Statistical Analysis
Given the small sample size, the primary statistics for the study were reported as descriptives with frequencies, means, and percentages. The data was maintained in an Excel (Microsoft Inc.) spreadsheet, and the analysis was generated from it.
Results
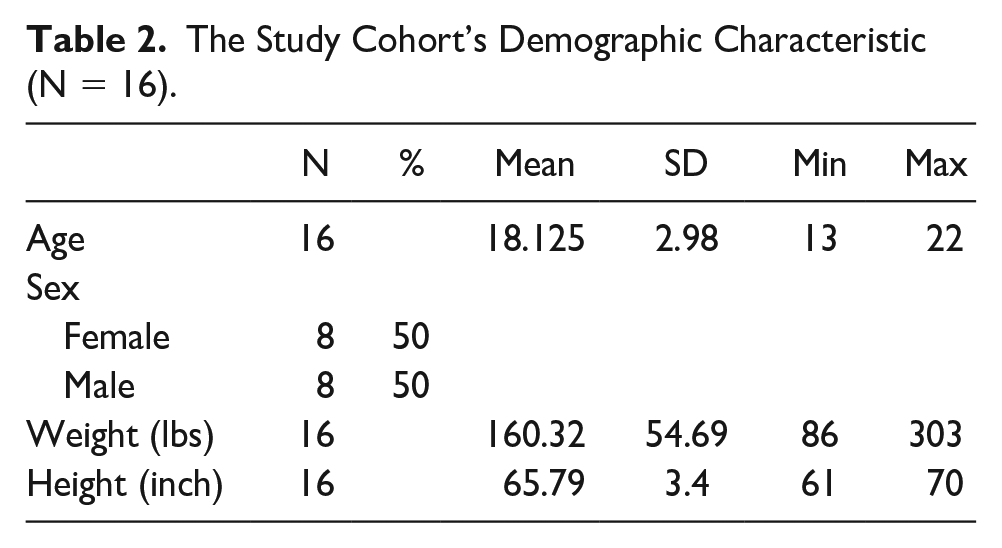
The results of the study were based on the 13 participants that consented to the study. A CONSORT diagram is provided that demonstrates the number of participants recruited (N = 25), the number consented (N = 16), and those that qualified for MRI (N = 13) (See Figure 3). The mean age of the participants was 18 years and was slightly skewed toward the upper end of the age range (See Table 2). This was due to some of the older siblings and college-age students who volunteered for the study. The image analysis was based on eight female and eight male participants, but only six females and seven males completed all study phases (See Table 2). The majority (81%) of the complete cohort reported having been symptomatic when they were diagnosed with COVID-19. Based on their self-reported symptoms, the most common continuing complaint among the cohort was slight fatigue (37%), slight difficulty breathing (25%), and slight difficulty with activities (18%).
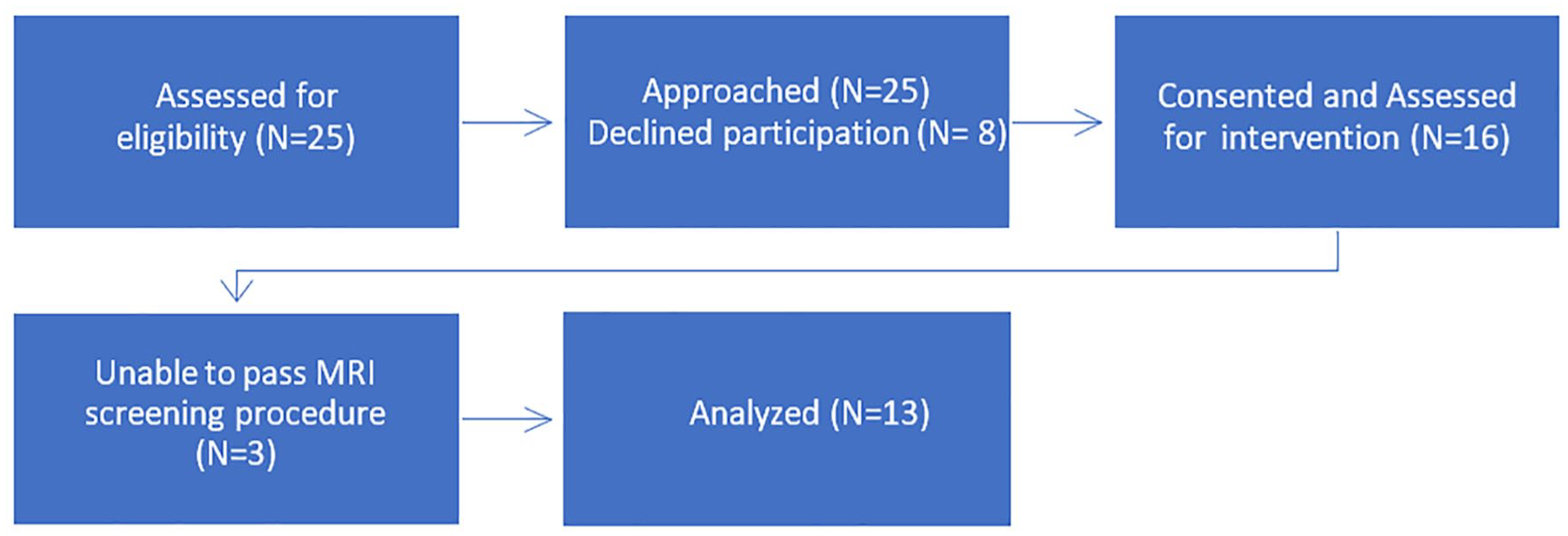
CONSORT diagram for a single-arm study that illustrates the enrollment, allocation, dropout, and analysis of study participants.
The Study Cohort’s Demographic Characteristic (N = 16).
Sonographic Modified Scoring Results
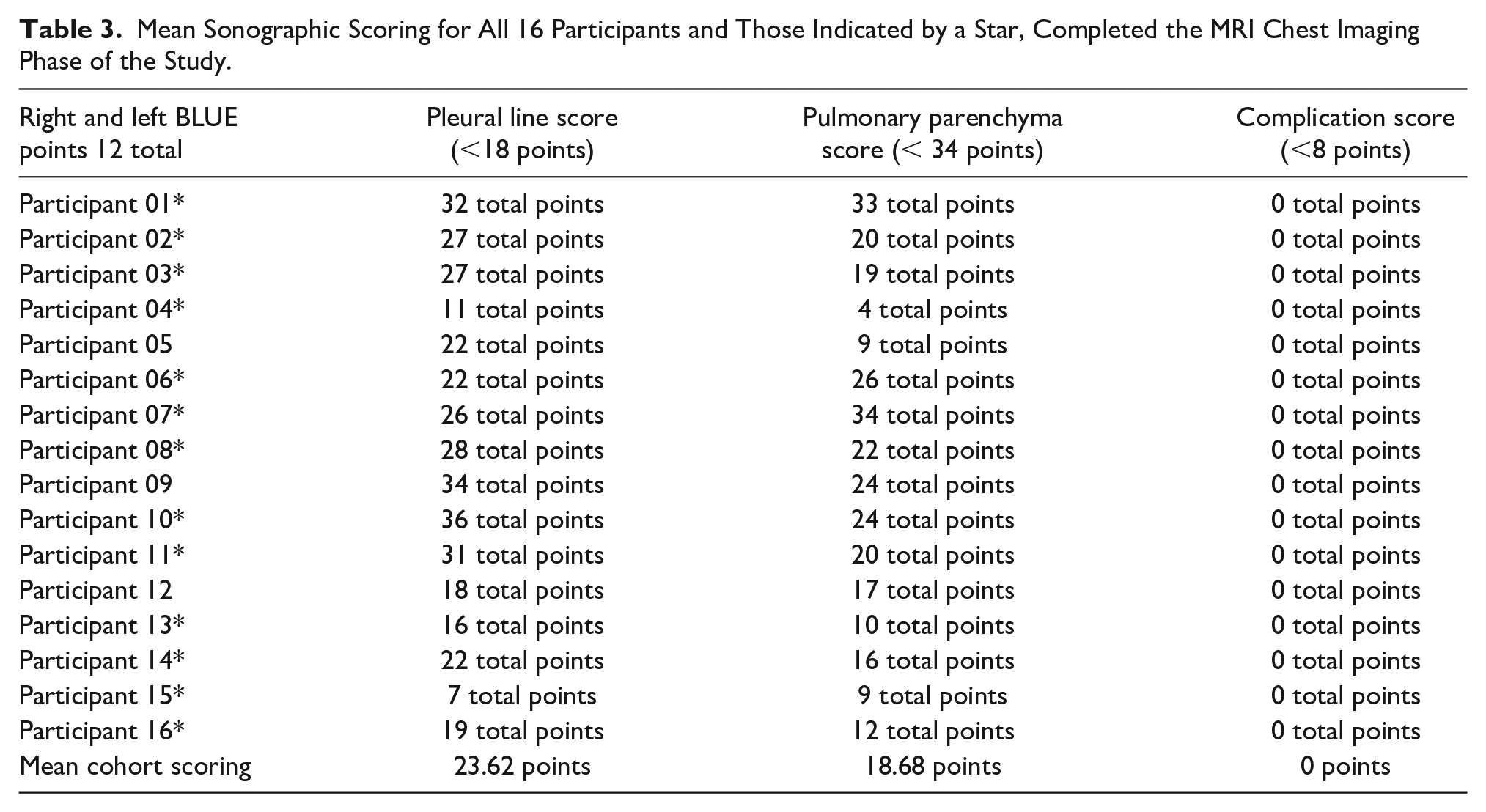
The entire cohort completed the lung sonography examination, and all 12 images were used to score the participants (N = 16). Utilizing the Tan et al’s scoring for the pleural line involvement and the lung parenchyma, the cohort demonstrated greater changes to the pleura compared with the lung parenchyma. Table 3 provides a sample of the results from the image scoring, and the most common description across the cohort was a fragmented and thickened pleura, with lung sliding (See Figures 4–7).
Mean Sonographic Scoring for All 16 Participants and Those Indicated by a Star, Completed the MRI Chest Imaging Phase of the Study.
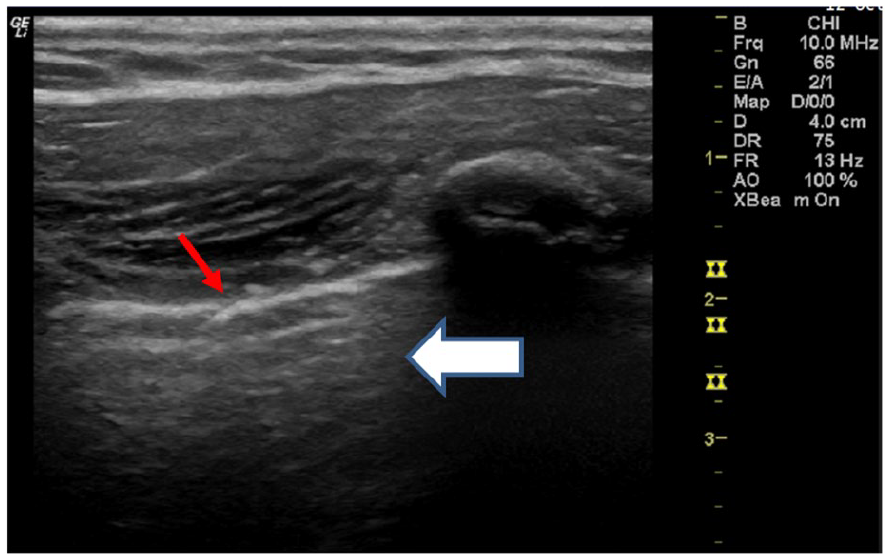
A right lung sonogram taken at the upper posterior (BLUE point 5) intercostal space of a 21-year-old male with lingering COVID-19 symptomatology. The red arrow indicates a fragmentation along the border of the pleural interface. The white arrow indicates normal A-line artifacts associated with normal pleural imaging. An additional concern is the irregular texture of the pleural line, which would be scored as pleural line = 3; pulmonary parenchyma = 4 (B-line partial merged).
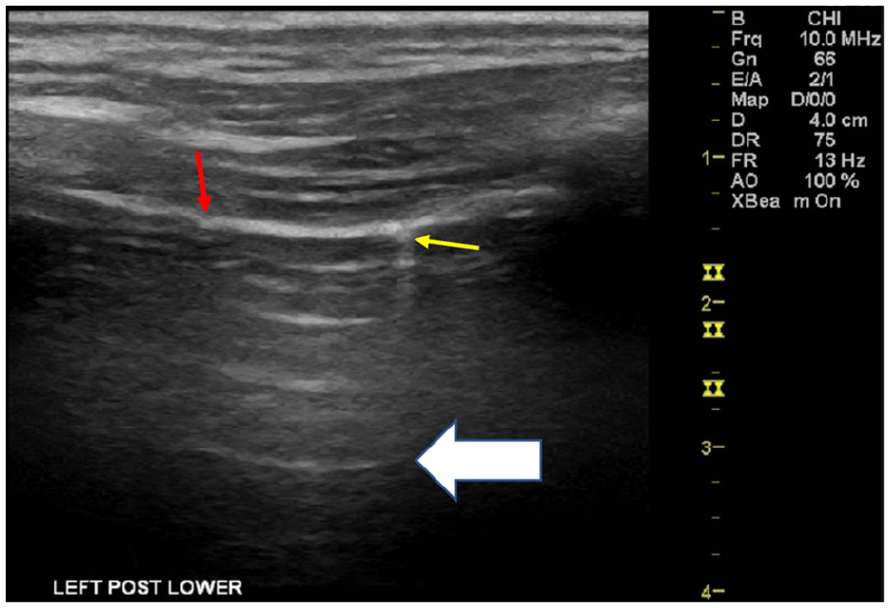
A left lung sonogram was taken at the lower posterior (BLUE point 12) intercostal space of a 21-year-old male with lingering COVID-19 symptomatology. The red arrow indicates fragmentation along the border of the pleural interface. The white arrow indicates normal A-line artifacts associated with normal pleural imaging. The yellow arrow indicates subpleural nodules or consolidation.
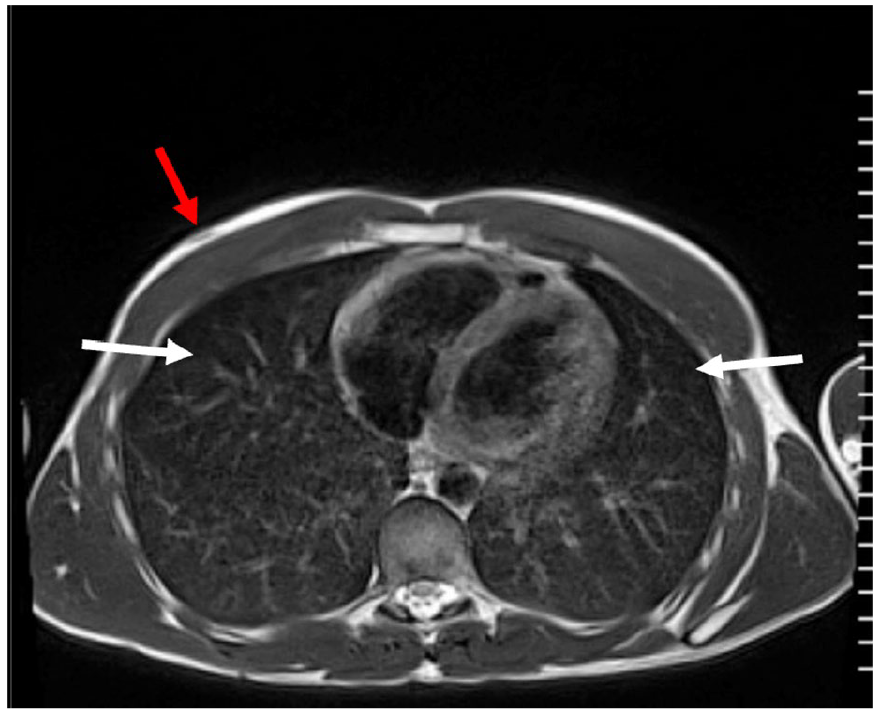
An axial T2-weighted MRI image of a 21-year-old male’s chest, heart, and lungs. The red arrow indicates this superficial landmark of the nipple, which allows this image to be matched with a lung sonogram taken at the right and left lower anterior (BLUE points 2 and 8), intercostal area (see white arrows).
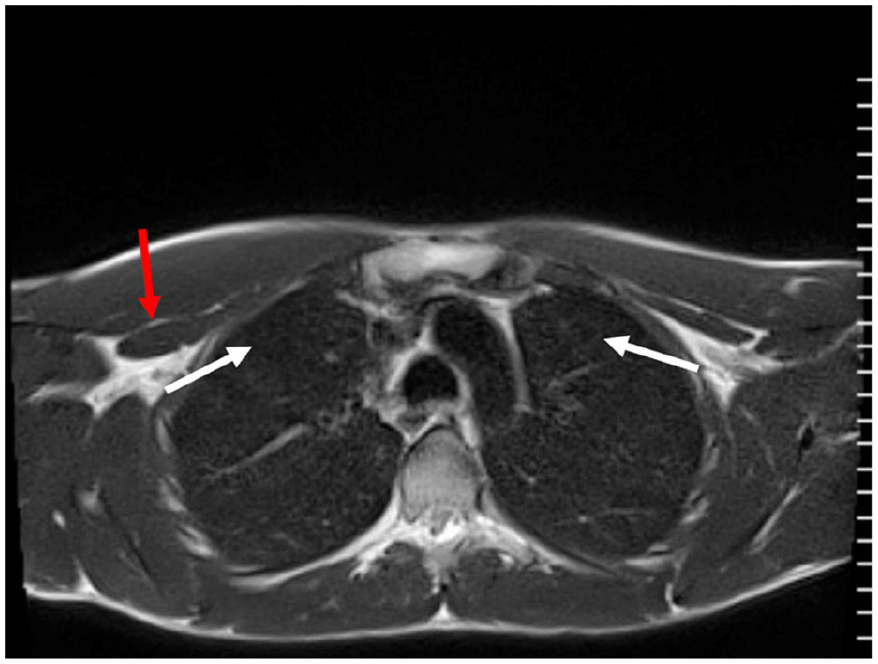
An axial T2-weighted MRI image of a 21-year-old male’s chest, heart, and lungs. The red arrow indicates this upper chest area, below the clavicle, which allowed this image to be matched with a lung sonogram taken at the right and left upper anterior (BLUE point 1), intercostal area (see white arrows).
Acoustic Mapping Completed With T2 Axial MRI Lung Images
Only 13 participants were able to complete the MRI lung examination. A male participant disclosed having scoliosis correcting rods (during the sonogram), a female participant had a permanent metal bracelet (after completing the sonogram appointment), and another female had dental braces and palate expander and did not feel comfortable completing the MRI portion of the study. Based on those who completed all phases of the study (N = 13), lung sonograms were matched and scored against the MRI lung images. The radiologist found no evidence of pleural or lung parenchymal changes on the reviewed images. A scheme of matching and not matching the images based on lung sonographic scoring was completed (See Table 4). The consensus of the diagnostic matching, using the acoustic mapping technique, demonstrated that abnormally scored lung sonograms were not found or confirmed at the same locations on the MRI lung images. The only diagnostic matches were those normal lung sonograms that corresponded with normal appearing MRI lung locations. All the MRI images were rechecked for landmarks and pathology using the coronal T2-weighted MRI images, even though that was not provided as part of the Smargiassi et al’s acoustic mapping technique. This was added during the analysis phase to reassure the identified anatomy and provided the added biplane evaluation of the lung tissue imaged.
The Acoustic Mapping Results Compare Lung Sonography Results With MRI Biplane Imaging.
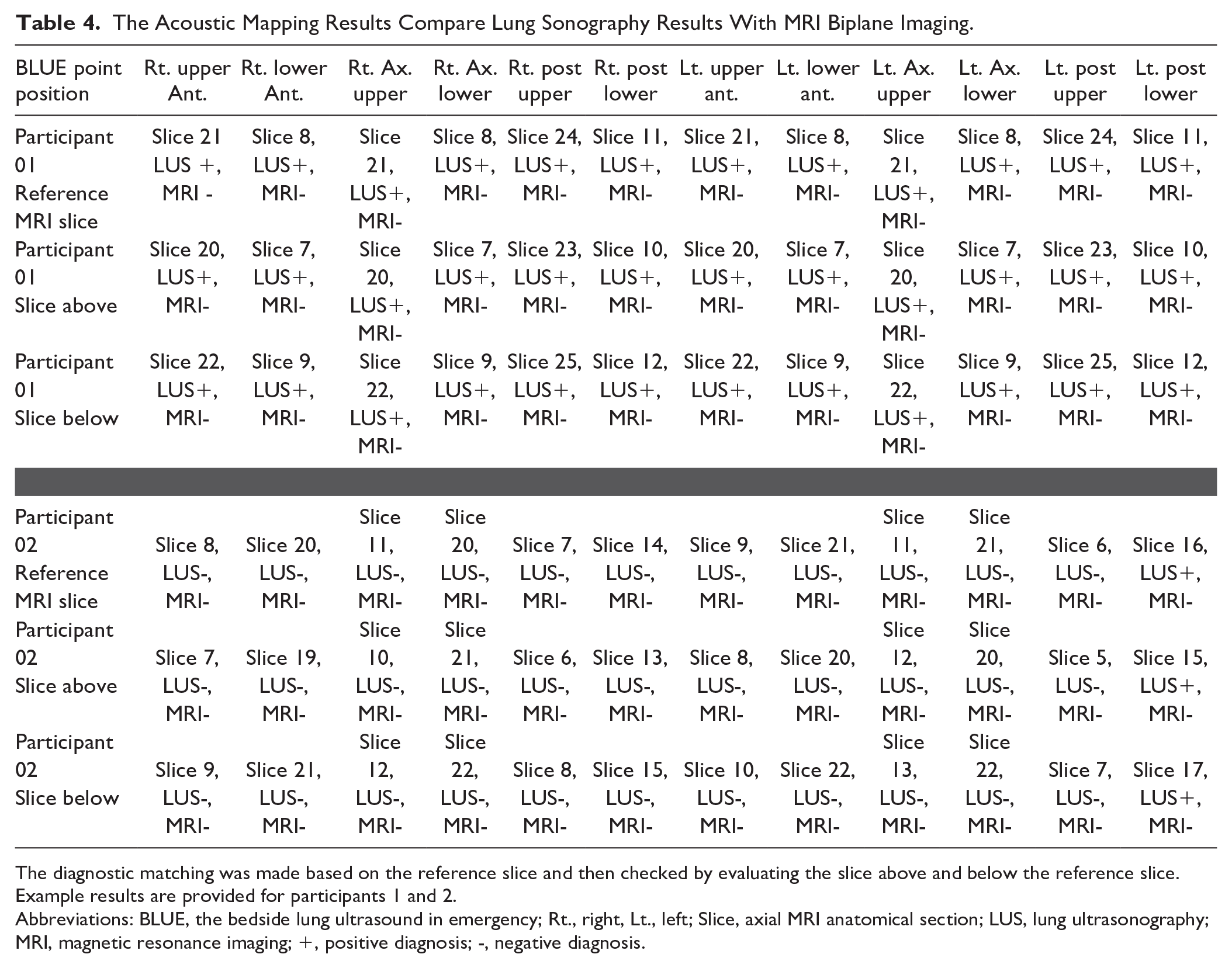
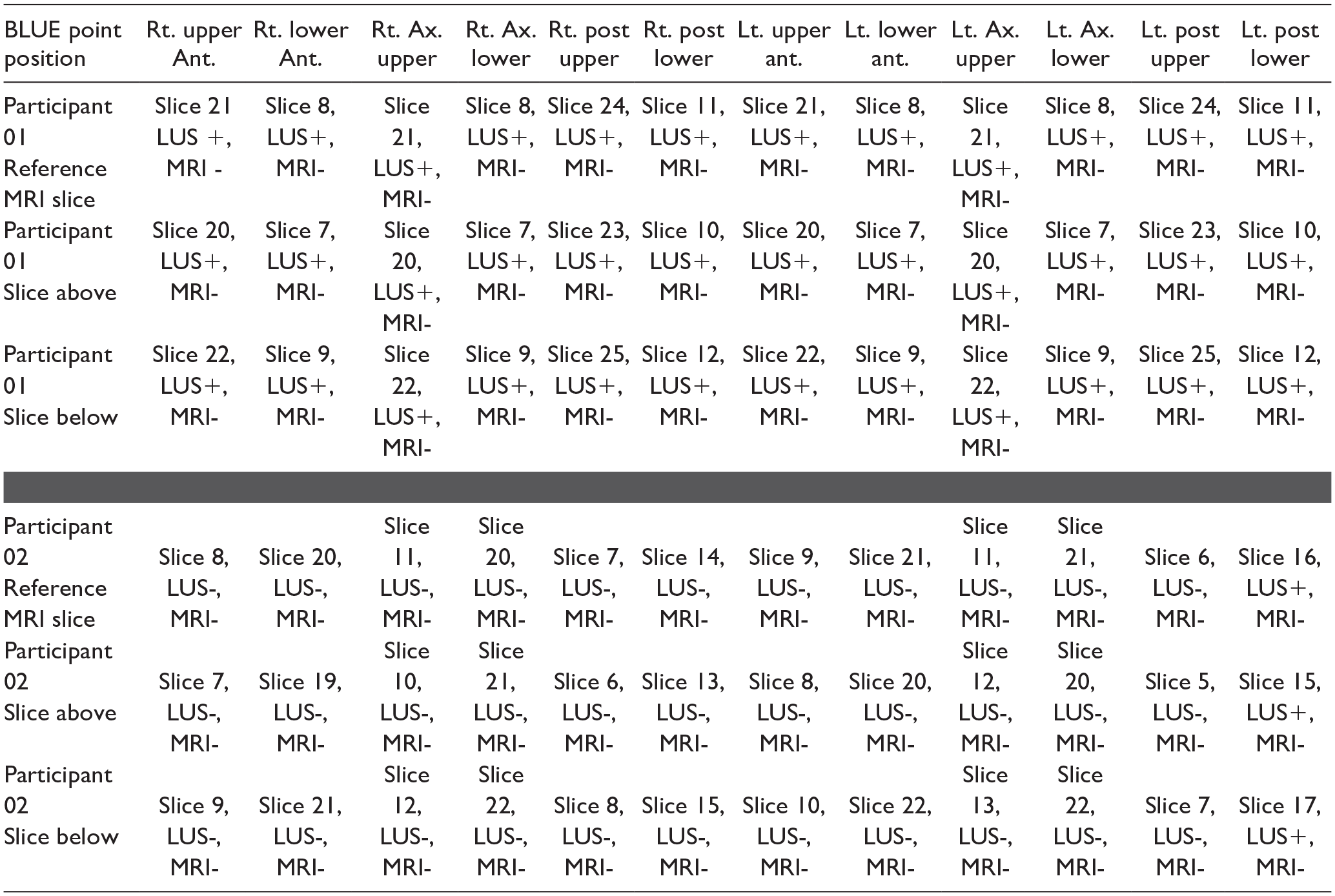
The diagnostic matching was made based on the reference slice and then checked by evaluating the slice above and below the reference slice. Example results are provided for participants 1 and 2.
Abbreviations: BLUE, the bedside lung ultrasound in emergency; Rt., right, Lt., left; Slice, axial MRI anatomical section; LUS, lung ultrasonography; MRI, magnetic resonance imaging; +, positive diagnosis; -, negative diagnosis.
Discussion
As providers and patients continue to navigate through a post-pandemic world, it has become apparent that many are suffering from the lingering effects of COVID-19. The most vulnerable are children and young adults, who may have difficulties expressing the chronic changes that they are experiencing from the virus. 17 Providers are faced with parents and children exhibiting signs of “long COVID-19” but very few diagnostic techniques to ascertain the chronic changes that may have occurred, due to SARS-CoV-2. This prospective cohort study was designed to address this gap in finding the proper diagnostic tools to assist in diagnosing the possible lung tissue damage that may be contributing to the self-reported challenges facing young people living with the aftereffects of COVID-19. In this study, the use of non-ionizing imaging was proposed to determine the contribution of each technique to assessing possible changes in the lung tissue of adolescents struggling with ongoing health concerns. In the current study, the lung sonograms demonstrated a propensity for chronic changes in the pleural line that likely was due to the virus. In a recent study by Palacios et al, a larger cohort of pediatric patients was examined with non-imaging tools to assess lung damage from COVID-19. In their work, 82 pediatric participants reported fatigue and dyspnea with exertion. More than half of their participants demonstrated a lack of abnormalities on their chest radiograph, lung function testing, and physical exam. 18 Nevertheless, this cohort was followed for 6.5 months and the group continued to report difficulty with breathing, after exercise (67%); however, the majority (67%) had a normal spirometry testing and (31%) a positive bronchodilator response. 18 The Palacios study is very comparable to this study as both groups reported slight fatigue, slight difficulty breathing, and slight difficulty with activities. It is interesting that the diagnostic testing provided to the Palacios cohort was not definitive as a surveillance set of techniques. Although population studies have pointed to a lower rate of hospitalization, due to COVID-19, for adolescents, the guiding principle has been that the illness was generally milder for children, compared with adults. 19 However, the lingering symptoms among pediatric patients appears to be elusive regardless of their viral load. This study may suggest that a combination of MRI and sonography is needed to properly evaluate both the superficial pleural lining, as well as the deeper lung tissue changes.
In a study by Kalkanis et al, they evaluated the diagnostic acuity of using CT and sonography to assess non-critical hospitalized patients. In that study, their cohort (N = 38) had a median age of 64 and were mainly male patients. However, the use of lung sonography scoring was highly correlated with the chest CT results. 20 The results from that study are limited due to the convenient sample of adult patients, but it does suggest that multi-modality imaging may be useful in detecting chronic lung changes due to COVID-19. When addressing the surveillance of adolescents recovering from COVID-19, CT is not advisable due to the ionizing radiation dose; however, this study would suggest that MRI could be a diagnostic substitute. To that end, the Austrian Society of Pneumology and the Austrian Society of Ultrasound in Medicine have endorsed lung sonography to assess patients suffering from “long COVID-19” symptoms. 21
In the present cohort of adolescent participants with long COVID-19, a possible association between increased body mass index (BMI) and increased pleural thickness was noted. This finding aligns with existing literature that identifies overweight and obesity as risk factors for severe COVID-19 and intensive care unit (ICU) admission. 22 Specifically, a study has found a nonlinear relationship between BMI and COVID-19 severity, with risks increasing with higher BMI. 23 Patients with higher BMIs have been found to have a higher risk of ICU admission, requiring mechanical ventilation, or dying from the disease. 22 The current findings suggest that possible similar mechanisms may contribute to increased pleural thickness in adolescents with long COVID-19 who have a high BMI. However, further research is needed to confirm these findings and elucidate the underlying mechanisms.
A systematic review provides the highest level of scientific and clinical evidence; however, those that have been published are centered on the use of a chest radiograph and CT for detecting acute disease and a lack of evidence on diagnostic tools for pediatric surveillance.24,25 This underscores the need for more studies on the use of non-ionizing diagnostic techniques for adolescents that provide comparable results. A systematic review that pulled data from adults and children found lung sonography was a highly reliable diagnostic tool, given that COVID-19 pneumonia has demonstrated effects on the periphery of lung tissue. 26 In addition, this review stresses the correspondence of lung sonography with axial imaging, such as CT, which is supported by this study. The implications of this systematic review were that more studies are needed on using lung sonography with adolescents to provide surveillance and how it might be used in conjunction with other diagnostic testing. 26 This study provides some unique data that respond to the call for additional studies with pediatric survivors of COVID-19 and demonstrates the importance of using an acoustic mapping process to provide both a superficial and deep diagnostic evaluation of chronic lung tissue changes.
Limitations
This cohort study does have major limitations due to the research design and convenient sampling strategy. Therefore, the threats to internal and external validity do not allow for generalizations. In addition, this cohort was predominately Caucasian and more representative of teenage youth who have recovered from COVID-19. The participants in this study were also limited in scheduling their lung sonogram in one facility and the MRI in an adjacent imaging center. In some cases, the wait time between the two appointments varied from two months to two weeks. Another issue that was encountered was that a few participants had a second episode of SARS-CoV-2 infection, and in one case, this occurred during the time of recruitment to schedule the first diagnostic appointment. It is unclear how many of the participants had more than one infection episode. Finally, this cohort was largely vaccinated (at least once) for COVID-19 prior to their infection episode. One participant was not vaccinated due to his diagnosis of autism and was hospitalized due to his SARS-CoV-2 infection.
Conclusion
The use of acoustic mapping with lung ultrasound and MRI for pediatric survivors of COVID-19 was explored and has demonstrated diagnostic utility. In this cohort, lung ultrasound demonstrated diagnostic effectiveness for evaluating the periphery of the lung tissue but was limited in viewing deeper structures.
Conversely, MRI provided definitive views of the entire chest cavity and lung tissue, which is important for properly evaluating deeper pulmonary parenchyma. The potential diagnostic synergy of using lung ultrasound and MRI as non-ionizing diagnostic tools for assessing pediatric patients is important, and it could provide physicians with a more complete evaluation of those patients who report “long COVID-19” symptoms. More research is needed on the proper combination of diagnostic testing, such as cardiac output, that could also contribute to some of the lingering symptoms reported. Considering the overlap in the self-reporting of slight fatigue, slight difficulty breathing, and slight difficulty with activities by adolescents, a comprehensive diagnostic evaluation needs to include blood biomarkers and cardiac output, as well as chronic lung tissue changes.
Footnotes
Acknowledgements
The authors thank Drs Simon Lee, Eric Mull, and Benjamin Kopp, who were critical in recruiting pediatric participants from their clinics.
Ethics Approval
Ethical approval for this study was obtained from Ohio State University (OSU IRB: 2021H0448).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding was provided by a grant from the ASRT Foundation.