Abstract
Objective:
As the global pandemic resulting from this virus continues, surviving patients continue to report chronic symptoms long after a diagnosis of coronavirus disease (COVID-19). A pilot study was conducted with a convenient sample to ascertain the proper diagnostic testing for detecting chronic disease.
Materials and Methods:
A convenient group of nine patients were directed by their primary care physician to be evaluated based on a variety of symptoms being reported by those surviving the acute phase of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The patients who volunteered provided sonographic imaging data of the lungs, kidney, and heart. This modified sonographic assessment was completed in a Family Practice office, with a laptop ultrasound equipment system, a blood draw, as well as completion of the EuroQol (EQ-5D) questionnaire. The data were collected as part of the patients 3- to 4-month follow-up primary care visit.
Results:
In this cohort, six patients were diagnosed and were able to recover at home. The other three patients chose to undergo monoclonal antibody therapy. The two subgroups’ data are provided and describe the types of residual effects that some individuals continue to suffer.
Conclusion:
Those patients with residual symptoms of SARS-CoV-2 have been dubbed “COVID long-haulers.” This combination of diagnostic tests, specifically renal sonography, flow cytometry, and dynamic sonographic assessment, may hold promise for guiding the treatment and surveillance of these patients.
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has caused a global pandemic, with millions of people worldwide suffering from the coronavirus disease (COVID-19). Extraordinary efforts have been made by health care providers to try and save as many patients as possible from COVID-19. The Centers for Disease Control and Prevention (CDC) has estimated that there have been more than 120 million total SARS-CoV-2 infections, of which more than 1 million patients had symptomatic illnesses.
1
In addition, during the period of February 2020 to September 2021, the CDC estimated that in the age range of 18 to 49 years, 3% of symptomatic cases were hospitalized. Likewise, in the age range of 50 to 64 years, 9% of symptomatic cases resulted in hospitalization.
1
The age range most commonly hospitalized was 65 years and older, of those hospitalized, 22% of those reporting symptoms.
1
This would suggest that many people between the ages of 40 and 60 years had symptomatic COVID-19 and recuperated outside the medical system. In a study by Pei et al,
2
they found that their Chinese COVID-19 acute patients had pneumonia as well as evidence of kidney pathology, demonstrated with an abnormal urine test. In the study of 333 patients with acute COVID-19 pneumonia, 75.4% had abnormal urine tests and only 45.7% of those patients regained their renal function.
2
This would suggest that those who survived the acute phase of the virus may have some chronic loss of renal function. In a retrospective review of 100 German COVID-19 survivors (43–53 years of age), it was revealed that 67% recovered at home, but magnetic resonance imaging (MRI) demonstrated that 60 patients had cardiac inflammation.
3
Those 60 COVID-19 survivors had chronic cardiac inflammation independent of any previously documented cardiac conditions.
3
These limited cohort studies of COVID-19 survivors would suggest that residual disease may be present and may be insidiously nested in those patients aged 40 to 60 years. Given this concern, a prospective effort was made to consent patients who had contracted COVID-19 and determine the residual disease burden in 40- to 60-year-olds, reporting to a primary care physician’s office. The research objective was to
Materials and Methods
A convenient sample of outpatients were initially identified as candidates for an institutional review board (IRB)-approved study on assessment for cardiovascular disease. These patients were consented at a local outpatient Family Practice office and met the study’s inclusion criteria of adult patients, 40 to 60 years of age. Although they did not have a history of cardiovascular disease at chart review, they did have a positive COVID-19 diagnosis, between September 2020 and January 2021. Although excluded from the main cardiovascular study, they were recruited to provide an extended set of diagnostic data, current blood work, and responses to a survey questionnaire. The data collected on these patients are part of their 3- to 4-month follow-up physician visit post-COVID diagnosis.
Sonographic Imaging of Participants
These nine COVID-19 survivors were invited to a 20-minute examination that included sonography and a blood draw, to detect chronic changes in the cardiovascular, pulmonary, and renal systems. An incentive for participation was provided to each patient for the extended time spent after their physician visit. The patients were examined with a GE Logiq laptop (Milwaukee, Wisconsin) ultrasound equipment system, a 3.0 MgHz curvilinear and 7.0 MgHz phased array transducer. The assessment protocol for this pilot study leaned heavily on the expertise of the physician/scientists in Wuhan and Hangzhou, P.R. China. The diagnostic algorithm used by physicians treating COVID-19 in China, specific for lung and renal disease, was provided to prospectively examine this cohort. The diagnostic steps provided were advocated as the clinical “road map” and was endorsed by the Chinese Medical Association.3 –6 Given these diagnostic steps used in treating many of the first COVID-19 patients in China, it was important to replicate methods, extend the data collection, and gauge the long-term effects of this disease.
A modified echocardiogram was performed to assess any structural or functional defects associated with the heart (Figure 1A and B). In addition, a bilateral lung sonogram that is focused on A-lines, B-lines, and lung sliding was completed using the BLUE-protocol (Figure 2A and B; Lichtenstein’s sonographic assessment technique for intercostal lung examination). 7 Furthermore, a bilateral renal sonogram was performed to evaluate size and cortical thickness (Figure 3). All imaging data collected at the Family Practice office underwent secondary image analysis at the research lab. Images, cine clips, and M-Mode Tracings were uploaded to the Philips DICOM viewer for intensive review and measurement. A set of credentialed sonographers completed both the imaging and the secondary image analysis.
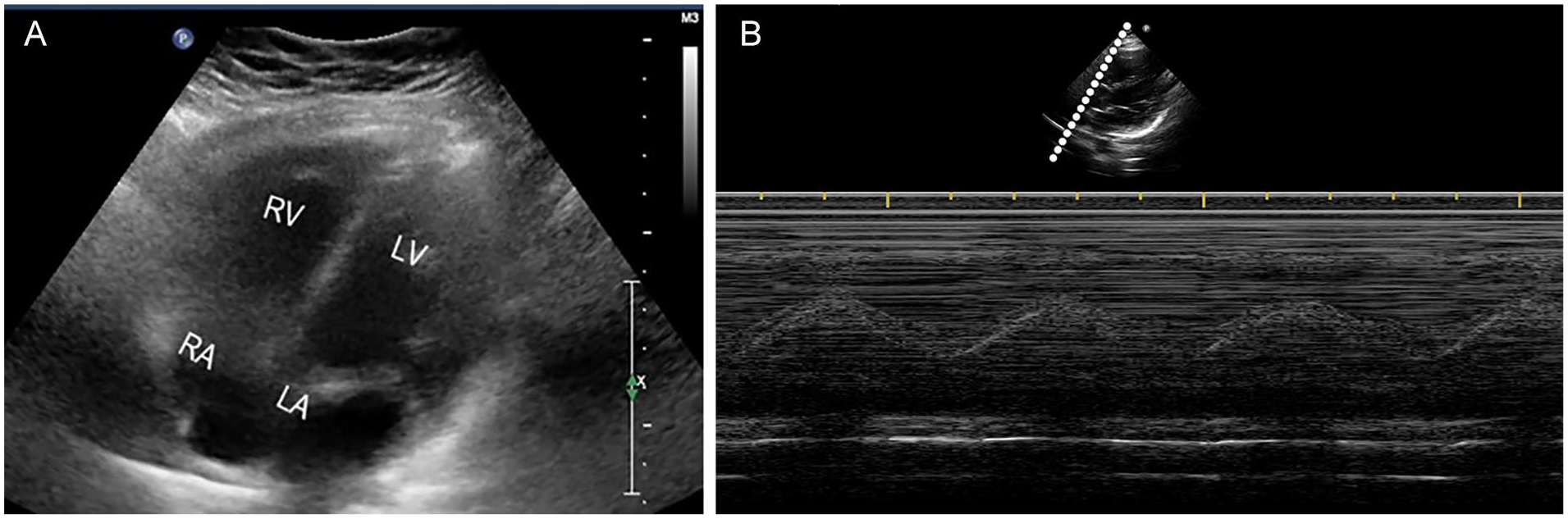
(A) Example patient’s apical four-chamber view of the heart. (B) Patient’s tricuspid annular plane systolic excursion (TAPSE) assessment, as part of a 3-month follow-up to his diagnosis. RV: Right Ventricle, LV: Left Ventricle, RA: Right Atrium, LA: Left Atrium.
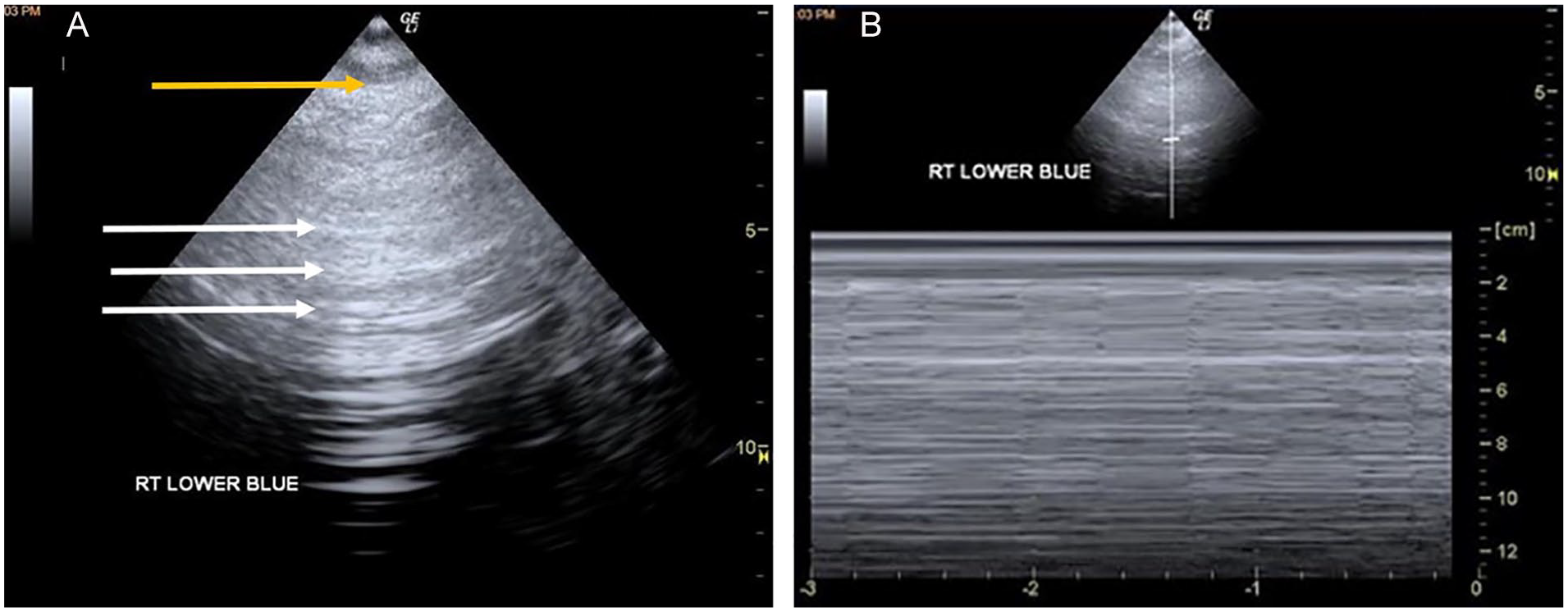
(A) Qualitative sonographic assessment of the right lower BLUE point on a 34-year-old COVID-19 female survivor, who rated her breathing as a slight difficulty. The gray-scale image demonstrates the presence of A-lines (horizontal) and the lack of B-line (vertical). White arrows indicate A-lines and the yellow arrow indicates the pleural line. (B) This M-mode image documents this same patient’s lung sliding and the M-mode tracing that shows the “seashore sign,” a demonstration of lung slide. This image was taken at the right lower BLUE point and helps to assess whether this area contributes to her rating of fatigue as slightly affecting her health.
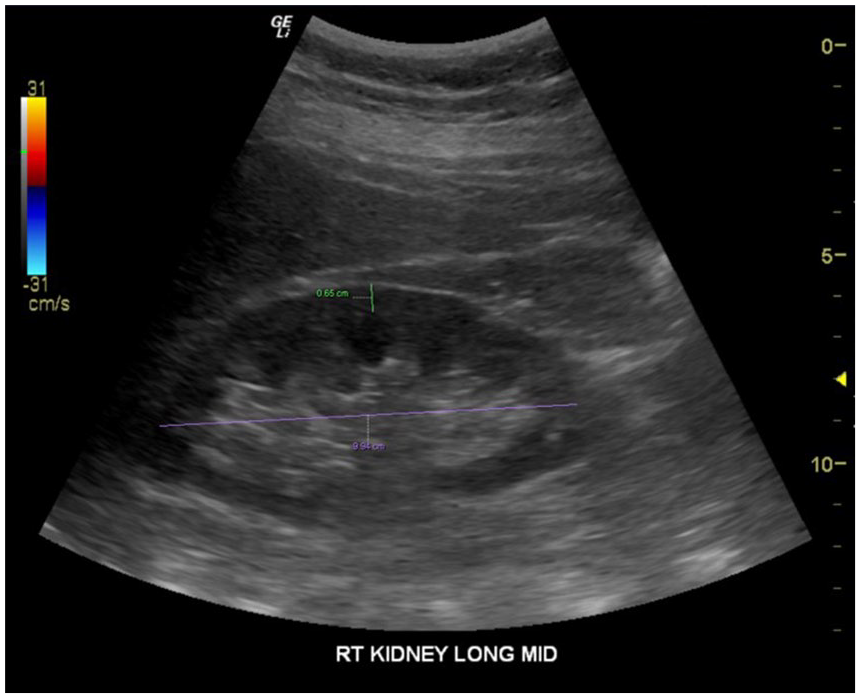
Right renal sonogram of a 50-year-old COVID-19 male survivor who had borderline cortical thickness (labeled in green), as part of the imaging assessment.
Whole Blood Collection and Flow Cytometry Analysis
In addition, patients underwent a blood draw performed by a trained phlebotomist. Peripheral blood from healthy donors and patients were collected into CPT tubes (BD, Franklin Lakes, NJ) and whole blood staining was performed. After collection, 120 μL of whole blood was separated into polystyrene FACS (fluorescence-activated cell sorting) tubes and red blood cells were lysed using ACK buffer (Thermofisher, Waltham, Massachusetts) for 5 minutes followed by a wash with R10 media (RPMI-1640 supplemented with 10% FBS [fetal bovine serum], 2 mM
Flow Cytometry Data Analysis
Flow cytometry data were analyzed using the following standardized workflow. The first step was to integrate the flow cytometry marker data from all the patients and healthy donors for implemented compensation and scaling using OMIQ (https://www.omiq.ai/). Then, it was necessary to visualize cells in a reduced two-dimensional space using the Uniform Manifold Approximation and Projection (UMAP) algorithm implemented in the OMIQ. The next step was to annotate cell subsets by visually investigating heatmaps of median marker expressions across clusters and expressions of these markers on the UMAP space. From the data set, the optimal number of cell clusters were chosen by selecting the model with the minimum Bayesian information criterion (BIC) score. Then, it was important to annotate cell types by visually investigating heatmaps of median marker expressions across clusters and expressions of these markers on the UMAP space.
Administration of the EuroQol
In addition to the modified diagnostic imaging protocol, patients were also asked to complete the EuroQol (EQ-5D) questionnaire. The EQ-5D is used to measure five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. 8 Patients were able to quickly provide their responses to the EQ-5D adult version. In this series, the EQ-5D dimensions were extended from three levels to five: no problems, slight problems, moderate, severe, and extreme problems. Each patient was asked to rate their health by checking the box next to the most appropriate statement, for each of the five dimensions. In addition, patients were asked about the cognitive lapses in concentration that have been called “COVID foggy brain.” 9
Results
This pilot study was composed of a cohort of outpatients who had survived the acute phase of COVID-19. There were ten qualified patients invited for this data collection; however, only nine patients consented. All of the consented patients had recovered from COVID-19, without hospitalization. Six patients stated that they had post-COVID-19 conditions. The CDC describes these lingering chronic complaints as “long-haulers” or “Long COVID.” 10 With Long COVID, patients can experience a range of symptoms that can last weeks or months after first being infected. Long COVID can happen to anyone who has had COVID-19, even if the illness was mild or asymptomatic. 11 The second subgroup of three patients also recovered from COVID-19; however, they were offered monoclonal antibody therapy (MA) within the first 7 days of their initial diagnosis. This cohort of patients had their diagnosis made between September 2020 and January 2021. The complete cohort was composed of six women and three men who had an average age of 43 years. The results for these two subgroups are presented separately to display their data appropriately.
“Long COVID” Group 1
These six patients (one male and five females) completed all of the required elements for the data collection. The modified sonographic imaging provided the following metrics: renal dimensions, dynamic and qualitative lung assessment, and a modified dynamic cardiac assessment. The data frequency of renal measurements, in group 1, is provided in the unshaded portion of Table 1. Renal measurements for normal adults were referenced against the guidelines provided by Fiorini and Barozzi. 10 They suggest that renal length should be between 9 and 14 cm and a discrepancy of greater 2 cm between bilateral kidneys is noteworthy. In addition, the renal cortical thickness for normal adults has been reported to fall in the range of 8 to 10 mm (see Figure 1). 12 Those patient measurements that fall outside of these normative ranges are highlighted in yellow in Table 1. An additional measure of renal function, estimated glomerular filtration rate (eGFR), was estimated on each patient via blood work. Each of the six individuals in group 1 had a normal eGFR of >60 mL/min/1.73m2. The dynamic sonographic assessment of the lungs, based on the three standardized BLUE points, is provided in the unshaded portion of Table 2. In addition, M-mode was utilized to demonstrate lung sliding (breathing) and captured what is known as the “seashore sign” (see Figure 2). Cardiac dimensions are provided for this cohort in the unshaded portion of Table 3. The parasternal long-axis view was used for both the dynamic and static assessments (see Figure 3). The cardiac assessment was based on the guidelines provided for making measurements with hand-held ultrasound equipment. 13 Ultizing the parasternal long-axis cardiac view allowed for the assessment of pericardium, the presence of pericardial effusion, ventricular, and atrial dimensions. The quantitative measurements used to take ventricular and atrial measurements were completed as outlined by Kou et al. 12 Given the limited time allotted to complete the sonographic examination of the bilateral kidneys, bilateral lungs, and heart, only a few patients were able to also allow for a tricuspid annular plane systolic excursion (TAPSE) measurement. TAPSE is obtained by placing the M-mode cursor over the lateral aspect of the tricuspid annulus. On the M-mode tracing, we measured the distance of the maximal longitudinal displacement. The guideline used for a normal TAPSE was 20 mm 14 (see Table 3).
COVID-19 Survivors’ Date of Diagnosis and Date of Examination for Bilateral Renal Dimensions.
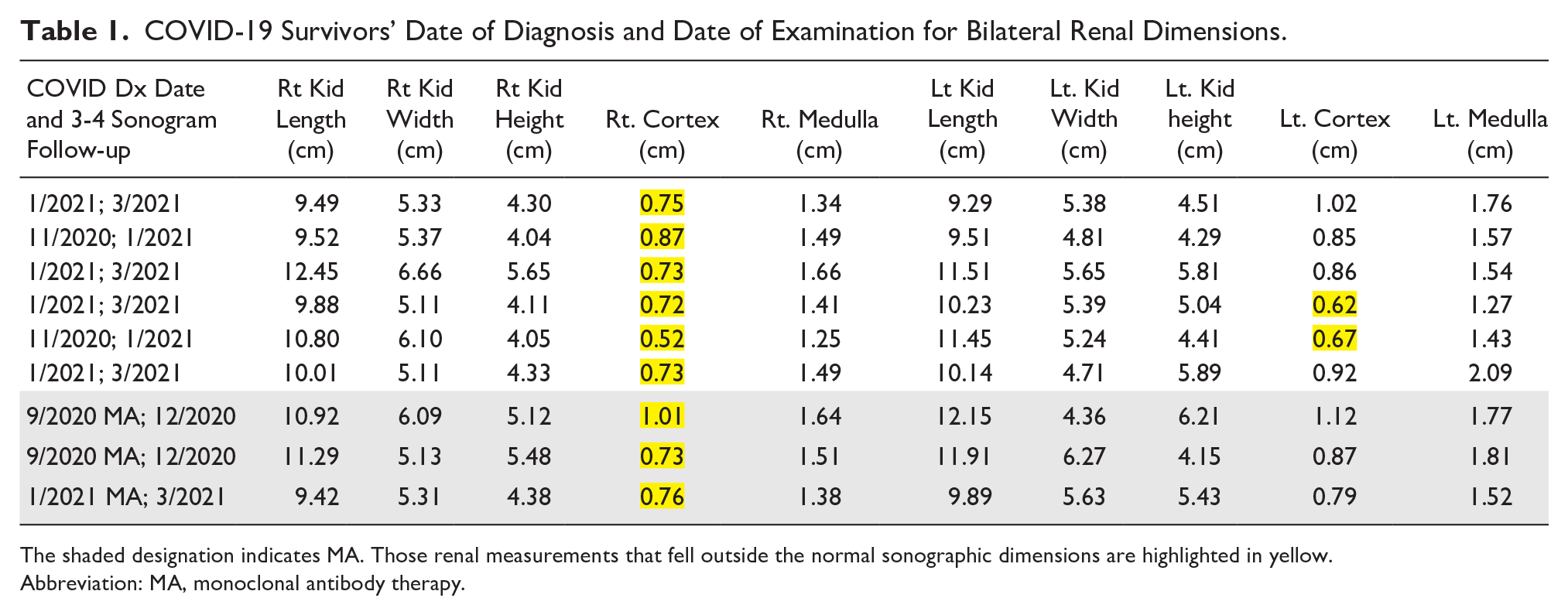
The shaded designation indicates MA. Those renal measurements that fell outside the normal sonographic dimensions are highlighted in yellow.
Abbreviation: MA, monoclonal antibody therapy.
COVID-19 Survivors’ Qualitative Sonographic Lung Assessment.
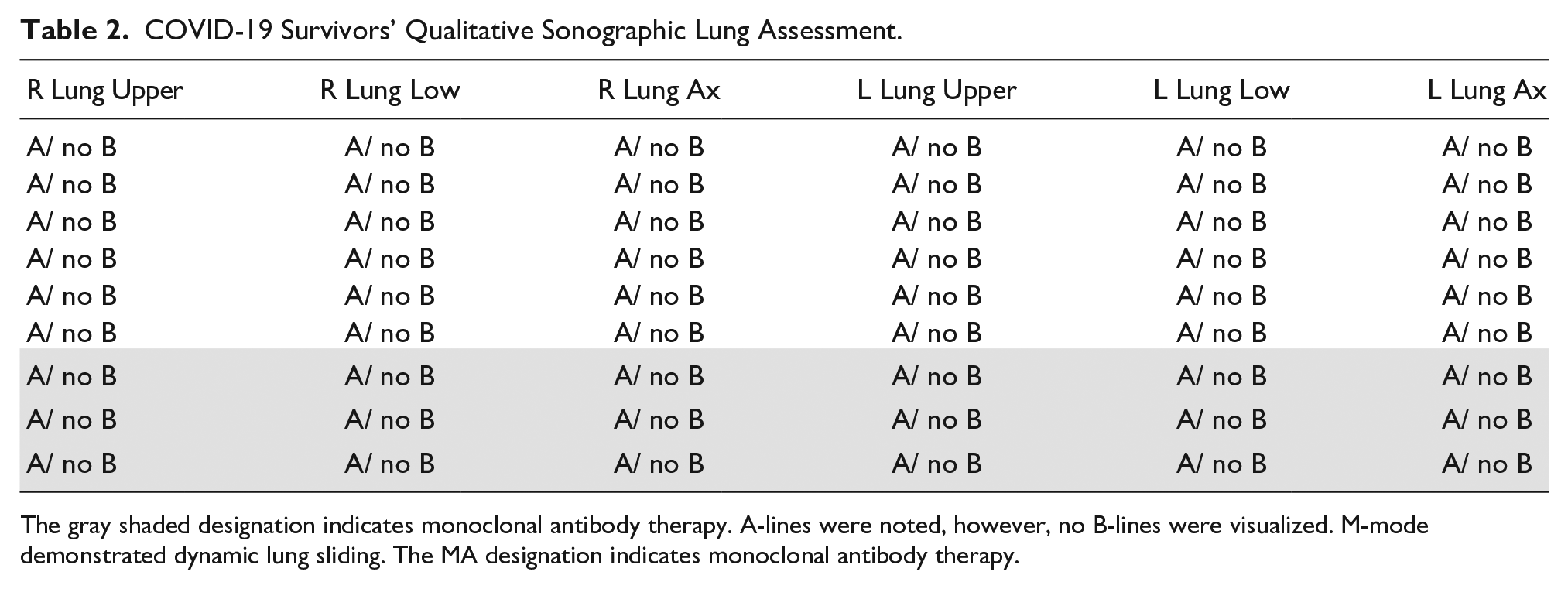
The gray shaded designation indicates monoclonal antibody therapy. A-lines were noted, however, no B-lines were visualized. M-mode demonstrated dynamic lung sliding. The MA designation indicates monoclonal antibody therapy.
COVID-19 Survivors’ Sonographic Qualitative and Quantitative Cardiac Assessment.
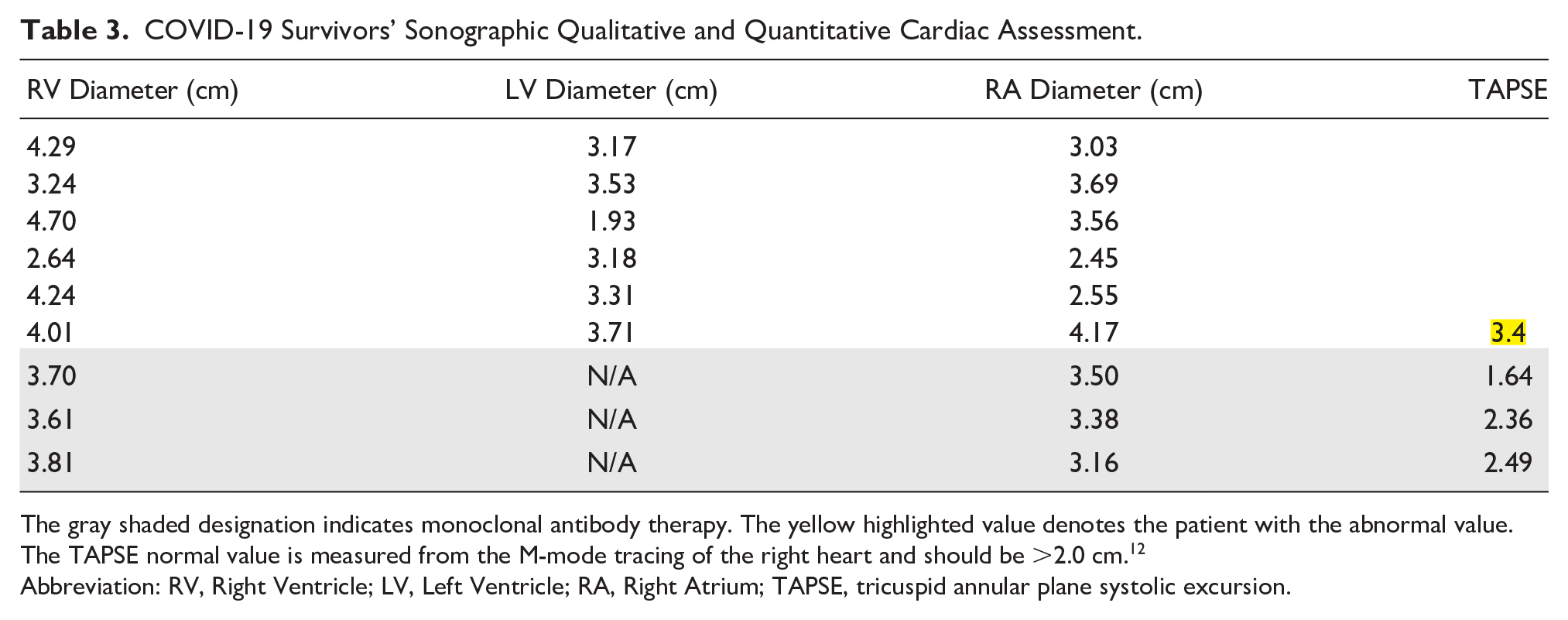
The gray shaded designation indicates monoclonal antibody therapy. The yellow highlighted value denotes the patient with the abnormal value. The TAPSE normal value is measured from the M-mode tracing of the right heart and should be >2.0 cm. 12
Abbreviation: RV, Right Ventricle; LV, Left Ventricle; RA, Right Atrium; TAPSE, tricuspid annular plane systolic excursion.
MA Group 2
These three patients (two males and one female) were recruited from the family practice office, but it was not until the history-taking session that it was discovered that they have been offered and completed MA. 15 MA has been offered to patients with COVID-19 and is most effective if the intravenous (IV) infusion is started as soon as the patient is diagnosed. MA is composed of the antibodies casirivimab and imdevimab and is administered IV. Given that the imaging and data collection were completed with these patients, post IV infusion, their results were isolated.
As with group 1, the MA group 2 patients were sonographically imaged to assess their bilateral kidneys. Their renal dimensions, cortical thickness, and medullary thickness are provided in Table 1. Group 2’s specific data are shaded gray to indicate those specific rows of data. The eGFR was again estimated on each patient via blood work. Each patient in group 2 had a normal eGFR of >60 mL/min/1.73m2. Group 2 patients also underwent sonographic assessment of their bilateral lungs. This quantitative and qualitative data are provided in Table 2. Likewise, the subcostal cardiac view was also completed on these three patients and their data are provided in Table 3. It is worth noting that patient no. 7 had the only abnormal TAPSE measurement, which was recorded.
Finally, these patients provided a blood sample for a normal metabolic panel and for measurement of monocyte activation by advanced flow cytometry. Interestingly, there were some COVID-19 survivors who had an increase in total, intermediate, and classical monocytes compared with COVID-19–negative individuals. Given that this was a cross-sectional, convenience cohort of COVID-19 survivors, the changes in monocyte phenotype, proportional representation, and activation induced by COVID-19 are difficult to assess. Figure 4 demonstrates the activation, exhaustion, and chemokine expression with the different monocyte subsets in three different sample groups: the control group from the recruited cardiovascular disease study; group 1, six participants; and group 2, three participants, who underwent MA. This qualitative analysis of the activation markers indicated a slight increase in the patients diagnosed with COVID-19 compared with the healthy controls. Ideally, a quantitative analysis would be a preferred method moving forward.
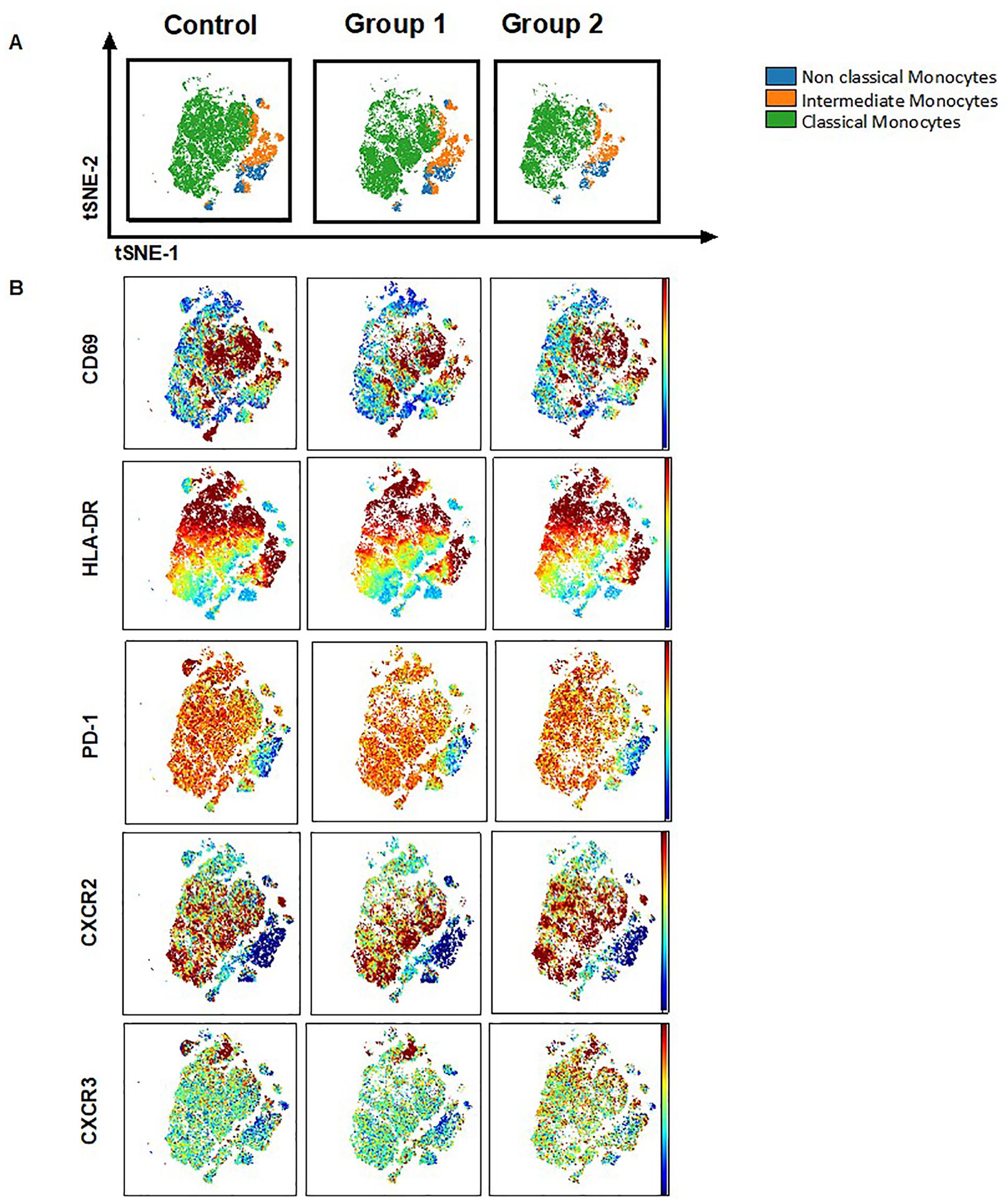
Columns 1, 2, and 3 are the control group, group 1, and group 2, respectively. All groups demonstrate monocyte subsets and their activation, exhaustion, and chemokine receptor expression. (A) The horizontal row demonstrates global tSNE projection of monocyte subsets for healthy donors (n = 19), group 1 (n = 6), and group 2 (n = 3) patients concatenated and overlaid. (B) The horizontal rows demonstrate tSNE projections of indicated protein expression in the monocyte subsets.
This cohort was also asked to respond to the modified EQ-5D survey and the extra question about whether they had experienced COVID “foggy brain.” The results of the questionnaire are provided in Table 4. The self-rated health responses for fatigue have the most concerning responses with group 1 reporting an average of 2 (slight impairment). The next variable of concern for group 1 was the average value of 1.83 for pain/discomfort (see Table 4).
COVID-19 Survivors’ Responses to the EuroQol 5D Adult Survey With Expanded 5-Point Responses (1 Indicating No Problem Associated With Category, 5 Indicating Extreme Problems).
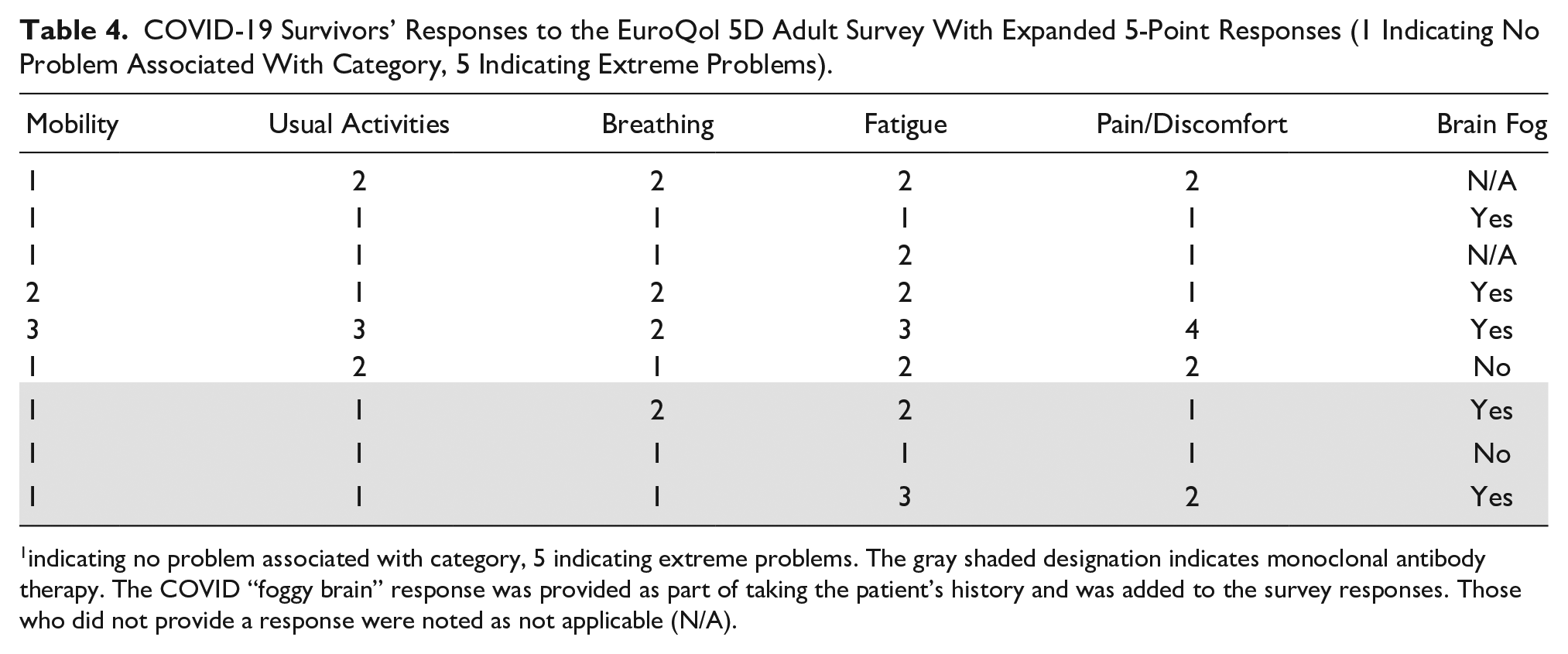
indicating no problem associated with category, 5 indicating extreme problems. The gray shaded designation indicates monoclonal antibody therapy. The COVID “foggy brain” response was provided as part of taking the patient’s history and was added to the survey responses. Those who did not provide a response were noted as not applicable (N/A).
Group 2 responses for the “foggy brain” phenomenon and modified EQ-5D are reported in Table 4, specifically the rows denoted with gray shading. Note that patient no. 7 also reported slight fatigue and multiple episodes of COVID “foggy brain.”
Discussion
SARS-CoV-2 is a viral infection that causes COVID-19, with both acute and chronic symptoms. As of April 14, 2021, the global pandemic had resulted in more than 136 million confirmed cases with more than 2.9 million deaths worldwide. 16 This novel coronavirus results in a wide-ranging disease course, with some individuals requiring hospitalization and supplemental oxygen, while others experience mild or no symptoms. Age and underlying medical conditions including cardiovascular disease, obesity, and diabetes have been identified as risk factors for a severe disease course. 5 Yet, young healthy individuals have also become critically ill. A significant proportion of individuals with COVID-19 experience damage to their lungs, heart, or kidneys,3,6 even if their disease course is not severe. Globally, diagnostic data taken from hospitalized patients have helped to unravel some of the mystery of this disease. What remains elusive is the degree to which residual disease and chronic pathologic changes are present in those adults who were infected but not hospitalized. “Long COVID” is a new term17 –19 that has developed to describe those patients, reporting to primary care physicians, with the lingering effects of this disease. A study performed by Logue et al 20 found that 32.7% of outpatients with mild COVID-19 infection were experiencing at least one persistent symptom between 3 and 9 months after illness onset. Likewise, the present cohort (groups 1 and 2) is also emblematic of these same sequelae. Furthermore, Logue et al 20 reported that 29.3% of their outpatients also reported having a worsened quality of life in the months after COVID-19 infection. Surprisingly, both groups in the present cohort (groups 1 and 2) reported fairly high quality of life. Nevertheless, the results of this pilot study demonstrate the need of further investigation into patients with “Long COVID” symptoms, specifically those within an outpatient medical practice.
This cohort demonstrates the feasibility of assessing COVID-19 survivors with a combination of sonography, flow cytometry, lab tests, and survey responses, in an outpatient environment. As more and more patients are recovering from COVID-19, the need for further investigation into the residual health of survivors is needed. It is unclear as to the severity of organ damage that may have occurred from COVID-19, after the virus has run its initial course. To compare this cohort and its possible lingering disease, the following keywords were used to search a variety of databases: SARS-CoV-2, COVID-19, coronavirus, chronic, follow-up, clinical sequelae, long term, survivor, recovered, hospitalized, hospitalization, post-COVID, kidney function, renal, heart, cardiac function, cardiovascular, pulmonary function, and lungs. These systematic searches resulted in obtaining articles on potential chronic changes to various organ systems. It also indicated that some early investigations had been completed, based on hospitalized patient data. According to Puntmann et al, 3 MRI cardiac investigation of patients recovering from COVID-19 showed that there were long-term cardiovascular implications, in a cohort of patients believed to be free of the virus. In addition, pulmonary function appeared to be decreased post-COVID-19, based on a study by Zhao et al. 6 Their work demonstrated that 3 months after discharge, a significant percentage of the patients had abnormalities demonstrated on their chest computed tomogram (CT). In the present study cohort, lung sonography provides a less ionizing method for providing surveillance of lung tissues changes. Finally, a study performed by Hirsch et al 21 found a high incidence of acute kidney injury (AKI) in hospitalized COVID-19 patients. Based on this limited literature search, there appears to be limited information regarding the renal function of discharged COVID-19 patients.
After reviewing the research published by the aforementioned authors, there continues to be a concern regarding the extent and prevalence of cardiovascular, pulmonary, and renal system damage, due to COVID-19. Both of the current outpatient groups that comprise this cohort demonstrated limited sonographic and no eGFR evidence of chronic disease, but did have a variety of monocyte activation profiles, which should be explored in greater detail in a larger cohort. In group 1, the borderline measurement of renal cortex thickness could be indicative of what was reported by Hirsch et al. The ability to use a hand-held ultrasound equipment system to do a targeted assessment can be important to determine whether patients with “Long COVID” symptoms need further diagnostic evaluation. Taking a blood sample from these patients is minimally invasive and has the potential to yield very important diagnostic results. In both cohorts (groups 1 and 2), the flow cytometry data could be important in determining whether the patient was still experiencing an inflammatory immune response throughout their body. One could theorize that with immune activation and inflammation some of the EQ-5D responses might also normalize. The ongoing surveillance of COVID-19 survivors has also been recommended based on the case series of German adult survivors. 3
Limitations
The major limitation of this study is the research design which is pre-experimental. Given this research design, the results are exclusively applicable to this cohort. Next, a small convenient sample was recruited, which further constrains the generalizability of the results. Furthermore, the qualitative assessments used limit reproducibility. Future work would be enhanced by a random sample of a larger population that uses these techniques with a stronger reliance on quantitative measurements.
Conclusion
As this is a virus that has affected and will continue to affect millions of people, it is imperative that a better understanding of the residual changes in organ systems be explored. Due to this slowly resolving health emergency, it would seem important to discover whether there has been damage to the vascular perfusion and structural integrity of the heart, lungs, and kidneys, among post-COVID-19 patients. Outpatient medical practices continue to handle patients with “Long COVID” symptoms. The use of additional diagnostics such as a modified sonographic examination coupled with a blood draw could help in screening those patients who need more medical attention. More data are needed to determine the role of immune cell activation within COVID survivors who continue to report prolonged symptoms or are concerned about breakthrough infections. The possible loss of renal cortical thickness among patients with “Long COVID” symptoms is also worth further diagnostic investigation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Peer Reviewer Guarantee Statement
The Editor/Associate Editor of JDMS is an author of this article; therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Associate Editor had no involvement in the decision-making process.
