Abstract
Thrombosis of the portomesenteric veins is an uncommon condition that can lead to serious complications, if left untreated. Bariatric reduction surgeries increase the probability for occurrence of this rare condition. This case study focuses on a patient who underwent a biliopancreatic diversion with duodenal switch procedure and consequently developed an occlusive thrombus in the superior mesenteric vein (SMV), which required stent placement. The patient ultimately developed a secondary thrombus, within the stent. Sonographic imaging aided in the diagnosis of occlusion of the SMV stent.
Keywords
Thrombosis of the portomesenteric veins is a relatively uncommon but serious complication following bariatric surgeries, particularly after laparoscopic sleeve gastrectomy (LSG). 1 The incidence of portomesenteric vein thrombosis (PVT) has been reported postoperatively from 0.3% to 1%, across multiple published studies. 2 While mild cases of PVT can be asymptomatic, there is a risk of potentially fatal effects if left untreated. Some of the clinical presentations include ascites, portal hypertension, gastrointestinal (GI) bleeding, and bowel ischemia. 3 One of the most serious complications, intestinal infarction, occurs as a result of venous obstruction, which leads to an impairment of arterial perfusion and thus tissue necrosis.4,5
Bariatric surgical procedures have increased in incidence in recent years to deal with the rise in global obesity. Obesity is defined as a body mass index (BMI) of greater than 30. 6 There are multiple health complications that go along with obesity and treating this epidemic is not an easy task. There are multiple variations of bariatric surgeries that either limit the size of the stomach or reduce intestinal absorption capabilities. The most common procedure is the LSG. This procedure reduces the size of the stomach and therefore limits the number of calories that can be consumed. 6 However, for severely obese patients with a BMI of greater than 50, there is another procedure that may be more effective in dramatic weight loss, with long-term success, in this population. The procedure is the biliopancreatic diversion with duodenal switch (BPD/DS). The surgical treatment consists of a partial gastrectomy with preservation of the pylorus and a bypass that connects a distal portion of the small intestines (ileum) to the duodenum. This complex intervention both limits the quantity of food that can be ingested and reduces the absorption capacity of the digested food due to a major bypass of the small intestines. 6
There are many health concerns that accompany a diagnosis of morbid obesity. Not only do these patients suffer from comorbidities such as type 2 diabetes, heart disease, obstructive sleep apnea, and stroke, but they are also considered high risk for development of venous thromboembolus. 7 To combat this, some patients are given prophylactic anticoagulants to help reduce the incidence of a postprocedural thrombotic event. Some of the causes of PVT after bariatric surgery have been identified as trauma to the portomesenteric structures, decreased flow in vessels, and inflammation, because of the surgical procedure. In addition, increased abdominal pressure with a subsequent decrease in venous return during laparoscopy plays a role in thrombus development. 7
It has been reported that most cases of PVT occur within 2–4 weeks postoperatively. 1 Diagnosis of PVT is usually based on clinical presentation and preadmission imaging studies, such as computed tomography (CT) and sonography. Although patients may present with varied and transient symptoms, most will experience abdominal pain, nausea, vomiting, diarrhea, and GI bleeding. 7
Case Report
A 36-year-old woman presented to the emergency department (ED) with pallor, black stools, and difficulty being roused from sleep. A complex patient history revealed a BPD/DS procedure performed at an outside facility. Two weeks postoperatively, the patient developed an occlusive thrombus in the superior mesenteric vein (SMV), which was treated with a thrombectomy in the interventional radiology (IR) department and was put on the anticoagulant, Eliquis. The patient continued to have complications including anemia which required a blood transfusion. She also developed mesenteric inflammation without ischemia. During this time, an esophagogastroduodenoscopy (EGD) revealed a GI bleed which prompted cessation of the anticoagulants. One year after surgery, the patient presented with abdominal pain and an upper small bowel bleed. A CT angiogram (CTA) of the abdomen, with and without intravenous contrast, was performed. Severe wall thickening of the descending colon was noted. There were numerous venous collaterals seen due to the chronically occluded SMV, with diffuse mesenteric congestion. Collateral veins were also seen near the duodenum with extension into the duodenal wall. Additional varices were noted near the duodenoileostomy anastomosis and a bleed could not be excluded. Based on the CTA findings, the patient was sent to IR for an SMV stent placement.
Six months after SMV stent placement, the patient made a return visit to the ED. A CT was ordered on admission with presenting symptoms of acute abdominal pain and recurrent GI bleeds. The diagnostic findings showed edema of the ileal anastomosis and thickening of the right colon. Also noted was small amount of ascites and diffuse mesenteric edema. The SMV stent was noted in position with suggestion of recurrent in-stent stenosis (See Figures 1 and 2). However, patency was not well evaluated so an abdominal sonogram with Doppler assessment was recommended. The sonogram was performed and confirmed complete occlusion throughout the course of the stent and patency of the main portal vein (See Figures 3 and 4).
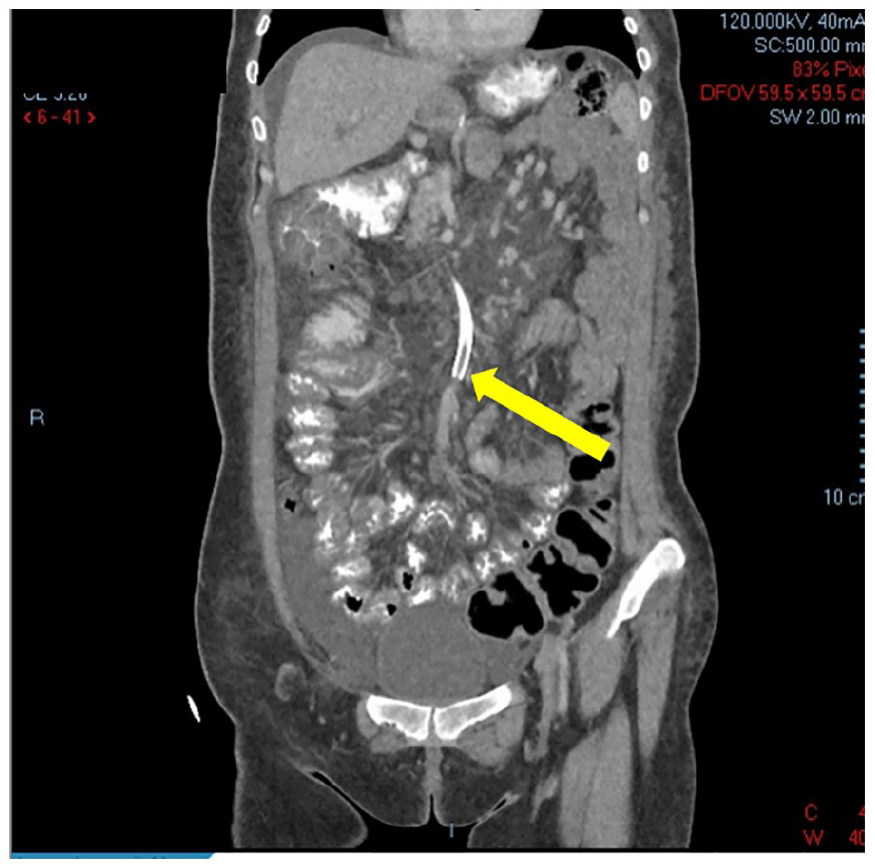
The patient’s computed tomographic coronal image demonstrated the thrombus within the superior mesenteric vein stent (See yellow arrow).
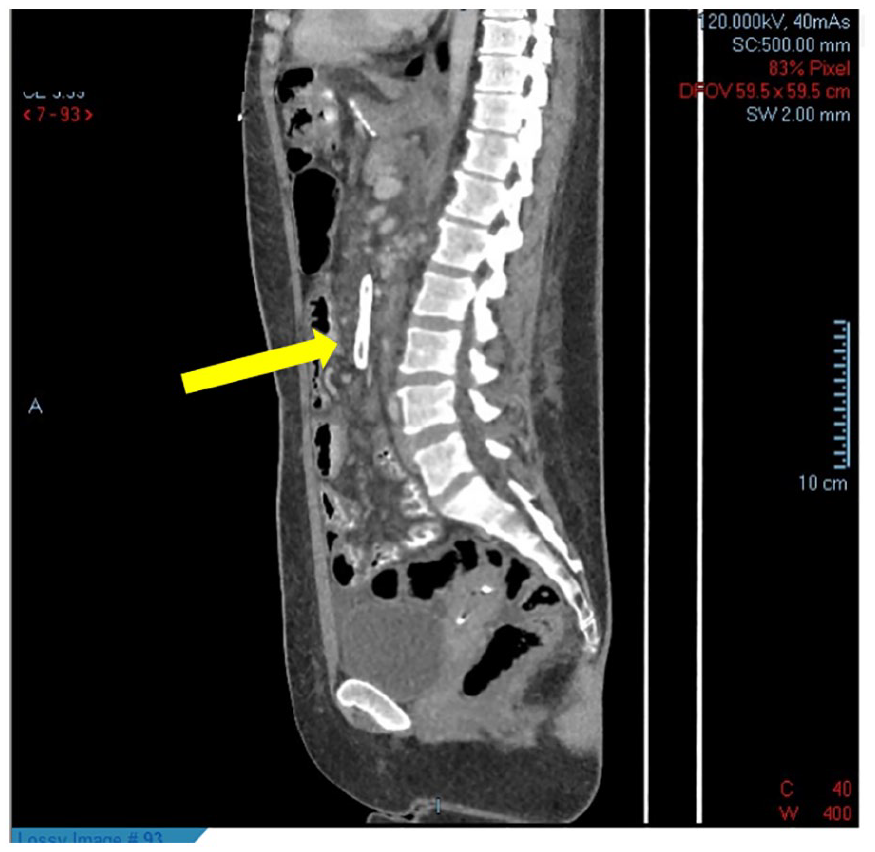
The patient’s computed tomographic sagittal image also showed thrombus in the superior mesenteric vein stent (See yellow arrow).
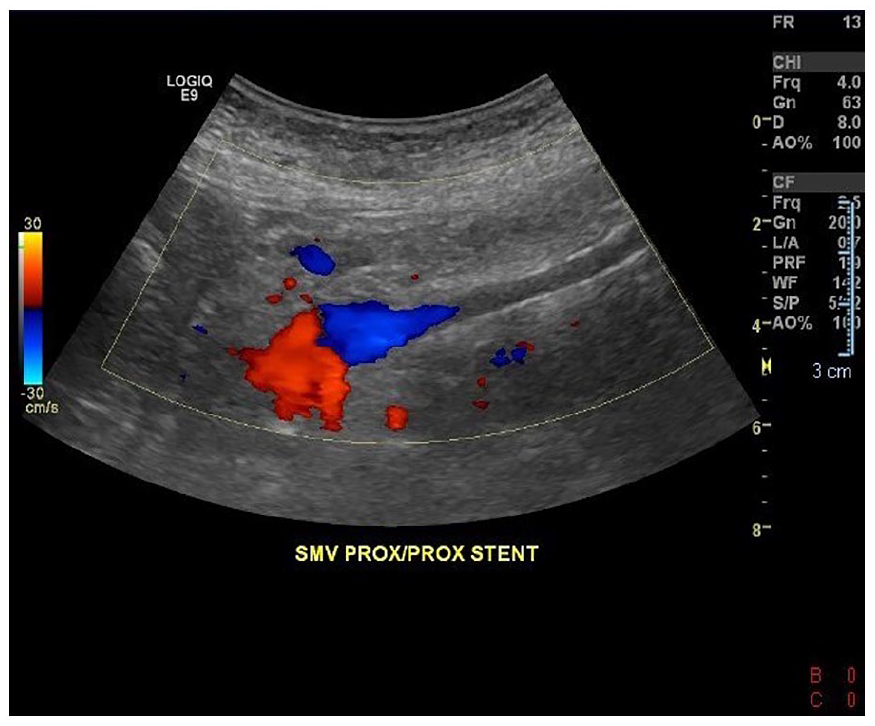
This color Doppler image demonstrates the absence of flow in the proximal portion of the stent, with flow present in the portal vein.
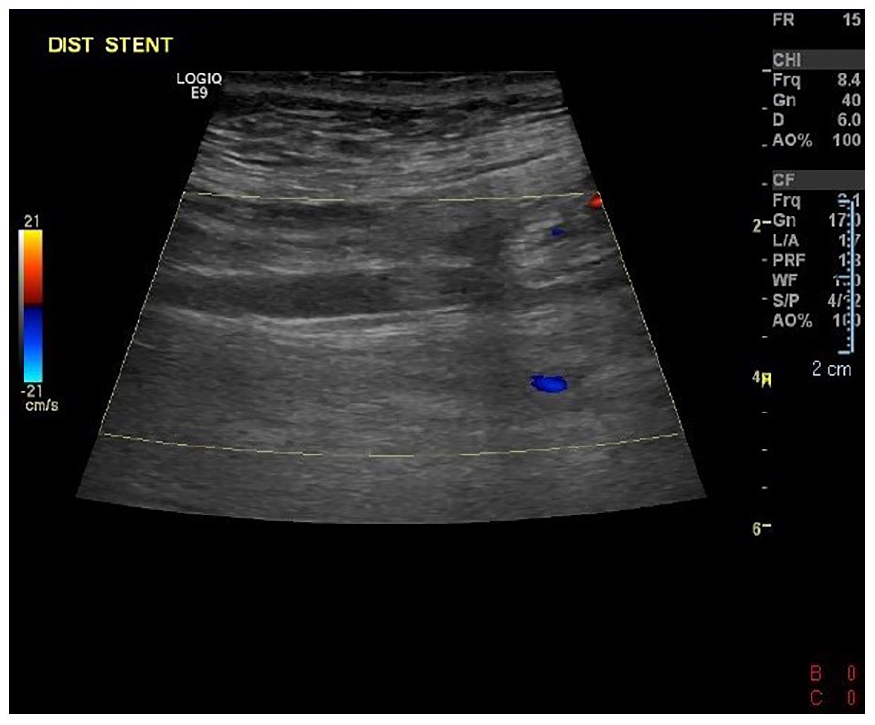
This color Doppler image also provides evidence of an absence of color flow in the distal stent.
The patient was then referred to the IR department for venography. Under direct sonographic guidance, the right internal jugular vein was accessed and a catheter was introduced into a right portal vein branch which allowed access to the SMV stent. The venogram confirmed complete occlusion of the stent with prominent mesenteric venous collateralization. Angioplasty was performed with a 6-mm balloon, but postvenography demonstrated only minimal flow through the stent. A 7-mm stent was then placed, which improved vascularization as the postvenogram demonstrated brisk flow through the stent. The new stent was evaluated with sonography repeatedly with a sonogram 2 days after placement, to check vascular flow and velocities. At that time, the stent velocities were normal in the proximal, mid, and distal portions and color Doppler documented the flow, in all portions of the stent and vessel. The portal vein remained patent with hepatopetal flow. One month after placement of the second SMV stent, the patient was reevaluated with sonography to determine patency. The SMV stent was reported as widely patent and had hepatopetal blood flow (See Figures 5 to 7). The velocities in the stent ranged from 36 to 76 cm/s. The peripheral branches of the SMV were also patent with a velocity of 29 cm/s. The portal vein also showed hepatopetal flow with a velocity of 81 cm/s.
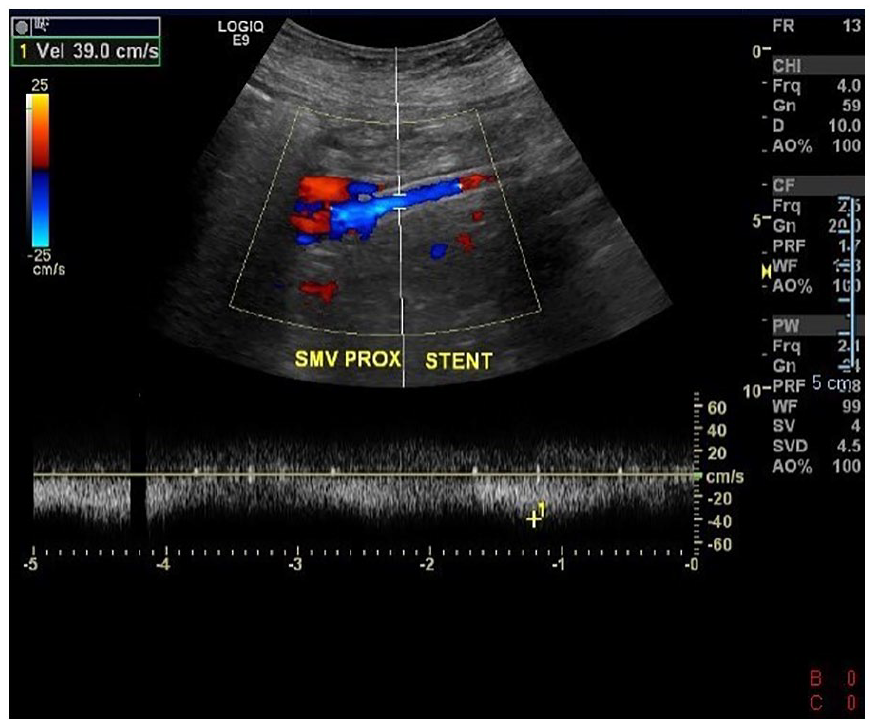
This triplex Doppler image and spectral analysis demonstrated patency of the proximal portion of the new stent.
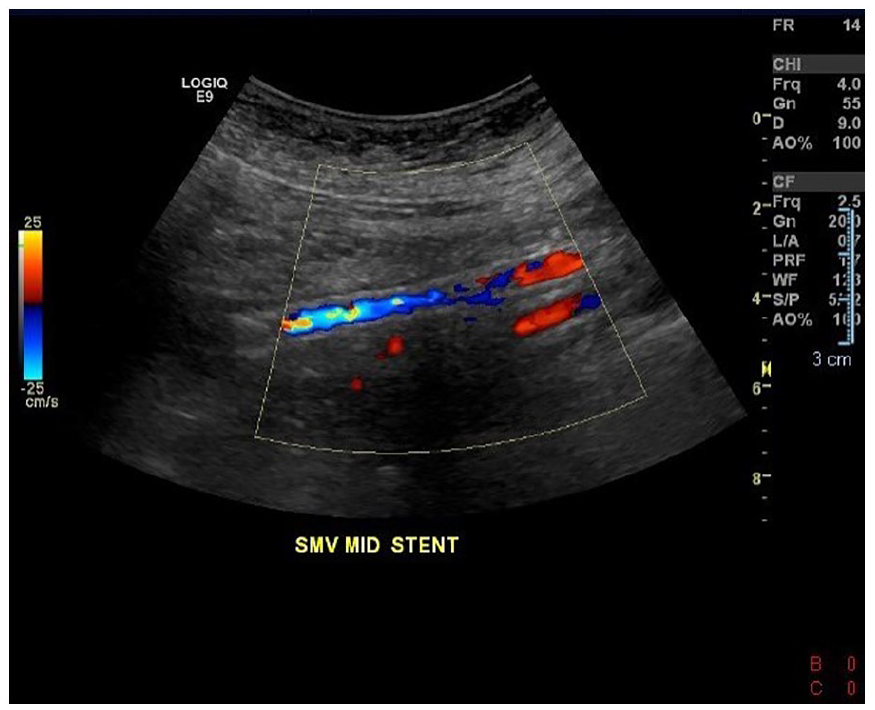
This color Doppler image provides evidence of vascular flow in the mid-portion of the new stent.
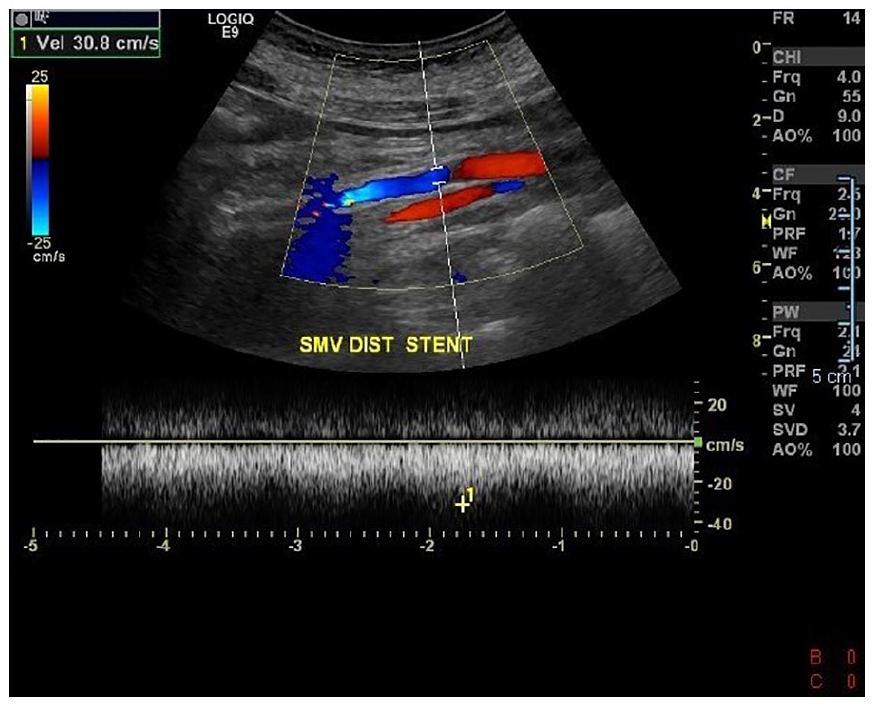
This Doppler triplex image and spectral analysis demonstrated the flow and patency of the distal portion of the new stent.
Discussion
Cases of occlusion of the SMV as reported are uncommon, and portomesenteric thrombus formation, after bariatric surgery, is even more rare.1,2 Asymptomatic cases may go undiagnosed, but patients who present with life-threatening complications require immediate intervention. Some of the more common causes of benign occlusion of the SMV include pancreatitis or surgical injury, that is, bariatric procedures. 3 One of the main treatments for PVT is therapeutic doses of an anticoagulant, such as low-molecular-weight heparin (LMWH). 2 Alternatively, some patients can be treated with and show improvement of symptoms after stent placement, in the occluded vessel. Stenting can be a minimally invasive intervention as compared with open surgery and the procedure can be performed by a percutaneous transhepatic approach, using sonographic guidance. 3 There is limited published documentation regarding stenting and revascularization of an occluded SMV. Most of the research available on portomesenteric stenting involves the portal vein following liver transplantation, but many of the complications and outcomes seem to be easily transferable to the SMV. Some of the risk factors for stent occlusion include residual stenosis >30% and collateral vein formation. 8 It is proposed that embolization of the collateral veins after revascularization should be performed to help maintain stent patency by forcing more blood to flow through the stent. 8
Diagnosis of SMV thrombosis is usually done through contrast-enhanced CT but may also include the use of duplex sonography. 2 CT diagnostic findings typically include thickening of the bowel wall, narrowing of the lumen of the SMV, and associated mesenteric congestion. 3 Sonography certainly has known limitations but does have the capability for direct visualization of the vessels and allows for Doppler waveform analysis. 2 Sonographic findings of a complete occlusion include absence of color or spectral Doppler in the targeted vessel. In most instances, sonography is used in conjunction with CT and not as a stand-alone imaging technique. There is divided literature as to whether Doppler should be employed as the first diagnostic approach for imaging followed by CT or vice versa. 2 A small, nonanonymized study showed that endoscopic sonography, used in the diagnosis of portomesenteric thrombus, had a sensitivity of 81% and a specificity of 93%. While these data are commendable, CT results indicated a higher sensitivity of 90% and is therefore considered superior diagnostic option. 2
Routine follow-up for stent placement is done by contrast-enhanced CT during the portal venous phase or with duplex sonography. This is usually done 1 month postprocedure and every 3 months thereafter. 8 One study suggested grayscale sonography and color Doppler be used for surveillance of all patients, the day after stent placement, to establish patency. This same study employed the use of contrast-enhanced ultrasonography (CEUS) on a single patient which confirmed stent functionality, as the sonographic findings were inconclusive. 3
Research regarding SMV stent success is limited, but the literature supporting portal vein stent placement shows a rate of patency of 79.1% at 1 year and 65.9% at 3 years. 8 Risk factors for patients with stent failure include a prior history of venous thrombosis, hypercoagulability, and postoperative complications such as obstruction, leak, or hemorrhage. 1 Most patients are put on anticoagulation treatments for the first 2–5 days following the procedure and potentially longer, as most cases of PVT occur within the first 4 weeks after surgery.1,3
Conclusion
The prevalence of occlusive thrombus of an SMV stent is uncommon; therefore, the research on it is lacking. In the current patient case, a completely occluded SMV stent was confirmed through the use of sonography. The stent was subsequently removed and a new one placed for revascularization of the vessel. Sonography played a vital role in this situation as the CT examination did not confirm patency or occlusion of the stent. Sonography was also important in assisting in the venogram and second stent placement, as well as postprocedural surveillance. Although sonography has limitations in mesenteric vessel visualization, this case highlights the importance and effectiveness of sonography when used in conjunction with other diagnostic imaging techniques.
Footnotes
Ethics Approval
Ethical approval was not sought for this study because it was a retrospective analysis.
Informed Consent
Informed consent was not sought for this study because all case data were de-identified and/or aggregated and followed ethics committee or institutional review board (IRB) guidelines (also referred to as the honest broker system).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
