Abstract
Vitamin A deficiency is an uncommon but serious complication of malabsorptive bariatric surgery in developed nations, which can manifest several years after the procedure. It can easily be overlooked, as patients often present with nonspecific signs and symptoms early in the disease course, and ocular manifestations typically appear once the deficiency reaches a severe stage. A female patient in her early 40s presented with acute vision loss 8 years after undergoing biliopancreatic diversion with duodenal switch bariatric surgery. Ophthalmic examination revealed Bitot’s spots and progressive keratomalacia. Laboratory tests showed micronutrient deficiencies of vitamins A, D, E, and K1, which were attributed to noncompliance with bariatric follow-up and vitamin supplementation. Treatment with high-dose intramuscular vitamin A and total parenteral nutrition resulted in significant visual recovery and resolution of keratomalacia at 2 months after hospital discharge. Therefore, this case highlights that vitamin A deficiency can manifest years after the procedure, particularly in patients with poor adherence to nutritional guidelines. Prompt recognition and aggressive multi-modal supplementation are necessary to reverse ocular pathology and prevent permanent vision loss in this population.
Keywords
Introduction
Vitamin A deficiency (VAD) remains a significant global public health concern, predominantly impacting low-income countries.1–4 In developed nations, such as the United States (US), VAD is uncommon but increasingly associated with malabsorptive conditions, with bariatric surgeries emerging as a significant contributing factor.5–7 Among these procedures, biliopancreatic diversion with duodenal switch (BPD-DS) is effective for weight loss; however, it involves a lifelong risk of micronutrient deficiencies due to its impact on nutrient absorption. 8 This risk persists and can manifest years after the initial surgery, often because of inadequate adherence to lifelong nutritional monitoring and supplementation regimens. 9 This report highlights a case of bilateral keratomalacia in a patient who presented with severe VAD 8 years after BPD-DS bariatric surgery.
Case presentation
The Institutional Review Board of the University of North Carolina (UNC) at Chapel Hill does not require ethical approval for reporting individual cases (https://policies.unc.edu/TDClient/2833/Portal/KB/ArticleDet?ID=132243). All patient details have been de-identified. The reporting of this study conforms to the Case Report (CARE) guidelines. 10
A female patient in her early 40s presented with a 1-week history of burning sensation and progressive vision loss in both eyes at the UNC Kittner Eye Center. Her ocular history was notable for right-eye herpes simplex virus endotheliitis and severe dry eye syndrome, which was more prominent in the right eye. Her complex medical history included a resected astrocytoma, anemia, hyperlipidemia, hypertension, uncomplicated type 2 diabetes mellitus, fibromyalgia, anxiety and depression, systemic lupus erythematosus managed with chloroquine, and BPD-DS bariatric surgery performed in early 2016. One month prior to presentation, she sustained a head injury from a fall. A noncontrast head computed tomography (CT) without contrast revealed no evidence of acute intracranial pathology.
Upon initial ophthalmic examination, her uncorrected pinhole visual acuity was 20/400 in the right eye and 20/100 in the left eye. Slit lamp examination (SLE) of both eyes revealed a low tear lake, nasally and temporally dry conjunctiva, 4+ punctate epithelial erosions (PEEs) with 1+ corneal edema, and a nasal central corneal scar in the right eye. All other slit lamp and dilated fundus examination findings were unremarkable. She was instructed to continue aggressive lubrication with preservative-free artificial tears every hour, initiate erythromycin ointment 3–4 times daily, continue 0.05% cyclosporine ophthalmic emulsion every 12 h, and take 500 mg valacyclovir twice daily.
At her 2-week follow-up, her vision had significantly deteriorated, with visual acuity declining to hand motion in the right eye and counting fingers at 3 feet in the left eye. Confrontational visual fields demonstrated total bilateral visual field deficits. SLE revealed evidences of Bitot’s spots, severe PEEs in both eyes, and corneal haze and keratinization. These ocular manifestations were consistent with keratomalacia, which is highly suggestive of VAD (Figure 1). She was prescribed vitamin A ointment and advised to initiate a regimen of 100,000 IU of intramuscular (IM) vitamin A daily for 3 days, followed by 50,000 IU for the subsequent 2 weeks, along with oral zinc supplementation.

Slit lamp photography of the patient’s right eye during the second visit. (a) Bitot’s spot in the inferotemporal region. (b) Fluorescein staining demonstrates severe PEEs and (c) evidence of corneal haze and keratinization. PEEs: punctate epithelial erosions.
Two days later, the ophthalmology team contacted the patient telephonically and advised her to visit the UNC Rex emergency department (ED) for administration of a high-dose IM vitamin A injection to prevent impending corneal perforation. During the ED visit, the patient reported a 1-week history of blurry vision, dizziness, and progressive right lower extremity weakness. A noncontrast head CT revealed an acute/subacute cerebrovascular accident, and she was subsequently admitted to the UNC Rex Hospital at Raleigh in November 2024.
Laboratory analysis upon admission confirmed profound micronutrient deficiencies including vitamin A, total vitamin D, vitamin E, and vitamin K1 (Table 1). Continuous total parenteral nutrition (TPN) comprising vitamin and mineral additives, including vitamin A and zinc, was initiated at a rate of 75 mL/h. Over the course of her hospital stay, she also received three doses of 30,000 µg IM vitamin A palmitate, 2 mg intravenous (IV) phytonadione (vitamin K) weekly, and 50,000 IU IV cholecalciferol twice weekly for 2 weeks, followed by once weekly for 6 weeks. Subsequently, the bariatric surgery team was consulted for the management of severe nutritional deficiency. Further investigation into the patient’s medical history revealed significant noncompliance with her bariatric regimen and vitamin supplementation. She had not followed up with a bariatric surgeon since 2018 or a registered dietician since 2019.
Laboratory results on hospital admission.

A peripherally inserted central catheter line was placed, and TPN was administered through this port, maintaining the same rate. Her vision reportedly improved during her hospital stay. Per the registered dietitian’s recommendations, she was discharged with home health services for continued TPN infusions, which included additives of 1 mL trace elements, 200 mg thiamine, 3.7 mg copper, and 120 µg selenium, at a rate of 60 mL/h for at least 3 months. Additionally, she was prescribed 50,000 IU of IM vitamin A daily for 2 weeks, one capsule of 10,000 IU vitamin A palmitate daily for 60 days, one capsule of 1250 µg cholecalciferol twice a week for 1 month, and one tablet of 5 mg phytonadione once weekly for 1 month.
At the 2-week ophthalmology follow-up following hospital discharge, she reported continued vision improvement with some blurriness. Her uncorrected visual acuity was 20/100 in the right eye and 20/30 in the left eye. SLE revealed improvement in keratomalacia of the cornea and conjunctiva (Figure 2). Optical coherence tomography of the retina was unremarkable (Figure 3). At the 2-month post-discharge follow-up, she reported complete resolution of keratomalacia and corneal keratinization, with her uncorrected visual acuity recovering to 20/30 in the right eye and 20/20 in the left eye.
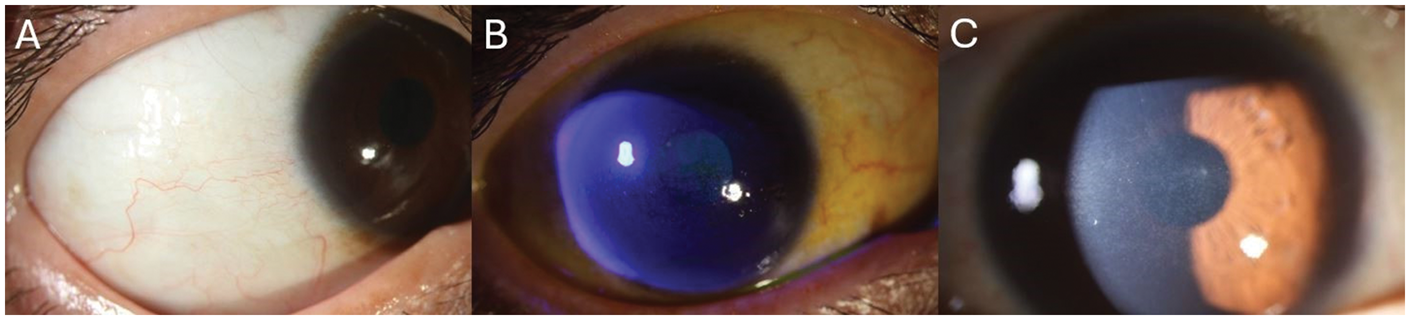
Slit lamp photography of the patient’s right eye at the 2-week post-discharge follow-up. (a) Absence of Bitot’s spot in the inferotemporal region. (b) Fluorescein staining demonstrates a decrease in PEEs and (c) decrease in corneal haze and keratinization. PEEs: punctate epithelial erosions.

Optical coherence tomography 5 line raster taken at the 2-week post-discharge follow-up. Optical coherence tomography demonstrates no abnormalities in the retinas of both eyes.
Discussion
The role of vitamin A in ocular health and vision
Vitamin A, a fat-soluble vitamin, plays a crucial role in various physiological processes, including vision, immune function, cell growth and differentiation, and reproduction. 11 In terms of vision, it helps maintain ocular surface health, facilitates the rhodopsin cycle, supports mucin expression in the conjunctiva, and promotes epithelial repair. 12 In the retina, vitamin A is essential for phototransduction, where it converts into 11-cis-retinaldehyde, a key component of rhodopsin in photoreceptor rod cells.13,14 Consequently, VAD can result in vision loss due to ocular surface drying and keratinization as well as impaired visual function. 15 Ocular manifestations of VAD include Bitot’s spots, nyctalopia, and xerophthalmia as well as keratomalacia and corneal perforation in severe cases. 16
Challenges in early diagnosis
Early detection of VAD is challenging because of its nonspecific signs. Patients may experience fatigue, which is often associated with coexisting anemia in patients with VAD. VAD impairs iron mobilization and hematopoiesis, reducing iron availability for erythropoiesis and erythrocyte progenitor cell development. 17 Dry skin can also be an indication of VAD, as vitamin A regulates skin keratin expression. 18 Additionally, VAD increases susceptibility to infections, which can further deplete vitamin A stores. 19 Diagnostic testing for VAD also presents challenges. Although serum retinol levels act as a marker of vitamin A nutritional status, they may not accurately reflect the body’s total vitamin A status until the deficiency becomes pronounced. 11 This is because the liver stores substantial vitamin A reserves, which are mobilized to maintain circulating levels until significantly depleted. 20 Therefore, a normal serum retinol level does not always rule out suboptimal vitamin A status or early deficiency. Moreover, inflammatory or infectious conditions can temporarily lower serum retinol levels, further complicating their interpretation. 19
Epidemiology
Globally, VAD is a leading cause of preventable childhood blindness and increases susceptibility to infections contributing to child morbidity and mortality, particularly in sub-Saharan Africa and South-East Asia. 21 In these developing countries, high rates of VAD are attributed to inadequate dietary intake, high infection rates, and poor maternal nutrition, often worsened by poverty, food insecurity, and limited access to healthcare. 22 Many low-income countries rely heavily on staple foods lacking in vitamin A, such as rice, wheat, and maize. Although some diets may include provitamin A carotenoids from plant-based foods, their bioavailability varies, which can lead to insufficient vitamin A intake compounded by a lack of dietary diversity.21,23,24
Although VAD remains a significant public health concern in low-income countries, it is generally uncommon in developed nations such as the US owing to abundant food resources.1–4 The Centers for Disease Control and Prevention (CDC) has previously reported VAD rates of <1% in the general US population; however, recent analyses suggest that up to 45% of the US population may have inadequate vitamin A intake, indicating a potentially suboptimal status rather than apparent clinical deficiency. 25 In developed countries, VAD is predominantly associated with underlying restrictive diets and medical conditions, with malabsorption disorders being the primary cause. Disorders impairing fat absorption (e.g. celiac disease, Crohn’s disease, cystic fibrosis, chronic pancreatitis, and bile duct obstruction) can severely limit the uptake of this fat-soluble vitamin. 26 Disorders of the liver, the body’s primary vitamin A storage organ, as well as conditions such as chronic alcoholism can also contribute to VAD. Additionally, bariatric surgery and restrictive diets, including vegan diets and selective eating, without proper supplementation can increase the risk of VAD.5–7,26,27
Bariatric surgery and VAD
Among bariatric procedures, BPD-DS is highly effective for weight loss; however, it is associated with the risk of micronutrient deficiencies. This procedure significantly reduces the absorptive surface of the small intestine, resulting in malabsorption of macronutrients and fat-soluble vitamins. 8 Studies have reported that patients undergoing BPD-DS consistently exhibit lower levels of vitamin A, calcium, and iron compared with those who undergo other types of bariatric procedures such as Roux-en-Y gastric bypass and sleeve gastrectomy. 9 Moreover, BPD-DS is increasingly recognized as a leading cause of VAD in adults in developed nations. Lifelong nutritional monitoring and supplementation are essential after BPD-DS, as deficiencies can develop years following surgery often due to noncompliance, with forgetfulness being the primary cause. However, some studies have reported that nutritional deficiencies may occur despite presumed compliance.8,11 Batar et al. investigated the nutritional status of patients in Turkey following sleeve gastrectomy at postoperative 1, 3, and 6 months. The patients demonstrated compliance with daily oral intake of tablets or chewable vitamin-mineral supplements, which included but were not limited to 20 μg vitamin D, 30 mg vitamin E, 160 μg vitamin K, 1000 μg vitamin A, and 15 mg zinc from the postoperative day 1 to month 6. However, their study demonstrated a decrease in nutrient intake at 3 months postoperatively compared with that at 1 month postoperatively: vitamin A intake decreased from 826.0 to 461.8 mg, vitamin D from 381.9 to 188.8 mg, vitamin E from 17.9 to 4.5 mg, and zinc from 10 to 5 mg. The study further highlighted the importance of nutritional monitoring and long-term follow-up. 28
Unfortunately, the significantly low serum vitamin A level (<5.0 µg/dL) in our patient underscored a severe, clinically significant deficiency, likely exacerbated by her multifactorial noncompliance with bariatric follow-up and vitamin supplementation. Notably, this patient had made many ED visits and hospital admissions since her bariatric procedure in 2016, which likely overshadowed her nutritional care.
Recommended supplementation regimens
According to the American Society for Metabolic and Bariatric Surgery Integrated Health Nutritional Guidelines for the Surgical Weight Loss Patient 2016 Update, the recommended daily vitamin supplementations for patients who have undergone specific bariatric surgery are as follows: (a) Laparoscopic adjustable gastric banding. 5000 IU vitamin A dose, 90–120 μg vitamin K, and 1200–1500 mg vitamin D; (b) Roux-en-Y gastric bypass or sleeve gastrectomy. 5000–10,000 IU vitamin A, 90–120 μg vitamin K, and 1200–1500 mg vitamin D; and (c) BPD-DS. 10,000 IU vitamin A, 300 μg vitamin K, and 1800–2400 mg vitamin D. For patients with VAD without corneal changes, a regimen of 10,000–25,000 IU vitamin A for 1–2 weeks or until clinical improvement is recommended. In cases with corneal changes, a more aggressive management with 50,000–100,000 IU IM injections daily for 3 days, followed by 50,000 IU daily for 2 weeks, is advised. For patients with vitamin D deficiency, 2000–6000 IU vitamin D3 per day or 50,000 IU vitamin D2 1–3 times weekly should be administered for repletion. For vitamin E deficiency, there is no clearly defined therapeutic dose for repletion. For vitamin K deficiency, a parenteral dose of 10 mg is recommended for acute malabsorption and 1–2 mg per day orally or 1–2 mg per week parenterally is prescribed for chronic malabsorption.29–30
Additionally, in developing countries with high rates of VAD, the World Health Organization (WHO) recommends a one-time dose of 100,000 IU for children aged 6–11 months, followed by doses of 200,000 IU every 4–6 months until the age of 5 years. 31 For at-risk pregnant women, a daily dose of 10,000 IU or a weekly dose of 25,000 IU for 12 weeks is recommended. The WHO no longer advises vitamin A supplementation for children aged <6 months or postpartum women.32–35
Efficacy of oral vs. parenteral supplementation
The effectiveness and implementation strategies for vitamin A supplementation differ between developing and developed countries owing to the varying deficiency etiologies and prevalence rates. In developed nations, particularly among patients with malabsorption, oral vitamin A supplementation can be challenging because of its fat-soluble nature and dependence on proper digestive system function. 36 In such cases, oral supplementation may be insufficient or ineffective due to impaired absorption. Therefore, IM or IV vitamin A supplementation may be necessary, as these routes bypass the digestive system and directly deliver the vitamin into the bloodstream, avoiding malabsorption issues and ensuring higher absorption and faster effect.37,38 In developing nations where VAD is endemic and affects a large proportion of children, high-dose oral vitamin A supplementation is recommended, as the primary cause is inadequate consumption of vitamin A-rich foods. 21
The importance of zinc supplementation
Zinc supplementation is crucial in patients with VAD. Zinc is essential for several aspects of vitamin A metabolism, including its absorption, transport, and utilization.39,40 Zinc is specifically required for the synthesis of retinol-binding protein, the primary carrier protein that transports vitamin A from the liver to target tissues. 41 It is also involved in the enzymatic conversion of retinol into retinal, a critical step in the visual cycle. 39 Zinc and vitamin A act synergistically, with zinc enhancing the effectiveness of vitamin A therapy. 42 Studies have shown that zinc supplementation has beneficial effects on vitamin A metabolism in malnourished children, preterm infants, and adults with alcoholic cirrhosis.43,44
Limitations
The lack of detailed information regarding the patient’s noncompliance with her bariatric follow-ups and vitamin A supplementation is a limitation of this study.
Conclusion
This case report highlights the severe ocular consequences of neglected micronutrient deficiencies following bariatric surgery, particularly for malabsorptive procedures such as BPD-DS. In our patient, severe dry eye symptoms progressed to keratomalacia and significant vision loss years after BPD-DS, underscoring the importance of long-term multidisciplinary follow-up, routine nutritional screening, and lifelong supplementation after bariatric surgery. Clinicians should maintain a high index of suspicion for VAD in patients who have undergone bariatric surgery or have malabsorptive disorders, especially if they present with ocular surface disease or unexplained vision loss. Early VAD is often overlooked because of its nonspecific signs and symptoms, making it imperative to closely monitor these patients. Prompt diagnosis and aggressive, multi-faceted supplementation are key to preventing permanent blindness and other systemic complications associated with severe multi-micronutrient deficiencies.
Footnotes
Acknowledgments
None.
Author contributions
Jaron Sanchez: chart review, literature review, writing-original draft, and writing-review and editing; Omar Nusair: writing-review and editing; Dipen Kumar: writing-review and editing; and Mohammad Soleimani: conceptualization, project administration, supervision, and writing-review and editing.
Data availability statement
The original contributions presented in the study are included in the article and further inquiries can be directed to the corresponding authors.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Funding
The authors declare that they received no funds, grants, or other support for this manuscript.
