Abstract
Objective:
This study was designed to assess the relationship between cases of vertigo and Hashimoto’s thyroiditis (HT) using vascular flow measured with vertebral Doppler ultrasonography (US).
Materials and Methods:
Patient cases that underwent vertebral Doppler US, thyroid hormone, and autoantibody tests between January 2020 and November 2021 were retrospectively reviewed. Hashimoto’s thyroiditis cases with associated vertigo were classified as the patient group, and other cases without vertigo and a known disease were the control group. A total of 184 patient cases were reviewed.
Results:
The HT patient group comprised 82 cases, and the control group comprised 102 cases. The median age was 43 years, and the interquartile range was 59 for all cases. The median total vertebral artery flow rate was 289.30 mL/min in the patient group and 278.35 mL/min in the control group, with no statistical difference (P > .05).
Conclusion:
This study is the first to compare patients with HT and vertigo and measure vertebral vascular flow using Doppler US. This study demonstrates that vertigo in HT cases may not be due to vertebrobasilar insufficiency. The vertebral flow in the cohort was normal, and vertigo may have resulted from the autoimmune system.
The thyroid gland has a regulatory effect on the endocrine system, resulting from its interaction with the hypothalamus and pituitary and has important functions due to its potential to affect various organs in the body. 1 Owing to this interaction, obesity, type II diabetes, and metabolic syndrome are associated with thyroid diseases. 2 Similar to other organ-specific autoimmune diseases like Addison’s disease, type I diabetes, adrenocorticotropic hormone deficiency, and long-term active hepatitis, autoimmune thyroid diseases are believed to be linked to other autoimmune diseases, such as rheumatoid arthritis, systemic lupus erythematosus, and systemic sclerosis. The association between autoimmune thyroid disease and other autoimmune diseases is believed to be influenced by pathophysiology and genetic predispositions shared among family members.2,3 The general population is estimated to have a 1% to 5% prevalence of autoimmune thyroid disorders, with Hashimoto’s thyroiditis (HT) being one of the most common. 4
The most typical cause of hypothyroidism is HT (approximately 50%). 5 Although deterioration in thyroid function mostly occurs as hyperthyroidism in Graves’ disease and hypothyroidism in HT, the pathophysiology of both is similar, with genetic and family factors contributing to thyroid autoimmunity.4,5 The overall population of Western countries is estimated to have a 1% to 3% prevalence of hypothyroidism. Hypothyroidism is also frequently encountered after radioiodine therapy, subtotal or total thyroidectomy, radiation to the neck, or surgery for cancer treatment. 6 Insufficient iodine use can also cause hypothyroidism, and the standard treatment is thyroid hormone replacement with levothyroxine. 7 Clinical symptoms of hypothyroidism are mainly fatigue, drowsiness, bradycardia, weight gain, and intolerance to colds. However, hypothyroidism has been recently linked to vestibular diseases, including vertigo, tinnitus, and hearing loss.6,7
Vertigo often results from central or peripheral dysfunction of the vestibular organ. Approximately one-third of persons worldwide report having balance issues or dizziness as common symptoms. 8 The primary causes of peripheral labyrinthine vertigo are associated with inner ear conditions such as benign paroxysmal positional vertigo (BPPV) or Meniere’s disease (MD), although nonlabyrinthine vertigo causes can also include neurological problems or cardiovascular illnesses.8,9 Meniere’s disease may potentially be connected to autoimmune disorders such as rheumatoid arthritis, Sjogren’s syndrome, or Cogan’s syndrome. 10 The association between HT and vertigo, the most common cause of hypothyroidism in addition to the described pathologies, has also been recently investigated.11 –15 During the inflammatory processes that develop due to HT, a response of autoreactive lymphocytes, antithyroid peroxidase (TPO), and antithyroglobulin (TG) autoantibodies may occur against other tissues, including the receptor cells of the vestibular organ in the inner ear.16,17
Only the relationship between autoimmune processes in hypothyroidism and vertigo has been explored in the literature. However, vertebrobasilar flow has not been evaluated with radiologic imaging in cases with a history of HT presenting with a diagnosis of vertigo, which is a significant limitation. This study assessed the relationship between vertigo and HT using vascular flow measured using vertebral Doppler ultrasonography (US) and the novelty within the literature.
Materials and Methods
Study Design and Participants
This study was conducted in a tertiary health care institution, and ethics committee approval was obtained beforehand (IRB: 2021/514/214/14). Written consent forms were available for all patients for vertebral Doppler US procedures. Every patient who underwent vertebral Doppler US and thyroid hormone and antibody tests between January 2020 and November 2021 was evaluated retrospectively. Patients with HT presenting with a diagnosis of vertigo were defined as the patient group, and other cases without vertigo and other diseases were the control group. The diagnosis of HT in the patient group was confirmed by thyroid US in addition to laboratory findings.
Inclusion and Exclusion Criteria
Patients with no history of previous cranial surgery or head trauma, who did not smoke or drink alcohol, who had long-term vertigo (BPPV) for at least 3 months (for the patient group), and who were ≥18 years old were included. Patients with a history of MD, diabetes, and hypertension, presence of carotid plaque or stenosis, known autoimmune diseases other than HT (for the patient group), missing measurement parameters in vertebral Doppler US, and age <18 years were excluded from the study.
Vertebral Artery Imaging and Laboratory Examinations
All Doppler US procedures for the carotid system were performed by a radiologist experienced in sonography. For each patient included in the study, the measurements of the vertebral artery (VA) included the diameters (mm) and flow volumes (mL/min) within both VAs (Figure 1). The total flow rate (mL/min) was also calculated. An Aplio 500 ultrasound equipment system (Toshiba MS, Tochigi, Japan) equipped with an 11L4 MHz linear array transducer was used for imaging and Doppler measurements. By turning the patient’s neck first to the right and then to the left, the V2 segment of the VA (C2-6 vertebrae transverse foramens) was made ready for measurement. 18 The procedures were repeated for each case in the supine position for both VAs. The total flow rate was finally obtained after measuring the right and left VA parameters (Figure 2). Regarding the laboratory findings, the anti-TPO, anti-TG, TSH, fT3, and fT4 values in the patient and control groups were noted for each case.
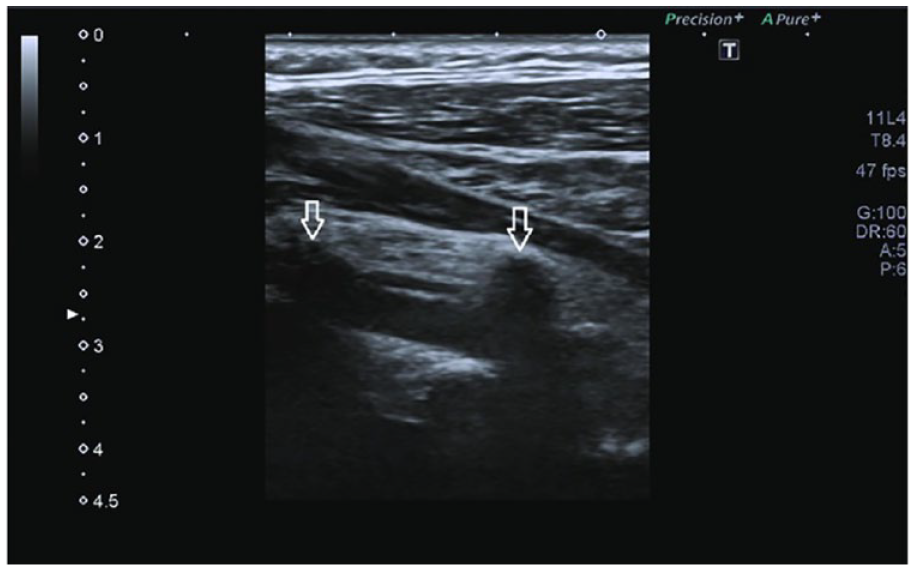
This sample sonographic image of the vertebral arteries was of a 42-year-old male in the control group. The V2 segment of the right vertebral artery is indicated with white arrows.
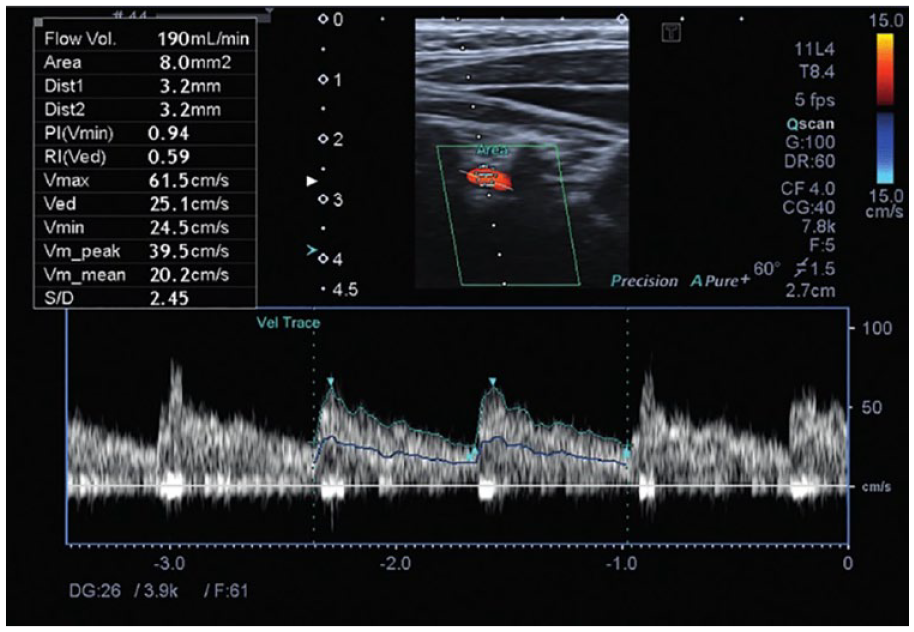
Doppler ultrasonography of the V2 segment of the left vertebral artery was performed in a 53-year-old female patient with vertigo and Hashimoto’s thyroiditis (+), and the diameter and other flow values of the vertebral artery are observed in the box in the upper left corner.
Statistical Analysis
Data analysis was processed using version 18.0 SPSS (IBM, Chicago, IL, US). The Kolmogorov-Smirnov test was used to examine data distribution. Descriptive statistical methods (minimum, maximum, median, range, frequency, and percentage) were used for the central tendency. The chi-square test of independence was used to determine the relationship between categorical data. Mann-Whitney U test was used to compare differences between sexes and laterality and identify the group causing these differences. Statistical significance was defined as P < .05 and a 95% confidence interval.
Results
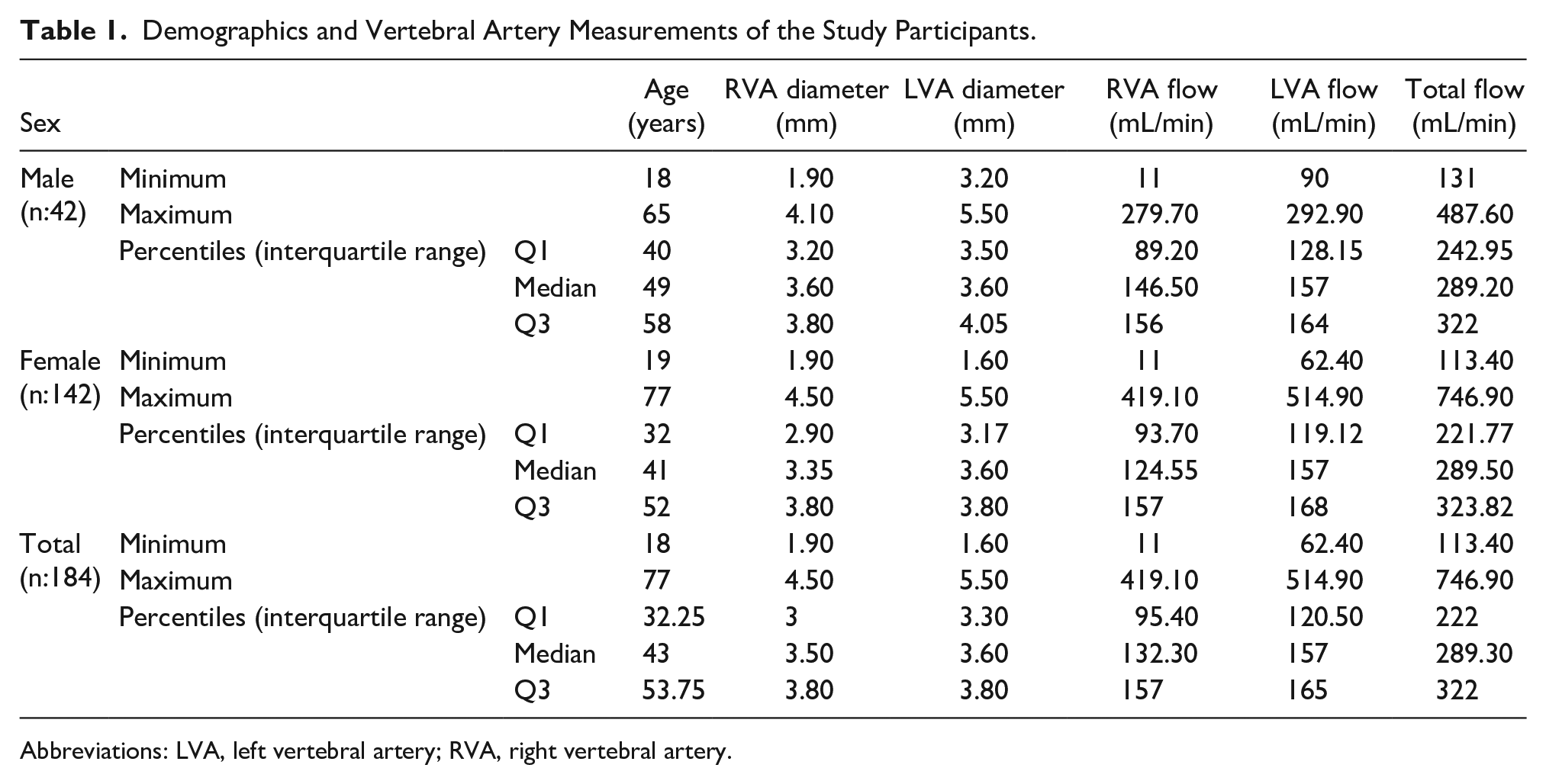
The study included 184 patient cases, 42 (22.8%) males and 142 (77.2%) females. There were 82 (44.6%) individuals in the patient group and 102 (55.4%) in the control group. The youngest patient was an 18-year-old male, and the oldest was a 77-year-old female, both in the patient group. In the control group, the youngest patient was a 19-year-old female, and the oldest was a 63-year-old male. The oldest male case was a 65-year-old in the patient group (Tables 1 and 3). The overall median age was 43 years, and the interquartile range was 59 years (Table 1). No statistical significance was found for the relationship between sex and age (P > .05).
Demographics and Vertebral Artery Measurements of the Study Participants.
Abbreviations: LVA, left vertebral artery; RVA, right vertebral artery.
Vertebral artery measurements were also calculated based on sex. The minimum, maximum, and median right vertebral artery (RVA) diameters for male cases were 1.90, 4.10, and 3.60 mm, respectively. The minimum, maximum, and median RVA diameters for female cases were 1.90, 4.50, and 3.35 mm, respectively. The minimum, maximum, and median total flow rates for males were 131, 487.60, and 289.20 mL/min, respectively. In females, the minimum, maximum, and median total flow rates were 113.40, 746.90, and 289.50 mL/min, respectively. The parameters obtained from VA measurements according to sex are presented in Table 1. There was no statistical difference when sex and VA parameters were compared (P > .05).
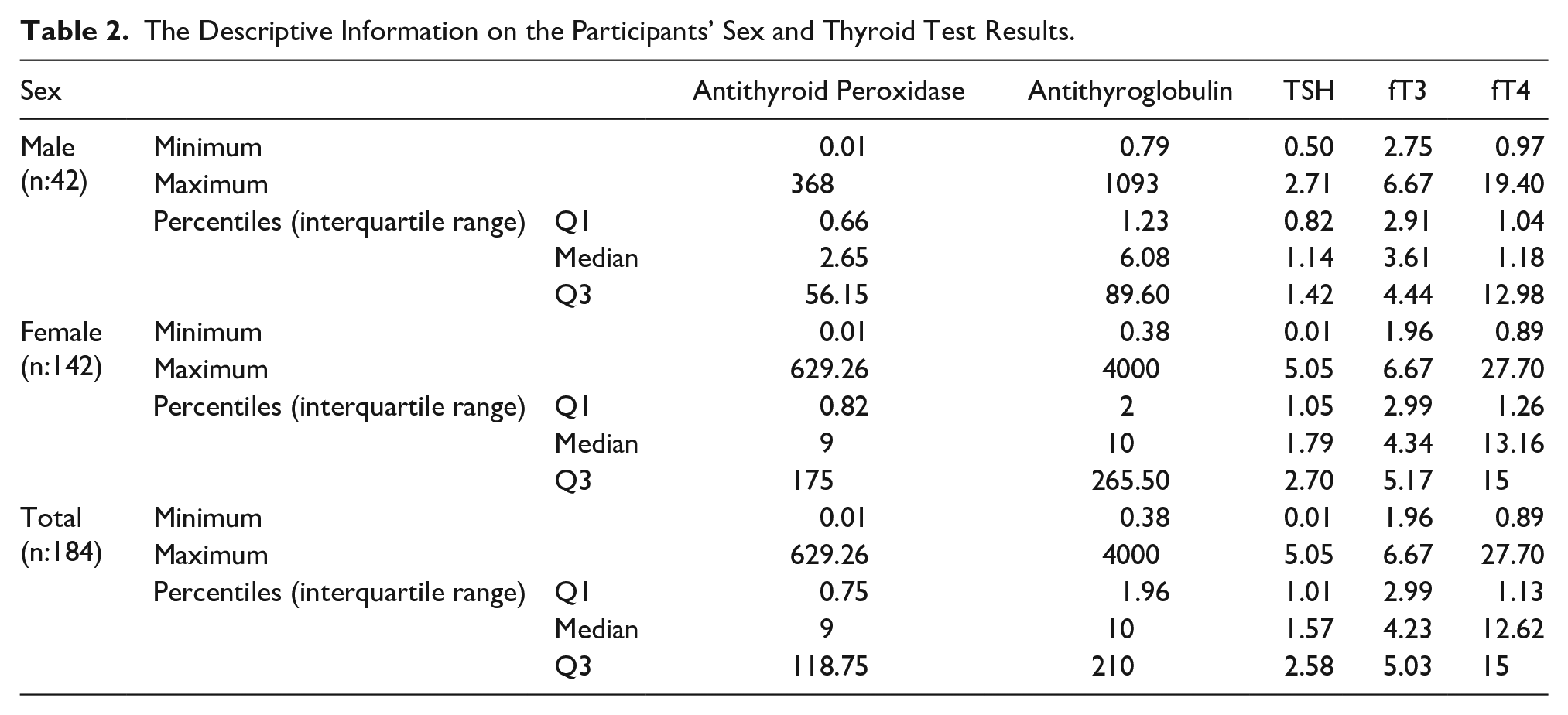
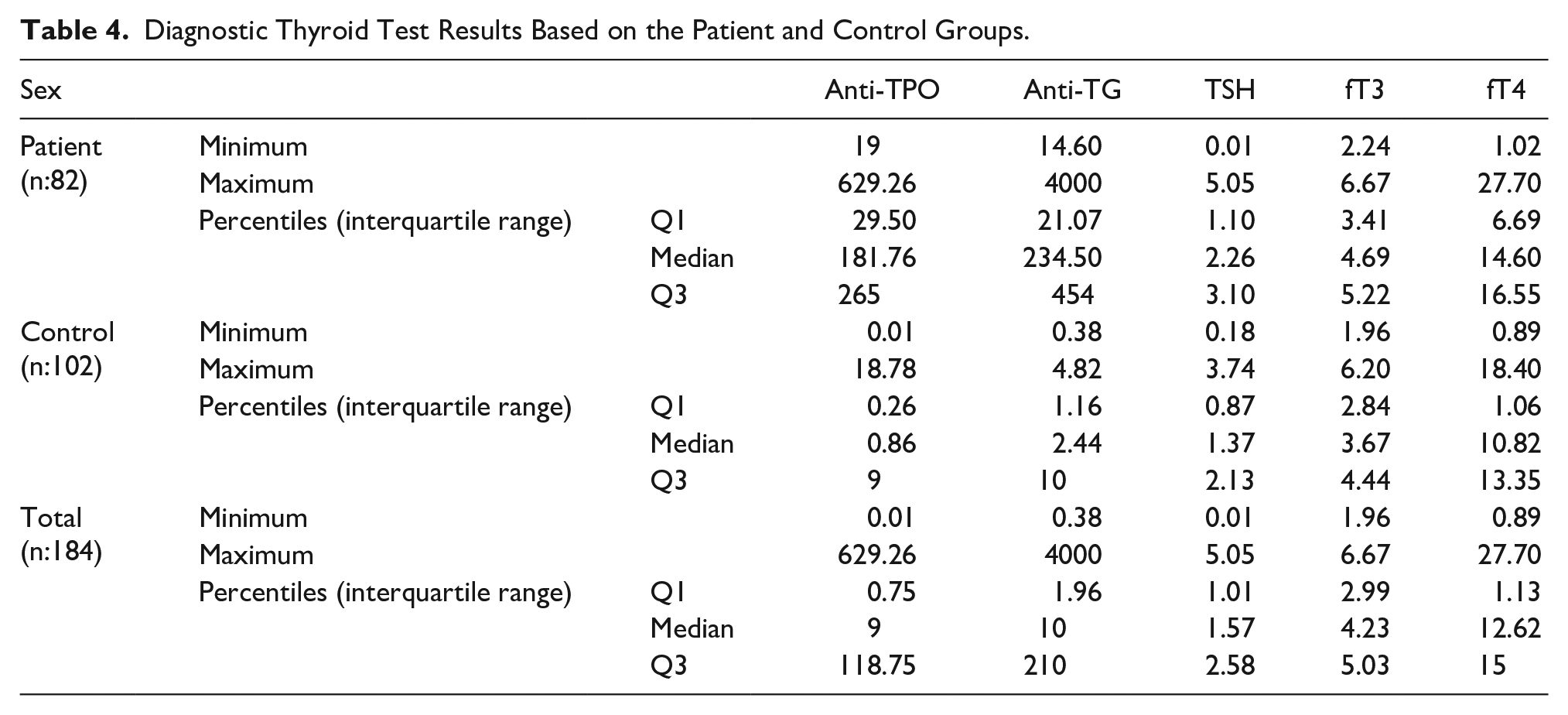
The relationship between sex, thyroid hormones, and antibodies was also examined. The minimum, maximum, and median anti-TPO levels were 0.01, 368, and 2.65 in the 42 males; 0.01, 629.26, and 9 in the 142 females; and 0.01, 629.26, and 9 in all cases, respectively. The minimum, maximum, and median anti-TG levels were 0.79, 1093, and 6.08 in the 42 males; 0.38, 4000, and 10 in the 142 females; and 0.38, 4000, and 10 in all cases, respectively (Table 2). The minimum, maximum, and median TSH values were 0.50, 2.71, and 1.14 for the males and 0.01, 5.05, and 1.79 for the females, respectively, with statistical significance (p:.005). Moreover, the minimum, maximum, and median fT4 values were 0.97, 19.40, and 1.18 for males and 0.89, 27.70, and 13.16 for females, respectively, with statistical significance (p:.035).
The Descriptive Information on the Participants’ Sex and Thyroid Test Results.
The relationship between VA measurements in the patient and control groups was also evaluated. The minimum, maximum, and median RVA diameters were 2.20, 4.10, and 3.30 mm in the patient group and 1.90, 4.50, and 3.50 mm in the control group, respectively. The minimum, maximum, and median left vertebral artery (LVA) diameters were 2.20, 4.10, and 3.30 mm in the patient group and 1.90, 4.50, and 3.50 mm in the control group, respectively. The minimum, maximum, and median total flow rates were 113.40, 746.90, and 289.30 mL/min in the patient group and 134.40, 381.50, and 278.35 mL/min in the control group, respectively (Table 3). Only the LVA diameters showed statistical significance between both groups (p:0.035). No statistical difference was detected when other VA parameters were compared among the groups (p > .05). Thyroid tests were also evaluated according to the patient and control groups (Table 4). As expected, there were statistically significant differences between the groups for all thyroid hormones and thyroid autoantibodies (p < .001).
Descriptive Measurements Within the Vertebral Artery Based on the Patient and Control Groups.
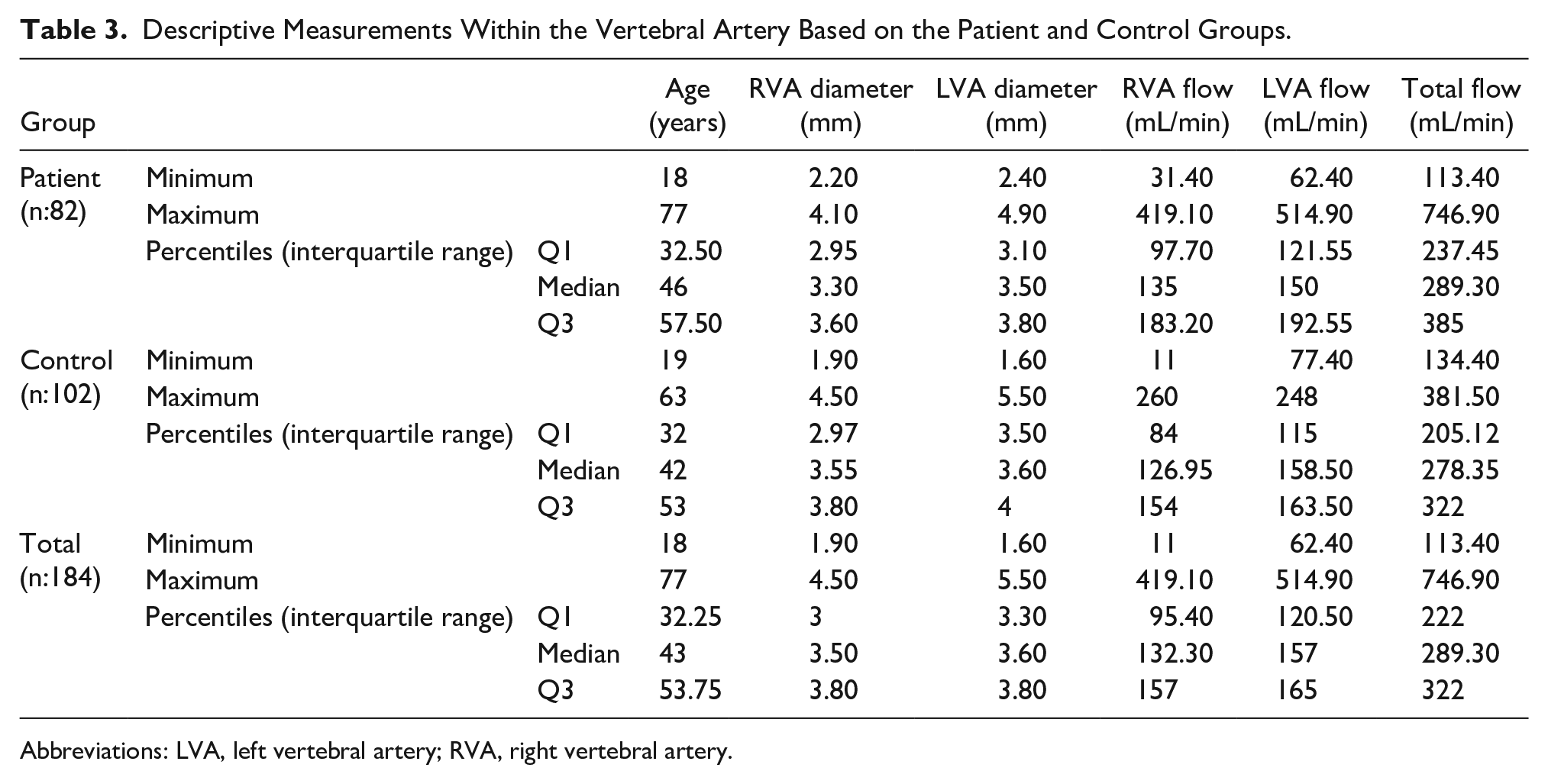
Abbreviations: LVA, left vertebral artery; RVA, right vertebral artery.
Diagnostic Thyroid Test Results Based on the Patient and Control Groups.
Discussion
Hashimoto’s thyroiditis, the most common cause of hypothyroidism, occurs as a result of autoimmune mechanisms. The production of autoantibodies against the thyroid gland results from abnormal immune system stimulation, which regards the thyroid parenchyma as foreign. 19 Hashimoto’s thyroiditis is detected by monitoring anti-TPO and anti-TG antibodies and autoreactive lymphocytes in the thyroid parenchyma. These autoantibodies play a role in primary myxedema, which causes hypothyroidism and subsequent atrophy by inhibiting the TSH receptor and the formation of cAMP, which stimulates TSH. 20 Hypothyroidism develops owing to the decreased production of hormones as a result of secondary damage to the thyroid gland.19,20
An immunological background has been demonstrated in many inner ear diseases, including MD, vestibular neuritis, and BPPV.8 –10,16,17 Inner ear inflammation and endolymphatic flow balance may be impacted by inflammatory or metabolic changes in the thyroid gland caused by various medical conditions. A relationship between inflammation and thyroid dysfunction has been established. 21 Based on inflammatory and metabolic cause, thyroid diseases have also been linked to metabolic disease leading to obesity. 22 The autoimmune process of HT may explain the inner ear disorders and vertigo that develop in this disease. 11
A few studies have evaluated the interaction between HT and vertigo.11 –14 The first study, a literature review conducted in 2017, evaluated the relationship between HT cases and vestibular dysfunction. Thyroid hormones and autoantibodies were also examined in MD and BPPV cases, and the autoantibodies were higher than normal levels. A shortcoming of the study is that there was no appropriate control group in most cases, and the effect of thyroid functional status could not be determined precisely. Patients with MD or BPPV are susceptible to HT development. Therefore, the study emphasized the importance of investigating even mild symptoms or signs associated with vestibular lesions and vertigo in patients with HT. 13 In another review article in 2020, the question of whether HT could cause vertigo was investigated. Vertigo and balance problems are prevalent health issues affecting 15% to 20% of people globally and can be caused by central or peripheral vestibular dysfunction. In otolaryngology practice, thyroid abnormalities are commonly found to be the cause of vertigo, with established evidence linking hypothyroidism to injury to the peripheral vestibular organs. Although HT is encountered in peripheral vertigo cases, especially BPPV and MD, new studies with larger patient groups are needed to evaluate the vestibular organ objectively in cases of autoimmune thyroiditis. 11
Miśkiewicz-Orczyk et al 12 evaluated vestibular organ function in 28 females with HT and long-term vertigo. Audiological evaluation, Dix–Hallpike maneuver, caloric test, and kinetic tests were performed in all cases, and normal hearing was observed in 15 cases. Nineteen cases had a history of BPPV, seven had a history of MD, and two had a history of vestibular neuritis. The excitability of the labyrinth in the vestibular organ was reduced in 15 cases. Thyroid function test findings and vestibular organ evaluation did not correlate with age, weight, height, body mass index (BMI), or thyroid autoantibodies despite being elevated in all patients. In addition, the negative effect of the increase in thyroid antibodies on abnormal results of the caloric test was not confirmed. Miśkiewicz-Orczyk et al 14 also performed head impulse testing and cervical vestibular-evoked myogenic potentials (cVEMPs) in the same group of patients with HT. Similar to the previous study, the effect of the increase in thyroid hormone levels and antithyroid antibodies on cVEMP results could not be confirmed.
Another study investigated the relationship between the risk of BPPV recurrence and hypothyroidism treated with hormone replacement therapy (HRT). 15 In a large series of 797 patients with idiopathic BPPV, 250 had recurrent BPPV. Sixty-one patients in the recurrent BPPV group were administered long-term HRT due to hypothyroidism, and HT was associated with recurrence (P < .001). The risk of BPPV recurrence is increased in patients with hypothyroidism on HRT, and this risk is especially strong for patients with HT and positive thyroid antibodies. Fattori et al 17 focused on the relationship between MD and thyroid autoimmunity. Healthy volunteers and patients with MD were compared, and thyroid hormone levels and autoantibodies were examined. Thyroid autoantibodies were significantly higher in the MD group, indicating a significant relationship between thyroid autoimmunity. These data also support that an autoimmune factor plays a role in the etiopathogenesis of the disease.
This study contains significant data and advantages compared to previous ones.11 –15 For the first time in the literature, HT cases with vertigo were evaluated for vertebrobasilar flow using Doppler US. Hashimoto’s thyroiditis cases were compared with controls to determine whether vertigo was due to vertebrobasilar insufficiency. In previous studies, the relationship between thyroid autoimmunity and vertigo was not evaluated from this perspective using Doppler US.14,15,17 This study demonstrates that vertigo in HT cases is not due to vertebrobasilar insufficiency. The usability and effectiveness of VA Doppler US for vertigo in such patients were also demonstrated.
Limitations
The major limitation was the research design and the use of a convenient sample of patients. These limitations result in threats to internal and external validity. The next limitation is the relatively small number of patients (N = 82) cases. Another limitation is the relatively small number of male cases (N = 42). Furthermore, only two groups were compared, and no comparison was made with other vertigo disease groups. The final limitation is that there is no previous study on HT cases with vertigo to make comparisons. Therefore, VA parameters and measurements were interpreted only by comparing the patient and control groups. The results are not generalizable due to these limitations.
Conclusion
The relationship between HT and vertigo continues to attract the attention of clinicians and researchers. In this study, HT cases with vertigo were compared with controls and evaluated using Doppler US in terms of vertebral flow amount. Vertebrobasilar insufficiency was ruled out in the measurements; thus, the possibility of vertigo caused by thyroid autoantibodies was further strengthened. Despite these valuable findings, new studies with higher case numbers and different groups are needed to explore these potential associations further.
Footnotes
Ethics Approval
Ethical approval for this study was obtained from Istanbul Kartal Dr. Lutfi Kirdar City Hospital Ethics Committee (IRB:2021/514/214/14).
Informed Consent
All patients included in this study gave written informed consent to participate in this research. Because human data were available, the Declaration of Helsinki was adhered to throughout the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
