Abstract
Thyroid hormone resistance syndrome (THRS) is a rare disease characterized by reduced sensitivity to thyroid hormones. Mutations in the thyroid hormone receptor beta (THRB) gene are considered as contributing to the pathogenesis. This report describes a Chinese pedigree with THRS and Hashimoto’s thyroiditis (HT) due to novel point mutation in the 11th exon of the THRB gene (c. 1378 G > A). The proband complained of goitre with increased thyroid hormone and normal thyroid stimulating hormone levels. Gene sequencing was performed to confirm the diagnosis. HT was also diagnosed based on positive thyroid autoantibodies and diffuse, grid-like changes in the thyroid on ultrasound examination. Additionally, a comprehensive examination of the proband’s pedigree was conducted. The patient’s father exhibited the same gene mutation site and was diagnosed with THRS and HT. No mutation site was detected in three patients with HT only and three healthy volunteers. Thus, gene sequencing should be considered the gold standard for diagnosing THRS. Furthermore, treatment should be individualized to control the patient’s symptoms rather than normalizing thyroid hormone levels. Further studies that determine the relationship between THRS and TH are warranted.
Introduction
Thyroid hormone resistance syndrome (THRS) is characterized by reduced sensitivity of target organs or tissues to thyroid hormones. 1 The patient’s thyroid function is typically characterized by increased levels of circulating thyroid hormones, while the serum concentration of thyroid stimulating hormone (TSH) remains normal or slightly elevated.2,3 Most patients might simply exhibit elevated blood thyroid hormone levels with no obvious clinical manifestations.2,4 However, others potentially exhibit symptoms of hyperthyroidism, hypothyroidism or non-toxic goitre. 4 It is a rare disease with an incidence of 1:40 000 to 1:50 000. 5 This disease has a genetic predisposition, but a few cases are sporadic. The main cause of this disease is thyroid hormone receptor deficiency due to genetic mutations, which can be autosomal dominant or occasionally appear as recessive inheritance. 5 Since resistance to thyroid hormones was initially observed in 1967, 6 more than 1000 cases originating from over 370 families have been reported. 7 Most cases (approximately 80–90%) are caused by mutations in the thyroid hormone receptor beta (THRB) gene on chromosome 3, 7 while other cases are caused by mutations in the thyroid receptor α gene located on chromosome 12 or genes related to the transport or metabolism of thyroid hormones. 4 Research has demonstrated that the mutation sites of the THRB gene are predominantly located on exons 7–10. 8 This current case report presents a paediatric female patient with THRS harbouring a novel mutation site on exon 11 of the THRB gene and concomitant Hashimoto’s thyroiditis (HT). The current literature on the relationship between THRB and autoimmune thyroid disease has also been reviewed.
Case report
Proband
On 25 August 2017, the proband (III:1), a 10-year-old Chinese girl, was admitted to the Department of Endocrinology, Beijing Luhe Hospital Affiliated to Capital Medical University, Beijing, China, for goitre. Six months prior, she visited the doctor because her neck had thickened. Thyroid function tests were performed and the results were as follows: elevated total triiodothyronine (TT3, 2.59 ng/ml; normal range 0.61–1.77 ng/ml); elevated total thyroxine (TT4, 20.00 ug/dl; normal range 5.13–14.06 ug/dl); elevated free triiodothyronine (FT3, 6.55 pg/ml; normal range 2.02–4.33 pg/ml); elevated free thyroxine (FT4, 2.37 ng/dl; normal range 0.93–1.71 ng/dl); and a normal TSH level (4.0 µIU/ml; normal range 0.27–4.2 µIU/ml) (visit 1; Table 1). Thyroid autoantibody tests showed that the levels of thyroid peroxidase antibody (>1300 U/ml; normal range ≤34 U/ml) and thyroglobulin antibody (248 U/ml; normal range ≤115 U/ml) exceeded the normal range. No specific treatment was administered to the patient. Since then, the patient’s mother had observed a gradual enlargement of her daughter’s thyroid. Through careful medical history inquiry and comprehensive physical examination, this patient was found to have no clinical manifestations of hyperthyroidism or hypothyroidism, such as tachycardia or palpitations, heat intolerance, excessive sweating, fatigue, tremors, sensitivity to cold or unusual bowel habits. The patient’s body weight did not change significantly during the first half of the year. Additionally, the girl was born full-term, weighing 2.5 kg, with no history of radiation exposure or a family history of thyroid disease. Physical examination revealed the following: body temperature, 36.3 °C; pulse, 100 bpm; blood pressure, 105/70 mmHg; height, 138.6 cm; weight, 37.7 kg; normal development, vision, and hearing; a diffuse goitre with no palpable nodules, tenderness, tremor or vascular murmur; and no hand shaking or oedema in the lower extremities. A thyroid ultrasound revealed an enlarged thyroid volume and diffuse changes without thyroid nodules (left lobe, 3.9 × 1.4 × 1.2 cm; right lobe, 4.4 × 1.5 × 1.5 cm; isthmus, 0.3 cm). Thyroid echoes were neither even nor reticular. Routine blood and urine tests, blood glucose levels, blood lipid profiles and liver and kidney function were all within normal ranges. Gonadotropin hormone level was normal. The bone age was the same as the chronological age (Figure 1a). No occupying lesions were detected on the hypothalamic-pituitary magnetic resonance image (Figure 1b). Based on the clinical features and laboratory tests, THRS with concomitant HT was highly suspected in this patient. Gene sequencing was performed to confirm this hypothesis. Sequencing revealed a novel point mutation on the 11th exon of the THRB gene (c. 1378 G > A). This mutation causes the conversion of glutamic acid to lysine at position 460 (p. Glu460Lys). Thus, the patient was diagnosed with THRS based on genetic analysis and no medication was administered. After obtaining consent from her parents to treatment, 25 µg levothyroxine sodium was administered orally per day for 1.5 months to control goitre. During a follow-up visit (visit 2; Table 1), thyroid function was stable with no evidence of worsening thyrotoxicosis. There was no significant change in the goitre.
Thyroid function and thyroid autoantibody test results of the proband, a 10-year-old Chinese girl admitted for goitre.
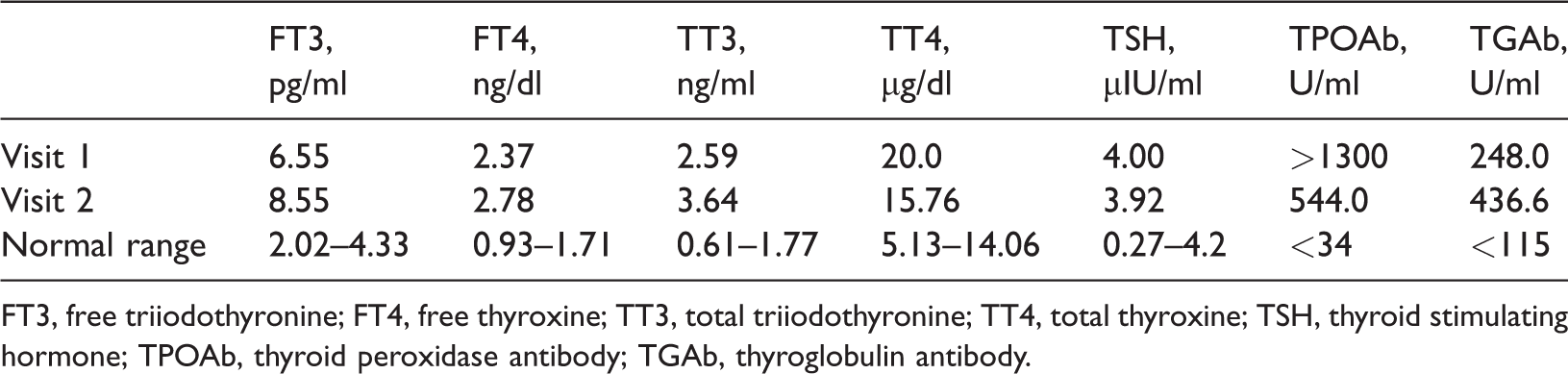
FT3, free triiodothyronine; FT4, free thyroxine; TT3, total triiodothyronine; TT4, total thyroxine; TSH, thyroid stimulating hormone; TPOAb, thyroid peroxidase antibody; TGAb, thyroglobulin antibody.

(a) X-ray imaging of the proband (III:1), a 10-year-old Chinese girl admitted for goitre, showing the that the bone age was the same as the chronological age based on the wrist anterior segment; (b) magnetic resonance imaging of the pituitary of the proband; (c) the proband’s family pedigree and (d) partial sequencing results for the proband and her father for exon 11 in the thyroid hormone receptor beta (THRB) gene. The red arrows indicate the mutation site. The colour version of this figure is available at: http://imr.sagepub.com. The colour version of this figure is available at: http://imr.sagepub.com.
Two kindreds
Patients with thyroid hormone resistance typically possess a family history of the disease. Therefore, the proband’s pedigree was comprehensively examined. Three generations of the pedigree are presented in Figure 1c. The proband’s grandmother had passed away and her parents had no siblings. The family exhibited no history of consanguineous marriages. Neither the proband’s parents nor grandfather had a history or symptoms of thyroid dysfunction. Thyroid function tests revealed that only the father (II:1) had an abnormal thyroid function. The results indicated increases in serum FT3, TT3 and FT4 levels, along with positive thyroid autoantibodies, while TSH levels remained normal (Table 2). Thyroid ultrasound examination revealed diffuse, grid-like changes in the proband’s father and normal changes in her mother and grandfather. Based on the same thyroid function, thyroid antibody, and thyroid ultrasound results as those of the proband, gene sequencing was performed on the father, with results revealing the same point mutation on the 11th exon of the THRB gene (c. 1378 G > A) (Figure 1d). To rule out the possibility of this gene mutation appearing in patients with HT or in healthy individuals, gene sequencing was also performed on three patients with HT and three healthy volunteers; and no gene mutations were detected.
Thyroid function and thyroid autoantibody test results of the proband’s family members.

FT3, free triiodothyronine; FT4, free thyroxine; TT3, total triiodothyronine; TT4, total thyroxine; TSH, thyroid stimulating hormone; TPOAb, thyroid peroxidase antibody; TGAb, thyroglobulin antibody.
This study was approved by the Ethics Committee of Capital Medical University, affiliated with Beijing Luhe Hospital (no. 2016L-H-KS-08[ST]). Verbal informed consent was obtained from all study participants for publication of this report. The reporting of this case and family pedigree conforms to the CARE guidelines. 9
Discussion
Thyroid hormone resistance syndrome is a disorder characterized by a suppressed response to thyroid hormones due to mutations in the thyroid hormone receptor gene. 1 There are two subtypes of human thyroid hormone receptors: THRα and THRβ.10,11 Each subtype has different isoforms, such as THRα1, THRα2, THRβ1, THRβ2 and THRβ3.10,12 These receptors possess specific organ distribution characteristics.8,11 Additionally, genetic defects involving TH cell transport and metabolism have been reported, broadening the understanding of impaired TH sensitivity. 2 All these features determine the diversity of the clinical manifestations of THRS. Based on clinical manifestations, THRS can be divided into the following three categories: global resistance (GRTH), pituitary resistance (PRTH) and peripheral resistance (PrRTH) to thyroid hormones. 8 GRTH, which affects the majority of patients, has no distinctive manifestations, except for goitre. 3 Patients with PRTH predominantly experience mild-to-moderate hyperthyroidism without ophthalmopathy or pretibial myxoedema. 3 PRTH potentially causes symptoms or signs of hypothyroidism. 3 The multiplicity of symptoms without a typical model renders it difficult to diagnose this disease.3,8,11 To date, gene sequencing is considered the gold standard for diagnosing THRS.8,11
To date, numerous mutations have been identified.8,11 Of these mutations, 85% are located in the THRB gene, which comprises 10 exons. 3 Most studies on THRB gene mutations have revealed that these mutations often occur in three hotspot regions between exons 7 and 10;13–16 that is, codons 234–282, 310–353 and 429–461. Only a few cases have presented mutation sites at codons 384–425, which constitutes the ‘cold region’. 17 Point mutations are prevalent, causing missense mutations, while insertion and deletion mutations are relatively rare.8,11 In this current report, the proband exhibited no distinctive manifestations, except for goitre. Thyroid function tests revealed increased levels of circulating thyroid hormones, with normal TSH concentrations. Gene sequencing revealed a novel point mutation on the 11th exon of the THRB gene (c. 1378 G > A). A comprehensive examination of the proband’s pedigree was conducted and the patient’s father was found to have the same thyroid function and gene mutation site. Thus, the patient’s father was diagnosed with THRS due to a new point mutation on the 11th exon of the THRB gene (c. 1378 G > A). In the patient’s father with THRS, elevated thyroid hormones potentially compensated for hormone resistance to a certain degree and the patient presented no clinical manifestations, except for goitre, and no treatment was administered to the father.
In this current pedigree report, both the proband and her father were found to be positive for thyroglobulin and thyroid peroxidase antibodies. In early 2008, a causal link between thyroid hormone resistance and primary autoimmune hypothyroidism was proposed. 18 The authors claimed that chronic TSH elevation in thyroid hormone resistance stimulates lymphocytes to produce the proinflammatory cytokine tumour necrosis factor-α, which mediates thyroid cell destruction by binding to its receptors on thyrocytes, or indirectly, by potentiating antibody formation. 18 Increased thyroid hormones may promote a high inflammatory burden. 19 In contrast, HT is considered to be an autoimmune inflammatory disorder, which has been linked with inflammation.20–22 Thus, the inflammation might explain the coincidence of THRS and HT. Although the diagnosis of HT in patients with THRS has been reported in a number of cases,11,23,24 whether HT is more prevalent in THRS 25 or whether it merely coexists in the same individual by coincidence remains debatable. 26 Current research has demonstrated that both HT and THRS exhibit certain genetic tendencies. 27 Considering the above theory and this current pedigree report, we believe that THRS increases the risk of HT. Thus, in the future, focus should be directed on the incidence of HT in patients with THRS. In addition, its pathogenesis needs to be determined.
In conclusion, THRS has no specific clinical features. Most patients with THRS exhibit normal growth and present only with goitre and high TH levels, rendering the diagnosis of this disease difficult. Gene sequencing is considered the gold standard for diagnosing THRS. Furthermore, treatment should be individualized to control the patient’s symptoms rather than normalizing elevated TH levels. The advances in our knowledge about THRS raise novel questions regarding the susceptibility of patients with THRS to TH. Further studies that explore the relationship between these two diseases in terms of molecular and genetic pathogenesis are warranted.
Footnotes
Author contributions
Z.Y.X. and X.H.W. designed the study. C.F.L. and Y.X.A. collected the data. Z.Y.X. analysed the data and wrote the case report. D.Z. and X.H.W. contributed to the discussion of the results and review of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
