Abstract
Objective:
To assess the correlation between fetal umbilical artery (UA) and middle cerebral artery (MCA) Doppler indices in a cohort of preeclamptic and normotensive pregnancies.
Materials and Methods:
This cross-sectional analytical study recruited 70 pregnant women (28–40 weeks of gestation) using a non-probability convenient technique. The study included 33 patients with preeclampsia and 37 normotensive participants.
Results:
In the normotensive group, a significant (P = .00) strong correlation (r = 0.72) was observed between UA velocity, systolic/diastolic (S/D) ratio, and pulsatility index (PI), and MCA velocity; a significant (P = .00) correlation (r = 0.56) between UA, S/D ratio, and the MCA resistive index (RI); a significant (P = .00) moderate correlation (r = 0.66) between S/D ratio, UA velocity, and MCA velocity; a significant (P = .00) negative correlation (r = −0.50) between UA velocity, RI, and MCA PI; a significant (P = .01) negative correlation (r = −0.44) between UA velocity, RI, and MCA S/D ratio. In contrast, in the preeclamptic group, we observed a substantial (P = .01) moderate correlation (r = 0.43) between UA, PI, and MCA velocity; a significant (P = .04) correlation (r = 0.368) between UA, RI, and MCA PI; a significant (P = .05) moderate correlation (r = 0.35) between UA, PI, and MCA RI; a significant (P = .00) strong correlation (r = 0.83) between UA S/D ratio UA and MCA velocity.
Conclusion:
This study suggests potential diagnostic value in Doppler measurements for assessing preeclampsia-related vascular changes. However, caution is warranted owing to a split sample and limited statistical power, necessitating further research for validation.
Keywords
Preeclampsia is a significant global health concern, affecting 2% to 8% of pregnancies and leading to maternal and fetal morbidity and mortality. 1 Various risk factors, including maternal age, body mass index (BMI), pre-existing health conditions, chronic renal disease, or preeclampsia in a previous pregnancy, can influence the prevalence of hypertension during pregnancy. 2 Pakistan is the sixth most populous country with the third highest rates of maternal, fetal, and child mortality, which poses considerable challenges in managing these conditions. 3
Oxidative stress and inflammation contribute to preeclampsia development by reducing blood flow and oxygen supply to the fetus. 4 Preeclampsia symptoms include proteinuria, edema, headaches, vision changes, and epigastric pain. 5 Routine prenatal care and blood pressure (BP) monitoring are crucial for early diagnosis and management. 6 Risk factors for preeclampsia include being a first-time mother, advanced maternal age, weight gain during pregnancy, a history of preeclampsia, overweight, or a family history of preeclampsia or hypertension. 7
Duplex sonography can evaluate blood flow anomalies in the umbilical artery (UA) and middle cerebral artery (MCA), which can be indicative of fetal distress. 8 It can also assess the placental and fetal blood flow in high-risk pregnancies, especially those with a diagnosis of preeclampsia, fetal growth restriction (FGR), or multiple gestations. 9 Doppler indices, such as the resistive index (RI) and pulsatility index (PI), can provide valuable information about blood flow velocity and resistance. Abnormal velocity indices may indicate fetal anemia, hypoxia, acidosis, or placental insufficiency. 10
Monitoring Doppler indices allows for the identification of fetuses at risk of adverse outcomes and aids in making clinical decisions about timing and mode of delivery. In cases of impaired pregnancies, early delivery may be necessary to prevent fetal mortality or irreversible impairments, such as cerebral palsy. 11 Duplex sonography and Doppler indices have become essential in assessing fetal health, particularly in high-risk pregnancies. Adequate antenatal care can help reduce the risk of iatrogenic premature birth. 12
This study aimed to correlate fetal UA with MCA in patients with normal BP and preeclampsia and develop more effective prevention, early detection, and maternal-fetal management strategies.
Materials and Methods
This cross-sectional analytical study was conducted at a university clinic in a village within Lahore. The study spanned 9 months after receiving approval from the Ethical Review Committee of the University of Lahore (IRB70034851). A non-probability convenient sampling technique was used. The study included 70 patients, with 37 assigned to the normotensive group and 33 to the preeclamptic group.
The normotensive group comprised pregnant women aged 18 to 40 with singleton pregnancies, normal BP (120/80 mm Hg), gestational age between 28 and 40 weeks, and the ability to record fetal UA and MCA Doppler parameters. The preeclamptic group comprised pregnant women aged 18 to 40 with singleton pregnancies, abnormal BP (140/90 mm Hg), gestational age between 28 and 40 weeks, and the ability to record fetal UA and MCA Doppler parameters. Patients with fetal congenital abnormalities or abnormal placenta, such as placenta previa, placental abruption, or any other significant structural anomalies and those with uncertain dates that could affect the interpretation of fetal Doppler parameters were excluded from the study. Abnormal values were defined as systolic/diastolic (S/D) ratio, PI, and RI > 2 standard deviations (SDs) for a specific gestational age.
Data Analysis
Patient data were analyzed using SPSS (IBM corp. Armonk, New York) software version 27.0. Continuous variables were expressed as mean ± SD. Categorical variables were expressed as frequencies and percentages. The Shapiro-Wilk test was used to test the normality of continuous data. Pearson’s correlation coefficient was used to assess the correlation between UA and MCA Doppler velocity indices. Statistical significance was set at a P value of < .05.
Results
Of the 70 participants, 37 (52.9%) were categorized into the normotensive group, whereas the remaining 33 (47.1%) were classified as the preeclamptic group. Among the participants, 5 (7.1%) had a history of preeclampsia, 35 (50%) had a low-resistance velocity waveform, and 35 (50%) had a high-resistance velocity waveform.
In the normotensive group, the mean age was 26 ± 6.1 years, with a mean BMI of 33.69 ± 2.43, a mean systolic BP of 111.24 ± 11.57, a mean diastolic BP of 72.68 ± 8.59 mm Hg, and a mean gestational age of 33.69 ± 2.43 weeks. In the preeclamptic group, the mean age was 28.97 ± 5.1 years, with a mean BMI of 27.94 ± 2.42, a mean systolic BP of 149.55 ± 5.64 mm Hg, a mean diastolic BP of 96.70 ± 6.22, and a mean gestational age of 33.01 ± 3.01 weeks (Table 1; Figures 1 and 2).
The Descriptive Statistics of the Study Cohort’s Normotensive and Preeclamptic Patients.
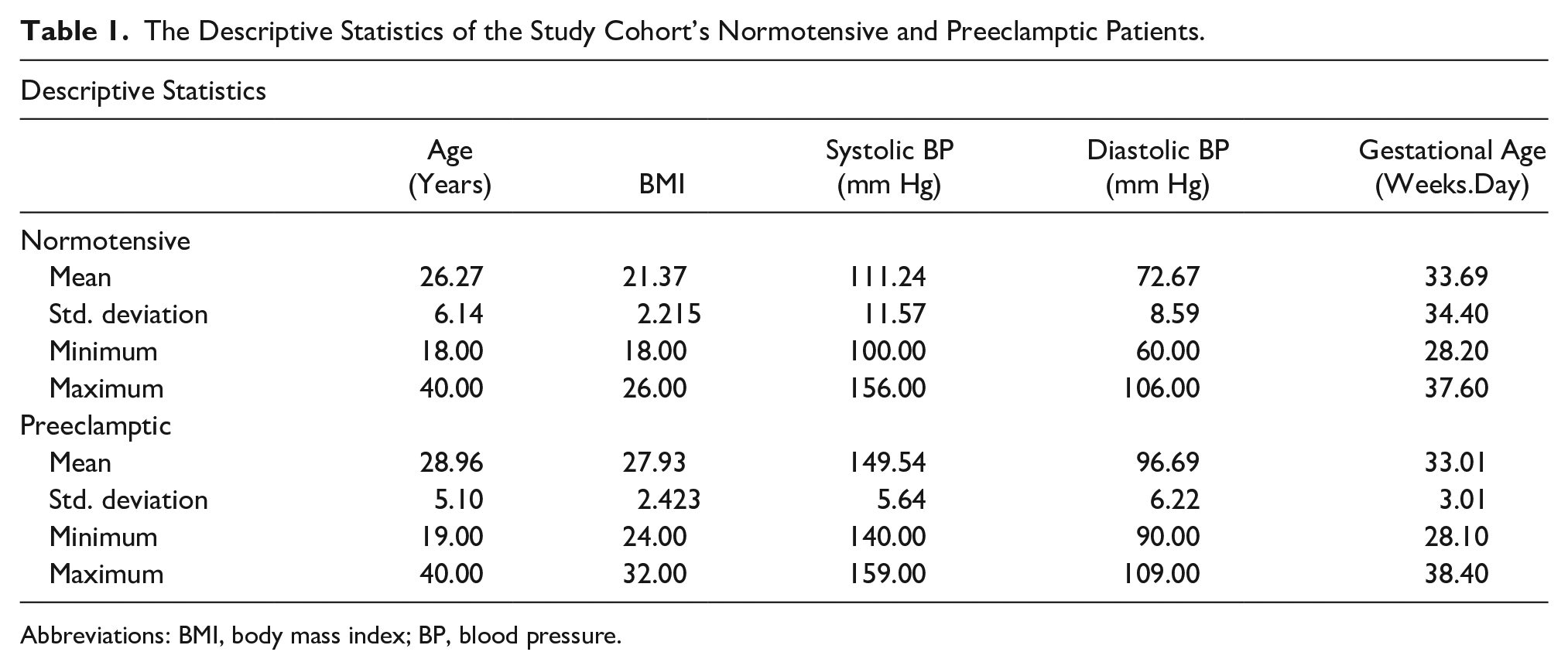
Abbreviations: BMI, body mass index; BP, blood pressure.
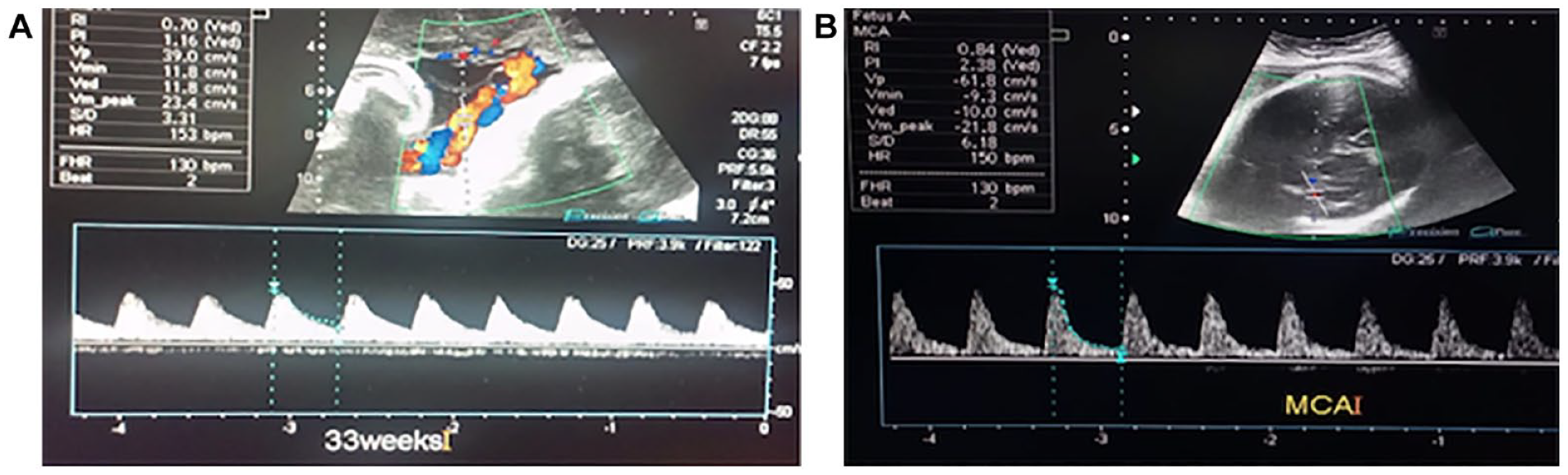
A 26-year normotensive pregnant patient with a single viable fetus, at a gestational age of 33 weeks and 2 days (A) Umbilical artery Doppler showing forward flow with a good diastolic component with PI = 1.16, RI = 0.70, and S/D ratio = 3.31. (B) Middle cerebral artery Doppler showing typical spectrum with no evidence of brain sparing with PI = 2.28, RI = 0.84, and S/D ratio = 6.18. PI, pulsatility index; RI, resistive index; S/D, systolic/diastolic.
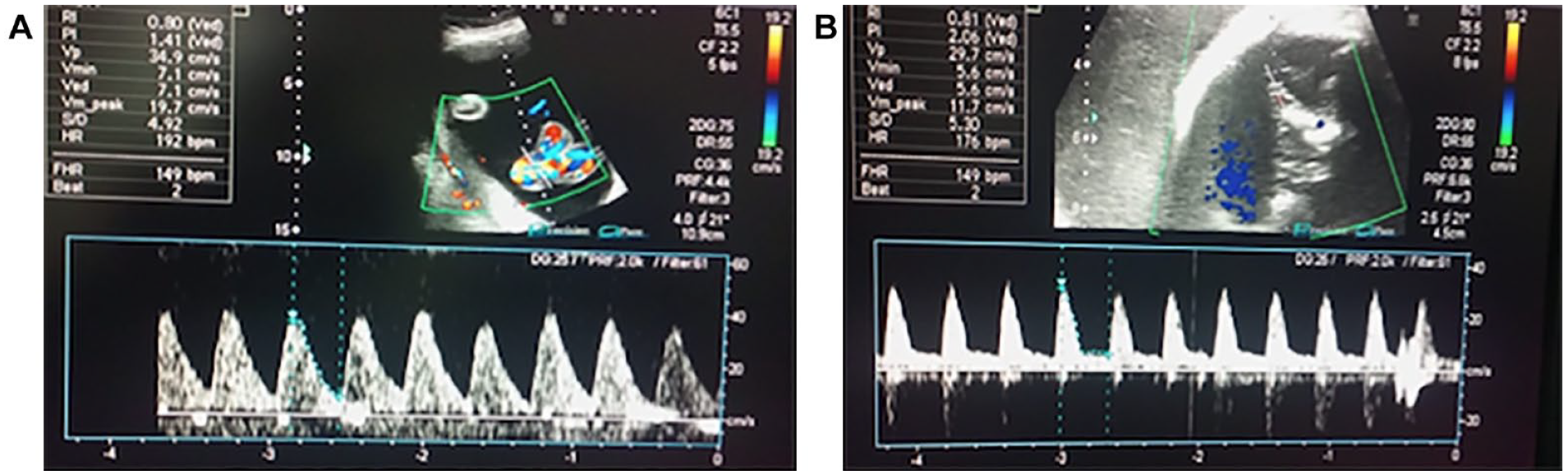
A 30-year preeclamptic multiparous pregnant patient with a single viable fetus at a gestational age of 31 weeks 2 days—(A) Umbilical artery Doppler showing high-resistance flow with a PI = 1.41, RI = 0.80, and S/D ratio = 4.92. (B) Middle cerebral artery shows high-resistance flow with a PI = 2.06, RI = 0.81, and S/D ratio = 5.30. PI, pulsatility index; RI, resistive index; S/D, systolic/diastolic.
The mean UA PI UA was 1.29 ± 0.19, with a UA RI of 0.82 ± 0.09, a UA S/D ratio of 2.37 ± 0.45, an MCA PI of 1.34 ± 0.36, an MCA RI of 0.74 ± 0.06, an MCA S/D ratio of 2.95 ± 1.55, and a mean cerebroplacental ratio (CPR) of 0.88 ± 0.16.
The preeclamptic group had a UA PI of 1.28, a UA RI of 0.67, a UA S/D ratio UA of 2.05, an MCA PI of 1.59, an MCA RI of 0.72, an MCA S/D ratio of 2.52, and a mean CPR of 1.26 ± 0.41.
In the normotensive group, the mean UA PI was 1.29 ± 0.19, the mean UA RI was 0.81 ± 0.09, the mean UA S/D ratio was 2.37 ± 0.45, the mean MCA PI was 1.34 ± 0.36, the mean RI was 0.74 ± 0.06, and the mean S/D ratio of 2.95 ± 1.55. Significant correlations were observed between UA and MCA Doppler velocity indices. We observed a significant (P = .00) strong positive correlation (r = 0.72) between UA S/D ratio and MCA PI, a significant (P = .00) positive correlation (r = 0.56) between UA S/D ratio and MCA RI, a significant (P = .00) moderate positive correlation (r = 0.66) between the UA and MCA S/D ratios, a significant (P = .00) negative correlation (r = −0.50) between the UA RI and MCA PI, and a significant (P = .01) negative correlation (r = −0.441) between the UA RI and MCA S/D ratios. These correlations suggest a relationship between the blood flow velocities in the UA and MCA of this fetal cohort and potential fetal well-being indicators (Table 2).
The Associations Measured Between the Umbilical Artery and Middle Cerebral Artery Doppler Indices, Within the Normotensive Patient Group (N = 37).
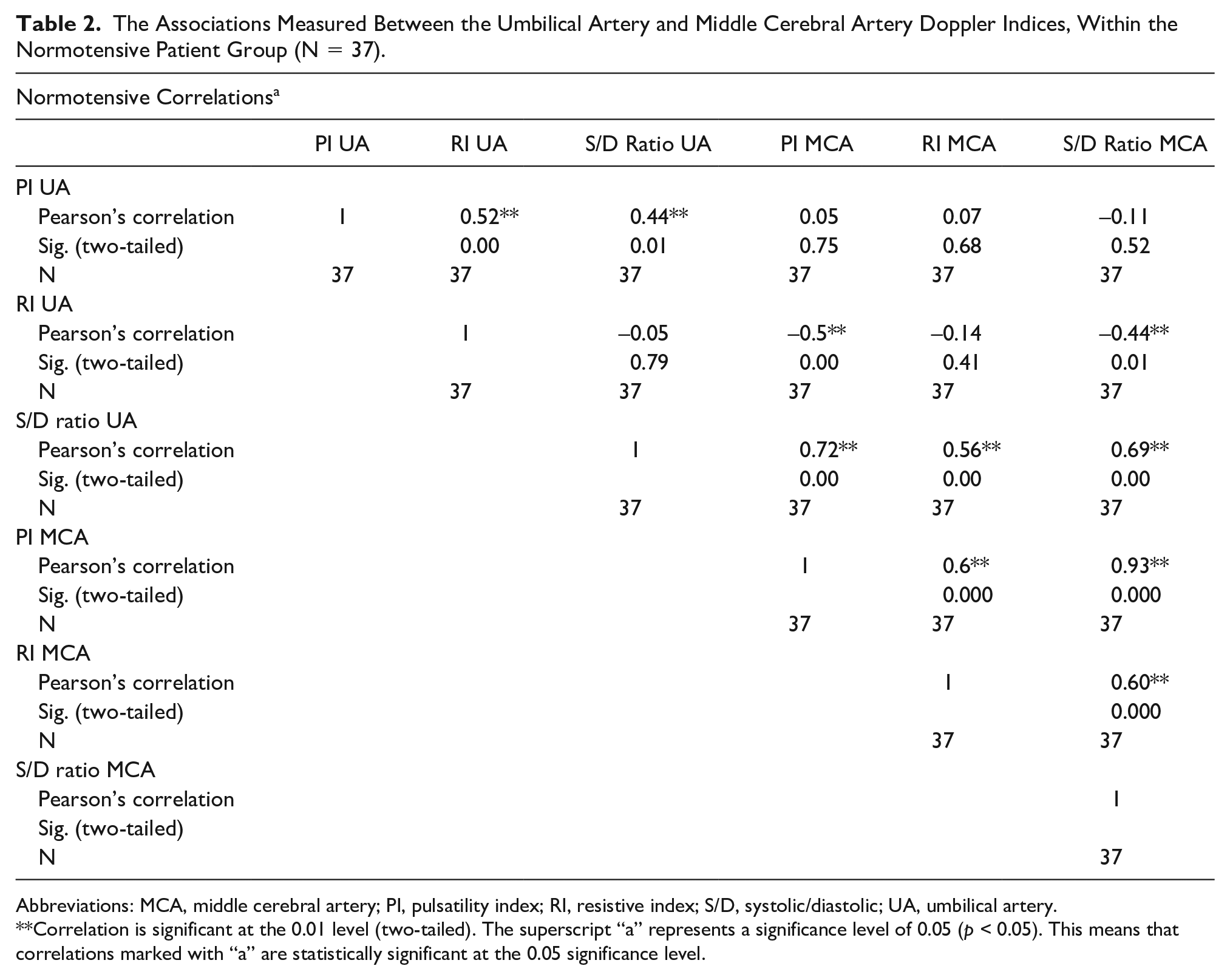
Abbreviations: MCA, middle cerebral artery; PI, pulsatility index; RI, resistive index; S/D, systolic/diastolic; UA, umbilical artery.
Correlation is significant at the 0.01 level (two-tailed). The superscript “a” represents a significance level of 0.05 (p < 0.05). This means that correlations marked with “a” are statistically significant at the 0.05 significance level.
In the preeclamptic group, the mean UA PI was 1.28 ± 0.23, the mean UA RI was 0.66 ± 0.10, the mean UA S/D ratio was 2.05 ± 2.14, the mean MCA PI was 1.59 ± 0.48, the mean MCA RI was 0.72 ± 0.14, and the mean MCA S/D was 2.52 ± 2.38. Between UA and MCA Doppler indices, a significant (P = .01) positive moderate correlation (r = 0.433) existed between PI, UA, and MCA (Table 3).
The Associations Measured Between the Umbilical Artery and Middle Cerebral Artery Doppler Indices, Within the Preeclamptic Patient Group (N = 33).
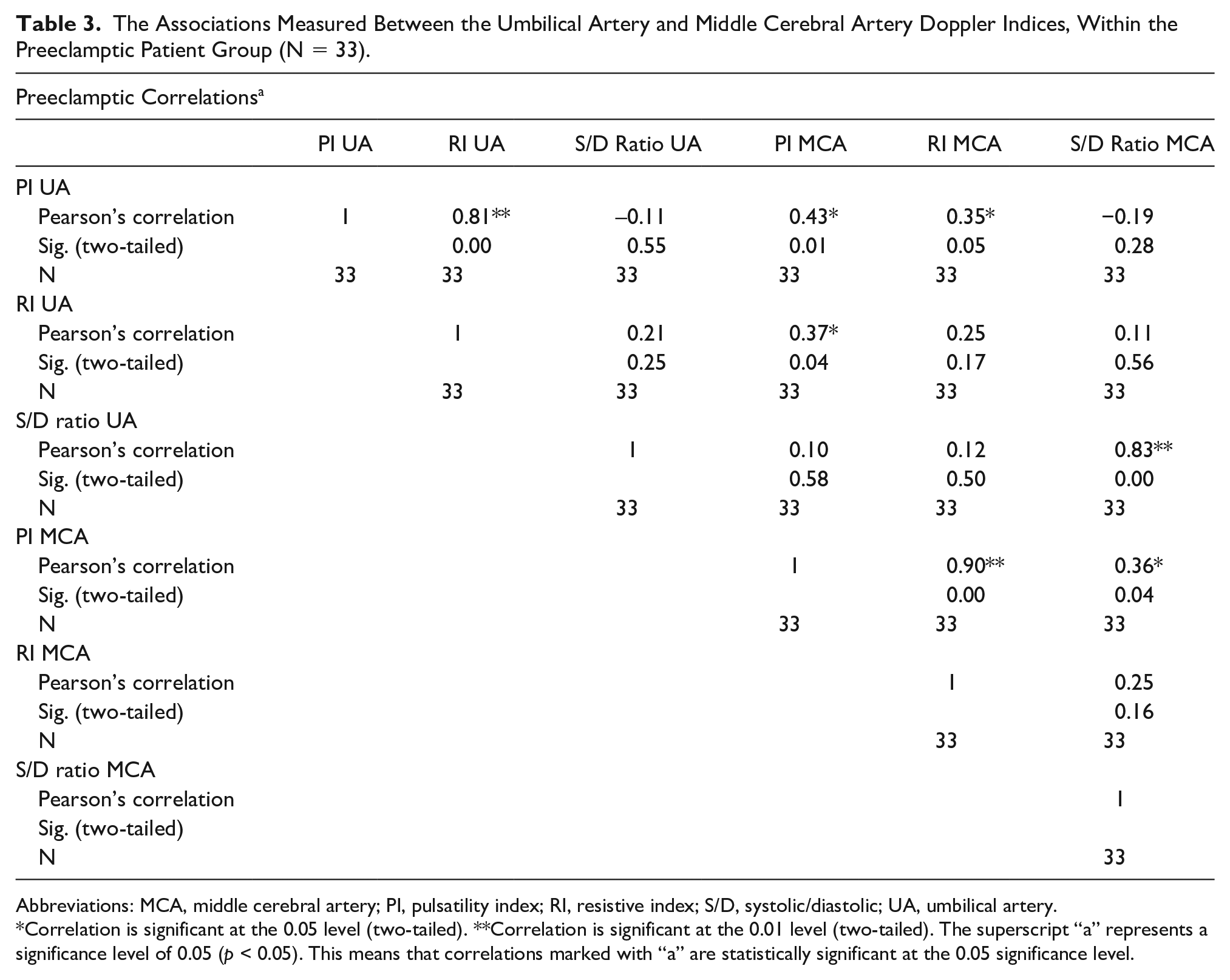
Abbreviations: MCA, middle cerebral artery; PI, pulsatility index; RI, resistive index; S/D, systolic/diastolic; UA, umbilical artery.
Correlation is significant at the 0.05 level (two-tailed). **Correlation is significant at the 0.01 level (two-tailed). The superscript “a” represents a significance level of 0.05 (p < 0.05). This means that correlations marked with “a” are statistically significant at the 0.05 significance level.
Discussion
Pregnancy-induced hypertension is a significant cause of mortality and morbidity of the mother and fetus, occurring in 3% of pregnancies. In addition, all hypertensive disorders occur in approximately 5% to 10% of pregnancies. 13 Preeclampsia is hypertension related to the gestation period, accompanied by proteinuria and edema. 14
In this cohort study, 37 women were assigned to the normotensive group, whereas 33 women qualified for the preeclamptic group. In the normotensive group, the mean maternal age was 26 ± 6.1 years, the mean gestational age was 33.69 ± 2.43 weeks, the mean systolic BP was 111.24 ± 11.57 mm Hg, the mean diastolic BP was 72.68 ± 8.59 mm Hg, and the mean BMI was 21.38 ± 2.21. The results of this study are comparable with that of a cohort study by Lopez-Mendez et al, 15 which included 51 normotensive patients with a mean age of 26.15 ± 5.53 years, a mean gestational age of 35.52 ± 3.33 weeks, a mean gravida of 2.50 ± 1.34, a mean systolic BP of 103.94 ± 15.25 mm Hg, and a mean diastolic BP of 69.81 ± 6.49. Concurrently, in the preeclamptic group, the mean maternal age was 28.97 ± 5.1 years, the mean gestational age was 33.01 ± 3.01 weeks, the mean systolic BP was 149.55 ± 5.64 mm Hg, mean diastolic BP was 96.70 ± 6.22 mm Hg, and mean BMI was 27.94 ± 2.42. These results can be compared with those obtained by Rafiq et al. Their study included 126 patients with preeclampsia with a mean age of 27.48 ± 5.61 zyears, a mean gestational age of 35.05 ± 2.95 weeks, a mean systolic BP of 151.98 ± 8.88 mm Hg, and a mean diastolic BP of 96.90 ± 4.64 mm Hg. 16 Abdelrazik Abdelfattah et al 17 reported on 100 patients with preeclampsia, who had a mean age of 28.62 ± 4.27 years and a mean gestational age of 36.24 ± 1.66 weeks. Lopez-Mendez et al 15 reported on 52 patients with preeclampsia with a mean age of 30.06 ± 5.32 years, a mean gestational age of 33.39 ± 3.46 weeks, a mean gravida of 3.43 ± 1.93, a mean systolic BP of 148.33 ± 14.1 mm Hg, and a mean diastolic BP of 93.63 ± 7.94 mm Hg. The findings of our study revealed notable differences between the preeclamptic and normotensive groups based on various parameters. The preeclamptic group had higher mean maternal age, systolic and diastolic BP, and BMI than the normotensive group. However, both groups had similar mean gestational ages. The hypothesis explored in this study was whether a significant correlation exists between UA and MCA Doppler velocity indices in patients with preeclampsia and normotensive patients. The null hypothesis was that no significant correlation existed, whereas the alternative hypothesis proposed was that a significant correlation existed. However, the findings of this study support the alternative hypothesis, indicating a significant correlation between the Doppler velocity indices within the UA and MCA in this cohort of preeclamptic and normotensive maternity patients.
In the normotensive group, the mean UA PI was 1.29 ± 0.19, the mean UA RI was 0.81 ± 0.09, and the mean UA S/D ratio was 2.37 ± 0.45. Khalid et al 18 reported a cohort mean with a UA PI of 1.02 ± 0.69, a mean UA RI of 0.67 ± 0.52, and a mean UA S/D ratio of 3.08 ± 2.1. Lopez-Mendez et al’s 15 study revealed that 51 of 103 normotensive pregnancies exhibited a mean UA PI of 0.96 ± 0.15, a mean UA RI of 0.52 ± 0.06, and a mean UA S/D ratio of 2.89 ± 0.48, consistent with the findings of our study. In contrast, the preeclamptic group had a mean UA PI of 1.28 ± 0.23, a mean UA RI of 0.66 ± 0.10, and a mean UA S/D ratio of 2.05 ± 2.14. This finding is comparable with the findings of a report published by Khalid et al, 18 which revealed a decline in mean values with advancing gestational age, although individual values consistently exceeded the two S/D ratio. Lopez-Mendez et al 15 reported on 52 of 103 preeclamptic pregnancies, and the UA PI mean of the cohort was 1.31 ± 0.18, a UA RI mean of 0.77 ± 0.09, and a UA S/D ratio mean of 4.4 ± 0.54. In addition, Zhou et al 19 reported higher blood flow parameters (PI, RI, and S/D ratio) in the preeclamptic group than in the normotensive group, with all differences being significant (P < .05). Zhou et al’s findings align well with our study findings.
In the normotensive group, the mean MCA PI was 1.34 ± 0.36, the mean RI was 0.74 ± 0.06, and the mean was S/D ratio of 2.95 ± 1.55. Khalid et al 18 reported a mean MCA PI of 2.09 ± 1.4, an MCA RI of 0.84 ± 0.75, and an MCA S/D ratio of 6.28 ± 4.20. In Lopez-Mendez et al’s 15 study, 51 of 103 normotensive pregnancies had a mean MCA PI of 2.07 ± 0.48, a mean MCA RI of 0.88 ± 0.08, and mean MCA S/D ratio of 6.90 ± 1.29. This result is comparable with that of their preeclamptic group; the mean MCA PI was 1.59 ± 0.48, the mean MCA RI was 0.72 ± 0.14, and the mean MCA S/D was 2.52 ± 2.38. 15
In the study by Khalid et al, 18 all the indices exhibited a decline in mean values with advancing gestational age. However, individual values at every gestation were consistently < 2 S/D ratio, which indicated vasodilation due to hypoxia. Lopez-Mendez et al 15 revealed a mean MCA PI of 1.47 ± 0.29, a mean MCA RI of 0.69 ± 0.06, and a mean MCA S/D ratio of 5.34 ± 0.67 in the preeclamptic group. In the study by Zhou et al, 19 blood flow parameters (PI, RI, and S/D ratio) of MCA gradually decreased in the preeclamptic group compared with the normotensive group, and all differences were significant (P < .05). These findings align closely with the results reported in this study.
In this study, the mean CPR ratio in the normotensive group was 0.88 ± 0.16, whereas in the preeclamptic group, the mean CPR ratio was 1.26 ± 0.41. A study by Konwar et al 20 revealed that in normotensive pregnancies, the mean CPR was 1.3 ± 1.4. Moreover, in the preeclamptic group, the mean CPR was 1.02 ± 0.94.
In this study, we observed significant correlations in the normotensive group between UA and MCA Doppler indices and a significant (P = .00) strong positive correlation (r = 0.72) between the UA S/D ratio and PI MCA. We also observed a significant (P = .00) positive correlation (r = 0.56) between the UA S/D ratio and the MCA RI. A significant (P = .00) moderate positive correlation (r = 0.66) was observed between the UA S/D ratio and the MCA velocity, and a significant (P = .00) negative correlation (r = −0.50) was observed between the UA RI and MCA PI. A significant (P = .01) negative correlation (r = −0.44) was observed between the UA RI and MCA S/D ratio. These correlations suggest a relationship between the blood flow patterns in the UA and MCA, potentially serving as an indicator of fetal well-being. Simultaneously, in the preeclamptic group, between UA and MCA Doppler indices, a significant (P = .01) positive moderate correlation (r = 0.43) was observed between the UA PI and MCA velocity. In contrast, a study by Borges et al revealed no significance (P = .07) and a negative correlation (r = −0.19) between the UA PI and MCA velocities in their patient cohort. Here, we observed a significant (P = .04) positive correlation (r = 0.37) between the UA RI and the MCA PI and a significant (P = .05) positive moderate correlation (r = 0.35) between the UA PI and the MCA RI. Similar results were reported by Borges et al, indicating a strongly significant (P = .00) negative correlation (r = −0.75) between the UA RT and MCA velocities in their group. No correlation was observed between RI in the UA and MCA within the preeclamptic group of our study, which differs from the findings of another study that demonstrated a significant and strong negative correlation between these two groups. This implies that as the RI in the UA increases, leading to decreased fetal-placental blood flow, resistance in MCA decreases and results in increased blood flow to the brain, considered the brain-sparing effect. 21 These results add to the body of knowledge and contribute to the ongoing discourse on the correlation between these arteries in the context of preeclampsia, aligning with similar findings in previous studies. Some studies have explored the ratio value between UA and MCA RI; however, none have specifically investigated the correlation between these two parameters.22,23 These potential correlations may provide valuable insights into compromised fetal blood flow and the potential brain-sparing effect observed in preeclampsia. 24 In this study, a significant correlation was observed between PI in the UA and the RI in MCA within the preeclamptic group. However, based on a limited search of the literature, no previous studies have specifically addressed the correlation between these two parameters.
This study revealed a significantly (P = .00) strong positive correlation (r = 0.83) between the S/D ratios within the UA and MCA. In contrast, the study by Borges et al demonstrated a significant (P = .00) and moderately negative correlation (r = −0.33) between the S/D ratios with the UA and MCA. When the S/D ratios within the UA and MCA were correlated in the preeclamptic group, a significant and moderate correlation existed between the two. An increasing UA S/D ratio indicates a more significant difference in peak systolic and diastolic flows and occurs in conditions that cause increased vasoconstrictions, such as in pregnancy-induced hypertension. In response, the MCA starts dilating to preserve the brain, thus exhibiting a low MCA S/D ratio. Similar results have been documented in a previous study. 21 Yalti et al 25 demonstrated the ratios between UA and MCA S/D.
Our study adds to the existing literature by demonstrating significant correlations in both preeclamptic and normotensive pregnancies. 26 This study aligns with existing literature on preeclampsia and UA velocimetry, revealing the intricate relationship between these factors and hemodynamic shifts in fetoplacental circulation. Previous studies have revealed a correlation between UA impedance and the development of the fetoplacental compartment, emphasizing the impact of tertiary villi and arterial channels. In the normal course, these findings indicate a progressive decline in UA indices with advancing gestational age. However, in FGR, high resistance in small arteries and arterioles of the tertiary villi leads to decreased diastolic flow, increasing Doppler indices. Moreover, studies have explored the trajectory of placental insufficiency in FGR, detailing the progression from decreased diastolic flow to its absence and subsequent reversal. In addition, these studies introduced the concept of the brain-sparing effect in FGR, highlighting adaptive blood flow redistribution to the brain and myocardium in response to chronic hypoxia. Our study elucidates normal and pathological variations in fetoplacental circulation. The consideration of FGR adds complexity, emphasizing the complex physiological responses in preeclampsia and contributing to the broader understanding of hemodynamic changes in high-risk pregnancies.
These findings also highlight the importance of considering maternal demographic and lifestyle characteristics, such as age, parity, and BMI, as these factors can influence the correlations between Doppler indices. In addition, the differences in methodologies, along with the number of waveforms studied across different studies, can contribute to variations in reported values. Hence, our study supports the alternative hypothesis by demonstrating a significant correlation between fetal MCA and UA Doppler indices in preeclamptic and normotensive patients. These findings contribute to the understanding of the vascular changes associated with hypertension in pregnancy and emphasize the potential utility of Doppler measurements in assessing fetal well-being.
In numerous instances, pregnant women may remain asymptomatic even in the presence of severe preeclampsia. Research has revealed that 38% of women diagnosed with preeclampsia exhibit no signs of hypertension or proteinuria. Despite the absence of these typical indicators, preeclampsia cases have been associated with severe maternal complications, underscoring the need for a precise and comprehensive definition. The challenge lies in recognizing fetal compromise in these situations, as failure to do so can lead to adverse outcomes, including fetal demise. One in 20 stillbirths without congenital anomalies can be intricately attributed to preeclampsia. 16
Limitations
A significant limitation of the study lies in its non-randomized research design and dependence on a convenient sample of maternity patients. Consequently, the results are specific to this cohort, and caution should be exercised when attempting to generalize findings to a broader population. The study also contains a small and potentially non-representative sample size, limited statistical power, and caution against making definitive statements. The findings may lack generalizability, and further research with larger, more diverse samples is crucial.
Conclusion
This study suggests the potential diagnostic value of Doppler measurements in assessing preeclampsia-related vascular changes. However, caution is warranted owing to the split participant sample and limited statistical power, particularly in the smallest group (N = 33). Further research with larger samples is crucial to validate and extend the findings, emphasizing the need for ongoing exploration of Doppler measurements in preeclampsia. In addition, integrating well-established Doppler indices of the uterine artery may offer a promising avenue to enhance the practicality of assessing the UA and MCA Doppler velocities in patients with preeclampsia.
Footnotes
Ethics Approval
Ethical approval for this study was obtained from Ethical Review Committee of the University of Lahore (IRB70034851).
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
