Abstract
Acute cerebral infarction, or stroke, is one of the leading causes of death and disability among adults worldwide, and the key to its treatment is achieving early recanalization of occluded blood vessels, methods for which include intravenous thrombolysis and mechanical thrombectomy. This article reports a case of a 43-year-old female who suddenly suffered acute cerebral infarction and was treated with tissue plasminogen activator intravenous thrombolysis. During the thrombolysis process, transcranial Doppler ultrasound was used to evaluate acute occlusion of the left middle cerebral artery, which was confirmed via an emergency head magnetic resonance angiography and digital subtraction angiography. Before mechanical thrombectomy, repeat angiography showed that the occluded left middle cerebral artery had regained blood flow, and after postoperative follow-up transcranial Doppler ultrasound, the blood flow of the left middle cerebral artery and anterior cerebral artery was found to have fully recovered. The patient’s symptoms were completely relieved, and they were discharged after 7 days of treatment, with follow-up at 2 weeks and 3 months showing no abnormal symptoms or intracranial blood flow. This case not only provides evidence that tissue plasminogen activator intravenous thrombolysis can salvage the ischemic penumbra, dissolve thrombi, and encourage reperfusion in occluded intracranial large arteries, but it also emphasizes the critical role of transcranial Doppler ultrasound in the rapid assessment of acute large vessel occlusions. As such, our report highlights the importance of transcranial Doppler ultrasound in clinical interventions to optimize outcomes in acute stroke management.
Keywords
Introduction
Stroke is an acute neurological deficit syndrome caused by cerebrovascular lesions, often characterized by “five highs,” namely, high incidence, high mortality, high disability rate, high recurrence rate, and high economic burden. It is one of the most common diseases causing death and disability in adults globally. 1 For treatment of acute cerebral infarction, early recanalization of occluded vessels is crucial, as the process can salvage ischemic penumbra nerve function and restrict expansion of the core infarct area. Currently, the most effective treatments are intravenous thrombolysis and mechanical thrombectomy within a limited time window, with tissue plasminogen activator (tPA) being the most representative thrombolytic agent. In clinical practice, intracranial large vessel occlusion can be confirmed through imaging examinations, such as magnetic resonance angiography (MRA), computed tomography angiography (CTA), or digital subtraction angiography (DSA).
More recently, however, transcranial Doppler ultrasound (TCD) has become an important imaging technique for assessing cerebral blood flow, as it allows direct scanning of the blood flow signals of cerebral arteries to obtain hemodynamic parameters, thereby depicting the functional status of cerebral blood vessels. TCD can also detect intracranial artery stenosis, occlusion, and collateral compensation, leading to its wide use in clinical practice. This article introduces a case of a patient with acute cerebral infarction who underwent tPA intravenous thrombolysis. In examining the patient, a TCD examination revealed large vessel occlusion, while subsequent cerebral angiography confirmed that tPA thrombolysis achieved recanalization of the occluded large vessels.
Case report
The patient, a 43-year-old woman, was admitted with a chief complaint of sudden speech impairment accompanied by right-sided limb weakness for 1 h. She was admitted to the hospital at 18:00 on October 16, 2023, presenting with a history of type 2 diabetes for 8 years, which she usually treated with metformin tablets orally. She self-reported good glycemic control and denied any history of other cardiovascular or cerebrovascular diseases, surgical procedures, trauma, smoking, or alcohol consumption. On admission for physical examination, her blood pressure was 160/100 mmHg and her heart rate was 93 beats per minute (bpm) with a regular rhythm. She was alert but presented with mixed aphasia, and her pupils were equal, round, and reactive to light, with normal ocular motility. Muscle strength was graded at III in the right upper limb and IV in the right lower limb, and a positive Babinski sign was elicited on the right side. However, during the ataxia physical examination, she was uncooperative. Auxiliary examinations revealed no obvious abnormalities on head computed tomography (CT; Figure 1). Electrocardiogram showed sinus rhythm at 93 bpm, while blood routine and coagulation tests were unremarkable, showing a final blood glucose level of 8.0 mmol/L. Preliminary diagnosis: Acute cerebral infarction. According to the 2019 AHA/ASA guidelines for the Early Management of Patients With Acute Ischemic Stroke, 2 within 4.5 h of onset, intravenous thrombolytic therapy is the first choice. Therefore, intravenous thrombolysis with 50 mg tPA was administered at 18:30, leading to an improvement in the patient’s right-sided limb weakness at 19:10. However, her mixed aphasia showed no significant relief, and her mental state remained poor. As large vessel occlusion was not discounted, a bedside TCD examination was performed at 19:15, indicating that the blood flow velocity of the M1 segment of the left middle cerebral artery (MCA) was significantly slower than that of the contralateral side, while the blood flow velocity of the left anterior cerebral artery (ACA) was increased (Figure 2(a) and (b)). This suggested acute occlusion of the M1 segment of the left MCA, and an urgent head magnetic resonance imaging (MRI) combined with MRA examination at 19:30 revealed multiple fresh infarctions (Figure 3(a)–(c)) in the left frontal, temporal, and parietal cortices, with poor visualization of the distal end and branches of the M1 segment of the left MCA (Figure 3(d)). After obtaining informed consent from the family, we planned to perform whole-brain angiography and mechanical thrombectomy, with the former procedure, performed at 20:20, showing distal occlusion of the M1 segment of the left MCA (Figure 4(b)), after which mechanical thrombectomy was prepared. However, prior to thrombectomy, another angiography revealed that the previously occluded vessel had spontaneously re-opened (Figure 4(c)). The cerebral angiography was completed at 20:45, and at this point, 75 min had elapsed since the completion of intravenous thrombolysis with alteplase. When leaving the catheterization room, the patient was articulate and had free movement of all limbs, and postoperative follow-up TCD found that the blood flow of the left MCA and ACA had returned to normal (Figure 5(c) and (d)). The patient’s postoperative course was stable, and she was discharged after 7 days of treatment, with follow-up at 2 weeks and 3 months showing no abnormal symptoms or intracranial blood flow.

CT scan results of the patient’s head after admission.
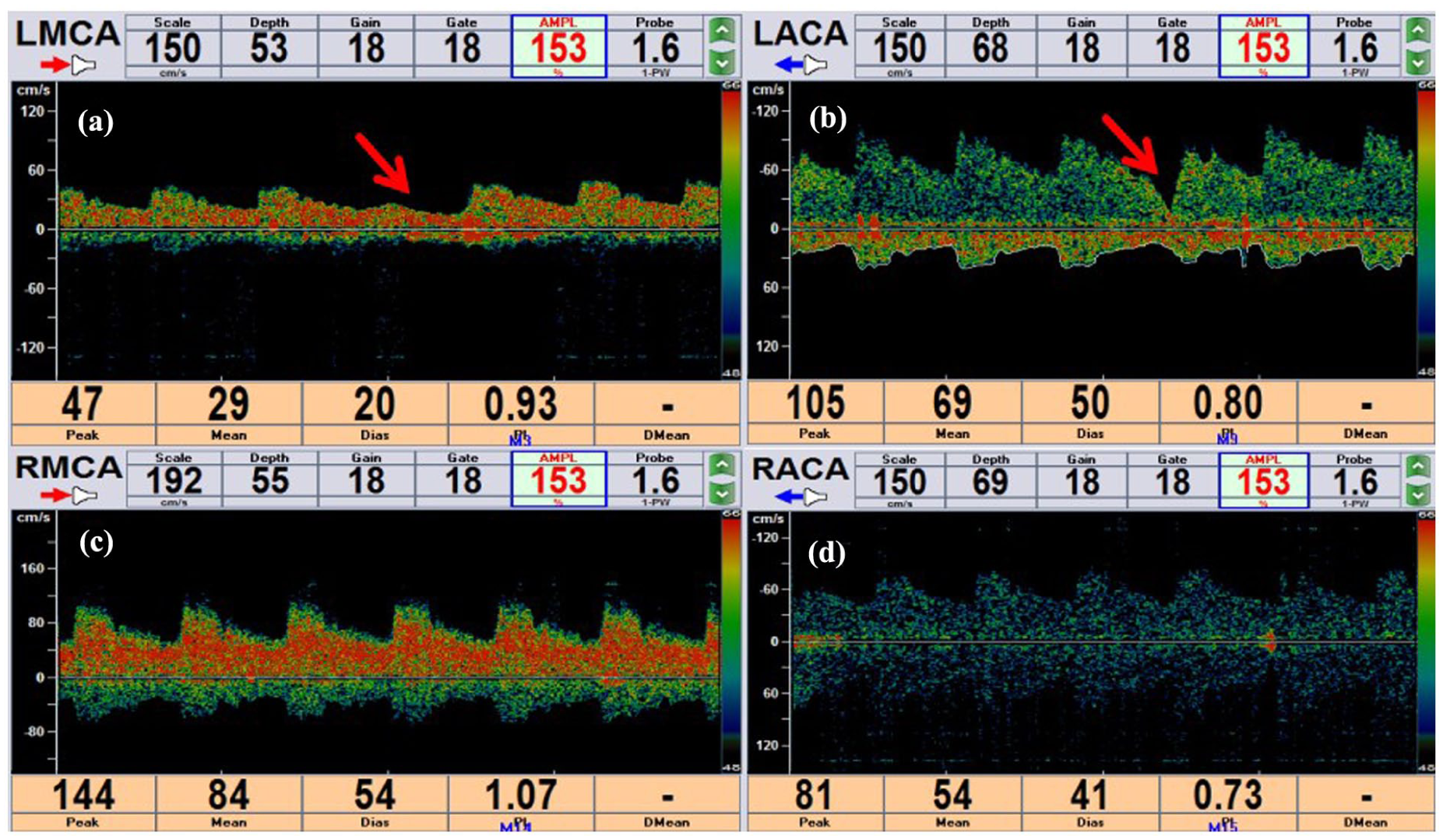
Results of TCD before intravenous thrombolysis. (a) A decrease in blood flow velocity in the left MCA. (b) A compensatory increase in blood flow velocity of the left ACA. The red arrow indicates the changes in blood flow velocity of the left CCA under compression. (c) The blood flow spectrum of the right MCA. (d) The blood flow spectrum of the right ACA.

Head (MRI) scan results of the patient after admission. (a) The diffusion-weighted imaging sequence, where the red arrow indicates the site of acute cerebral infarction. (b) The apparent diffusion coefficient, where the red arrow indicates the site of acute cerebral infarction. (c) The T2-weighted imaging. (d) The head MRA imaging sequence, where the red arrow indicates occlusion of the left MCA.
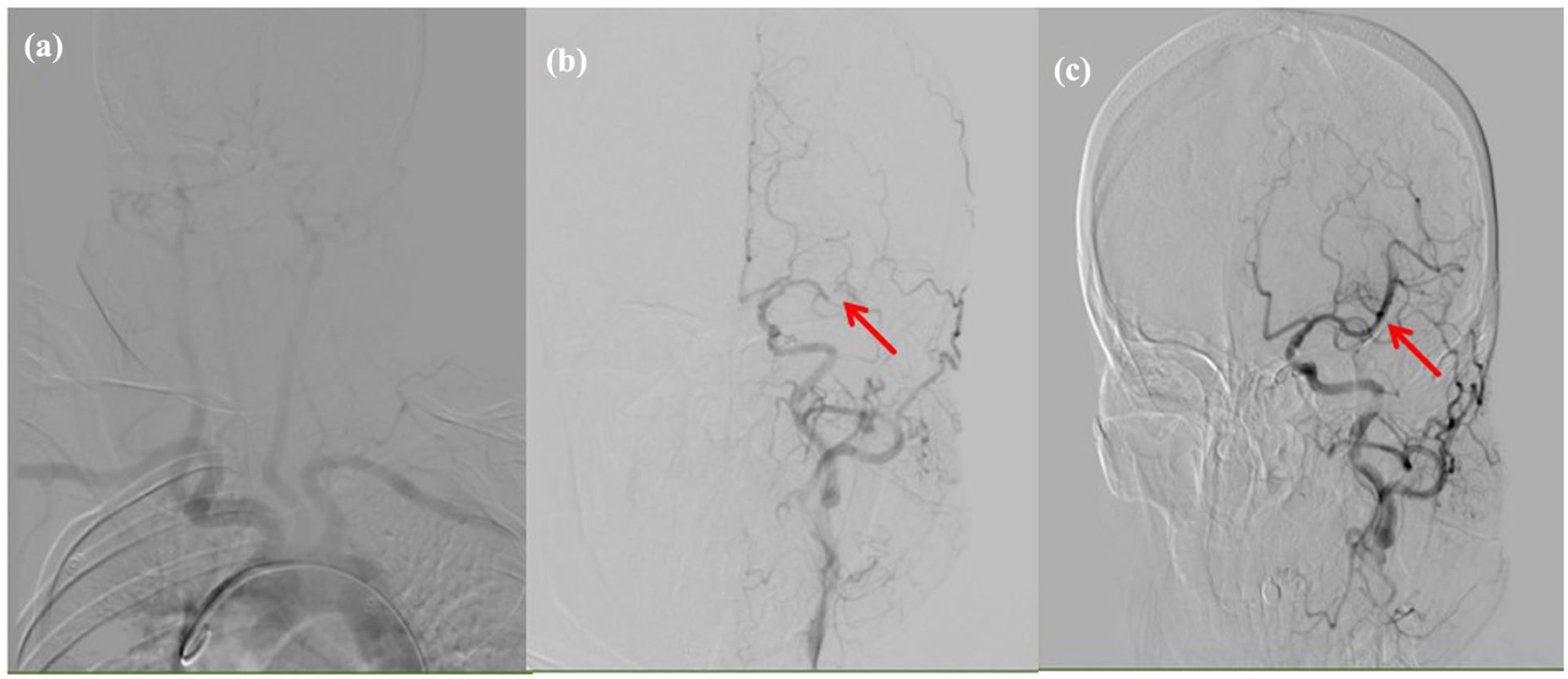
Results of the cerebral angiography examination. (a)The results of the brachiocephalic angiography, where the red arrow in (b) represents occlusion of the left MCA. The red arrow in (c) indicates the recovery of blood flow in the left MCA after the second cerebral angiography.

Comparison of preoperative and postoperative TCD results. (a) A decrease in blood flow velocity in the left MCA before cerebral angiography. (b) A compensatory increase in blood flow velocity of the left ACA before cerebral angiography, where the red arrow indicates the changes in blood flow velocity of the left CCA under compression. (c) The blood flow velocity of the left MCA increases after cerebral angiography, and it then returns to normal. (d) The blood flow velocity of the left ACA decreases after cerebral angiography, and it then returns to normal.
Discussion
A highly effective clinical treatment for acute cerebral infarction, tPA intravenous thrombolysis can effectively restore cerebral blood flow in the ischemic area, salvage the ischemic penumbra, and thus reduce the degree of neurological dysfunction. However, for cerebral infarction caused by an occlusion of the MCA, the reperfusion rate of intravenous thrombolysis is relatively low. 3 Bhatia et al. 4 showed that the vessel opening rate of M1 segment occlusion thrombolysis in MCA was 32.3%, while the occlusion opening rate in the internal carotid artery was only 4.4%. In the External-IA TNK trial, the reperfusion rate observed via DSA imaging after intravenous thrombolysis in the alteplase group was only 10% for patients with large vessel occlusion. 5 However, in this case report, we used TCD for real-time tracking of a patient with MCA-M1 occlusion to confirm complete vascular recanalization after tPA thrombolysis, though its benefits will decrease with a prolonged recanalization time, as confirmed by multiple studies.6,7 The reperfusion rate of blood vessels is affected by not only time, but also other factors, including thrombus. For instance, the thrombolytic effect of tPA on a red blood cell-enriched thrombus is better than that on a fibrin/platelet-enriched thrombus. Second, pathogenesis matters; for example, an atherosclerotic thrombus is easier to dissolve than a cardiogenic embolism. Third, collateral compensation can be determined by TCD, particularly when the ipsilateral ACA has good compensation, resulting in a relatively small infarct size. In addition, such factors as the ASPECTS score, thrombus burden, age, and coagulation function can also affect the vascular recanalization rate of intravenous thrombolysis in acute cerebral infarction. In this case, successful large vessel recanalization through tPA intravenous thrombolysis can be attributed to the short time from onset to thrombolysis, the patient’s young age, atherosclerotic thrombosis, a low thrombus load, and good collateral compensation.
In clinical practice, intracranial large vessel occlusion can be confirmed by imaging examinations, such as MRA, CTA, or DSA, though TCD has been proven an important imaging technique for examining cerebral blood flow maps, as it can directly scan the blood flow signals of cerebral arteries to obtain hemodynamic parameters, thereby reflecting the functional status of cerebral blood vessels. Compared with other vascular imaging examinations, TCD has multiple advantages such as non-invasiveness, simplicity, and a low cost, 8 and it can easily distinguish occluded blood vessels in patients with acute cerebral infarction leading to complete occlusion and partial occlusion, where the former refers to no blood flow or only weak blood flow signals and the latter to dull or low blood flow signals. Meanwhile, normal or narrowed blood flow after thrombolysis indicates vascular recanalization. Compared to angiography, TCD has good sensitivity (91%) and specificity (93%), especially for patients with complete vascular recanalization, for whom TCD has the best consistency with angiography. However, if TCD detects only partial blood flow improvement, DSA may still show occlusion, and when only weak blood flow signals are present, TCD is more sensitive than DSA.9,10
TCD not only has a significant advantage in assessing blood flow changes in patients with acute stroke, but it also plays a huge role in other cerebrovascular diseases. For instance, Ibraheem et al. 11 described a scalp arteriovenous malformation (AVM), a congenital or traumatic vascular abnormality where the arteries connect directly to veins, bypassing the capillary bed. Unlike stroke, AVMs are often identified due to a pulsatile mass or can be asymptomatic for extended periods, though they carry the risks of hemorrhage, seizure, or progressive neurological deficits. The tools for diagnosing AVMs also include CTA, MRI, and MRA, with DSA considered the gold standard for detailed characterizations. That said, TCD can diagnose medium to large AVMs and assess blood flow characteristics, providing a non-invasive means of evaluating hemodynamic significance.
Of course, TCD evaluation of acute MCA occlusion also has its limitations, 12 which are manifested in the following three aspects. First, some patients may have poor temporal window transmittance, which affects the quality of TCD detection of intracranial blood vessels such as MCA and reduces the accuracy and reliability of its diagnosis. For such patients, the blood flow signals of the contralateral MCA and ACA can be detected through the eye window, and blood flow signals can also be enhanced by intravenous injection of ultrasound contrast agents. If none of the above methods achieve the desired results, it may be necessary to perform a cranial MRA or CTA examination directly to clarify the vascular condition. Second, it is difficult to observe the structure of the vessel wall; because TCD mainly reflects the hemodynamic information in the vessel, it cannot directly display the structure and lesions of the vessel wall. For some MCA occlusions caused by vascular wall lesions, such as atherosclerotic plaque or dissections, the diagnostic value of TCD is relatively limited. At this point, it is necessary to combine TCD with other examination methods that can display the structure of the vascular wall, such as high-resolution MRI, intravascular ultrasound, etc., to evaluate vascular lesions comprehensively. Third, the results of a TCD examination require extensive operator experience and skill, as different operators may come to different diagnostic conclusions, which can affect the consistency and stability of the diagnostic results to some extent. Therefore, it is necessary to strengthen operator training and quality control to improve the accuracy and repeatability of TCD examination.
Conclusion
This case demonstrates that tPA intravenous thrombolysis is effective in recanalizing MCA occlusions, and the vascular recanalization initiated by intravenous thrombolysis can indeed significantly improve prognosis. Simultaneously, it highlights that TCD offers a timely assessment of the recanalization of blood vessels during intravenous thrombolysis for acute cerebral infarction. Due to its convenience, speed, and non-invasiveness, TCD plays an irreplaceable role in the rapid assessment of acute stroke and provides an important basis for neurologists to make clinical decisions quickly.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to the patient and the patient’s guardian for providing consent to include the case details and photographs in this publication.
Ethical considerations
This case report is exempt from ethical approval as determined by the ethical review board at our institution, given the nature of the article.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Author contributions
All authors contributed to this study. CXD and XG conceived and designed the manuscript; SYL was responsible for writing the manuscript with help from RHL and JHB; JPS and HML collected the clinical data. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The medical science research project of Hebei Province (No. 20232154, 20230984 and 20240529), the National Nature Science Foundation of China (No. 82301458), the Nature Science Foundation of Hebei Province (H2024206372), Hebei Province Government-funded Excellent Talents Project in Clinical Medicine (No. ZF2024147), the Project of Traditional Chinese Medicine of Hebei Province (No. 2023064), and the “Spark” scientific research project of the First Hospital of Hebei Medical University (No. XH202314).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
