Abstract
Renal transplant anastomotic pseudoaneurysms (RTAPs) are a rare vascular postoperative complication. The etiology of these pseudoaneurysms is attributed to infection or surgical vessel damage. RTAPs can lead to allograft dysfunction, allograft loss, or patient mortality due to rupture or sepsis. Because of these serious complications, expedient diagnosis is imperative for initiating surgical intervention and medical management. Sonography is a useful imaging modality for diagnosing and characterizing RTAPs. The sonographic findings, although not often described in detail in the literature, are definitive and unique. This case report describes the sonographic findings of a patient 22 days post renal autotransplant, who presented with excruciating abdominal pain. Sonographic examination identified multiple classic pseudoaneurysm findings and demonstrated intrarenal and extrarenal vascular abnormalities.
Keywords
Extrarenal anastomotic pseudoaneurysms are a rare pathology affecting renal transplant patients. These account for <1.0% of all vascular complications. 1 Significant allograft dysfunction or failure 2 results from impaired renal perfusion. Mortality from rupture or sepsis can result if the pseudoaneurysm is left untreated. Prompt diagnosis is challenging since symptoms are non-specific or absent. Sonography is a useful modality for diagnosing and characterizing pseudoaneurysms, and can provide imaging guidance for treatment. The following case report describes the clinical course, imaging, and outcome of a patient presenting with an anastomotic extrarenal pseudoaneurysm. This case demonstrates that sonography can be used to diagnose and characterize renal transplant artery pseudoaneurysms, follow up progression, and monitor the perfusion of the allograft.
Case Report
A 29-year-old man underwent a left nephrectomy and left lower quadrant renal autotransplant for a proximal 6 cm chronic ureteral stricture. Autotransplantation was chosen due to the location and length of the stricture, and the patient’s history of recurrent nephrolithiasis and bladder augmentation. The autotransplanted kidney had one artery and one vein. These were anastomosed to the left external iliac artery (EIA) and vein, respectively. The narrowed segment of the ureter was excised, and the ureteropelvic junction was reattached to the distal ureter. A postoperative Doppler examination demonstrated a well-perfused renal autotransplant, with evidence of a large renal pelvis from chronic obstruction. Samplings of the transplanted renal artery (TRA) measured velocities in the 270 cm/s range. The transplanted renal vein near the anastomosis and left iliac vein near the anastomosis showed elevated velocities (291 and 315 cm/s, respectively). This was attributed to immediately postoperative effects, with close attention at follow-up imaging recommended. Twenty days later, the patient’s ureteral stent was removed. The following day, the patient presented to the local emergency department. The patient’s symptoms included an episode of excruciating abdominal pain, causing loss of responsiveness, then continued abdominal pain. The patient was admitted for concern of pyelonephritis and surgical site infection, with elevated white blood count (WBC) of 16.9, (normal range = 3.4–9.6 × 109/L) and hemoglobin of 6.4 (normal range = 4.0 - 11.0 K/uL). A computed tomogram (CT) obtained that day displayed patchy enhancement of the left autotransplant and dilated loops of small bowel. The patient was initially treated with ceftraizone, an antibiotic, for pyelonephritis.
Three days after hospital admission, the patient developed increasing pain and redness over the surgical site. At that time, a sonogram was ordered. This showed a well-perfused autotransplant with hydronephrosis. It also demonstrated an adjacent 6.8 cm fluid collection without signs of gas, presumed to be a hematoma. Interestingly, the resistive indices (RIs) within the kidney remained in 0.6 range, but the velocities in the renal artery dropped to the 50 to 80 cm/s range. The same day, the patient underwent surgical irrigation, debridement, and wound vac placement at the left lower quadrant surgical site for concern of infection. The surgery confirmed soft tissue infection with areas of non-viable fascia. The surgeon removed necrotic soft tissue and fascia, and identified the retroperitoneal hematoma with intraoperative sonographic technique. However, there was difficulty aspirating the hematoma because of its thick texture. The care team elected to leave it in place, since the autotransplant was recent and the infection did not appear to progress throughout the fascia into the retroperitoneum. Cultures taken from surgical pathology grew Staphylococcus epidermidis, Staphylococcus hominis, Actinotignum schaalii, and Peptoniphilus asaccharolyticus.
The day following, the patient again experienced severe onset pain and tachycardia, prompting another CT. This CT examination showed a 3 cm pseudoaneurysm at the renal autotransplant artery anastomosis and left EIA, further enlargement of the pelvic hematoma, and heterogeneous nephrogram indicating impaired arterial perfusion (see Figure 1). The patient was airlifted to the tertiary medical center for further surgical management.
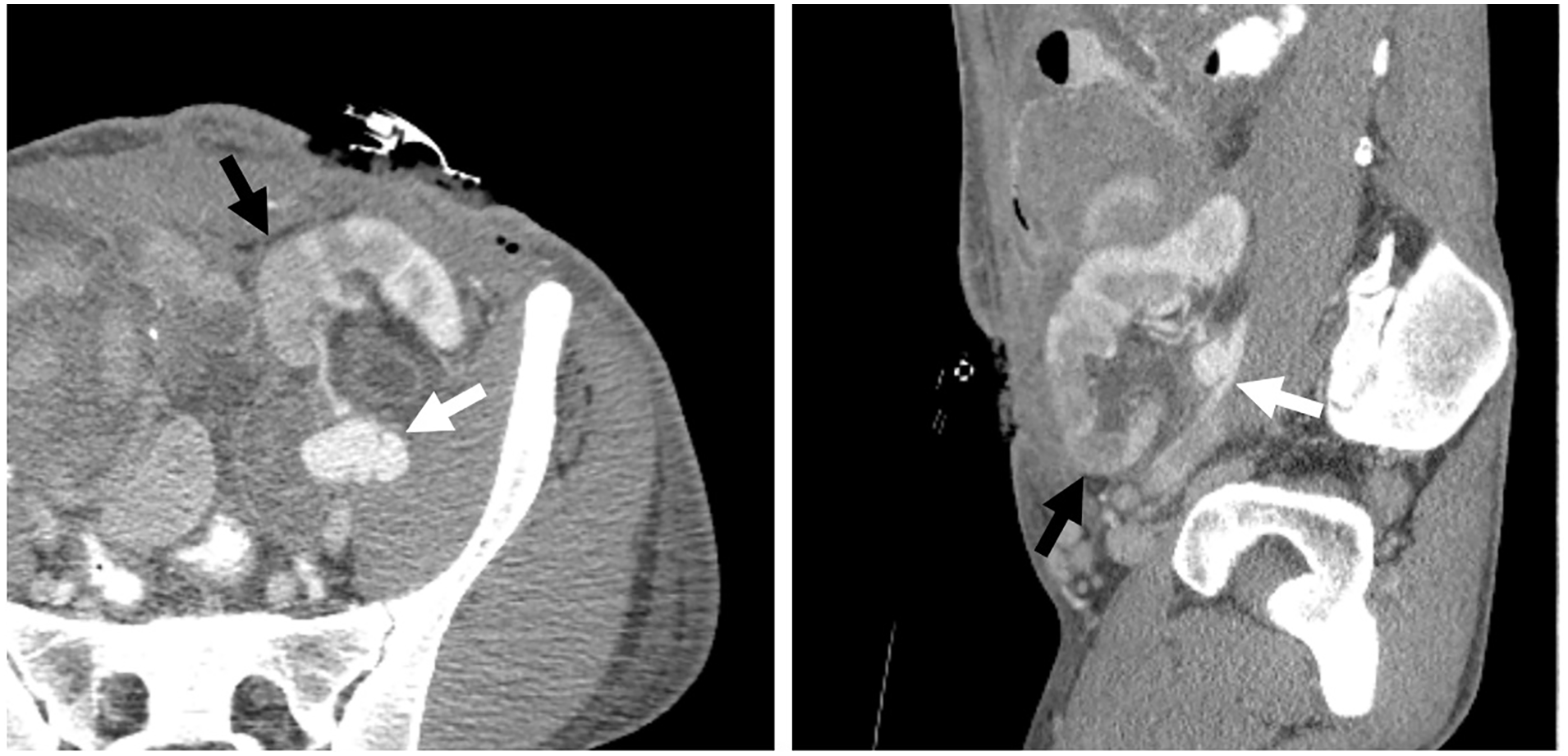
Computed tomography demonstrates the renal transplant anastomotic pseudoaneurysm (white arrows) arising from the left external iliac artery and renal autotransplant hypoperfusion (black arrows) in the sagittal and axial views.
An emergency sonogram was performed to evaluate the pseudoaneurysm and transplant perfusion. The examination was performed using a GE Logiq E9 (GE Health Care, Chicago, IL) ultrasound equipment system and a curvilinear 1–6 MHz transducer. Of note, the sonogram was limited by the overlying wound vac, adjacent hematoma, and patient’s pain. The autotransplant demonstrated normal RIs in the upper and mid-pole, but low arcuate artery RIs of 0.36 to 0.44 in the lower pole, suggestive of poor arterial perfusion (see Figure 2). In addition, the TRA demonstrated a decrease in velocities to 40 to 59 cm/s compared with 191 to 270 cm/s previously (see Figure 3). The pseudoaneurysm demonstrated an anechoic sac on gray-scale sonography, with a connecting neck to the renal artery and iliac artery anastomosis. On color Doppler, the pseudoaneurysm was filled with a swirling blood flow pattern, called the “yin-yang” sign (see Figure 4). Sonographically, the pseudoaneurysm measured 1.8 cm in largest dimension (see Figure 5), and the difference was attributed to an incomplete sonographic evaluation due to the aforementioned limitations or partial thrombosis of the pseudoaneurysm. No evidence of active hemorrhage was seen on the sonographic examination.
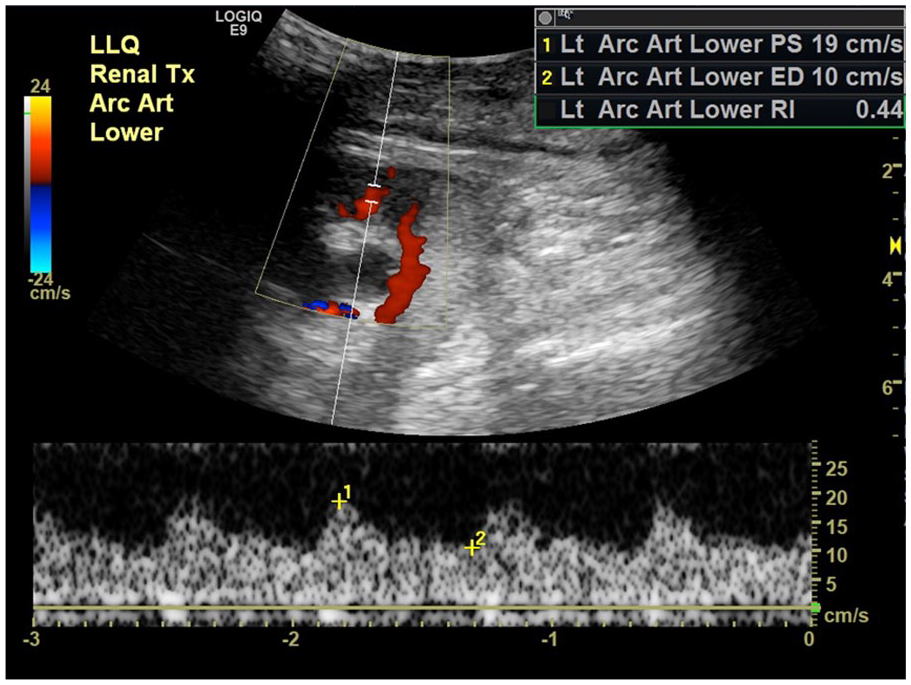
Sonographic spectral Doppler image demonstrating decreased resistive indices (<0.5) and a tardus parvus waveform within the arcuate arteries.
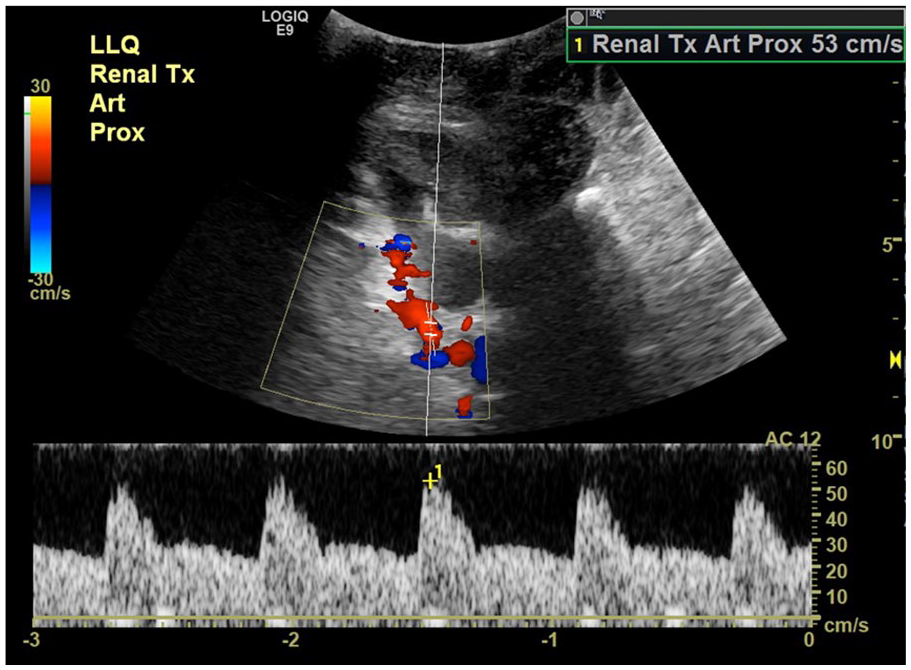
Sonographic spectral Doppler image of the renal transplant artery showing decreased velocities (53 cm/s, previously 248 cm/s).
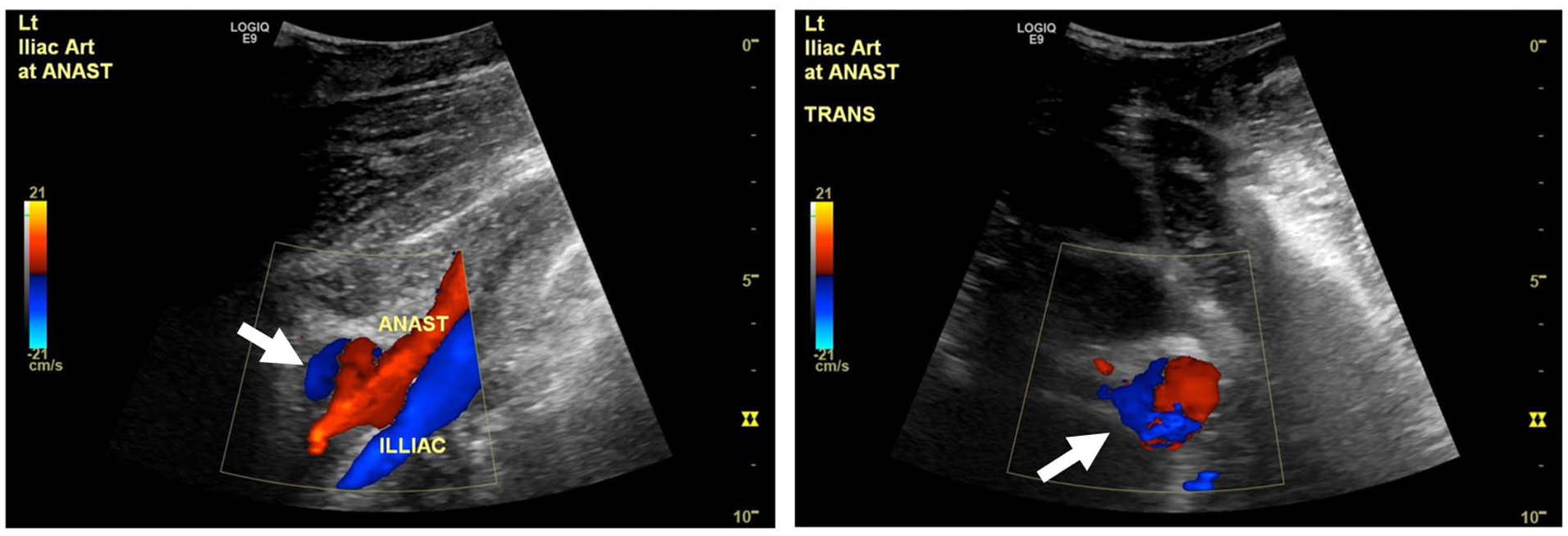
Longitudinal and transverse sonographic color Doppler images showing swirling, “yin-yang,” color flow at the renal transplant anastomotic pseudoaneurysm site (white arrows).
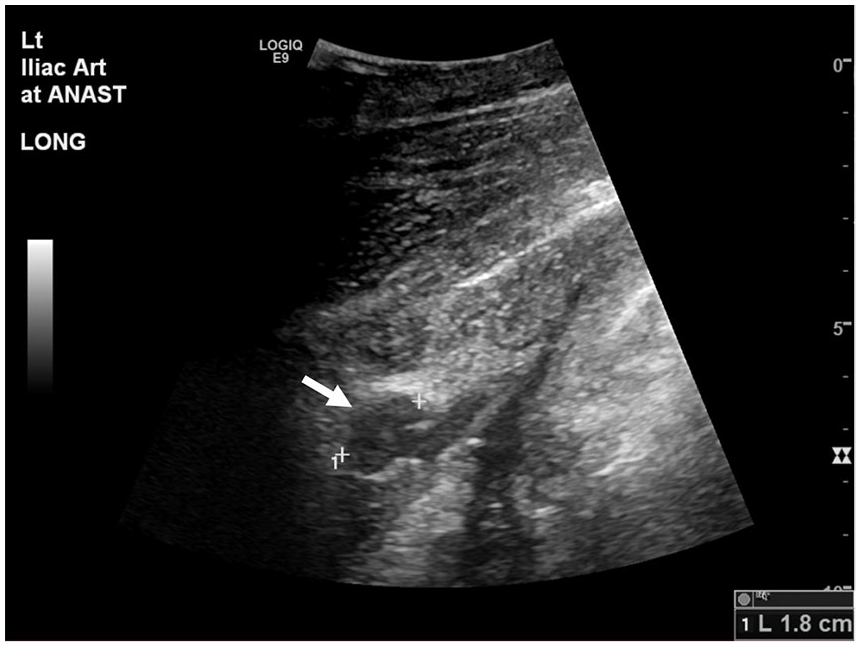
Longitudinal gray-scale sonographic image demonstrates an anechoic 1.8 cm sac (white arrow) connected by a neck to the external iliac artery.
The patient was taken to surgery for attempted repair of the pseudoaneurysm or nephrectomy if repair was not possible. A lack of discernible tissue planes, extensive scarring, and inflammation made dissection difficult. Because significant bleeding made visualizing the vascular structures difficult, the team proceeded with a nephrectomy. Postoperative pathology of the autotransplant indicated suspicion for pyelonephritis, chronic obstructive neuropathy, acute tubular injury, and multifocal organizing thrombi, primarily within arteries. The following day, interventional radiology (IR) treated the patient with two covered stents in the left EIA, successfully excluding the pseudoaneurysm (see Figure 6). The follow-up CT noted a fluid collection with gas at the nephrectomy site. Postoperatively, the patient required pain management, drain exchange and repositioning, and a sonographic guided aspiration of a left lower quadrant fluid collection. This fluid collection again grew S epidermidis on laboratory cultures. Sixteen days following the nephrectomy, the patient was discharged to home on antibiotics. At the last evaluation, the patient had normal renal laboratory results, an unremarkable postoperative CT, and continued presumed neuropathic pain in the left lower quadrant, which was present before the autotransplant.
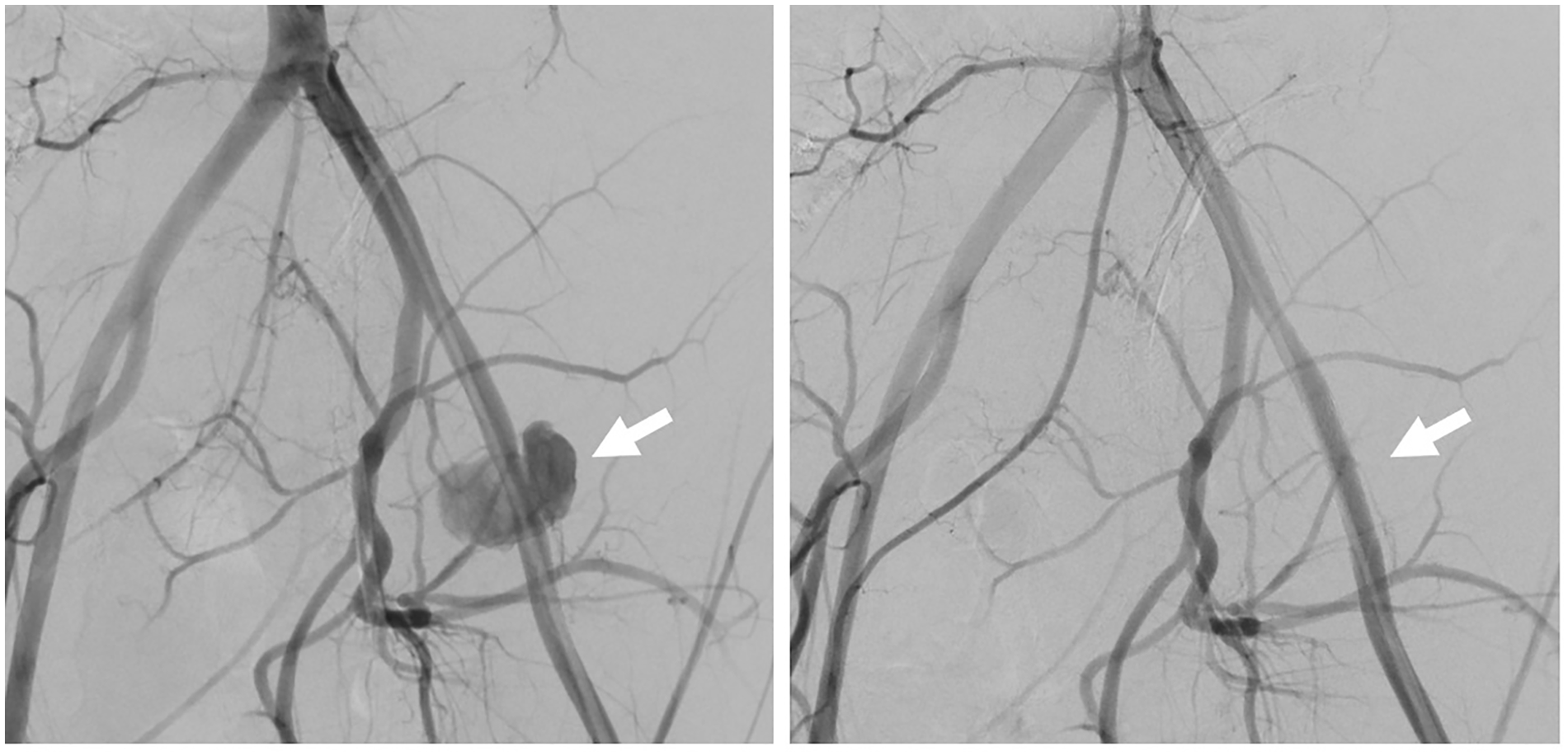
Left external iliac artery angiography image depicting the anastomotic pseudoaneurysm (white arrows) following a transplant nephrectomy, pre- and post-stenting performed in the interventional radiology suite. Note the successful exclusion of the renal transplant anastomotic pseudoaneurysm following intervention on the right image.
Discussion
Renal transplantation and autotransplantation are two outcomes of a similar surgical technique and produce similar imaging appearances, but have unique purposes. Renal transplantation is a well-established method of treating end state renal disease. 3 Renal autotransplantation is a method of treating proximal ureteral injury, renal artery aneurysms, chronic kidney pain, and some complex cases of renal tumors.4–6 It is a favorable alternative to conventional nephrectomy and prevents a patient from experiencing loss of renal function. 4 However, since autotransplantation is a challenging surgery and a patient’s recovery can be slow, the multi-disciplinary team must have excluded all other treatments of the underlying pathology. 4
A renal transplant is typically positioned in the extraperitoneal right or left iliac fossa (see Figure 7). 7 In cadaver donor transplants, the TRA is harvested along with a small patch of aorta. This patch, known as a Carrel patch, is attached end-to-side to the recipient common or EIA.8,9 In living donor transplants, the TRA is harvested without an aortic patch. The TRA is anastomosed end-to-side to the common or EIA, or, less commonly, end-to-end with the internal iliac artery. In either case, the transplanted renal vein is attached end-to-side to the recipient iliac vein. Normal variations in the donor renal vascular and recipient anatomy, such as multiple donor renal arteries and veins, are possible. Multiple TRAs may be anastomosed to the iliac artery separately, or harvested and attached with a common Carrel patch. 9 One artery may also be shortened and anastomosed to the other artery, forming a “Y” shape. This creates a single anastomosis to the iliac artery. The smaller of multiple renal veins are ligated. The donor ureter is typically implanted directly into the recipient’s bladder, a process called ureteroneocystostomy. 10 In renal autotransplantation, the surgical technique follows the same principles as living donor renal transplantation, except the patient’s own native kidney is relocated to the iliac fossa. In proximal ureteral injury and renal artery aneurysms, the damaged section is removed, and the kidney and newly shortened conduits are anastomosed more proximally.
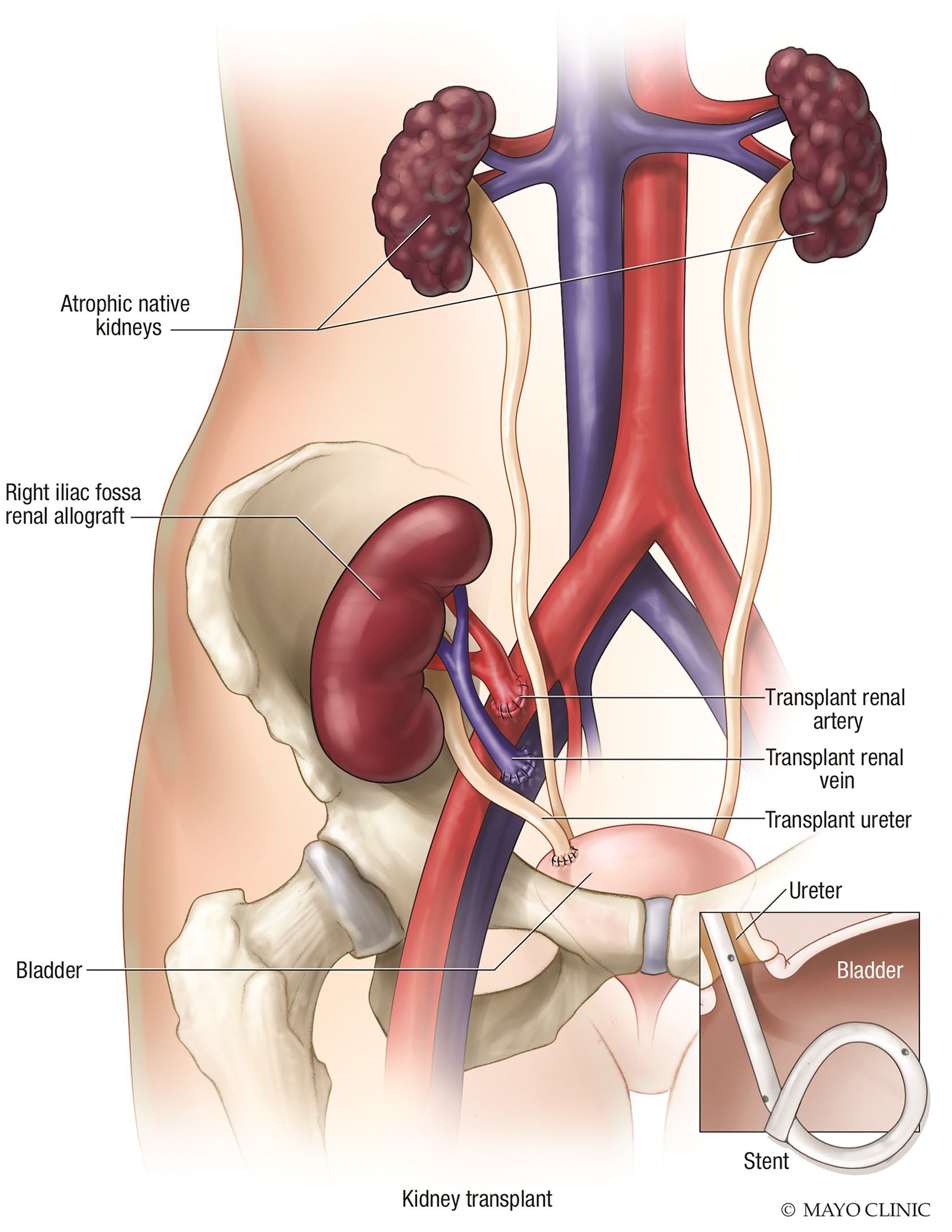
Medical illustration depicts the standard position and vascular connections of a renal transplant. Used with permission of Mayo Foundation for Medical Education and Research, all rights reserved.
Successful sonographic evaluation of a renal transplant is dependent on understanding the expected anatomy. Thus, it is imperative for the sonographer and radiologist to be familiar with their institution’s surgical protocols. 9 For immediately postoperative studies, it is helpful for the sonographer to review the surgical note or discuss the case with the surgical team or review previous imaging for follow-up studies.
The general formation of pseudoaneurysms begins with trauma to the layers of the vessel wall. Blood may leak through a defect in the first two layers of the wall, creating a sac contained only by the adventitia. Alternatively, it may leak through all three layers, accumulating into the surrounding soft tissues. 11 In contrast, typical aneurysms are a dilatation of all three uninterrupted layers of the wall (see Figure 8). Anastomotic pseudoaneurysms have a slightly different etiology. In this case, the connection between the graft and native vessel leaks, and a fibrotic capsule formed by the tissue around the anastomosis prevents complete rupture. 12
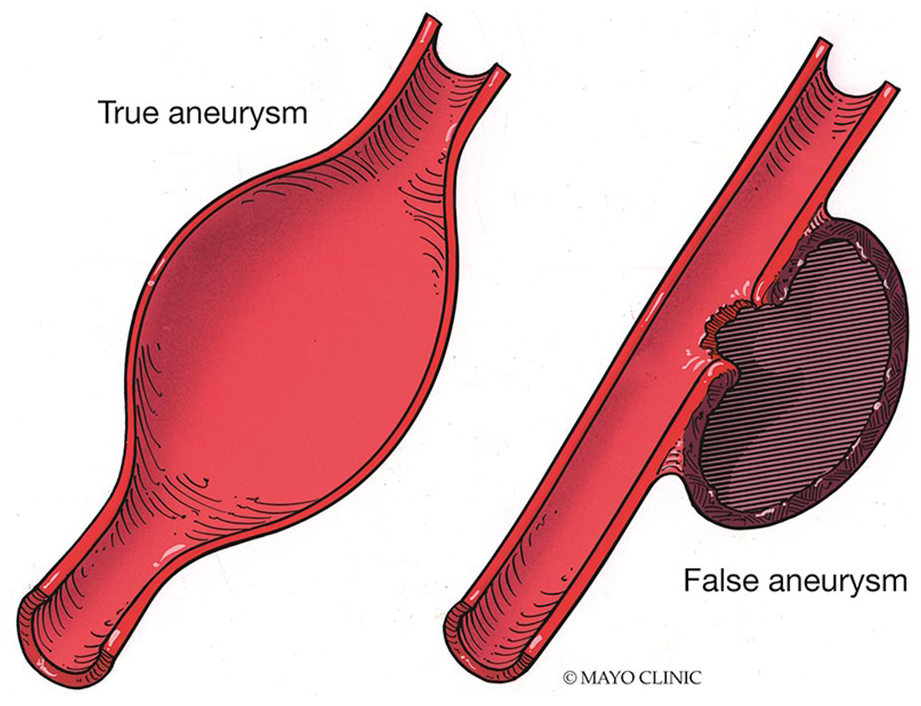
Medical illustration contrasting the differences in the wall structure of true and false (pseudo) aneurysms. Used with permission of Mayo Foundation for Medical Education and Research, all rights reserved.
In a renal transplant pseudoaneurysm, this vessel wall trauma has two known etiologies: surgical or infective formation. Arterial wall damage during surgery or suturing technique may disrupt the wall, leading to a pseudoaneurysm. This is the less common causation. An infective etiology causes degradation of the vessel wall, due to inflammatory changes. Infective etiologies account for more than 75% of cases, according to a recent literature review. 13 Transplant recipients are especially susceptible to infection because of their immunosuppressed state. 14 Infections can be divided into two groups: mycotic (fungal) versus non-mycotic (non-fungal) pseudoaneurysms. 15 The most common types of infective mycotic organisms include Candida albicans and Aspergillus species. 13 Non-mycotic organisms are much less common, with Psuedomonas aeruginosa and Methicillin-resistant Staphylococcus aureus (MRSA) being the most prevalent. 13
TRA pseudoaneurysms have been reported to occur in multiple segments throughout the vessel. They most commonly occur near the anastomosis: in the renal artery itself, or the EIA. However, they can also occur more distally in the TRA as well. Because of the irregular shape and close proximity of the structures in an anastomosis, it can be difficult to delineate exactly which vessel the pseudoaneurysm originates. 16
Clinical signs and symptoms of renal transplant anastomotic pseudoaneurysms (RTAPs) include large pulsatile mass, abdominal pain, increased serum creatinine, decreased urine output, hypotension, hemorrhagic shock, anemia, and ipsilateral lower extremity ischemia and swelling.13,17,18 Infection-specific symptoms include fever of unknown origin. Fungal infections specifically may not be symptomatic for several months. 13 However, surgically caused pseudoaneurysms and the majority of infective pseudoaneurysms seem to present within weeks. 18 Pseudoaneurysms may also present as an incidental imaging finding in asymptomatic patients. 14
Treatment of pseudoaneurysms requires surgical or interventional repair, and depends on the etiology of the pseudoaneurysm, as well as the severity and size of the defect. However, asymptomatic pseudoaneurysms, with low clinical suspicion for infection, and measuring less than 2.5 cm in diameter, have lower risk for severe complications.16,19 One case seems to suggest that surgery can be avoided in small, asymptomatic cases, if the patient is monitored regularly, although this pseudoaneurysm was not at the anastomosis. 16 Symptomatic, greater than 2.5 cm pseudoaneurysms, are an indication for surgery. 19 In addition, infective pseudoaneurysms will need to be treated with appropriate antibiotics. In a recent review of literature covering 30 cases, only one reported an infected pseudoaneurysm case, which was managed with antibiotics alone without surgical or endovascular intervention. 13
Open repair with allograft nephrectomy and vascular reconstruction has historically been the most common management choice. The damaged section of the native vessel is excised and an interposition graft or femoral-femoral bypass is placed, and the transplanted kidney is removed. However, in ideal scenarios, the transplant can be spared. Endovascular repair techniques with stenting continue to evolve and become more popular, but are only feasible in non-infected pseudoaneurysms. These show promise in transplant-sparing procedures as well.20,21 Sonography-guided thrombin injection can be used alone or alongside covered stent placement, if the pseudoaneurysm neck is small. 22
Left untreated, RTAPs create serious complications. RTAPs can disrupt the renal transplant’s blood supply, leading to allograft dysfunction and loss. Rupture of the pseudoaneurysm can lead to hemorrhagic shock or death. 23 Untreated infection can lead to sepsis. 13 Thus, the high-risk nature of pseudoaneurysm complications adds urgency to the need for an accurate imaging diagnosis.
A normal renal transplant and its associated vasculature have well-defined expected sonographic features. The transplant itself should be of similar size, echogenicity, and echotexture to a native kidney. 8 On color and power Doppler evaluation, it should display even perfusion throughout the parenchyma. Spectral Doppler of the arcuate arteries should display RIs in the range of 0.5 to 0.7 in the upper, mid, and lower poles. The arcuate arteries should have spectral Doppler tracings demonstrating sharp upstroke at systole. The TRA displays a normal anastomotic diameter and appearance on gray scale. On color, it does not display any areas of aliasing or turbulent flow on color Doppler. Spectral Doppler tracings throughout the anastomosis, proximal, mid, and distal TRA display a low-resistive pattern, with velocities less than 300 cm/s. 24 Velocity information may be inaccurate if the spectral Doppler angle is not parallel with the vessel wall or the angle exceeds 60°. The normal iliac artery displays normal gray-scale caliper, absence of aliasing or turbulent flow, and no doubling of velocities between segments. Postoperative fluid collections near the renal transplant may be an expected finding, although they should decrease in size over time.
A RTAP will demonstrate abnormal intrarenal and extrarenal sonographic findings. First, the arcuate arteries within the transplant demonstrate classic low-resistive waveforms with a delayed upstroke on spectral Doppler. Since the renal transplant’s blood flow has been disrupted by the pseudoaneurysm, the transplant’s vascular bed becomes more compliant. RIs measuring less than 0.5 indicate vascular compromise proximally in the renal artery. Gray-scale interrogation shows a saccular dilatation adjacent to the anastomosis site, with a connecting neck between the sac and the originating vessel. The length, width, and height of the sac, and length and width of the neck can be measured on gray-scale sonography. In addition, the presence or absence of septations or lobes may be observed. 11 Partial absence of color within the sac can suggest partial thrombosis. Although the application of contrast-enhanced ultrasound (CEUS) is not widely used, it has been helpful to evaluate the degree of thrombosis within pseudoaneurysms or differentiate between fluid collections and pseudoaneurysms in other areas of the body, and could prove value in the future. Adjacent postoperative fluid collections or exophytic cysts could mimic the anechoic saccular appearance of a pseudoaneurysm. However, the absence of a connecting neck to the vessel and lack of flow on color and spectral Doppler within the structure will quickly differentiate these from a pseudoaneurysm. On color Doppler, pseudoaneurysms classically demonstrate a “yin-yang” sign within the sac, and turbulent flow on spectral Doppler imaging. A “to-and-fro” flow spectral Doppler waveform is a classic finding within the neck (see Figure 9). In diastole, the “to” flow above the baseline represents blood entering the pseudoaneurysm. The “fro” flow represents blood exiting during systole. In the adjacent tissue, fluid collections seem to be common in infected pseudoaneurysms. Growth of a fluid collection over time, vascularity within the collection, or the presence of gas indicate a pathologic cause.
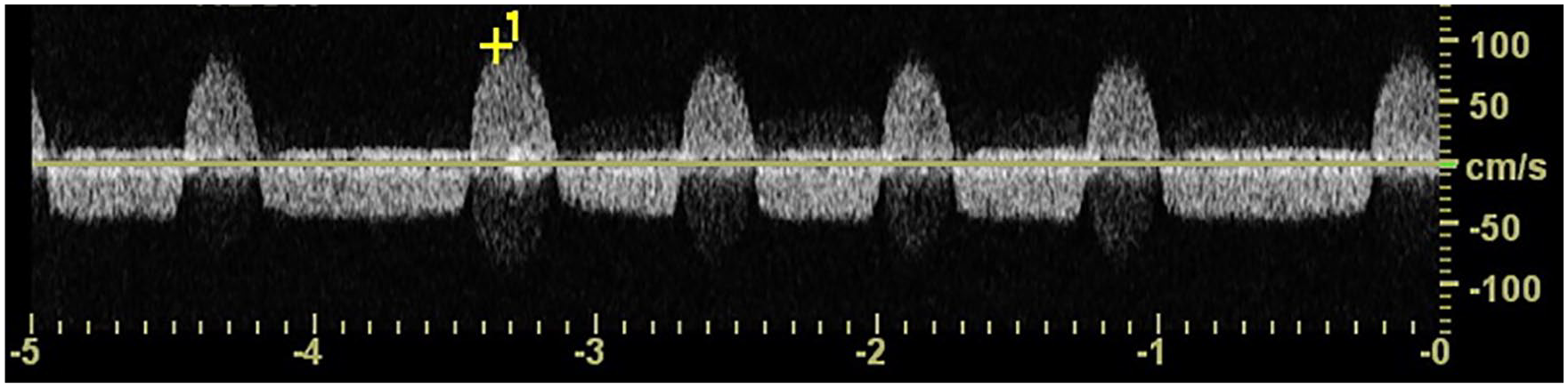
Sonographic spectral Doppler tracing demonstrating the classic to-fro appearance of blood flow within a pseudoaneurysm neck.
Atypical color and spectral Doppler pseudoaneurysm findings are also possible. One case report noted complete absence of color flow within the pseudoaneurysm, likely due to thrombosis and/or a wide aneurysm neck. 16 Multiple cases have reported elevated, stenotic velocities at the renal artery origin, adjacent to the pseudoaneurysm.15,18 It has been suggested that this might be caused by transplant renal artery compression from the adjacent pseudoaneurysm. 18
Sonography has been identified as a good initial modality for identifying anastomotic pseudoaneurysms, since it is effective and non-radiating. 14 It has few contraindications, and is time-efficient, noninvasive, and non-nephrotoxic. It provides specific information on the perfusion of the renal transplant in the form of intrarenal RIs and vascular velocities. The repeatability of sonography makes it a good choice to establish an immediate postoperative baseline and follow the progression of the transplant. 25 However, it is operator dependent. It may give suboptimal results when viewing deep vascular structures in the body, especially in patients with obese body habitus. Computed tomography angiography (CTA) and magnetic resonance imaging angiography (MRA) can provide detailed anatomical information to confirm the diagnosis, 14 and are especially well suited for characterizing fluid collections. 7 Although CTA is radiating and sometimes nephrotoxic, both MRA and CTA are less limited by body habitus. Finally, the gold standard of all imaging types used for vascular complications is catheter angiography. 26 It is also the most common modality for image-guided vascular interventions. During a patient’s treatment, sonography and at least two other modalities will likely play an important role in diagnosis and intervention.
Conclusion
RTAPs are rare, but serious, vascular complications. Expedient imaging diagnosis of an anastomotic pseudoaneurysm can prevent escalation to allograft loss, sepsis, hemorrhagic shock, or mortality. Sonography is a useful modality for diagnosing the presence of these pseudoaneurysms. It displays classic findings such as an anechoic sac connected by a neck to the transplant renal artery/iliac artery anastomosis in gray scale. This sac is filled with the swirling, “yin-yang” sign on color Doppler, and turbulent flow on spectral Doppler. Arcuate artery RIs provide information about the degree of vascular compromise inside the renal transplant. Sonography may be used as imaging guidance for thrombin injection. In the hands of a well-educated and technically competent operator, sonography plays an important role in the expedient diagnosis and management of this emergent complication.
Footnotes
Authors’ Note
The patient gave permission for the use of medical history and imaging for research purposes.
Declaration of Conflicting Interes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
