Abstract
Hepatic artery pseudoaneurysms (HAPs) are rare, life-threatening complications that arise in response to liver trauma or underlying inflammatory processes. The risk of rupture necessitates prompt diagnosis and treatment. Sonography is an important imaging modality used to evaluate and characterize suspected HAPs. In the case described below, sonography played an important role in the diagnosis and successful treatment of a patient with a large, high-risk HAP.
Rare vascular conditions have been known to develop as a complication of abdominal trauma or inflammatory disorders. Hepatic artery pseudoaneurysms (HAP) are one such complication. At one large institution, the incidence of HAP was reported as 0.03% of trauma and surgical admissions, and 1% of liver transplant patients over the course of 7 years. 1 While many HAPs are found incidentally in asymptomatic patients, others have presented in acute distress with symptoms such as abdominal pain, jaundice, hypovolemia, hematemesis, anemia, or melena.2,3 HAPs have the potential to rupture causing life-threatening complications. Sonography has many diagnostic benefits for these patients, especially when combined with other imaging modalities. Prompt diagnosis of HAPs leads to expeditious treatment, reducing the risk of rupture.
The following case report details the diagnosis and management of a large HAP. The use of sonography directly contributed to the characterization, diagnosis, and treatment of the HAP, in this patient.
Case Report
A 60-year-old female, with complex medical history including Marfan syndrome and prior descending aortic dissection, presented to an outside institution with chest pain and recent episodes of hematochezia. An abdominal computed tomogram (CT) was performed and revealed a large 9 cm enhancing mass in the liver (Figure 1), compatible with intact pseudoaneurysm or arteriovenous fistula (AVF) and possible gastrointestinal bleed. Shortly after the CT examination concluded, the patient was air lifted to a tertiary institution and directly admitted into the intensive care unit (ICU). The patient appeared to be in hemorrhagic shock upon arrival with low hemoglobin and hematocrit values at 3.6 g/dL (normal range 11.6–15.0 g/dL) and 12.5% (normal range 35.5%–44.9%), respectively. The international normalized ratio (INR) was 2.8 (normal range 0.90–1.10) and platelets were 46 x 109/ L (normal range 157–371 x 109/L). The patient was hypotensive, presenting with systolic blood pressure readings of 70 mm Hg which further lowered to 60 mm Hg.
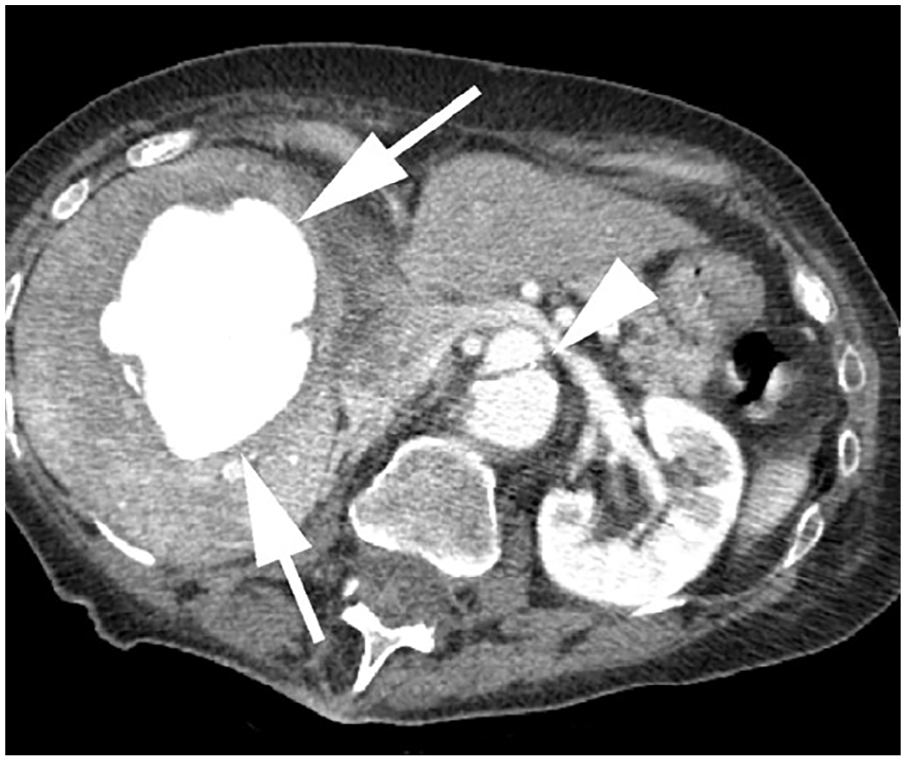
Axial contrast-enhanced abdominal computed tomogram shows a large, intensely enhancing mass centrally in the right hepatic lobe (arrows). Chronic aortic dissection is also present (arrowhead).
Upon arrival to the ICU, an emergent sonogram was ordered to further characterize the liver lesion. This examination was promptly performed (LOGIQ E9; GE Health Care, Chicago, IL) at the patient’s bedside, with a curvilinear 1 to 6 MHz transducer. The large vascular lesion was identified in the right lobe of the liver, using an intercostal approach. The application of color Doppler revealed a “yin-yang” sign within the lesion (Figure 2), consistent with pseudoaneurysm. Further interrogation of the lesion revealed a potential fistulous connection between the middle hepatic vein and the pseudoaneurysm (Figures 3 and 4). The examination was prematurely terminated by attending staff due to the declining status of the patient. It was decided the patient would be taken immediately to interventional radiology (IR), although a difficult endovascular access was expected.
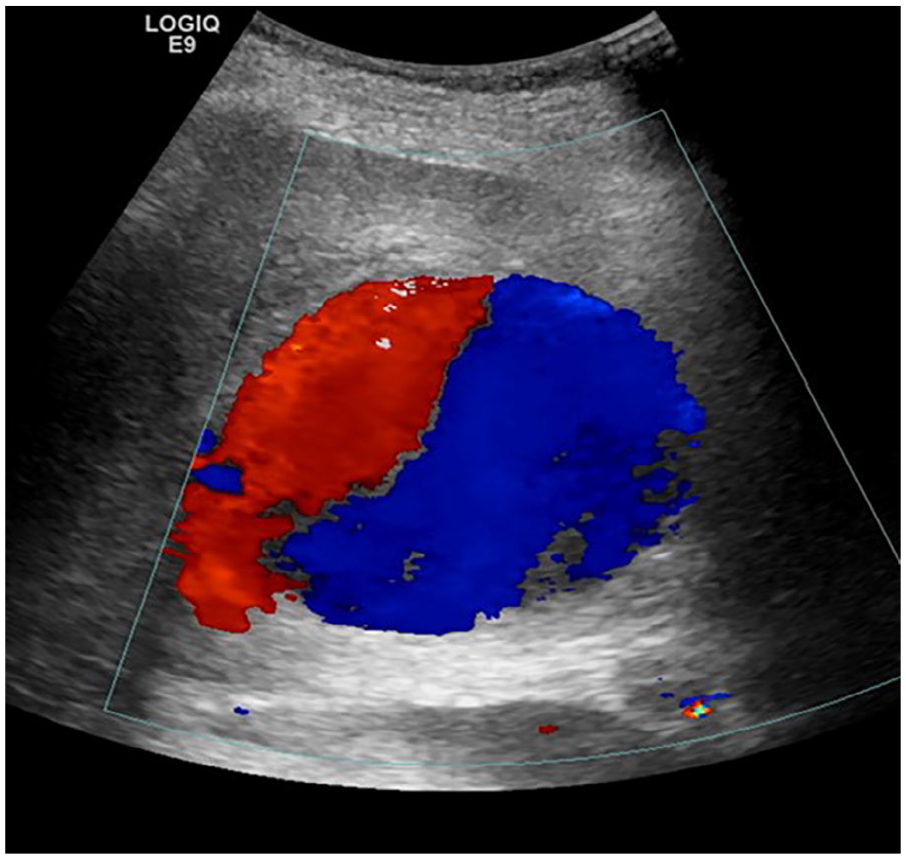
Color Doppler sonographic image shows classic “yin-yang” color flow within the large hepatic pseudoaneurysm.
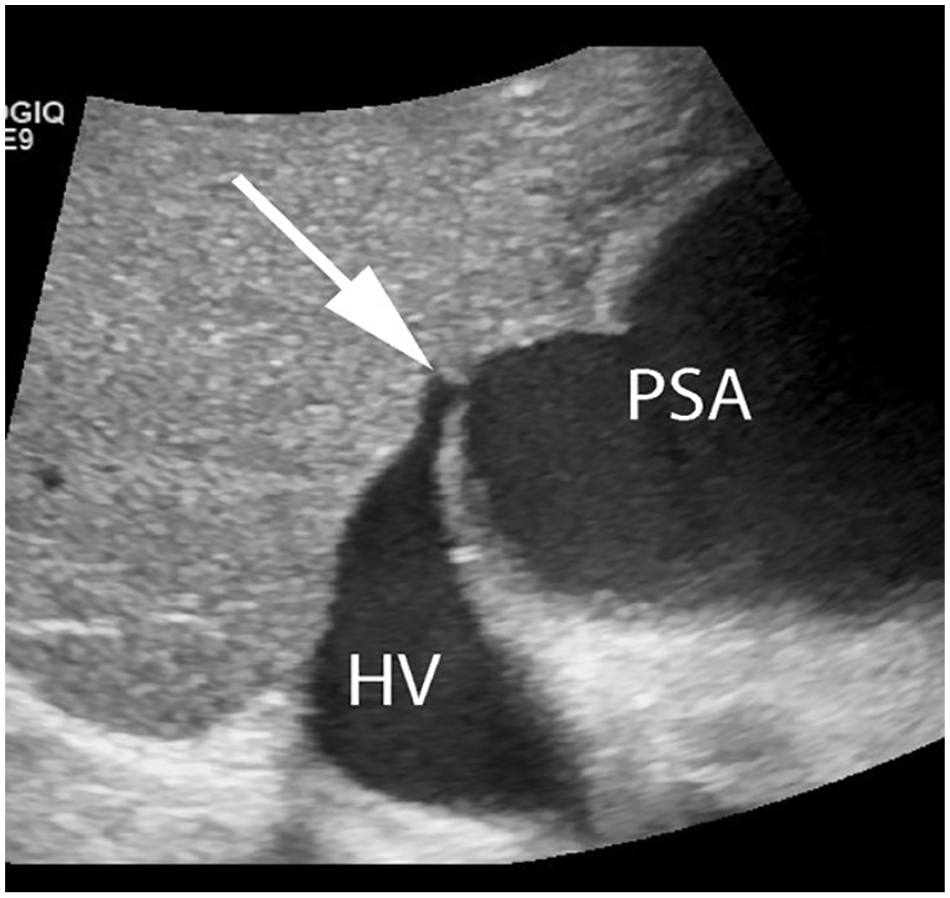
Sonographic gray-scale image depicting a fistula (arrow) between the pseudoaneurysm (PSA) and middle hepatic vein (HV).
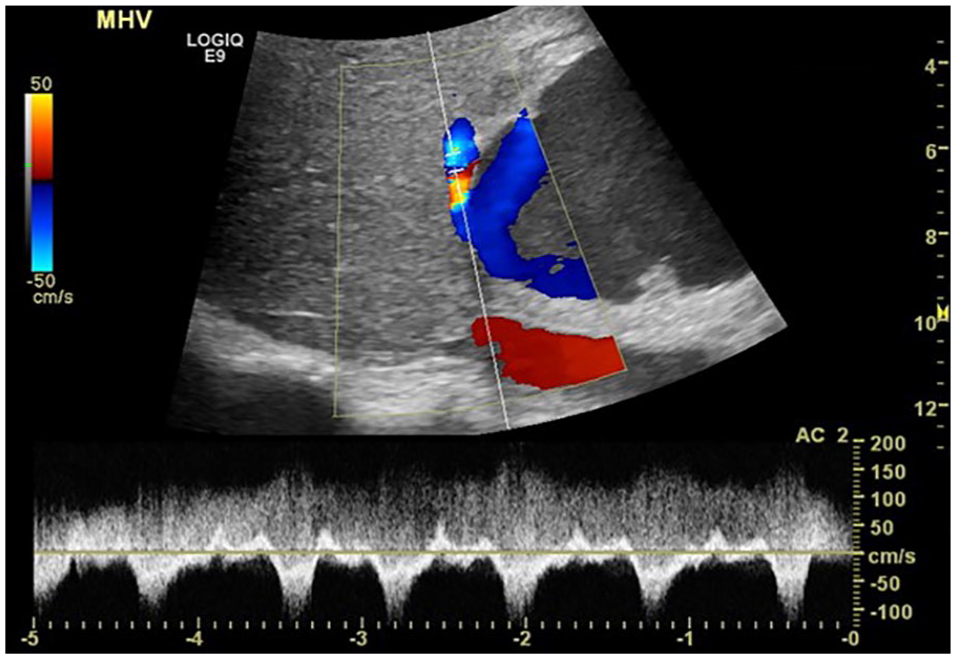
Sonographic color and spectral Doppler image of the middle hepatic vein (MHV) near the fistula with the pseudoaneurysm. Normal flow patterns in the hepatic vein are displayed below the baseline, while continuous flow from the fistula is depicted above the baseline.
In IR, direct puncture of the pseudoaneurysm was performed (Figure 5) with subsequent contrast injection confirming communication with hepatic veins. The size of the fistula and adjacent vein was large enough such that a mechanical occlusion device was needed. Such a device is placed using an approach similar to the Seldinger technique, where a wire is used to deploy or insert a device. Using sonographic guidance, the fistulous connection was crossed with a wire (Figures 6 and 7) and, using combined real-time sonographic and fluoroscopic guidance, an Amplatzer vascular plug (Abbot Laboratories, Abbott Park, IL) was percutaneously deployed across the middle hepatic vein fistula (Figures 8 and 9). Despite this maneuver, multiple additional venous fistulas remained present as well as additional small hepatic artery pseudoaneurysms. Multiple small pseudoaneurysms were embolized under fluoroscopy in conjunction with the large feeding branch of the dominant pseudoaneurysm. Despite aggressive intravascular treatment, persistent blood flow from multiple patent smaller feeding branches remained in the large pseudoaneurysm. Direct puncture thrombin injection of the pseudoaneurysm was then performed under sonographic-guidance, targeting the arterial inflow. Injection of 1000 units of thrombin resulted in complete thrombosis (Figure 10).
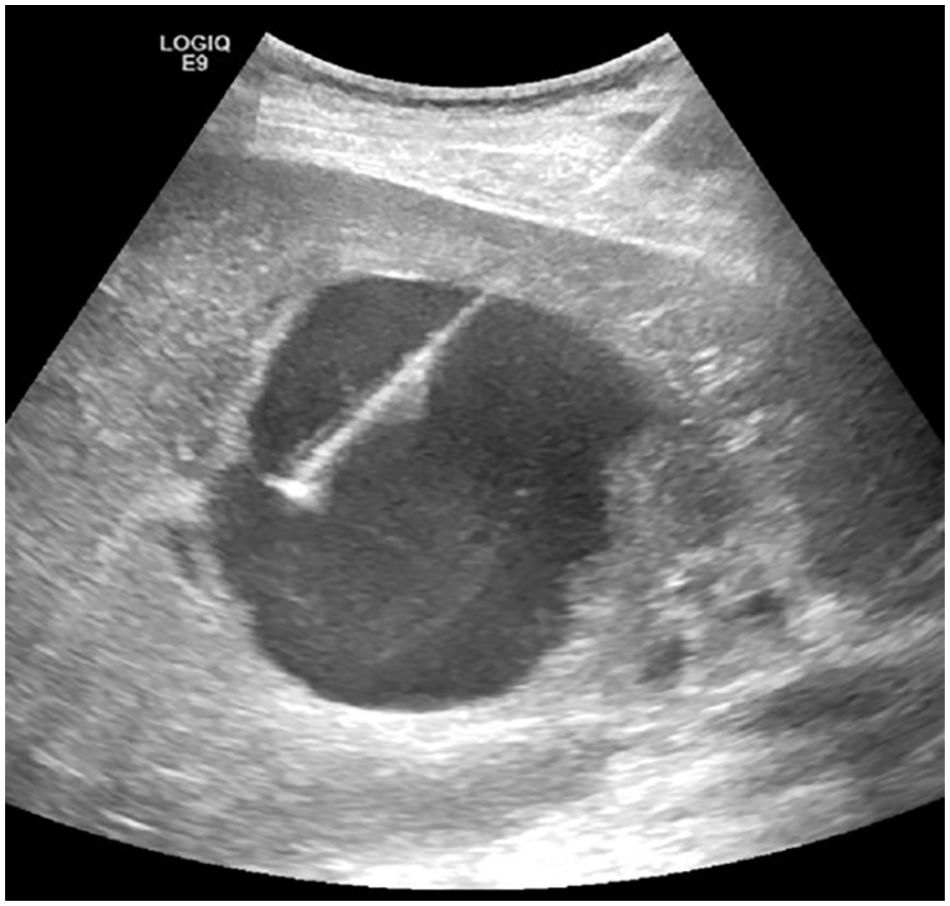
Sonographic gray-scale image obtained during direct puncture of the pseudoaneurysm to allow subsequent manipulation.
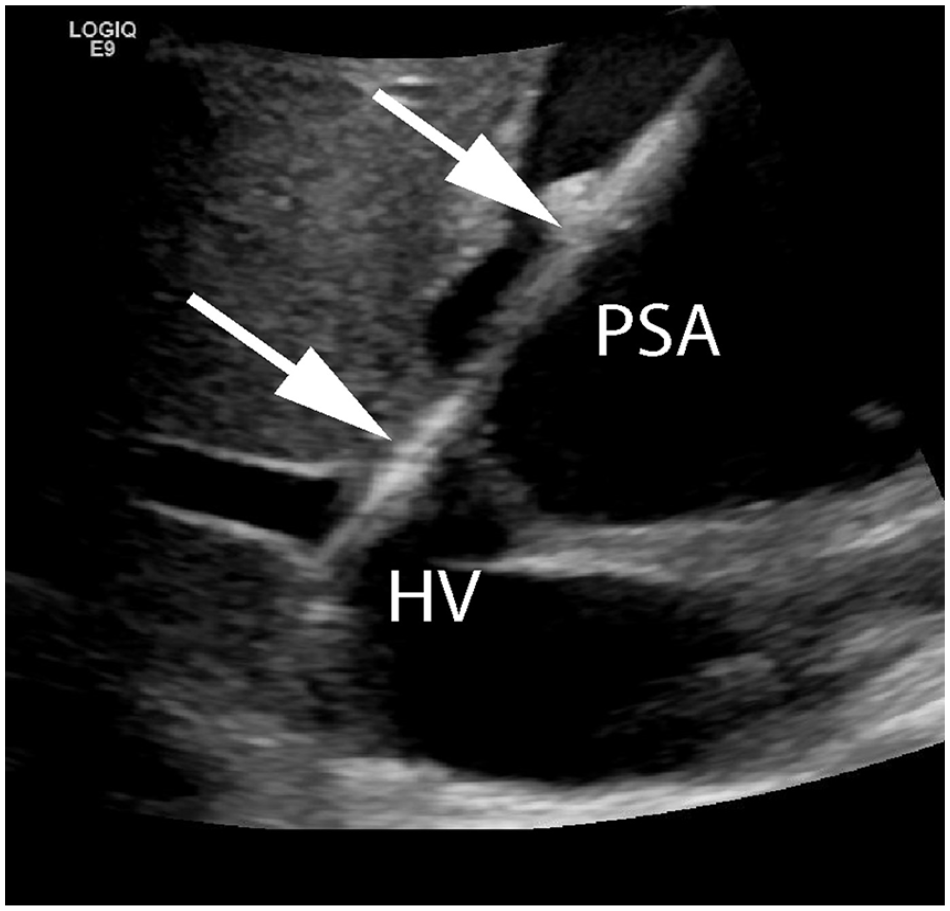
Sonographic gray-scale image shows a wire (arrows) crossing the fistulous communication between the pseudoaneurysm (PSA) and middle hepatic vein (HV).
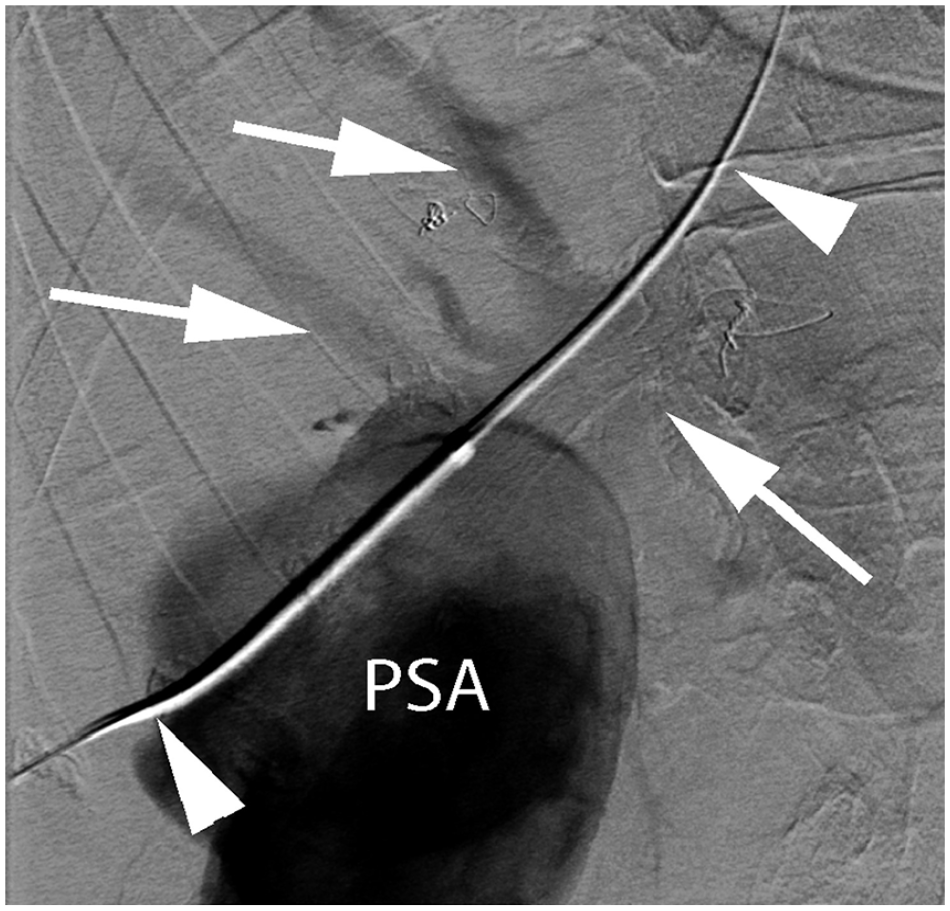
Fluoroscopic image shows a 0.35 mm wire (arrowheads) traversing the fistulous communication between the PSA and middle hepatic vein (vein and branches designated by arrows). PSA, pseudoaneurysm.
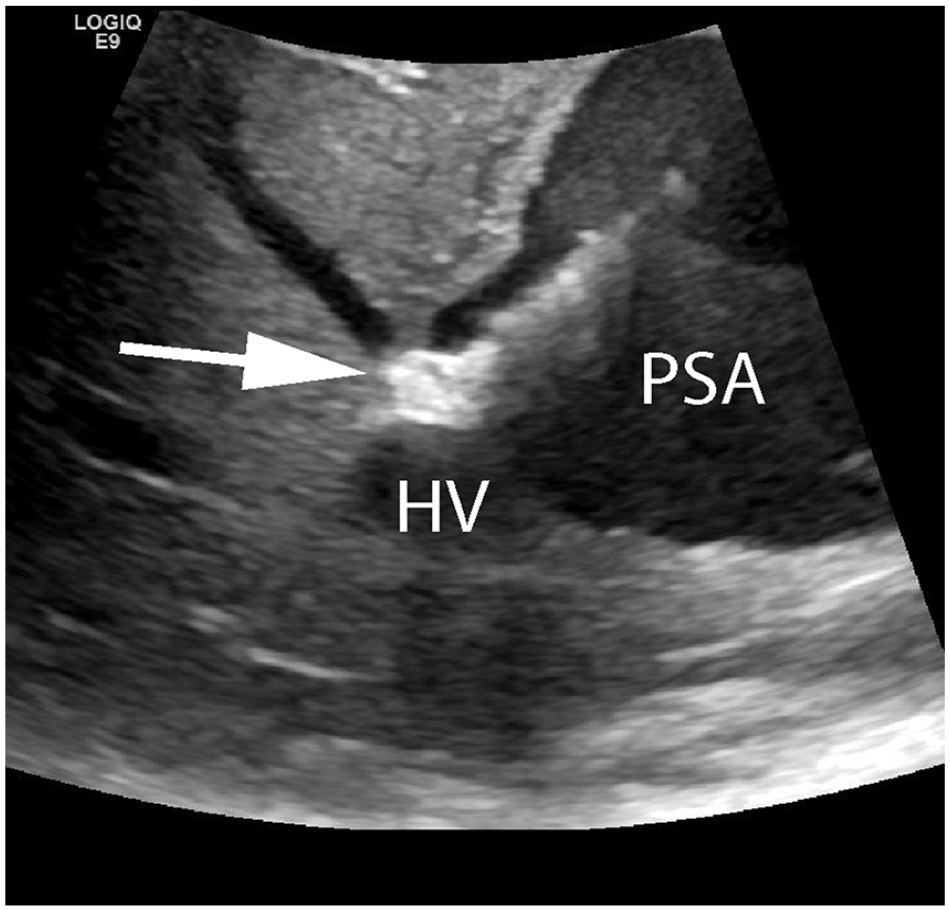
Sonographic gray-scale image shows the vascular occluder device (arrow) placed across the fistulous communication between the pseudoaneurysm (PSA) and middle hepatic vein (HV).
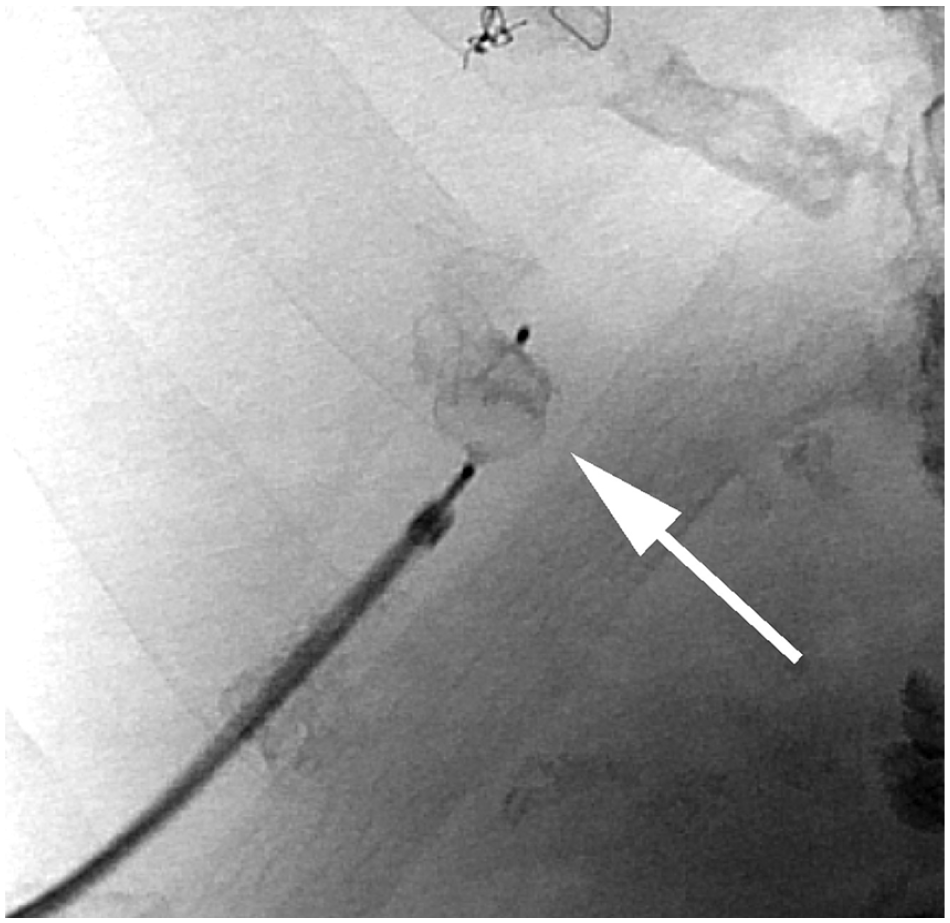
Fluoroscopic image shows the vascular occluder device (arrow) immediately following deployment from the vascular sheath.
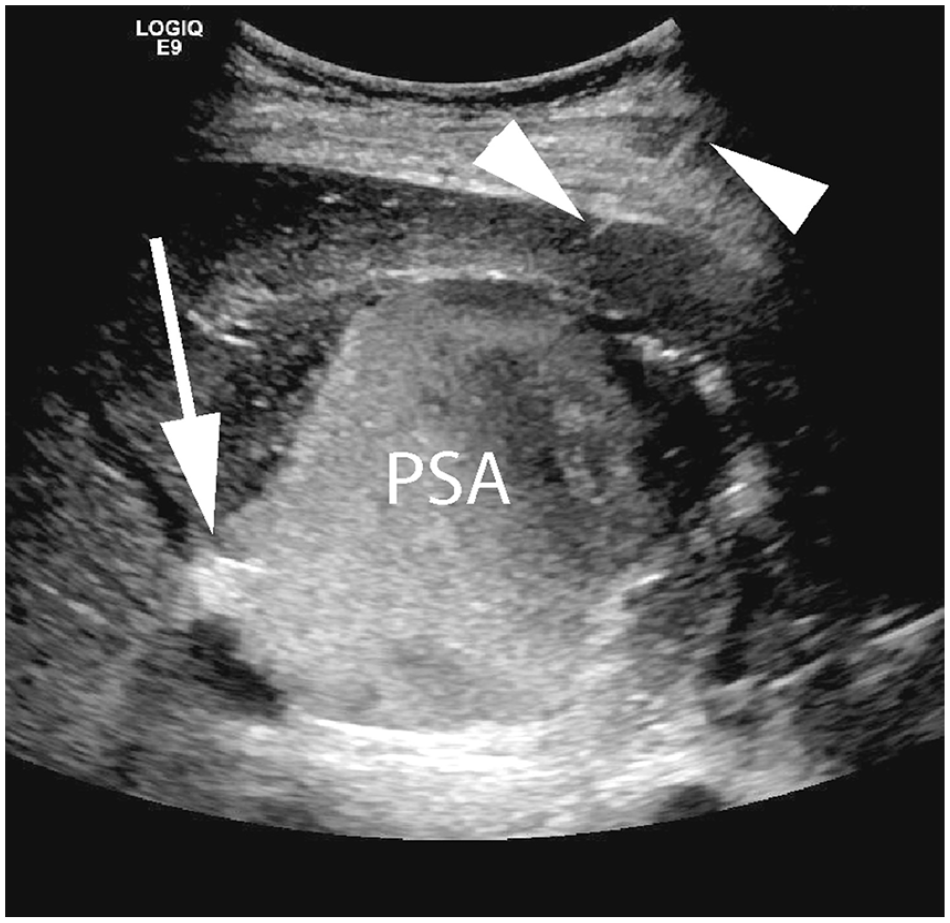
Sonographic gray-scale image obtained following successful thrombin injection shows that the pseudoaneurysm (PSA) is filled with acute, echogenic thrombus. The vascular occluder plug (arrow) and injection needle (arrowheads) are partially visualized.
Additional sonograms the following day and 11 days post treatment (Figure 11) showed persistent thrombosis of the pseudoaneurysm. With last known follow-up three months following treatment, the patient has not experienced any related medical complications.
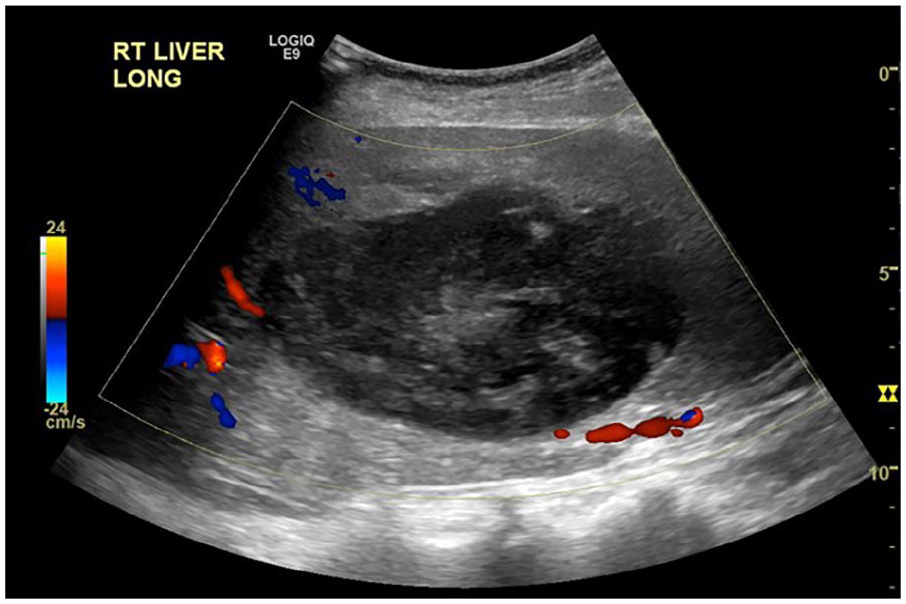
Sonographic color Doppler image obtained 11 days after right hepatic lobe pseudoaneurysm treatment showing persistent thrombosis.
Discussion
Initial differential diagnoses for the case described above included hepatic artery pseudoaneurysm and arteriovenous fistula. AVFs are the abnormal connection between an artery and a vein. Spectral Doppler interrogation of AVFs classically display low resistive arterial flow proximal to the fistula, high resistive arterial flow distal to the fistula, and pulsatile venous flow in the communicating vein (Figure 12). The patient described herein presented with a complex, atypical pseudoaneurysm such that while no direct AVFs were discovered, venous fistulas were found in communication with the HAP. Spectral Doppler interrogation of venous fistulas can produce continuous, high-flow venous waveforms (Figure 4).
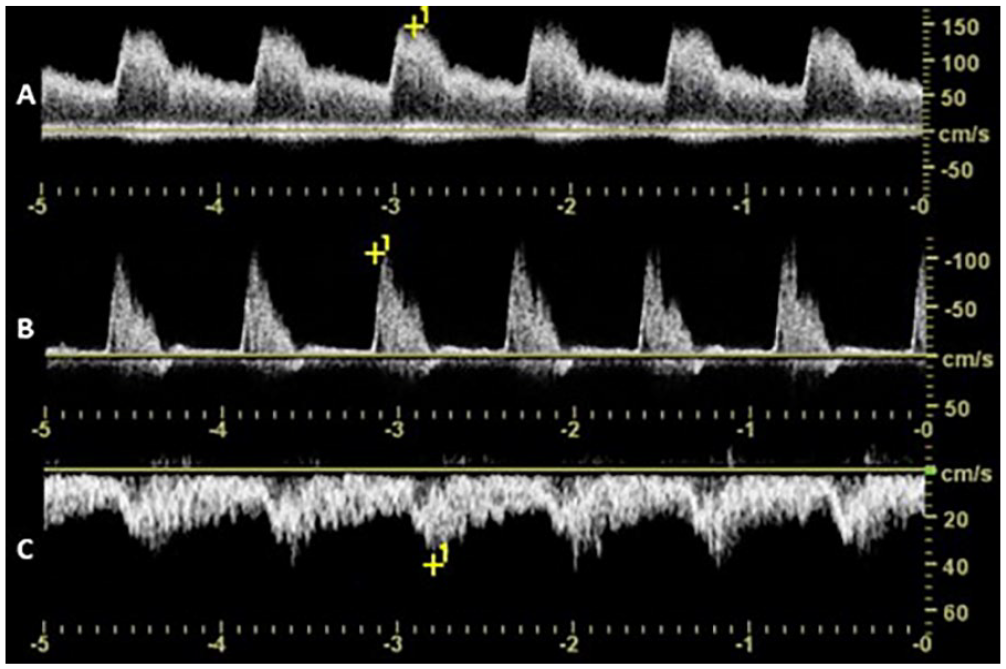
Spectral Doppler waveforms depicting classic flow characteristics of arteriovenous fistulas (AVFs). Low resistive arterial flow is observed proximal to an AVF (A), while high resistive arterial flow is detected distal to an AVF (B). Pulsatile flow is appreciated in the communicating vein (C).
Pseudoaneurysms can occur when there is damage or weakening to the inner layers of an arterial wall which consequently allows a blood-filled cavity to develop. This cavity is still surrounded by the adventitial arterial layer and remains in direct communication with the artery (Figure 13). Spectral Doppler interrogation of a pseudoaneurysm neck classically demonstrates a to-fro blood flow pattern (Figure 14) and a color Doppler “yin-yang” sign (Figure 2) is frequently appreciated. The “yin-yang” sign depicts the swirling, bi-directional blood flow in a pseudoaneurysm. These sonographic color and spectral Doppler characteristics aid in diagnosis and allow the patient to undergo prompt treatment.
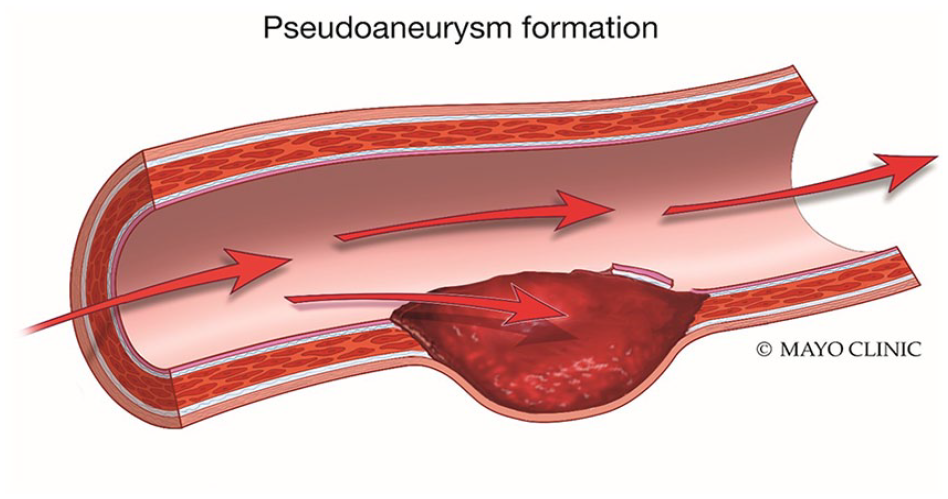
Medical diagram depicting the formation of a pseudoaneurysm from the damaged inner arterial wall layers. Used with permission of Mayo Foundation for Medical Education and Research, all rights reserved.
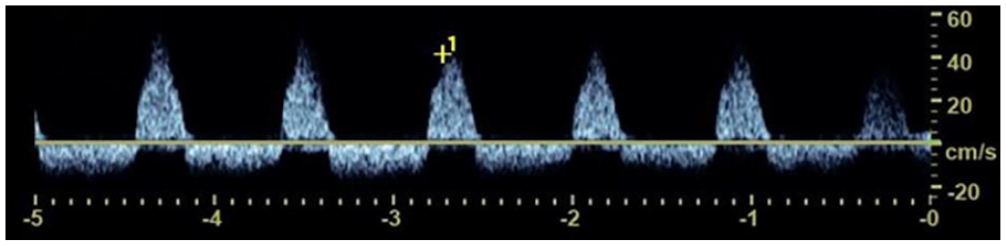
Spectral Doppler waveform depicting the bidirectional to-fro flow classically visualized in the neck of a pseudoaneurysm.
Sonography of the liver is typically performed from an intercostal or subcostal approach. The optimal scanning window is determined based on the patient’s anatomy and avoidance of overlying structures that may impede insonation. Typically, the liver is best evaluated with the patient in the left posterior oblique or supine position, but patient condition can limit mobility and visualization. Interventional procedures utilizing sonography provide real-time imaging guidance for wire or needle placement. In the IR setting, sonographers provide a supportive role to the interventional radiologist. Sonographers prepare the transducer with a sterile probe cover and assist with image optimization/acquisition throughout the procedure, often for both diagnostic purposes and to guide directed interventions.
HAPs are rare and potentially life-threatening if left untreated. These pseudoaneurysms can arise as a surgical complication of liver transplantation 4 or as a result of an underlying inflammatory process like polyarteritis nodosa.1,5 HAPs have also occurred in patients with a preceding diagnosis of fibromuscular dysplasia 6 and pancreatic pseudocysts. 1 HAPs can often be the result of blunt force or iatrogenic trauma to the liver, the latter consisting primarily of liver biopsies 1 and percutaneous biliary interventions. 7 In addition to HAPs, liver trauma can also cause additional vascular damage to the liver. Trauma is one of the most common causes of acquired hepatic fistulas. 8 Sources of other fistulous connections may be due to cirrhosis, hepatic neoplasms, or congenital vascular malformations. 9
HAPs account for 50% of all hepatic artery aneurysms. 7 Patients with true mesenteric aneurysms are frequently treated when diameters exceed 2 cm and often have arteriosclerosis as an underlying condition. 5 While hepatic aneurysms have been identified in patients with Marfan syndrome, they are not clearly known to be associated with the disease. 10 Marfan syndrome is a genetic disorder that affects the connective tissues of the body. Hallmark vascular abnormalities associated with Marfan’s include aortic dissection, ascending aortic aneurysm, and aortic root dilation. 11
The presentation of patients with HAP varies greatly. A subset of patients may present with symptoms of abdominal pain, hematemesis, jaundice, anemia, hypovolemia, or melena.2,3 Even though these symptoms have been associated with HAPs, the majority of cases are interestingly asymptomatic. HAPs arising from blunt trauma may go undetected at first. They may present incidentally at follow-up or emergently as rupture. Batur et al 12 describes the case of a 34- year-old man who died from complications of a HAP discovered 1.5 months after his blunt trauma. Rupture is the most acute and life-threatening complication of HAPs. Intrahepatic pseudoaneurysms frequently rupture into the biliary system resulting in Quinke’s triad of jaundice, hemobilia, and pain; extrahepatic pseudoaneurysms most frequently rupture into the peritoneum. 13 Portal hypertension, variceal bleeding, and high cardiac output failure can occur if cases are further complicated by fistulous connections. 8
There are several treatment options for HAPs. Standard treatments can include angiographic embolization, angiographic stent placement, surgical ligation, or resection. 13 The overall objective is to maintain perfusion of the liver while reducing the risk of rupture. The role of sonographic imaging in treatment is used at the discretion of the interventional radiologist or surgeon. Treatment via sonographic-guided percutaneous thrombin injections have been performed in select cases, and reduce the risk of tissue infarction.14,15 For cases that involve hepatic fistulas, both surgical and transarterial embolization treatments are typically considered. 9 However, the deployment of vascular plugs percutaneously has also been achieved. 16 Contraindications to angiographic treatment include contrast allergies to iodine, severe renal disease, and complicated vascular anatomy such as occluded feeding vessels or occluded femoral vessels for access. 7 Complications can include hepatic abscess formation, gallbladder ischemia, and progression of cirrhosis and liver failure. 7 Surgical treatment contraindications are typically based on patient comorbidity.
Conclusion
Hepatic artery pseudoaneurysms are a rare vascular pathology that most commonly arise as a complication of liver trauma, surgery, or inflammatory processes. While some patients present in acute distress, many are asymptomatic and present incidentally. The potential of pseudoaneurysm rupture remains a critical risk, and each patient should be evaluated and treated on a case-by-case basis. Sonography is frequently one of the first imaging modalities used to aid in diagnosis. Pseudoaneurysm interrogation with color and spectral Doppler reveal to-fro flow within the neck and the swirling “ying-yang” sign. These blood flow patterns are specific to pseudoaneurysms and diagnostic when appreciated sonographically. While the use of sonography during treatment is not applicable in every case, it has the potential to add considerable value in many circumstances and lead to a life-saving outcome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
