Abstract
We report a case of renal allograft dysfunction due to plasma cell-rich acute rejection (PCAR), which is an uncommon clinical entity with a wide range of differential diagnoses. Extensive diagnostic workup, treatment approach and outcome are discussed and we provide a brief summary of the current management dilemma. Nevertheless, the diagnosis of PCAR portends a poor prognosis and therefore timely diagnosis and intensification of treatment is crucial to prevent disease progression.
Introduction
Despite the continuing effort to reduce the rate of acute rejection episodes and to optimize allograft survival with potent immunosuppressive drugs, plasma cell-rich acute rejection (PCAR), an uncommon type of acute rejection characterized by the presence of mature cells comprising more than 10% of the inflammatory cells infiltrating renal allograft, 1 is associated with poor long-term outcome in renal transplantation and little is known about the management of this condition. We describe a case of biopsy-proven PCAR presented with acute renal allograft dysfunction and the clinical outcome of the treatment.
Case report
A 58-year-old man with past medical history of end-stage renal disease secondary to chronic glomerulonephritis with subsequent overseas living unrelated kidney transplantation a year prior to admission, presented with worsening renal allograft function. Notably he had an episode of borderline acute rejection (Banff score of t1, i1, v0, g1, c4d-negative) previously for which he was treated in a private institution with pulse intravenous (IV) methylprednisolone, with maintenance immunosuppression of everolimus 0.75 mg BD, mycophenolate mofetil (MMF) 500 mg BD and prednisolone 4 mg OM. Baseline serum creatinine was 230 µmol/L. Physical examination was unremarkable with no tenderness on renal allograft. Laboratory investigation demonstrated an elevated serum creatinine of 262 µmol/L, normal-range procalcitonin and trough everolimus level of 3.0 µg/L. Urinalysis showed pyuria with proteinuria of 0.62 g/day. Virology screen included Cytomegalovirus and BK virus which were negative. Doppler ultrasonography study of renal allograft showed mildly echogenic appearance with normal limit resistive index and no evidence of transplant renal artery stenosis. A transplant kidney biopsy was performed which showed diffuse interstitial infiltration of plasma cells mixed with lymphocytes, with some neutrophils within tubular lumens (Banff score of t2, i3, g1, v0, ptc0, ci1, ct2, cg1, mm1, cv1, c4d-negative; see Figure 1A). Further immunohistochemistry tests revealed both B- and T-cell markers (CD20 and CD3) were expressed in the lymphoplasmacytic infiltrate with predominant T lymphocytes (Figure 1B). Plasma cell markers (CD138) were present with polyclonal kappa and lambda chains detected. Immunostain for IgG4, EBERish and SV40 large T antigen was negative. The morphologic and immunohistochemical findings, correlating with clinical features, were not suggestive of a post-transplant lymphoproliferative disorder (PTLD), and a CT scan of chest and abdomen did not reveal any infiltrative lesion. A diagnosis of PCAR was made. A finding of neutrophils within the tubular epithelium was suggestive of concomitant urinary tract infection, which was consistent with subsequent urine culture of
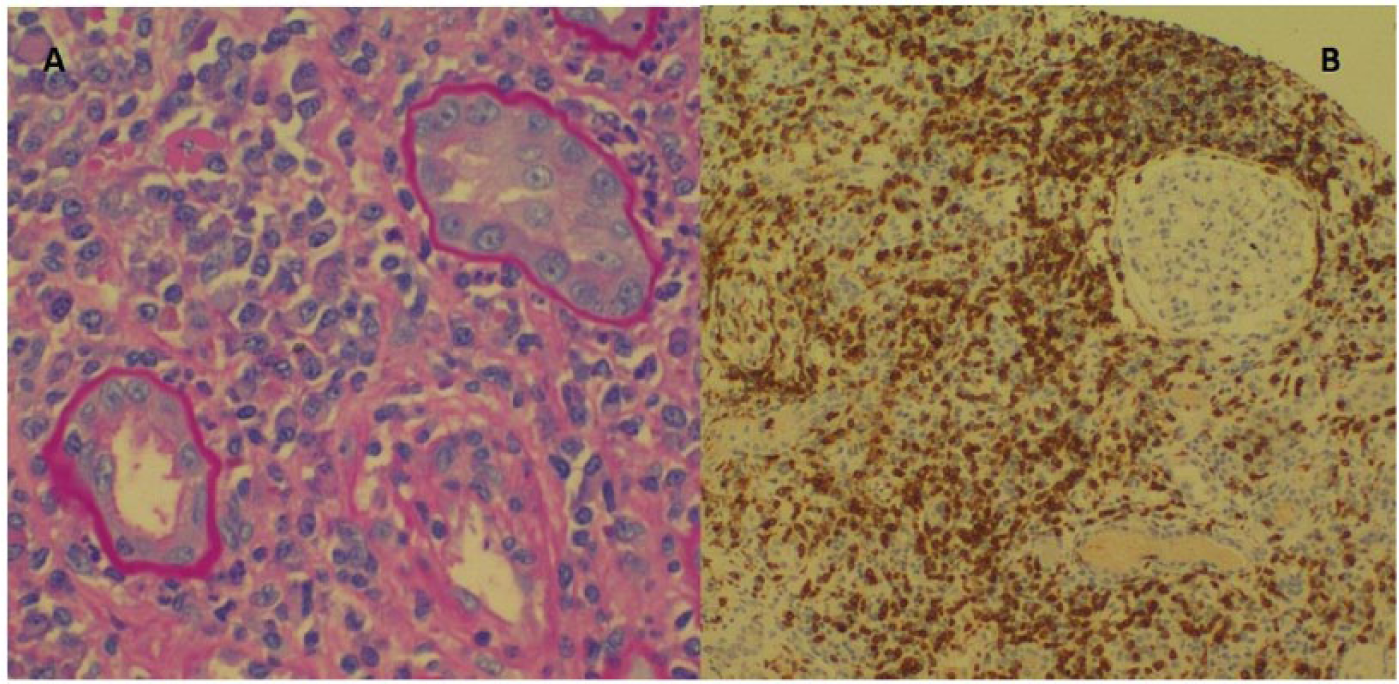
(A) Transplant renal biopsy showed diffuse interstitial infiltration of plasma cell mixed with lymphocytes, with occasional neutrophils seen within tubular lumens. (B) Immunohistological stain (Brown stain) showing predominantly positive T-cell marker (CD3).
Discussion
PCAR is an uncommon clinical entity independent of acute renal allograft rejection which is observed in approximately 5% patients with biopsy-proven acute rejection. 1 The time of onset is variable. In a case series of 27 cases over 10 years, 45% of PCAR occurred early (<6 months, mean 2.5 months) and 55% occurred late (> 6 months, mean 37.3 months) post renal transplantation. 2 Apart from PCAR, plasmacytic infiltration has been reported to be associated with viral infection, 1 antibody-mediated rejection (AMR), 3 PTLD 4 and patient non-adherence to immunosuppressive therapy. 2 Interestingly, the manifestation of PCAR can also mimic IgG4-related kidney disease. 5 Risk factors associated with PCAR included female gender, age >50 years, deceased donor kidney transplantation and simultaneous pancreas–kidney transplantation. 6 In a case series by Martin et al., plasma cell infiltration was reported to be associated with development of chronic allograft dysfunction. 7 Recent literature also suggested that detection of donor-specific antibodies (DSA) might be useful in determining whether the PCAR is of a subtype of AMR, which can be refractory to immunosuppression treatment with unfavorable outcome.3,6 In our case, however, DSA detection was not available in view of unknown donor profile.
Therapy for PCAR has not been entirely established and previous reports showed varied outcomes with treatment of baseline immunosuppression intensification, IV methylprednisolone pulses, IV immunoglobulin and TPE. Use of thymoglobulin, rituximab and bortezomib has been reported in PCAR with T-cell, B-cell predominance and monoclonal gammopathy, respectively.
8
In our case, although the clinical picture was consistent with PCAR, the effect from concomitant urinary tract infection could not be entirely excluded, and therefore the clinical improvement could be from both intensified immunosuppression as well as antimicrobial treatment of underlying infection. Nevertheless, the detection of PCAR has important prognostic relevance. Gartner et al. in his analysis of 109 renal transplant recipients over 13 years, found that the group rich in plasma cells (
In summary, the diagnosis of PCAR portends a poor prognosis with high rate of graft loss or severe allograft dysfunction,1,4,9 and therefore timely diagnostic workup in close collaboration with histopathologist followed by intensification of treatment is crucial to prevent disease progression. The issue in PCAR treatment remains unresolved and further cases with treatment outcome should be reported.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This report received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
