Abstract
Mitral valve (MV) pathology, along with its associated peripheral anatomy, is one of the most common categories of heart disease. Two-dimensional (2D) transthoracic echocardiography (TTE) plays a prominent role in the detection and management of various types MV disease, specifically mitral annular disjunction (MAD). MAD is defined as a structural abnormality of the mitral annulus fibrosus, causing a noticeable gap between the atrial wall-mitral valve junction and the basilar portion of the left ventricular free wall (LV). The integral role that cardiac sonographers play in identifying MAD cannot be underscored, as well as the risk of easily overlooking this unique abnormality. Often associated with mitral valve prolapse (MVP), accurate documentation of MAD amid an echocardiographic study can have positive implications on patient prognosis. This case series highlights the echocardiographic characteristics of MAD, to raise awareness of this often forgotten feature of myxomatous mitral valves, which can indeed cause adverse patient outcomes. It is important to demonstrate correlational features with additional imaging techniques such as magnetic resonance imaging (MRI) and computed tomography (CT).
Mitral valve prolapse (MVP) is a common finding that is found in 2% to 5% of the world’s population.1–3 Most patient cases go undetected and are usually discovered incidentally with diagnostic imaging techniques. Closely associated with myxomatous mitral valve (MV) disease and MVP, mitral annular disjunction (MAD) is a rarely documented phenomenon that is defined as a structural irregularity where a clear, fibrous separation exists between the left ventricular muscle and MV annulus. 4 However, in a recent study, MAD has been detected in patients with normal mitral valve structure. 5 Because of its ability to be detected with transthoracic echocardiography (TTE) and its clinical implications, sonographers play a vital role in the identification and assessment of MAD. Due to the potential risk that this abnormality poses to patients with myxomatous valve disease, it is important to review this challenging echocardiographic finding and by doing so, provide patients with the best possible outcome.
A Case Series
A case study is a comprehensive study of a single patient encounter with a goal of applying these techniques and findings to a larger cohort and translating these findings to naturally occurring events or phenomena, with an everyday context. 6 This collection of cases, describe four different patients with MAD and associated MVP, assessed with two-dimensional echocardiogram (2D) and TTE. The aim of this case series is to provide examples of this unique and under documented pathology through detailed encounters of each patient’s clinical history and sonographic images of MAD. The sonographic images provide measurements of the annular/posterior LV wall separation, to guide sonographers on what to look for and how to measure potential future cases. The summation of these patient cases and their subsequent discussion, shed light on the implications of MAD and what cardiac sonographers should expect, when discovering this pathology.
Patient Case #1
The first case is a 50-year-old female patient with a history of poorly documented MVP. A normal rhythm and rate were detected with no murmurs. She was referred for uncontrolled hypertension (HTN), based on her own personal documentation of systolic numbers increasing steadily over the past few months, due to stoppage of Lisinopril. The patient was prescribed Lisinopril and a 2-D TTE was ordered to assess cardiac anatomy and function and to fully define the patient’s history of MVP. The 2-D TTE demonstrated normal LV structure and systolic/diastolic function. LV wall motion was described as normal with a left ventricular ejection fraction (LVEF) of 65% via biplane Simpsons. There was borderline MVP noted with 2 mm of bowing of the leaflets into the left atrium (LA) cause just trace mitral regurgitation (MR). Additionally, the sonographer measured a leaflet thickness of 4.9 mm. Notably, from the parasternal long-axis (PLAX) window, there was noticeable MAD creating a free wall separation measuring 1.04 cm in size (See Figure 1).
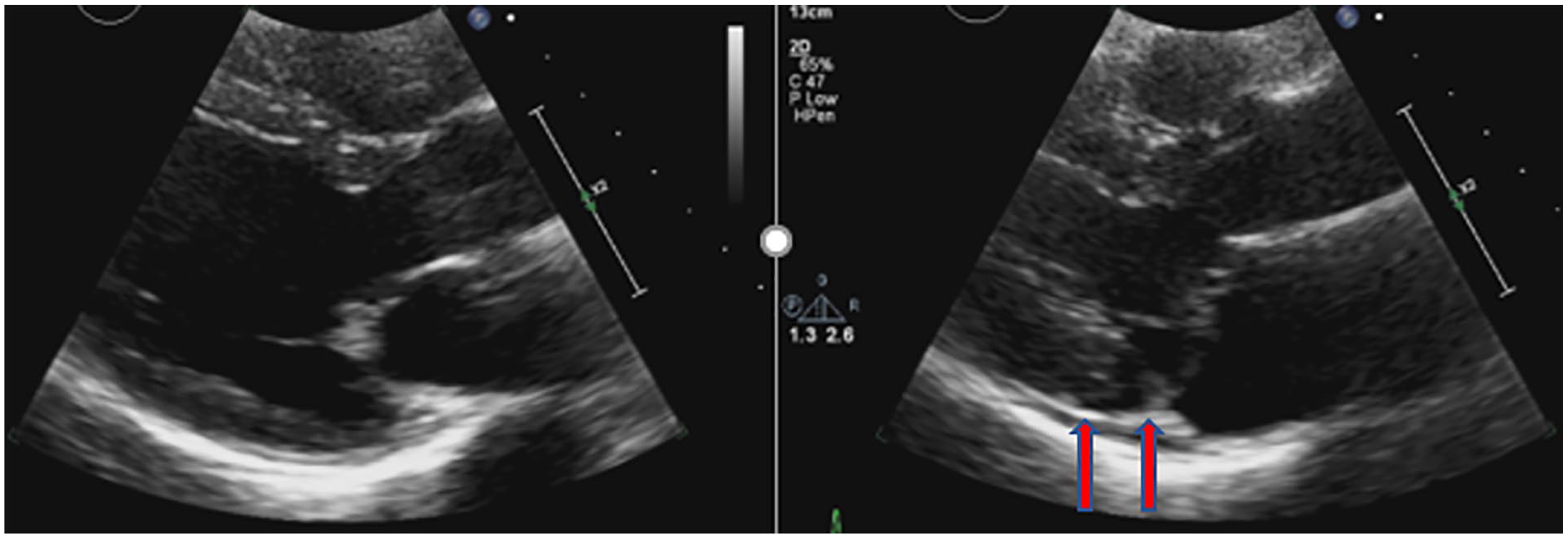
Case study #1 example of a parasternal long-axis on transthoracic echocardiography in diastole (left) and systole (right) demonstrating a separation between the left ventricular myocardium and mitral valve annulus indicative of mitral annular disjunction (see red arrows).
Patient Case #2
A 20-year-old male presented to the emergency department (ED) for continuous sternal and left-sided chest pain (CP) and shortness of breath (SOB) that lasted for 2 months. No palpitations were reported throughout the period of these symptoms and no cardiac murmurs were noted. As a child, the patient was seen for pectus excavatum and was told to seek medical attention if symptoms of pain persist as an adult. Upon seeking surgical evaluation due to his worsening symptoms, computed tomography (CT), electrocardiogram (EKG), 2-D TTE, and pulmonary function test (PFT) were ordered.
The CT demonstrated findings of severe pectus excavatum with a corresponding Haller index of 3.98. This value is the diameter of transverse chest measurements divided by internal antero-posterior (AP) internal thoracic diameter measured with 2-D TTE or CT. 7 The 2-D TTE revealed a flattened right ventricle (RV) and LV due to the pectus excavatum. Normal LV structure and function was noted with a LVEF of 56% by biplane Simpsons. MV interrogation demonstrated a mildly myxomatous structure with borderline MVP and mild MR. Upon closer interrogation of the MV, the cardiac sonographer measured a significant separation between the LV and MV annulus measuring 0.60 cms, which was later deemed to be MAD (See Figure 2). The characteristics of MAD in this patient was only visualized during systole as it appeared normal in diastole.
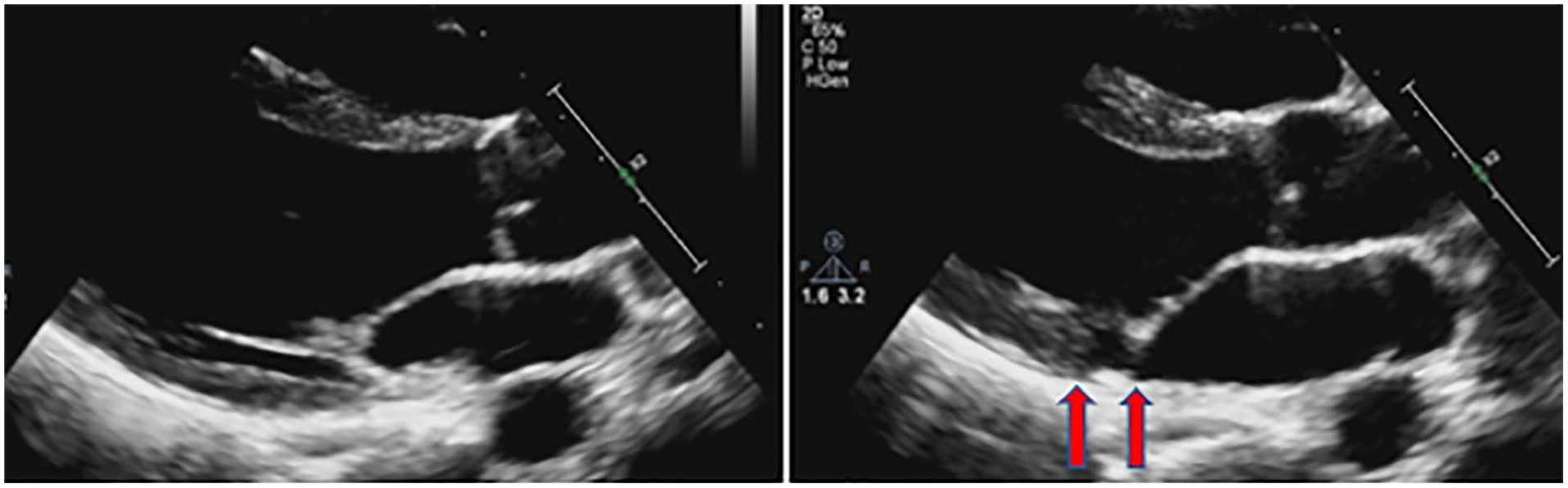
Case study #2 example of a parasternal long-axis on transthoracic echocardiography in diastole (left) and systole (right) demonstrating a separation between the left ventricular myocardium and mitral valve annulus indicative of mitral annular disjunction (see red arrows).
Patient Case #3
A 74-year-old male with known arrhythmias, normal blood pressure, and no chest pain presented to his cardiologist with syncope while walking his dog. The patient had a history of chronic obstructive pulmonary disease (COPD) and was an avid smoker. 2-D TTE revealed normal LV structure and function with an LVEF of 68% measured by bi-plane Simpsons. Interrogation of the MV demonstrated mild bileaflet prolapse with trace, late systolic, central MR. Additionally, the cardiac sonographer documented and measured an area of LV/annulus separation appearing as MAD measuring).70 cm (See Figure 3).
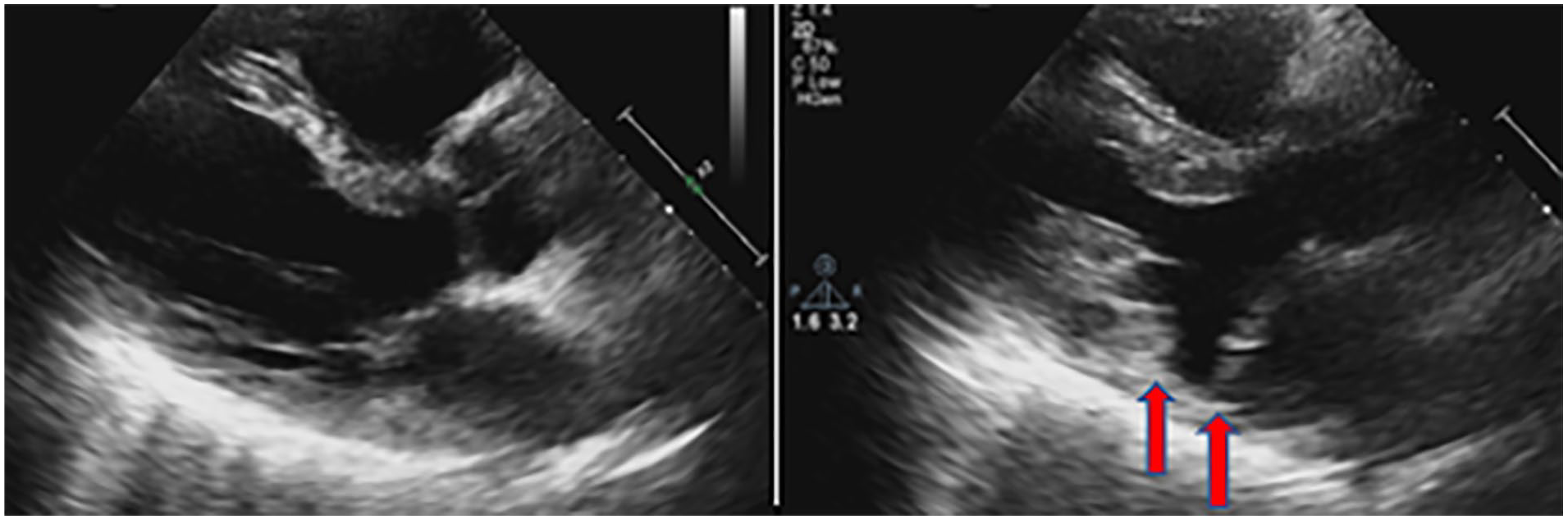
Case study #3 example of a parasternal long-axis on transthoracic echocardiography in diastole (left) and systole (right) demonstrating a separation between the left ventricular myocardium and mitral valve annulus indicative of mitral annular disjunction (see red arrows).
Patient Case #4
A 74-year-old male with known history of essential hypertension, hyperlipidemia current every day smoker with a 120 per year pack history and diabetes mellitus. Presented to the ED with a compliant of intermittent left-sided chest discomfort that has been ongoing for some time. The discomfort is not associated with activity or rest, no aggravating or alleviating factors. A normal sinus rhythm with a first-degree AV block, and 2/6 SEM murmur at the apex. The 2D TTE, completed for chest discomfort, had an EF of 55% to 60% and the MV integration was unremarkable. However, there was an area of suspicious separation between the annulus and posterior LV free wall, which was not clearly distinguishable (
Case study #4 example of a parasternal long-axis on transthoracic echocardiography demonstrating a suspicious area of potential mitral annular disjunction along the posterior left ventricular free wall.
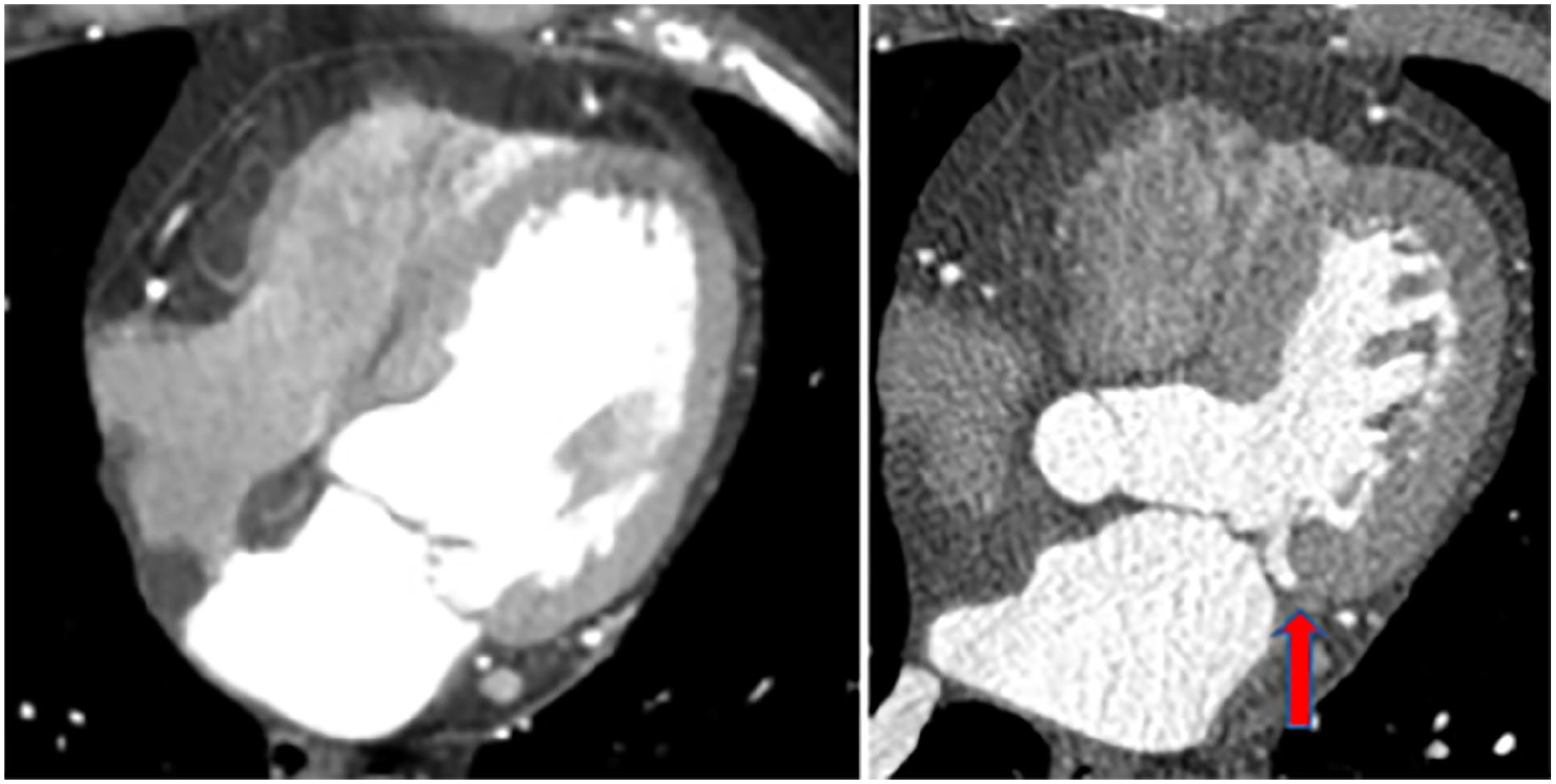
Case study #4 example of a cardiac computed tomography in diastole (left) and systole (right) confirming the presence of mitral annular disjunction (see red arrow).
Patient Case #5
A patient case report is presented of a 54-year-old male with a past medical history of hyperlipidemia, hypertension, non-rheumatic MVP with MR, and obstructive sleep apnea. Through a culmination of efforts in testing utilizing multiple imaging modalities, it was determined that the patient has mitral valve disjunction. His baseline EKG was noted to have inferior T-wave inversion without additional abnormalities. A routine TTE demonstrated an LVEF and right ventricular ejection fraction (RVEF) of 59 and 48%, respectively. There were no LV regional wall motion abnormalities (RWMA) and left ventricular diastolic dysfunction (LVDD), classified as grade II. A 2-D and color Doppler inspection of the MV showed posterior MVP with mild to moderate MR. In the PLAX window, there was an area of clear separation between the LV and mitral valve annulus (See Figure 6). Global longitudinal strain (GLS) imaging showed a decrease in basal inferior strain of -13 (See Figure 7). A transesophageal echocardiogram (TEE) confirmed similar findings of potential MAD in addition to a flail leaflet (See Figure 8). A magnetic resonance imaging examination (MRI) was ordered for further investigation of possible MAD.
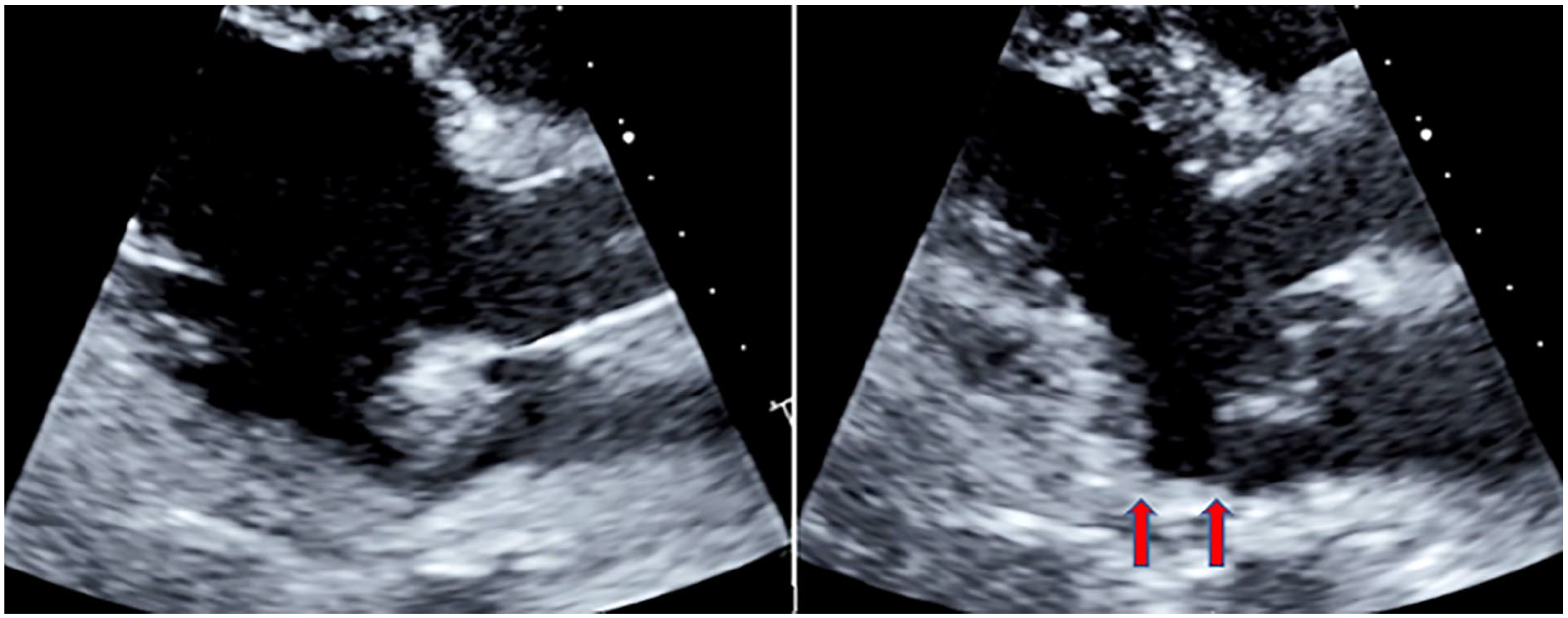
Case study #5 example of a parasternal long-axis in diastole (left) and systole (right) demonstrating a separation between the left ventricular myocardium and mitral valve annulus indicative of mitral annular disjunction (red arrows).
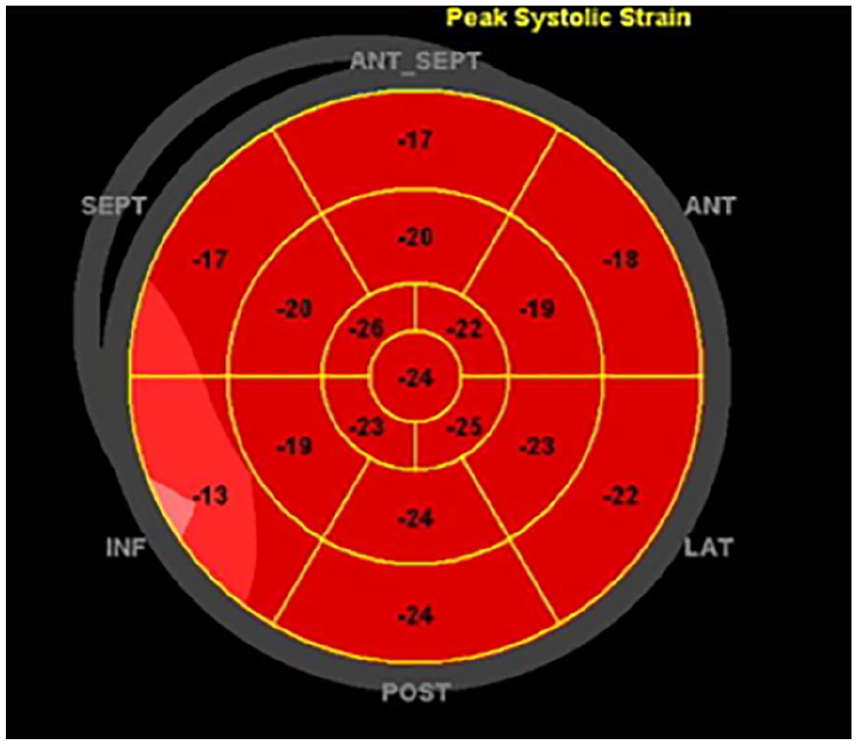
Case study #5 left ventricular global longitudinal strain map depicting a decrease in basal inferior strain pattern in a patient with confirmed mitral annular disjunction. ANT, Anterior; INF, Inferior; LAT, Lateral; POST, Posterior; SEPT, Septal.
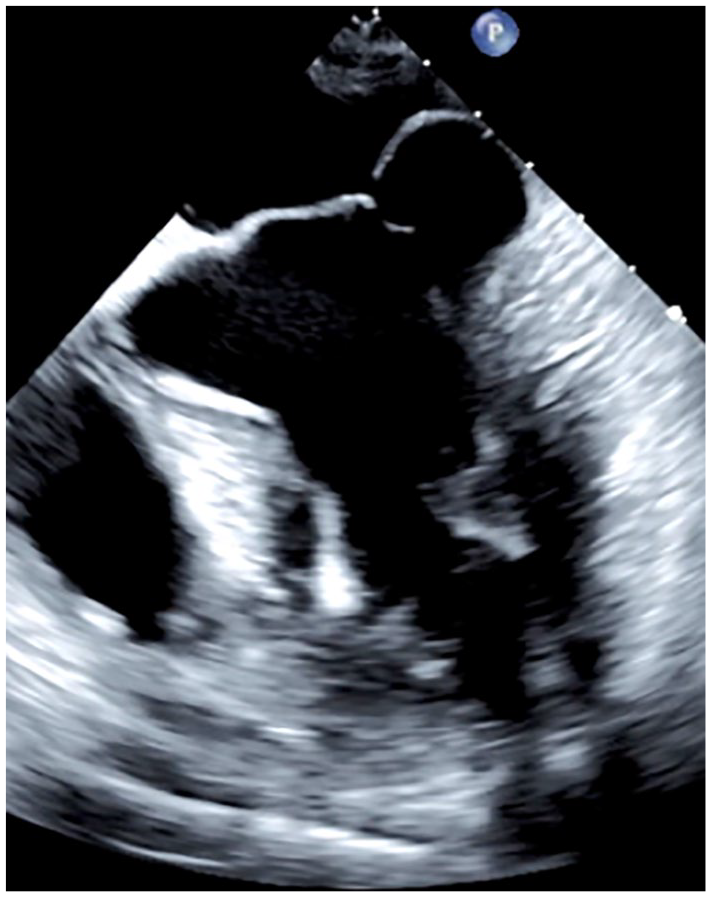
Case study #5 transthoracic echocardiography demonstrating the area of annular disjunction where the mitral valve annulus and left atrium attachment site is significantly disconnected from the basal left ventricular muscular structure.
The MRI demonstrated severe posterior MVP (P1 and P2) with eccentric anterior direction from the A2—P2 commissures. It was noted that there was a significant gap adjacent to the mitral annulus associated with features of MAD, which measured 4.5 mm inferiorly (
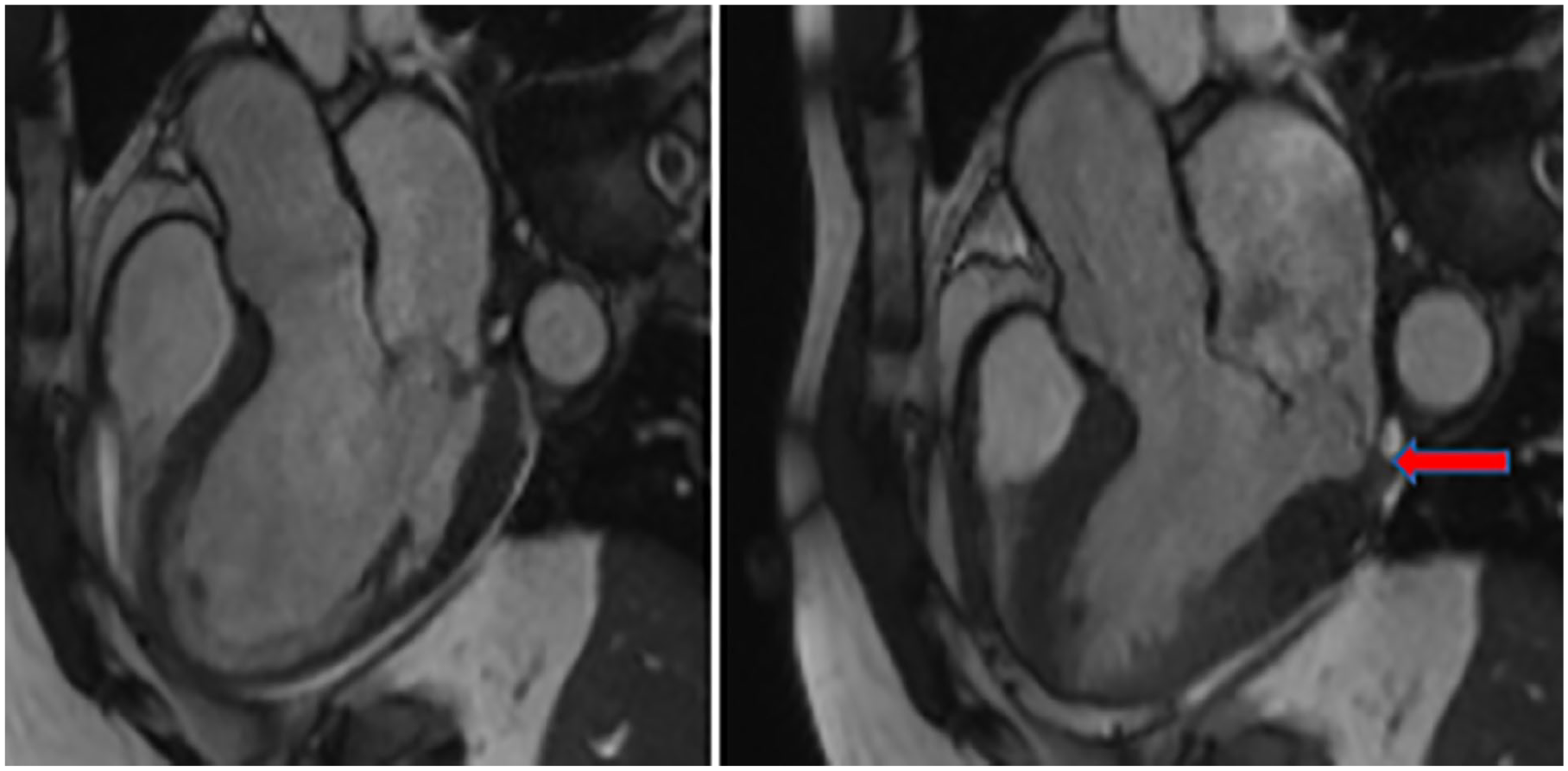
Case study #5 correlating and confirming the presence of mitral annular disjunction (see red arrow) with magnetic resonance imaging which was initially discovered by transthoracic echocardiography.
Discussion
MV disease is a frequent cause of heart failure and mortality worldwide. Through intensive study and empirical evidence, the MV been found to be susceptible to active changes, leaving it vulnerable to increased disease patterns over time. 8 The mitral annulus anatomy is a specific area of the heart that is subject to hemodynamic and mechanical forces, during the cardiac cycle. One dynamic structural abnormality of the mitral annulus that does not get the attention it deserves is MAD. MAD is often described as a clear separation between the MV annulus and the basal portion of the posterior wall of the LV. In normal mitral annulus function, LV longitudinal contraction pulls the lateral and medial, creating uniform curvature starting at the basal posterior wall. This paradoxical annular enlargement and flattening during systole conforms with the movement of the LA. 9 Typically, this disjunctive area of the MV annulus predominantly involves the P1 and P3 scallops of the posterior mitral leaflet and will classify as MAD with a disjunctive distance of >2 mm.10,11
TTE and TEE are imaging modalities that allow great spatial and temporal resolution to identify this annular separation/gap. Cardiac sonographers have a unique and important responsibility in identifying this lesion and relaying this information to reading cardiologists. Because mitral valve prolapse (MVP) is closely correlated with this MAD, sonographers maintain the responsibility of distinguishing between these two pathologies or noting both of their presence at the same time, while also measuring the given size of the area of disjunction. 12 Dejgaard et al found that 78% of patients with MAD had associated MVP. In these same patients, with MAD, the magnitude and extent of annular disjunction was positively associated with increasing degrees of MR. In a similar 2020 study, MAD was present in 24% of participants with severe MR. 11 Because of this, it is paramount that sonographers keep abreast of MR quantification techniques to accurately quantify this associated pathology. Regarding TEE, assessment and documentation of MAD with TEE is a vital pre-surgical step for optimal MV repair as a modified repair technique can correct the disjunctive area, which has seen positive results in difficult cases. 13
Pseudo MAD vs True MAD
The history and documentation of MAD is comparatively new when compared to the presentation and documentation of other structural abnormalities of the heart. In 1981, Bharati et al
14
first pronounced this phenomenon in a case report within the
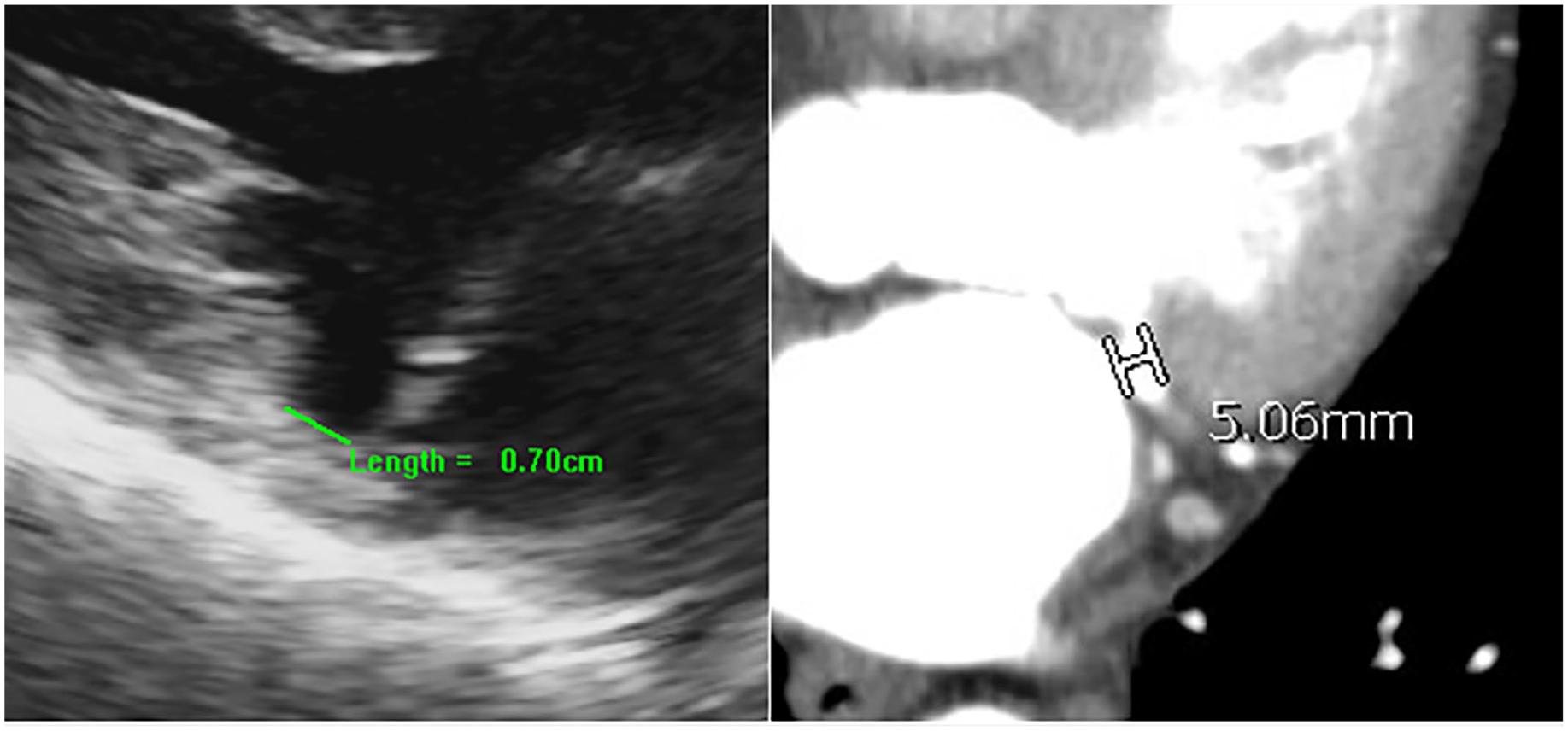
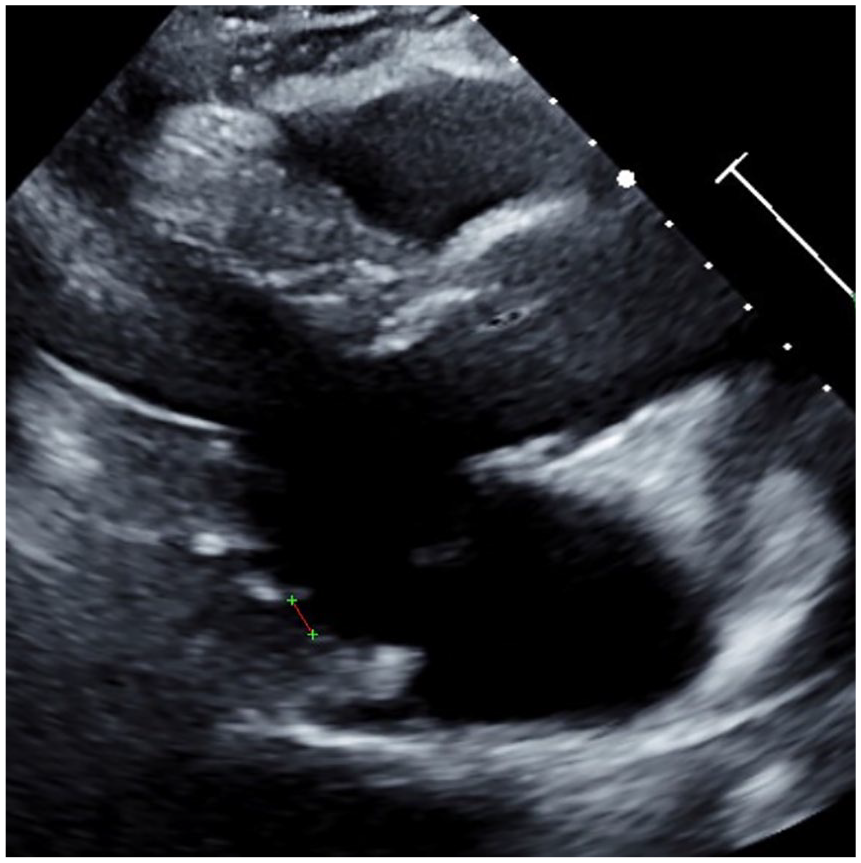
A zoomed parasternal long-axis on transthoracic echocardiography and computed tomography example demonstrating how to measure the separation between the mitral annulus and left ventricular myocardium, known as mitral annular disjunction.
CT and MRI Correlation
The implementation of CT characterizes a largely used imaging modality for structural heart disease. 17 When defining the characteristics of MAD with CT, increased spatial resolution and highly noninvasive acquisition facilitate and enables the reproducible assessment of MAD. Specifically, a CT clearly defines the MV apparatus to include the leaflet/annulus relationship during the cardiac cycle.18–25 Recent studies have demonstrated that MAD can be diagnosed and assessed on CT both retrospectively with EKG-gated CT data, but also prospectively with high-pitch CT angiography acquired at end-systole. 26 In addition to TTE and CT, MAD has routinely been detected with a MRI. Although MAD is commonly diagnosed with sonography, a MRI maintains a higher sensitivity for detecting this abnormality. Furthermore, additional information is gleaned from a MRI, including prognostic data associated with LV fibrosis. 27 Because different imaging modalities offer unique advantages, a multimodality technique is best practice in assessing patients with suspected MAD. 16
Arrhythmic Implications
The significance of MAD reaches beyond its mechanical presentation in patients with myxomatous valve disease. Although ventricular arrythmia research and correlation with the presence of MAD is still ongoing, recent studies on MAD have concluded that it is a common component of arrhythmic MVP and sudden cardiac death (SCD) in patients with LV fibrosis.16,28 In a recent study of 38 patients with myxomatous valve disease, the severity of MAD correlated closely with the occurrence of non-sustained ventricular tachycardia (NSVT) and was found to be more common in females.29,30 In a similar study of 36 patients, 67% of the study cohort with a mitral annular disjunctive length of >8.5 mm exhibited NSVT on follow-up Holter monitoring. 29
Important Points
The initial documentation and measurement of MAD with TTE 2-D images is crucial in the assessment and diagnosis of MAD. However, there are additional tools that the cardiac sonographer can implement to help confirm this diagnosis. Tissue Doppler imaging (TDI) can be used to demonstrate what is called a Pickelhaube sign. By placing a pulsed-wave (PW) Doppler sample in the area suspected of MAD, increased mid-systolic velocities will resemble the appearance of a 19th-century spiked German helmet. The postero-basal wall will be pulled up to the apex quickly demonstrating this spike and increase in lateral velocities. The Pickelhaube sign is a rare TTE finding which is suggestive of malignant MVP. 31 In addition to TDI, the cardiac sonographer can implement strain imaging to assess for any potential decrease in the basal segments of the LV, in addition to CMR strain techniques. Few studies have demonstrated the feasibility and accuracy of this practice as this should be taken into consideration when fully characterizing this phenomenon as research in this area continues. 32
Conclusion
Accurate sonographic detection of MAD maintains strong implications on patient prognosis. The cardiac sonographer holds an important role in the patient care continuum in identifying these lesions, measuring their size, and assessing associated pathology such as mitral regurgitation. This mitral annulus abnormality goes under documented, due to minimal research on progressive MAD and confusion with associated MVP. This case series provides an awareness of this under documented pathology. It also addresses what we can do as sonography advocators to interrogate the structure and function of MAD to give our patients that best chance at positive outcomes. Cardiac sonographers must continue to embrace and continue pushing for increased image quality, specific to unique pathology, to ensure that we are providing our patients and physicians with accurate data.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
