Abstract
Mitral annular disjunction is a structural abnormality characterized by atrial displacement of the mitral valve annulus or leaflet hinge point away from the ventricular myocardium. This anomaly is frequently associated with mitral valve prolapse, leaflet degeneration, and fibrosis of the papillary muscles and myocardium, and is increasingly recognized as a substrate for malignant ventricular arrhythmias. Mitral annular disjunction contributes to paradoxical systolic annular expansion and has been linked to arrhythmogenic mitral valve prolapse, with a higher risk of ventricular arrhythmias and sudden cardiac death. A disjunction length of ≥5 mm is considered clinically significant for predicting arrhythmic risk. Cardiac magnetic resonance imaging plays a pivotal role in identifying and quantifying mitral annular disjunction, assessing coexistent mitral valve prolapse, and detecting myocardial and papillary muscle fibrosis. These insights are vital for risk stratification and guiding patient management, particularly regarding electrophysiological interventions and surgical repair.
Keywords
Case
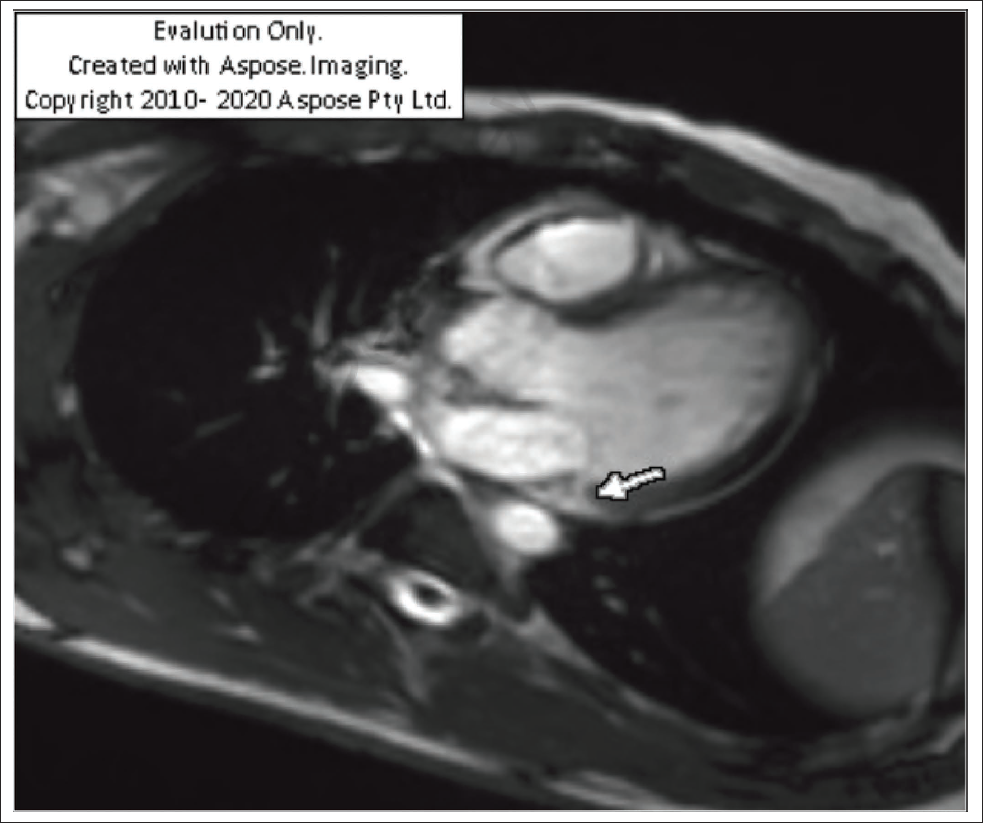
A 28-year-old male presented with complaints of palpitations. Cardiac magnetic resonance imaging (CMR) revealed normal left ventricular (LV) size and function, without late gadolinium enhancement (LGE). The right and left ventricles demonstrated normal diameters, wall thickness, and contractility, with no evidence of focal dyskinesia. The atria, interventricular and interatrial septa, as well as the mitral and tricuspid valves, appeared normal. Pulmonary venous drainage, the ascending aorta, and aortic cusps were unremarkable. An incidental 6 mm mitral annular disjunction (MAD) was identified (Figures 1–3).
Steady-state Free Precession Three-chamber Short-axis View in Cardiac Magnetic Resonance Imaging (CMR) Showing the 6 mm Gap Between the Mitral Valve Hinge Point and Left Ventricular (LV) Myocardium (Arrow Pointing to the Top of LV Myocardium).
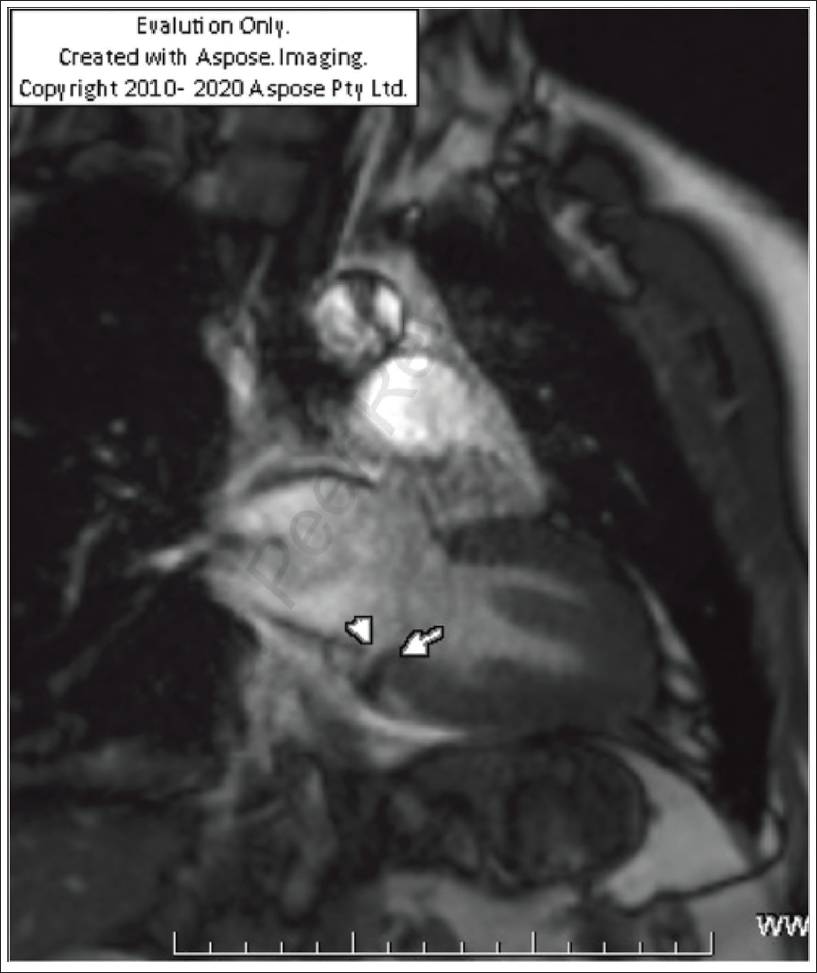
Vertical Long-axis Two-chamber View Showing Mitral Annular Disjunction (MAD) (Between Arrows).
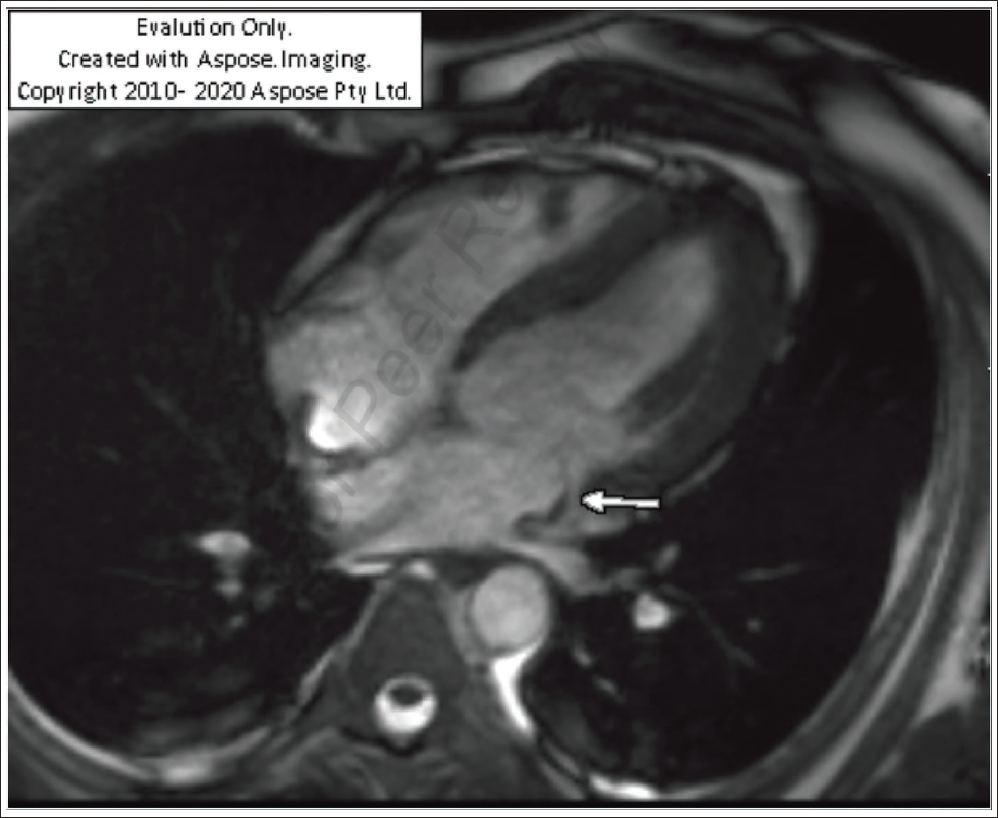
Horizontal Long-axis Four-chamber View Depicting Mitral Annular Disjunction (MAD).
Discussion
MAD usually located in the inferolateral basal wall of the left ventricle, may trigger complex ventricular arrhythmias (VAs) which are more in incidence in patients with wider disjunction.1 In 1981, Bharati et al. described a case of sudden cardiac death (SCD) linked to atrial displacement of the mitral annulus, identified through clinical, electrocardiographic, and histological evaluation. 2 The etiology of MAD remains uncertain, with ongoing debate over whether it is a congenital defect or an acquired abnormality. A disjunction measuring ≤3 mm is often considered a benign or incidental finding. 3 It is not yet clear whether all forms of MAD carry pathological significance or if some may represent a normal anatomical variant. The possible association between MAD and subvalvular membrane has also been proposed but remains unproven. 4 Typically located along the inferolateral basal wall of the left ventricle, MAD has been associated with complex ventricular arrhythmias, and the likelihood of arrhythmic events appears to increase with greater disjunction length. 1
There is an increasing role of MAD in the prognostication of patients with MVP. There is an association of MAD with severe mitral valve lesions. 5 There is a potential relation of MAD in sustained ventricular tachycardia and SCD. So it is imperative to recognize MAD in MVP patients with arrhythmias.
Mitral annular disjunction can be evaluated using transthoracic echocardiography (TTE), transesophageal echocardiography (TEE), cardiac computed tomography (CCT), or CMR. CCT, owing to its high spatial and temporal resolution, is particularly useful for defining the circumferential extent and severity of MAD. 6 CMR not only measures the degree of disjunction but also detects myocardial fibrosis through LGE. 7 By comparison, TTE is limited because of lower orthogonal resolution and restricted imaging views. 8
Mitral valve prolapse (MVP) is diagnosed when one or both mitral leaflets extend ≥2 mm beyond the annular plane during systole in the long-axis view. 9 Classic MVP is defined by leaflet displacement accompanied by thickness >5 mm, whereas leaflet thickening may be absent in non-classic forms. To prevent overdiagnosis, prolapse should be assessed in the parasternal long-axis (three-chamber) view. 10
Mitral annular disjunction length is measured as the separation between the basal LV myocardium and the hinge point of the posterior mitral leaflet at the atrial wall. It is most often identified in the inferolateral LV, where it commonly coexists with MVP and arrhythmic events.
According to Gulati et al., cine CMR should be assessed in multiple long-axis planes (two-, three-, and four-chamber) across the cardiac cycle, with the systolic three-chamber view recommended for precise measurement of MAD. 11 In certain cases, the posterior leaflet hinge point maintains attachment to the LV myocardium but appears to shift toward the atrial wall during systole. This phenomenon, termed “pseudo-MAD,” is frequently seen with MVP. 12
Cardiac magnetic resonance imaging offers multiplanar imaging, superior tissue characterization, and the ability to detect LV and papillary muscle fibrosis, which supports prognostic evaluation. Imaging planes that intersect the mitral annulus also allow accurate definition of the extent and distribution of disjunction. Mitral regurgitation volume has been shown to be similar in MVP patients regardless of MAD presence; however, LGE of the papillary muscles is more common in those with MAD, suggesting fibrotic remodeling. 13 Patients with MVP and LV LGE have also demonstrated longer MAD lengths.
Conclusion
Mitral annular disjunction in the setting of MVP is associated with LV remodeling, where chamber enlargement occurs independently of age or mitral regurgitation severity. The magnitude and distribution of MAD help differentiate normal anatomical variants from pathological changes, with arrhythmias arising from the combination of structural displacement and leaflet traction. Stretching of the inferobasal LV wall and posterior papillary muscle promotes fibrosis, providing an arrhythmogenic substrate. A disjunction exceeding 8.5 mm has been linked to nonsustained ventricular tachycardia. 14 In patients not undergoing mitral valve surgery, multimodality imaging and continuous rhythm monitoring remain essential for risk evaluation
Footnotes
Authors’ Contributions
S. V. K.—Conceptualization: Equal; Formal analysis: Lead; Writing—original draft: Lead; Writing—review & editing: Lead; N. S. N.—Conceptualization: Equal; Investigation: Equal; Supervision: Equal; P. K. P.—Investigation: Equal; Writing—editing.
Data Availability Statement
No new data were generated or analyzed supporting this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
Written informed consent was obtained.
