Abstract
Serous cystadenofibromas are benign surface epithelial stromal tumors of the ovary. Many of the sonographic features of serous cystadenofibromas mimic what are traditionally considered possible characteristics of malignancy. Depending on the size of the ovarian neoplasm, there can be increased risk for ovarian torsion. Meticulous sonographic evaluation and appropriate optimization are critical for establishing the risk for malignancy and likelihood of torsion. This case describes how sonography was used to help identify a serous cystadenofibroma in a postmenopausal woman.
Serous cystadenofibromas are a rare type of benign ovarian neoplasm, making up only 1.7% of the vast number of benign ovarian tumors. 1 They have a wide variety of appearances and can resemble many other benign tumors as well as ovarian cancer. The larger they grow, the higher the risk they pose for ovarian torsion which is a gynecologic emergency. Ovarian torsion is a twisting at the vascular pedicle, resulting in the obstruction of the ovarian artery, vein, and lymphatic system. Sonography plays a critical role in the evaluation of serous cystadenofibromas as well as the concern for torsion. The most common treatment for both serous cystadenofibroma and ovarian torsion is exploratory laparoscopy with potential salpingo-oophorectomy.
Case Report
A 73-year-old woman presented to the emergency department with acute right lower quadrant abdominal pain for 4 days, with nausea and vomiting. Her prior medical history included a total vaginal hysterectomy, left salpingo-oophorectomy, and ductal carcinoma in situ of the breast. Earlier in the day, an outside computed tomogram (CT) was performed that demonstrated a 13.6-cm septated cystic lesion arising in the right adnexa, suspicious for a benign cystic ovarian neoplasm (see Figure 1). Tumor markers, including CA 125, CA 19-9, and carcinoembryonic antigen (CEA), were confirmed to be normal and therefore reduced the concern for malignancy. The CT images also showed stranding of the retroperitoneal fat adjacent to the right adnexa with apparent twisting of the vascular pedicle. This was suggestive of superimposed torsion of the ovarian neoplasm (see Figure 2). The patient was referred for a sonogram to further evaluate and classify the suspected ovarian neoplasm. A pelvic sonogram was performed using a Philips iU-22 ultrasound system (Philips Medical, Bothell, WA, USA) with a C5-1 MHz curved linear transducer and a C10-3v MHz endovaginal transducer. This sonographic examination confirmed a 13 × 13.7 × 10.7 cm multiloculated mass with thin, vascular septations associated with the right ovary (see Figure 3). No free fluid was noted in the pelvis, and both arterial and venous blood flow were appreciated, within the right ovary (see Figure 4). Despite the presence of color Doppler, intermittent ovarian torsion could not be excluded from the diagnosis. Owing to the size of the mass, concern for malignancy, and the CT findings suggestive of ovarian torsion, the patient underwent exploratory laparotomy and a right salpingo-oophorectomy, 2 days later. The surgical pathology report confirmed a multiloculated mass filled with yellow serous fluid. There was a solid component that was made up of scattered firm areas with a brown-red, hemorrhagic section containing focal excrescences. This was consistent with the diagnosis of a benign serous cystadenofibroma. Despite the CT findings suggestive of torsion, no mention of this was made by pathology. There were no postoperative complications for the patient.
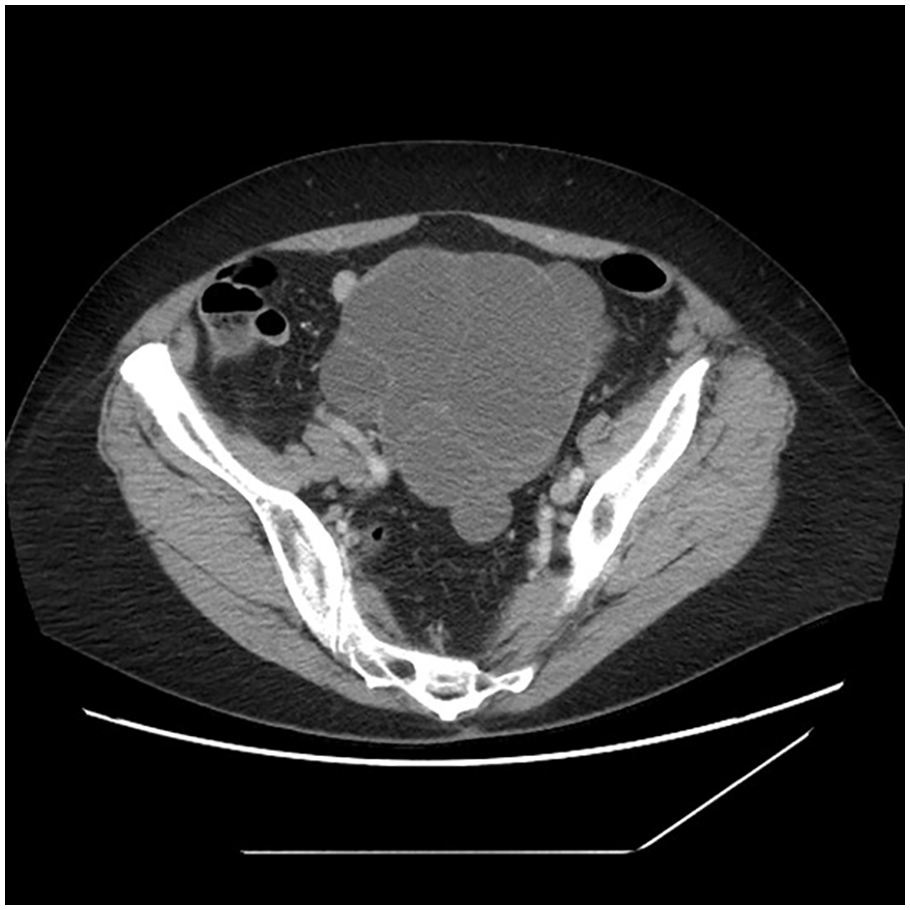
Axial computed tomography image demonstrating the multiloculated serous cystadenofibroma at its widest dimensions.
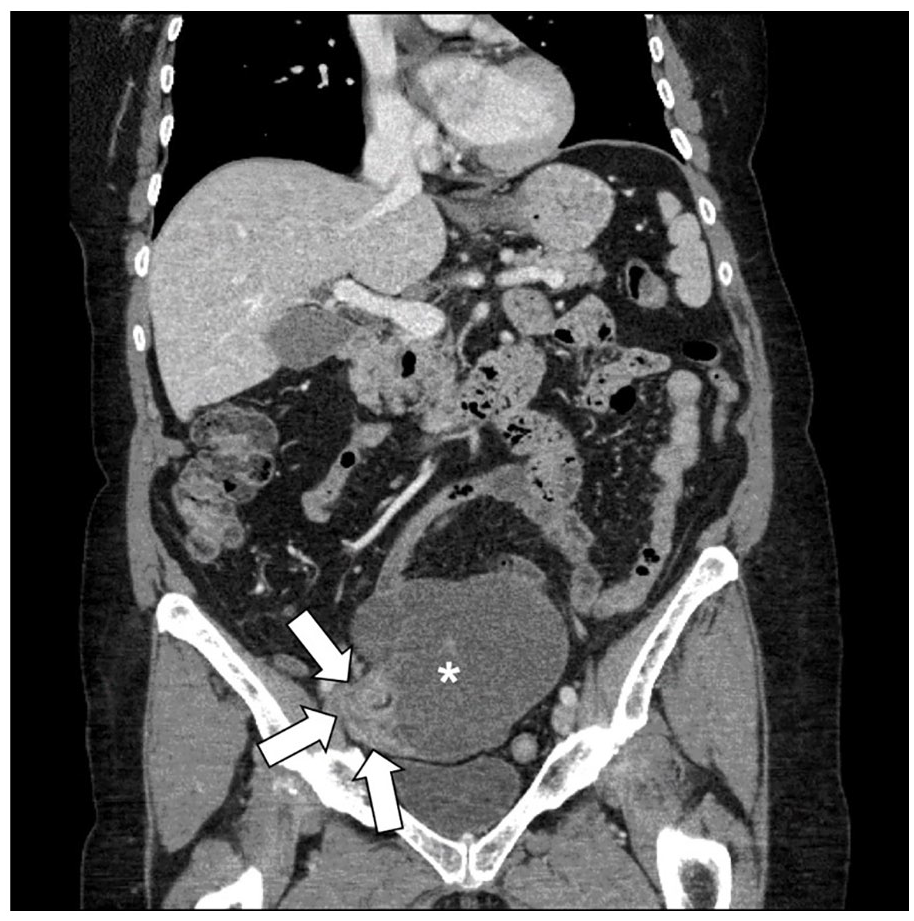
Coronal computed tomography image showing twisting at the vascular pedicle (solid white arrows) with adjacent serous cystadenofibroma (white asterisk).
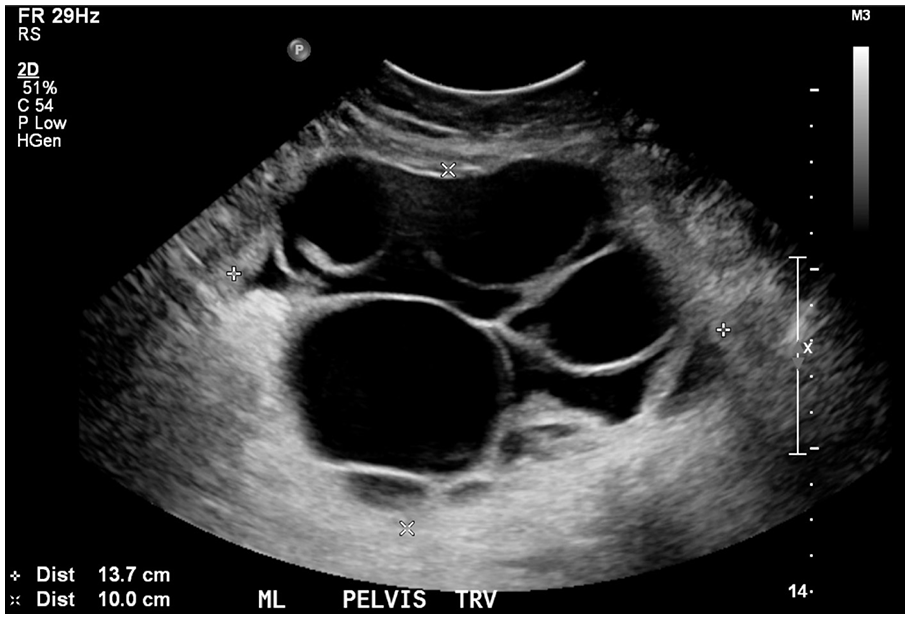
Transabdominal sonogram of the serous cystadenofibroma in a transverse plane with measurements at its largest dimensions.
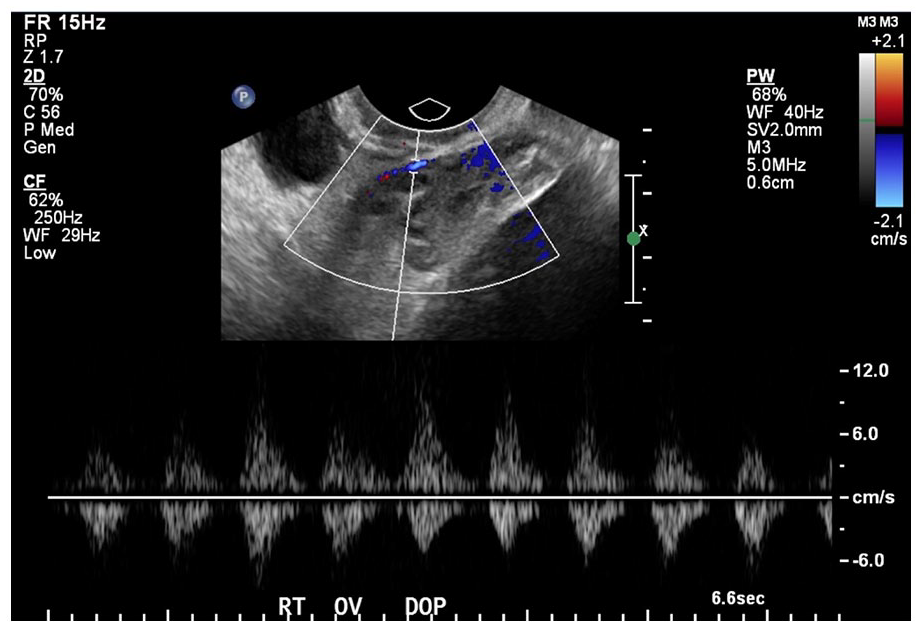
Endovaginal sonographic image of the right ovary demonstrating an arterial waveform on pulsed-wave Doppler.
Discussion
Sonographically, the normal ovaries are oval-shaped hypoechoic structures, containing small anechoic follicles. The appearance of the ovaries can change throughout a woman’s menstrual cycle as folliculogenesis occurs. During menopause, the ovaries atrophy and become harder to identify sonographically, due to decreased size and the absence of follicles. 2
Serous cystadenofibromas are part of a broad category of ovarian neoplasms called surface epithelial stromal tumors that make up approximately two thirds of all ovarian neoplasms. 3 Serous cystadenofibromas are rare and only comprise 1.7% of all benign ovarian neoplasms. 1 Embryologically, surface epithelial tumors come from the coelomic epithelium that lines the ovary and penetrates the mesenchyme to form the Mullerian duct. 1 The World Health Organization (WHO) defines serous cystadenofibromas as tumors that are primarily comprised of benign stromal proliferations that overshadow the epithelial element. This stroma can appear similar to the normal stroma of the ovary; however, it contains more fibrous and edematous elements. 4
Macroscopically, the appearance of serous cystadenofibromas can vary greatly. In previous studies, their size has been reported up to 30 cm, with an average diameter of 5–8 cm. 4 These neoplasms are complex, yet predominantly cystic. 5 The cysts are thin walled with a shiny exterior and are filled with clear, watery fluid or thin mucoid material.4,5 Due to the wide spectrum of appearances, many studies have classified the most common presentation of serous cystadenofibromas. Most commonly, they are a unilocular cystic mass that contains one or more solid, shadowing, hyperechoic papillary projection(s) without color Doppler blood flow.4-7 A papillary projection is specifically defined as solid tissue that protrudes into the internal components of the cyst. To be considered a papillary projection, its height must measure at least 3 mm. 4 The second most common pattern of serous cystadenofibromas was discovered to be a multiloculated cyst that did not contain papillary projections, but did have small, solid internal components. Although less common, the third presentation was determined to be bilocular or multilocular cysts, and the fourth was a unilocular cyst. 4 Approximately 47.8% of serous cystadenofibromas have internal vascularity. Blood flow tends to be on the peripheral portion of the cyst and has a high impendence. 5
Serous cystadenofibromas affect women of all ages. A comprehensive study by Virgilio et al surveyed 233 women ranging in age from 14 to 89 years, all of whom had histologically confirmed serous cystadenofibromas. Sixty-three percent of these women were postmenopausal. 4 The most common symptoms associated with serous cystadenofibromas are pelvic pain and discomfort. However, symptoms of this ovarian neoplasm are unspecific and can also include vaginal bleeding and increased girth. In addition, many patients are asymptomatic.1,4 Benign serous cystadenofibromas are typically unilateral, although they can occasionally be seen on both ovaries.4,5,8 Laterality can provide important information on the type of ovarian tumor as malignancies are more often seen bilaterally. 3
One of the biggest complications of the sonographic evaluation of benign serous cystadenofibromas is that they have many features that mimic those of malignant neoplasms.3,4 There is valid reason for concern as 90% of all ovarian cancers are the malignant form of surface epithelial tumors. 3 Some of the more common malignant neoplasms that mimic serous cystadenofibromas are serous cystadenocarcinomas and mucinous cystadenocarcinomas. 4 Although exceedingly rare, cystadenocarcinofibromas have the closest resemblance and are the malignant counterpart to cystadenofibromas. 9 Thorough and proper evaluation of ovarian neoplasms is essential for documenting malignant characteristics and establishing appropriate treatment plans. Risk for malignancy increases exponentially as age increases and is most common in perimenopausal and postmenopausal women. 3 Women younger than 45 years are at a 13% risk for ovarian cancer, while the risk for older women is increased to 33%. 3 Elevated tumor marker laboratory values, specifically serum CA 125, may be useful for determining malignant potential of neoplasms. 10 However, CA 125 can lack specificity, as it can also be elevated with pregnancy, uterine fibroids, normal menstruation, endometriosis, and liver disease. 10
One of the most commonly accepted sonographic signs suspicious of ovarian malignancy are papillary projections, which are also seen in the majority of benign serous cystadenofibromas.4,6 Blood flow within a papillary projection is concerning for malignancy, whereas a lack of internal vascularity is reassuring. 6 To identify blood flow, particularly in small vessels with extremely low velocities, it is crucial to thoroughly evaluate the papillary projection and to ensure that color Doppler settings are optimized appropriately. Using a low-velocity scale (pulsed repetition frequency), increasing color Doppler gain, and decreasing the size of the color box to the area of interest are important optimization techniques. In addition, it may be important to increase the color Doppler write priority and persistence.6,11 Power Doppler can also be a useful technique when blood flow is difficult to detect. Benign papillary projections within serous cystadenofibromas have fibrous, solid components that tend to reflect acoustic pressure waves making them appear hyperechoic, with posterior shadowing. 6 In contrast, papillary projections within ovarian cancer most often contain anechoic spaces and lack shadowing. 4 Other ominous sonographic features of malignancy include neoplasms greater than 4 cm, with wall septations greater than 3-cm thickness, and exudate ascites within the pelvis and abdomen.8,10 Although any of these conditions alone can be concerning, many risk factors combined raise the highest concern for malignancy.
In addition to mimicking malignancy, benign serous cystadenofibromas can appear similar to many benign ovarian pathologies, such as serous cystadenomas, fibromas, thecomas, and fibrothecomas. Serous cystadenomas are the most common ovarian epithelial tumor, accounting for approximately 20% of all benign ovarian neoplasms. 7 Like serous cystadenofibromas, serous cystadenomas have a Mullerian duct origin. They can range greatly in size, from 1 to 35 cm and affect women of all ages. The most common symptoms of serous cystadenomas are abdominal pain and vaginal bleeding. 12 Serous cystadenomas are predominantly cystic, filled with clear, watery fluid and occasionally blood, and can be unilocular or multilocular. They are encapsulated and have a smooth exterior wall. Sonographically, the best way to differentiate serous cystadenomas and serous cystadenofibromas is in the evaluation of their papillary projections. In contrast to the thick, fibrous, and short papillary projections within serous cystadenofibromas, those in serous cystadenomas are thin and delicate. The absence of papillary projections altogether can also be a good indication as they are far less common in serous cystadenomas than cystadenofibromas. 12
Fibromas are a rare form of sex cord stromal tumors that comprise 4% of ovarian neoplasms. 13 Histologically, fibromas are derived from the stroma of the ovary and contain collagen-producing spindle cells.13,14 Fibromas typically affect middle-aged women, the majority of whom are postmenopausal and asymptomatic.13,14 Sonographically, fibromas appear most often as large, solid, hypoechoic tumors that cast streak-like shadows. Although less common, fibromas can have cystic spaces within them. They tend to be unilateral, with limited vascularization, and have a round or oval shape with slightly lobulated borders. 14 In addition, fibromas are one component in the triad of symptoms associated with Meigs syndrome. The remaining two symptoms include ascites, most often seen in the pouch of Douglas, and pleural effusions.13,14
Thecomas are also a subtype of sex cord stromal tumors, accounting for less than 1% of ovarian neoplasms. 13 Thecomas comprise groups of spindle-shaped cells with lipid-laden cytoplasms that originate from the ovarian medulla, whereas serous cystadenofibromas have more fibrous components.4,13,15 Similar to fibromas, thecomas are most often unilateral, round, oval, or lobulated neoplasms that most commonly affect perimenopausal or postmenopausal women.15,16 They are typically benign, but can occasionally be malignant. 15 In general, thecomas tend to be solid appearing and smaller than fibromas, the majority of which are less than 5 cm. 16 Sonographic appearance of thecomas is nonspecific as it depends on the composition ratio of fibrous components to lipid components. Most often, thecomas appear as hyperechoic masses with distal acoustic shadowing, or a well-defined hypoechoic mass. 13 Cystic degeneration and necrosis are often seen with those thecomas that become larger, and occasionally, calcifications can be seen.15,16 Upon color Doppler evaluation, thecomas typically have little to no vascularity. 16 Women with thecomas can have a variety of symptoms ranging from asymptomatic to vaginal bleeding, postmenopausal bleeding, and/or abdominal or pelvic pain.15,16 Thecomas are functional, estrogen-producing neoplasms. Due to the increased estrogen produced by the thecoma, endocrine complications can occur including vaginal bleeding, endometrial hyperplasia, and endometrial cancer. 15 Androgenic thecomas have also been reported, most often manifesting as hirsutism. 16
Occasionally, a hybrid occurs resulting in a fibrothecoma which is a cellular combination of the fibroma and thecoma. 16 Sonographically, the fibrothecoma shares the typical solid appearance of the fibroma and thecoma. At the cellular level, fibrothecomas contain a large amount of collagen and edema. Fibrothecomas most closely resemble fibromas and are also associated with Meigs syndrome. 14
Due to the nonspecific appearance of serous cystadenofibromas, fibromas, thecomas, and fibrothecomas, they are not easy to distinguish by sonogram. Perhaps the biggest differentiation is that fibromas, thecomas, and fibrothecomas tend to be predominantly solid, while cystadenofibromas are mostly cystic with some solid nodules or papillary projections. Discernment can also be aided when there are secondary symptoms related to the neoplasm. For example, ascites and hydrothorax related to Meigs syndrome may help differentiate fibromas and fibrothecomas from other differential diagnoses, whereas symptoms related to estrogen production may help distinguish thecomas.
There are two main treatment approaches for serous cystadenofibromas. Owing to their benign nature, often times, conservative management is recommended with serial follow-up sonograms. 8 However, if there is concern for additional complications, the gold standard form of treatment is laparoscopic surgery to completely remove the tumor. 1 This is indicated when sonography confirms suspicion for malignancy, or potential for rupture or ovarian torsion. With prompt and proper medical treatment, patients with serous cystadenofibromas typically have an excellent prognosis. 1
One of the most concerning complications that can be associated with serous cystadenofibromas is ovarian torsion. Ovarian torsion is a rare gynecologic surgical emergency with a prevalence of 2.7%.11,17-19 It involves the ovary completely or partially rotating around the axis of its vascular pedicle which can lead to arterial, venous, and lymphatic compromise.17,18,20 Without proper blood flow, the ovary can become ischemic and necrosis can ensue, ultimately leading to ovarian death.11,18,21 Adnexal torsion can occur in any stage of a woman’s life, with the highest prevalence being in the second and third decades and in postmenopausal women.17,20,21
Serous cystadenofibromas pose a threat for ovarian torsion due to their potential to grow very large. Any ovarian cyst or mass larger than 5 cm creates a high risk factor for ovarian torsion because the added weight of the mass can act as a fulcrum for the fallopian tube to rotate around. 17 The primary risk factor for ovarian torsion is a history of adnexal torsion. 18 Other predisposing factors are widespread and can include assisted reproductive technology, especially in the setting of hyperstimulated ovaries, normal pregnancy due to large corpus luteal cysts, polycystic ovary syndrome, drastic changes in intra-abdominal pressure often from heavy lifting or pushing, and prior tubal ligation.18,21 Tubal ligation, along with hydrosalpinx, long fallopian tubes, and young age all contribute to a hypermobile adnexa which can lead to torsion at the mesosalpinx.11,21 Contrarily, adnexal torsion is rarely seen with pathologic conditions that present with pelvic adhesions such as pelvic inflammatory disease, endometriosis, and malignant neoplasms because the fibrous response helps anchor the ovary in place.11,18 Occurrence of left-sided adnexal torsion is far less common than right-sided which is about two thirds more frequent. 18 This is speculated to be because the left ovary sits among the fixed sigmoid colon, whereas the right ovary is in close proximity to the more mobile cecum and ileum.11,18
The most common clinical symptom of ovarian torsion is acute pain, occurring in 90% of patients. 18 Nausea, vomiting, fever, and tenderness to palpation are also commonly encountered symptoms, and occasionally, laboratory values indicate a high white blood cell count.17,18 The diagnosis of ovarian torsion is typically made by a combination of clinical symptoms and sonography together. The American College of Radiology recommends sonography as the primary imaging modality to diagnose ovarian torsion. 17 As the vascular pedicle twists upon itself, venous and lymphatic return is obstructed.11,18,21 This leads to an enlarged and edematous ovary which is the first and most accurate sonographic sign of ovarian torsion. Other gray-scale sonographic findings in this stage include hyperechoic central stromal edema and multiple peripheral follicles. Together, these 2D findings have a high predictive value as they occur in 74% of ovarian torsion cases. 18 As the ovary becomes larger and more congested, pressure within the vascular pedicle increases, thus leading to arterial thrombosis. 21 Once the ovary is no longer receiving blood flow, ischemia and necrosis result.18,21 Free fluid within the posterior cul-de-sac is seen with one third to two thirds of patients experiencing adnexal torsion. 21 Although color Doppler has traditionally been considered the preferred method to evaluate for ovarian torsion, special attention should be paid to these gray-scale indications. 20
Historically, the presence of arterial and venous blood flow has been considered pathognomonic to rule out torsion. 20 However, there have been many recent cases of surgically proven ovarian torsion where blood flow had been identified with sonography using color Doppler.11,20 A wide range of blood flow patterns were noted within these torsed ovaries depending on the chronicity and degree of vascular torsion. 22 Due to this variable nature, it is difficult to definitively characterize blood flow patterns. Reports have included normal arterial and venous blood flow in up to 60% of surgically confirmed ovarian torsion. 23 Other abnormal blood flow patterns have included reversed or absent end-diastolic arterial flow creating a high resistance waveform, 24 decreased venous with absent arterial flow, and decreased arterial with absent venous flow. 11 Color Doppler sample gate placement is crucial in these instances because it is possible to pick up blood flow just outside the ovary in the pelvic side wall. If vascularization persists within a questionable ovary, it is important to compare blood flow patterns with the contralateral side. Most often, even if there is still blood flow within the ovary, it is diminished compared with the opposite ovary.11,20 Persistent blood flow within a torsed ovary can be from a nonocclusive ovarian artery with a small residual lumen, from collateral branches off of the uterine artery, or from partial or intermittent torsion.18,20
Intermittent ovarian torsion involves the twisting and untwisting of the vascular pedicle. Signs and symptoms of intermittent torsion closely follow those of torsion including but not limited to severe pain, nausea, vomiting, and a diffusely large and edematous ovary with peripheral follicles. 11 Symptoms and color Doppler indications of intermittent torsion are only present when the ovary is actually torsed.11,25 This makes the diagnoses of ovarian torsion by color Doppler exceedingly difficult as the ovary may not be torsed during sonographic evaluation. Therefore, intermittent torsion can never be fully ruled out by sonogram. 25
The most reliable sonographic sign of ovarian torsion is the whirlpool sign, which has a 90% predictability rate.17,26 The whirlpool sign is a representation of the fallopian tube, broad ligament and/or branches of the ovarian artery, and vein twisting at the vascular pedicle. The reason the whirlpool sign is so effective is because it evaluates the specific area of torsion, rather than the secondary sonographic or clinical findings. The whirlpool sign is identified by moving the transducer back and forth in a transverse plane along the area of the vascular pedicle.26,27 The whirlpool sign can be detected in both gray-scale sonography and color Doppler. In 2D, it can be identified by hypoechoic concentric rings surrounding an echogenic focus forming a target or bulls-eye appearance.17,26,27 With color Doppler, the whirlpool sign’s signature appearance is a swirling of blood flow in a spiral pattern.26,27 Owing to the mobility of the ovary and the fallopian tube, the vascular pedicle and therefore the whirlpool sign can be variable in location. Typically, the whirlpool sign is detected either lateral to the ovary, between the ovary itself and the pelvic side wall, or medially, between the ovary and the uterus. 26 A study by Navve et al discovered a correlation between size of ovarian masses and location of the whirlpool sign, stating that the larger the ovarian mass, the higher the likelihood that the whirlpool sign will be located lateral to the ovary. 26 This is speculated to be because as the ovarian mass grows in size, the ovary is pulled downward with the weight of the mass; therefore, the fallopian tube and vessels can no longer fit between the uterus and the ovary. There is also a higher predictive value of right-sided lateral whirlpool sign, which is likely due to the increased mobility of the ileum and the cecum on the right. The whirlpool sign, with a predictive value of 94%, is also the most definitive way to determine whether the ovary is receiving perfusion, and therefore whether it is viable. 26 If there is an absence of blood flow to the ovary and ischemia and ovarian tissue necrosis have occurred, the ovary must be removed.26,27
Emergency laparoscopy is the preferred surgical treatment method for ovarian torsion because it is less invasive, and safer due to fewer incisions, less adhesions, and decreased side effects for the patient.17,18 Especially because ovarian torsion most commonly affects women of reproductive age, the ovary should be attempted to detorse and preserve the patient’s fertility. 17 If reperfusion does not occur after this attempt, an oophorectomy is indicated. An oophorectomy is the recommended treatment for postmenopausal women with ovarian torsion. 18
Diagnostic medical sonography with adnexal Doppler is the gold standard to evaluate the etiology of pelvic pain, ovarian masses, and adnexal torsion.5,17,18 A combination of transabdominal and endovaginal sonography should be used. Transabdominal sonography is beneficial to gain a wider field of view, whereas endovaginal sonography provides a closer and more detailed perspective. Endovaginal sonography has a sensitivity of up to 86% and a specificity of up to 99% in identifying ovarian masses. 17 A high correlation has been found between the sonographic features of ovarian masses and their microscopic characteristics, which confirms that sonography plays an important role in the classification of benign and malignant ovarian neoplasms. 5 CT and magnetic resonance imaging (MRI) can also be useful imaging modalities to further classify sonographic pathologic findings. CT, along with sonography, is often one of the first tests performed when a patient presents to the emergency department with abdominopelvic pain. MRI is especially useful for the patient suspected of being pregnant. 18 Although many imaging modalities can contribute to the characterization of ovarian masses, histopathologic examination is the only method that can confirm specific ovarian neoplasm diagnoses with certainty. 3
Conclusion
Owing to their nonspecific appearance, the classification of serous cystadenofibromas can be difficult to diagnose, solely with sonography. Sonographic attributes such as a lack of vascularity in the papillary projections and posterior shadowing are classic signs of serous cystadenofibromas and can help rule out malignancy. Ovarian torsion is an emergent complication that can be seen with serous cystadenofibromas and any large ovarian neoplasm. This case highlights the importance of using multiple imaging modalities to determine a diagnosis. It is also important to remember that intermittent torsion may be difficult to detect sonographically, due to the presence of ovarian flow.
Footnotes
Acknowledgements
Thank you to Emily Downs, BS, RDMS, RVT, RDCS, RT, and Britt Smyth, BA, RDMS, RVT, RDCS, for their guidance and feedback on this manuscript.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
