Abstract
Mature cystic teratomas are benign germ cell tumors and, depending on the size, may be at risk for torsion. These ovarian masses can cause the ovarian vasculature to twist upon itself and inhibit blood flow, resulting in partial to complete torsion. If detection and treatment are not prompt, a salpingo-oophorectomy may be needed impeding future fertility. This case describes a young woman with acute left adnexal pain where sonography aided in the diagnosis of ovarian torsion caused by a large mature cystic teratoma. Transabdominal sonography was used, aided by extended field-of-view technology for characterization of the mass. The sonographic attributes of mature cystic teratomas vary, but careful evaluations can lead to an accurate diagnosis to aid in surgical procedures. The patient underwent an exploratory laparotomy, which resulted in a left ovary oophorectomy and excision of the mass.
Mature cystic teratomas (MCTs) are benign germ cell tumors. These growths increase the risk for ovarian torsion, which is a gynecologic emergency. Torsion is an ischemic event that can require surgical intervention, possibly including oophorectomy. It is important to promptly identify an MCT and torsion sonographically to aid in surgical preparation. In many cases of MCT, the torsion can be surgically untwisted and the fallopian tube and ovary salvaged. This requires torsion to be detected early to minimize the time of ischemia. Since many torsion events happen to women of childbearing age, accurate and rapid sonographic detection can preserve the fertility of the patient. This case describes initial imaging of a large MCT causing acute ovarian torsion.
Case Report
A 24-year-old gravida 0, para 0 presented to the emergency department with approximately 12 hours of acute left lower quadrant pain. The pain progressively increased from the onset early in the morning. Physical examination revealed left adnexal tenderness. A pelvic sonogram was performed using a Philips iU-22 ultrasound system (Philips Medical, Bothell, WA) with a C5-1-MHz curved linear transducer and a C10-3v-MHz endovaginal transducer. Images revealed a large, heterogeneous, predominantly echogenic left adnexal mass measuring 17.7 × 16.9 × 8.2 cm, most likely arising from the left ovary with no internal vascularity. The size of the mass obscured any remaining ovarian tissue, and blood flow to the ovary could not be evaluated (Figure 1). A transabdominal panorama was used to obtain a measurement of the mass in the longest dimension (Figure 2). Sonographic findings were consistent with an ovarian teratoma. Further same-day evaluation using contrast-enhanced abdominal and pelvic computed tomography (CT) was obtained to confirm the sonographic finding and evaluate for additional signs of torsion. The CT showed a soft tissue and fat-containing mass with a peripheral dystrophic calcification measuring 14.8 × 6.0 × 17.7 cm (Figure 3). The CT findings were consistent with an ovarian teratoma with associated mass effect on the adjacent bladder and bowel. The left ovary was not well delineated, and torsion could not be confirmed. An exploratory laparotomy was performed the next day, 21 hours after the sonogram was performed. During surgery, a large fat-, hair-, and blood-containing cystic structure was identified. The cystic contents were drained using a veress needle attached to suction. Copious dark yellow thick material and hair were extracted. Additional evaluation confirmed left ovarian torsion and a normal right ovary. Therefore, a left salpingo-oophorectomy was performed. The patient tolerated the procedure well and was taken to recovery in stable condition. The pathology report determined grumous material with hair as well as focally calcified areas consistent with MCT and verified the ovary was torsed.
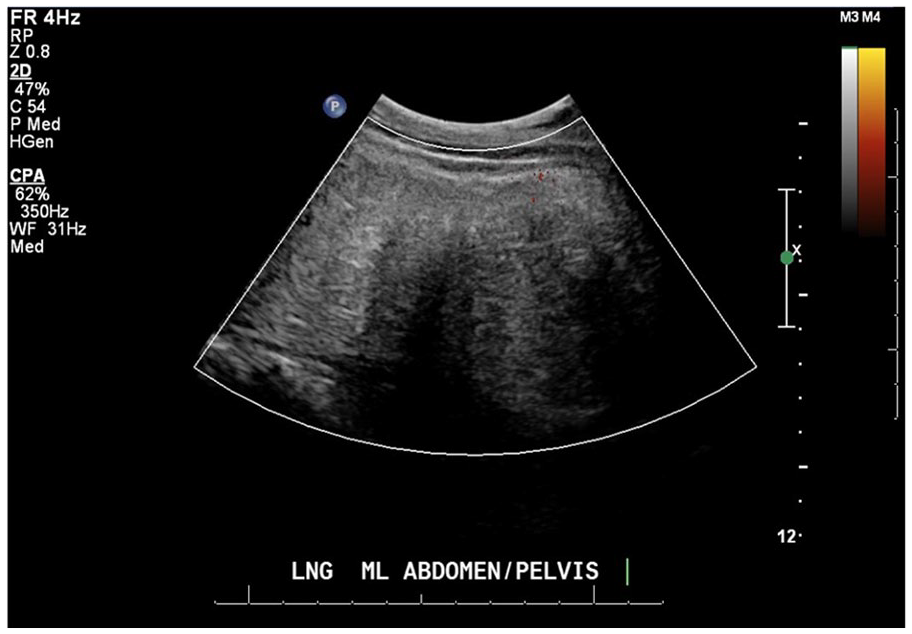
Power Doppler sonogram indicating no blood flow within the mass.
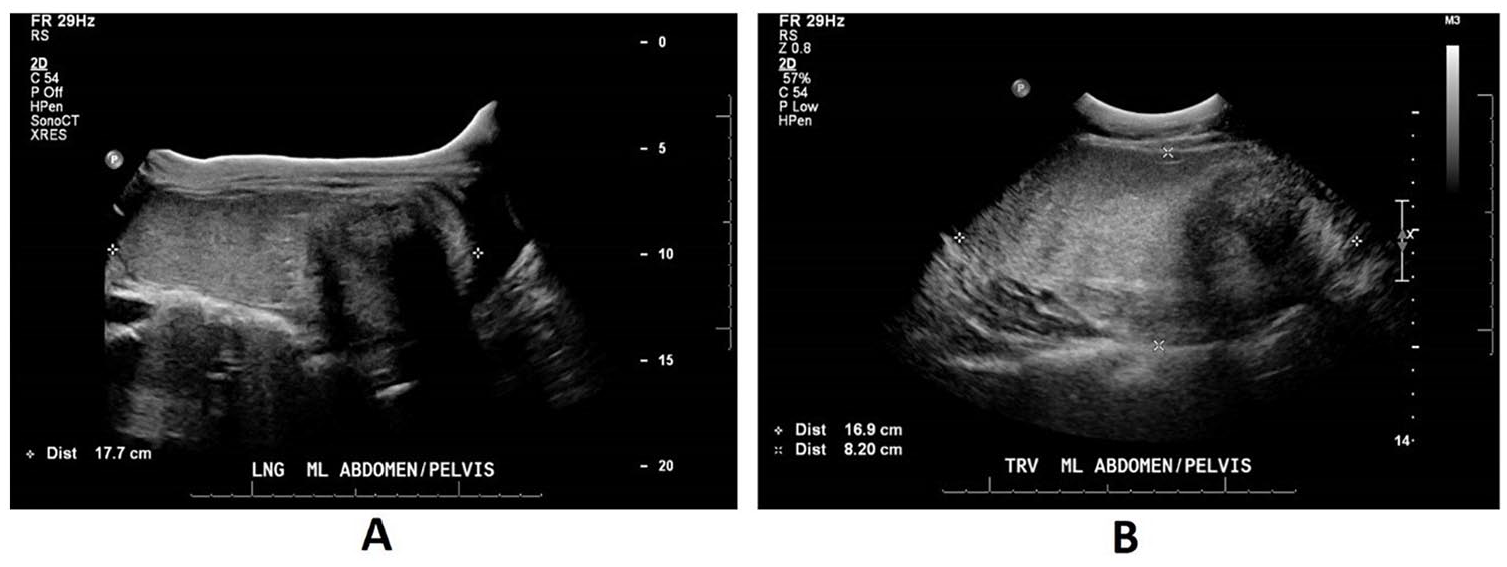
(A) Transabdominal panorama sonographic image. Multiple echogenicities representing the different tissues, including fat, blood, hair, and calcifications. (B) Transabdominal sonogram of the mature cystic teratoma in a transverse plane representing different tissue echogenicities.
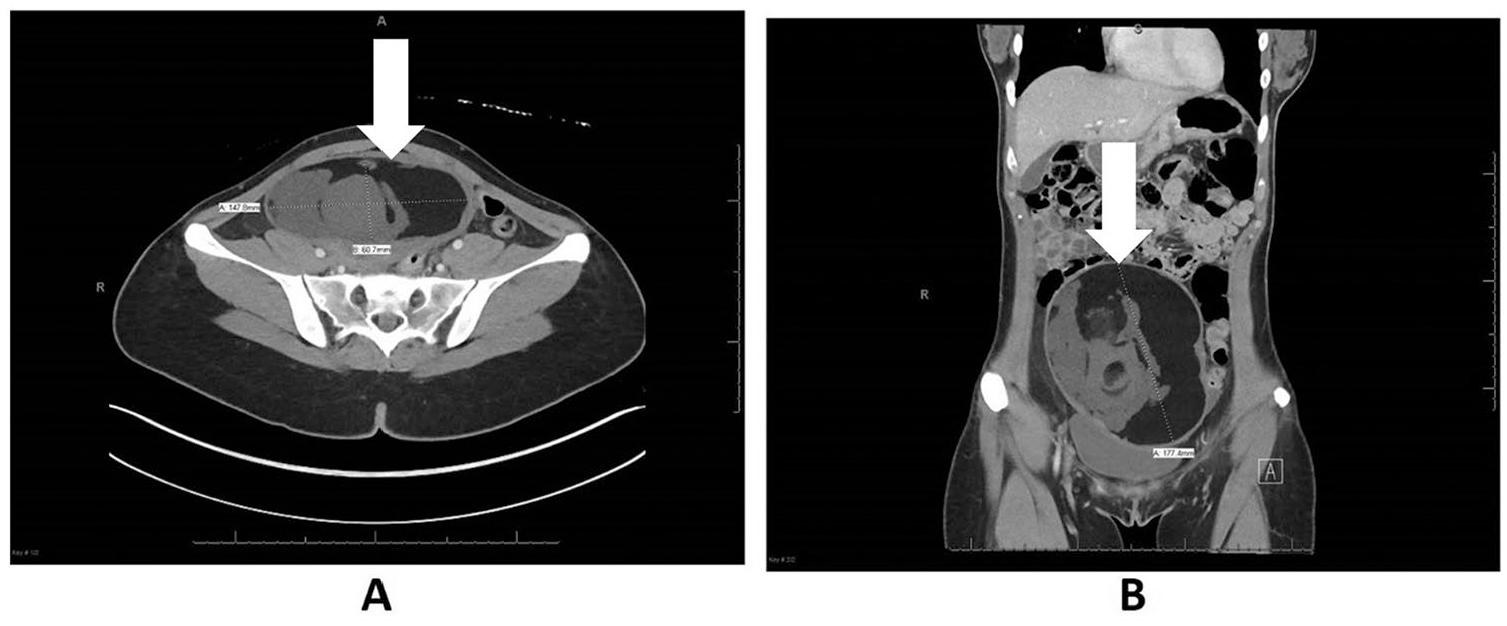
(A) Axial computed tomography (CT) image at widest measurement of the mature cystic teratoma (MCT) showing different densities of the tissues (solid white arrow). (B) Coronal CT image of the MCT (solid white arrow).
Discussion
Mature cystic teratomas are germ cell tumors containing tissue derived from the ectoderm, mesoderm, and endoderm. 1 More specifically, these malformations arise histologically from primordial mitotic germ cell proliferation and often include hair, fat, and bony components. 2 They are the most common form of a broader morphologic category known as ovarian teratomas. 3 Subcategories include, mature, immature, monodermal, and fetiform teratomas. MCTs must contain at least two well-differentiated layers, typically from the ectoderm and mesoderm. Furthermore, they may include skin and hair tissue from the ectoderm in addition to fat or muscle tissue from the mesoderm. 3 An MCT forms during the second meiotic division of cells, when specific cell lines are differentiating. These cells appear as deposits along the migration line from yolk sac to primitive gonads. Each MCT develops from a single germ cell and forms with the entrapment of skin structures. Thus, MTCs are present from birth, even though many are not diagnosed until the second or third decade of life. 2 These neoplasms are also referred to as benign mature cystic teratomas, dermoid cysts, ovarian dermoids (ODs), or ovarian teratomas in the literature.1,4
Mature cystic teratomas are the most common of all nonepithelial ovarian neoplasms, making up to 15% to 20% of ovarian tumors and up to 95% of germ cell tumors.4–6 Most MCTs are unilateral, but they can also present bilaterally. 5 The literature varies with numerical values for the incidence of typical MCT growth rates, but most of these masses are characteristically slow growing, around 1.8 mm per year.3–5,7 Occasionally, a much more accelerated growth has been reported.4,5 Growth rates have been stated to be as slow as 0.6 mm per year to as rapid as 25 mm a year.4,5 Size increase of 2 mm or more per year was historically used to rule out MCT as a possible diagnosis. However, histology negated this parameter by confirming multiple cases of rapidly growing benign dermoid cysts. 4 Growth can also be hormonally influenced by estrogen, and therefore, growth rates during childbearing years are reported to be higher than in postmenopausal timeframes. 4 The most common clinical presentation of patients with an MCT is asymptomatic but can also cause significant abdominal and pelvic pain.3–5 Approximately 15% of MCT cases may have abnormal menstration.1,8
Endovaginal sonography is highly accurate in detecting ovarian tumors. The reported sensitivity for identifying ovarian masses is up to 86%, with a specificity up to 99%. 5 While sonography can be very useful in characterizing an MCT, the neoplasm can only be definitively diagnosed via pathology examination. 5 Some limitations exist with visualization of MCTs by sonography—for example, misidentifying the tumor for bowel gas in the instance of a highly echogenic teratoma or a left-sided tumor concealed by sigmoid colon.2,3
Transabdominal sonography may be more useful if the mass is too large to visualize via a transvaginal approach. The utilization of an extended field-of-view imaging technology, or “panorama,” provided aid in showing and measuring this patient’s large pelvic structure. Application of this imaging technique is often used for detailed sonograms of superficial structures such as the neck, scrotum, breast, or musculoskeletal structures. 9 However, extended-view imaging can also prove useful when evaluating deeper abdominal and pelvic structures that exceed the transducer’s field of view. 9
There is a small risk of a misdiagnosis of an MCT when using a single imaging modality alone. It is an unlikely differential, but the sonographic appearance of an MCT can be similar to an immature cystic teratoma.4,10 Immature cystic teratomas are malignant and account for 1% to 3% of ovarian germ cell tumors. 10 Histologically, immature cystic teratomas contain nerve tissue in addition to embryonic tissue. Historically, tumor size has been a sonographic differentiating factor, and larger neoplasms along with the presence of solid components can indicate a malignant tumor. 10 The presence of a suspected isolated MCT may not be an indication for surgery, even though there is a small risk of malignancy. Expectant management may be an acceptable approach in cases where patients are asymptomatic. 5 Tumors with growth rates greater than 10 mm over a six-month follow-up interval should be considered for surgical intervention, as well as patients exhibiting symptoms of abdominal or adnexal pain.5,10 Noteworthy, the literature is somewhat divided on the strategy for MCT outside of the specific parameters mentioned above. A clear plan of action is essential due to the risk for serious complications such as torsion or misdiagnosis of a malignant immature teratoma.4,10 Other potential complications for an MCT, which are less likely to occur, are rupture (1%–4%), infection of the peritoneum secondary to rupture (1%), and malignant transformation of the MCT (1%–2%).1,3,4
Although sonography is the first choice for imaging the reproductive gonadal organs, computed tomography (CT) and magnetic resonance imaging (MRI) can also aid in characterizing MCT by adding information about tissue consistency, including the identification of fatty components.3,11 Sonography is the first imaging modality recommended by the American College of Radiology (ACR) for confirming ovarian masses, as well as MCT. 12
There are classic sonographic features that depict an MCT. The most common sonographic finding that can help with an ultrasound diagnosis of an MCT is a Rokitansky nodule, also called a dermoid plug (Figure 4). 3 It is a hyperechoic focus or foci protruding into a cystic space, causing shadowing.3–5 Another common sonographic finding with the highest positive predictive value (98%) of MCT is the dot-dash sign, appearing as hyperechoic dashes and dots in the image representing hair orientated in different planes.3,4,13 Another identifying aspect is the “tip of the iceberg sign,” which is a combination of fatty fluid, hair, and cellular components that together form an echogenic mass creating strong posterior shadowing (Figure 4).3,4 A mass with fat-fluid levels contains sebum, which appears anechoic and can demonstrate more hyperechoic layers of fatty fluid. These formations can be difficult to differentiate from hemorrhagic cysts. However, an accompanying classic MCT sonographic feature can aid in recognizing the diagnosis.3–5 Hyperechoic areas with posterior shadowing, contained to specific regions, can be considered calcifications.3,4 When two or more of these signs are found in combination, a positive predictive value for MCT is 100%.3,13 Slow growth is a classic characteristic of these benign masses as well, although fast-growing cases have been confirmed as MCT. 4 While literature describes these neoplasms similarly, there are endless combinations of sonographic findings with MCT.
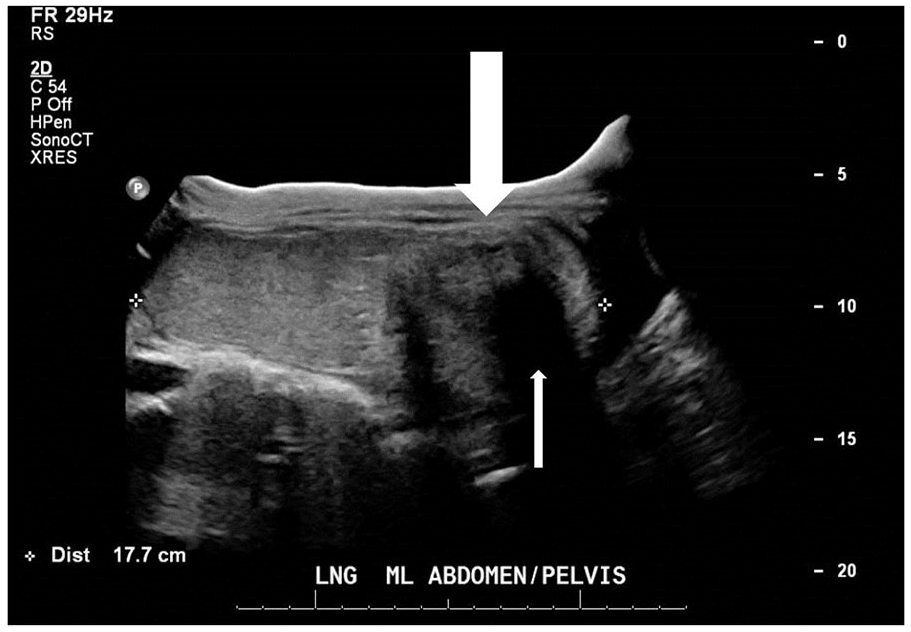
Transabdominal panorama sonographic image. Rokitansky nodule/dermoid plug (thick white arrow) producing a “tip of the iceberg sign” (thin white arrow).
An isolated MCT is typically an insignificant and incidental finding as they are most often benign.3,5 However, any ovarian mass greater than 5 cm increases the risk for ovarian torsion, which is a medical emergency.5,11,14,15 Ovarian torsion is a rare complication that disrupts the blood and lymphatic flow to one or both ovaries by total or partial twisting of the vessels supplying ovarian tissue. Approximately 2.7% of acute pelvic pain cases requiring surgery are due to torsion.1,14,16 Variable rates of torsion in the presence of acute pelvic pain have been reported as high as 16% and as low as 0.2%.3,5,17,18 Women’s age seems to influence the likelihood of torsion, with the greatest incidence of torsion in the second to third decade of life. Still, younger and postmenopausal women exhibiting acute abdominal pain should not be excluded from the possibility of torsion due to age alone. 1
Currently the ACR recommends sonography be used for characterizing torsion in real time. 19 Typically, patients with ovarian torsion present with acute abdominal and pelvic pain with nausea, vomiting, and tenderness upon palpation in the adnexal region.1,11 These symptoms are indications for transabdominal and transvaginal ultrasonography assessing for an adnexal mass and potentially torsion.
Transvaginal sonograms have become the preferred diagnostic imaging study to evaluate for ovarian torsion. 11 The combined use of color, power, and spectral Doppler can confirm the presence of blood flow to an ovary and therefore exclude torsion in most cases.1,11 In some instances, though, arterial flow can be appreciated due to collateral flow from the terminal uterine arteries or if the adnexal vessels are only partially twisted.11,14 If some flow is present, it may be diminished compared to the normal contralateral ovarian tissue. 11 Other sonographic findings associated with ovarian torsion include enlarged edematous ovarian parenchyma, free fluid within the posterior cul-de-sac (Figure 5), and the “whirlpool sign.” When the ovarian pedicle twists upon itself, a “whirlpool” or “bull’s-eye” appearance may be appreciated in gray scale or with color Doppler. The presence of this sign may increase the chance of positive torsion diagnosis by up to 90%.18,20 When associated with an ovarian mass, the presence of chronic ovarian ischemia should be considered, although it is rare (5%). 11
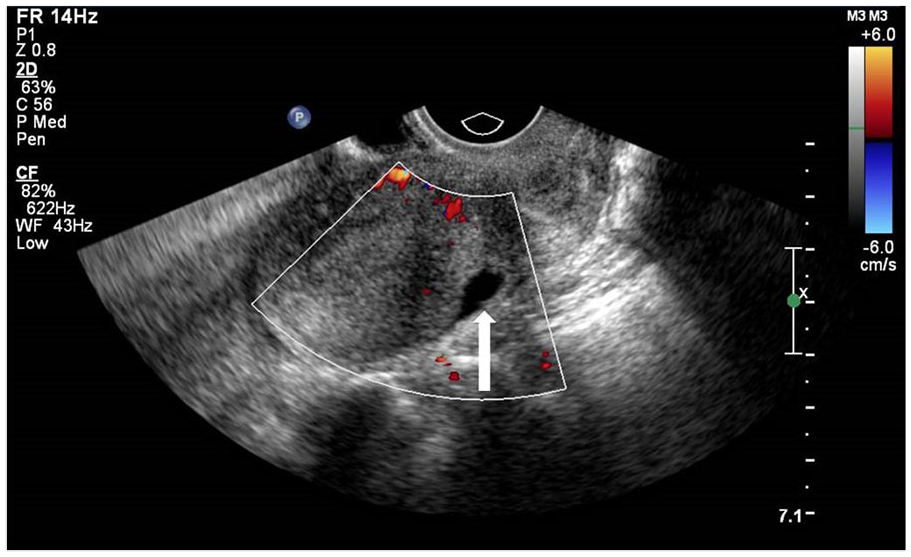
Transvaginal sonographic image. Free fluid within the posterior cul-de-sac (solid white arrow).
Laparoscopy is the surgical procedure of choice due to the decreased occurrence of incision complications, diminished pain, shorter patient stay, less incidence of adhesions, and a quicker recovery compared to more invasive surgical options. 1 Multiple case studies of ovarian torsion suggest an attempt should be made to restore blood flow to the ovary to preserve it at the time of the initial surgery, as opposed to an oophorectomy.1,14 In a study conducted on rodents, Taskin et al. 21 reported no long-term reperfusion injury was observed in the group of rats where torsion persisted less than 24 hours. Chu et al. 14 reported ovarian preservation in two separate patients’ posttorsion, which led to normal parenchymal color at the time of surgery, and upon follow-up examination by pelvic ultrasound, normal ovarian size and blood flow were appreciated. With the risk of torsion being highest during childbearing years, ovarian conservation after torsion is important for maintaining both fertility and biochemical benefits.1,14
An indication for oophorectomy is confirmed ischemia and ovarian tissue necrosis. 11 During surgical intervention, an ischemic or hemorrhagic ovary can have similar appearance indicating venous and lymphatic stasis, as opposed to necrosis. Therefore, detorsion can still be attempted to hopefully maintain the ovary. 14 In the event of an ovarian tumor causing torsion, detorsion and removal of the mass are preferred. 1
Conclusion
Sonography is the imaging study of choice for evaluating and characterizing MCT and ovarian torsion.3,5,11,12 In patients presenting with large adnexal masses, transabdominal sonography may be more useful that transvaginal sonography in characterizing the tumor, and extended field-of-view technology may provide an overall view of larger masses. Sonographic attributes such as Rokitansky nodules, the dot-dash sign, a “tip of the iceberg sign,” or fat-fluid levels are highly predictive of MCT, while size evaluation of suspected teratomas can aid in determining malignant potential of the mass. Since large MCTs can cause ovarian torsion, it is essential to show blood flow to the ovarian tissue using color, power, and spectral Doppler. The sonographic whirlpool sign is highly predictive of ovarian torsion.
Footnotes
Acknowledgements
I thank Emily Downs, RDMS, RVT, RDCS, RT(R), and Teresa Bieker, MBA, CRA, RDMS, RDCS, RVT, for their revisions, feedback, and support throughout this process. I also thank Eric Schaffer, MS, RDMS, RVT, RDCS, for his support and guidance, as well as Toshi Clark, MD, for his insight and consultation.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
