Abstract
During fetal development, abnormalities during Mullerian duct formation can lead to varied types of uterine, cervical, or vaginal anomalies. Herlyn-Werner-Wunderlich Syndrome (HWWS) is a rare, congenital Mullerian duct anomaly characterized by the triad of uterine didelphys, obstructed hemi-vagina, and ipsilateral renal agenesis. If the diagnosis is delayed or missed, permanent sequalae such as chronic pain, infection, and infertility can result. This is a case of a 16-year-old woman with heavy vaginal bleeding and menstrual cramping in which sonography was used in the diagnosis of HWWS. Sonography plays a vital role in this diagnosis, as it is the most common initial imaging examination. For this reason, it is important that the characteristics of uterine anomalies, like HWWS, be recognized with sonography. It is important that this diagnosis is made early and treatment can be initiated to prevent irreversible complications.
Keywords
Anomalies of the reproductive tract affect less than 5% of women. 1 The exact cause is not completely understood; however, most result from an arrested development of primordial embryologic structures. The development of the genital tract begins during the third week of pregnancy and carries into the second trimester. In 1957, Crosby et al proposed that the fusion of the two Mullerian ducts starts at their caudal ends in Muller’s tubercle and proceeds cranially up to the fundus. 2 Failure of ducts to develop leads to various types of uterine, cervical, or vaginal anomalies, while absent or incomplete fusion results in uterus didelphys. 2 The formation of the reproductive tract is associated with the development of the urinary system; therefore, woman with anomalies of the reproductive system can have concomitant anomalies of the renal system.
Herlyn-Werner-Wunderlich Syndrome (HWWS) is a rare, congenital Mullerian duct anomaly characterized by the triad of uterine didelphys, obstructed hemi-vagina, and ipsilateral renal agenesis (OHVIRA).1–3 The obstructed hemi-vagina causes the menstrual fluid to accumulate in the vagina otherwise known as hematocolpos. This leads to the classic presenting symptoms of menstrual cramping, abdominal pain, and a suprapubic mass.3,4 Less common presentations include abnormal vaginal discharge, urinary retention, pelvic infection, blood clots, infertility, recurrent miscarriages, and endometriosis.1–3 With an incidence estimated to be between 0.1% and 3.8%, a high level of clinical suspicion is necessary to make the diagnosis. 1
Symptoms typically start within a few months to a year following menarche and often prompt initial investigation with sonography.5,6 One of the main complications associated with this syndrome is endometriosis. Endometriosis is characterized by ectopic endometrial implants on the female pelvic organs. An obstructed outflow tract often leads to retrograde menstruation into the uterus (hematometra), fallopian tubes (hematosalpinx), and pelvic cavity (hemoperitoneum). The retrograde menstruation is the mechanism of action that leads to endometriotic implants within the pelvis. Other less common complications include pelvic inflammatory disease, tubo-ovarian abscess, and pelvic adhesions that may in turn adversely affect fertility.7,8 The goal of treatment is to create a patent opening to allow normal menstruation and optimize fertility. 7 While the treatment of choice is full resection of the vaginal septum via hysteroscopy, differences in technique and approach are based on variations in anatomy or secondary goals like preserving the hymen.1,4,6,7 Being able to recognize this syndrome early within the clinical course can prevent irreversible complications that may result in chronic pelvic pain and infertility. 9
Case Report
A 16-year-old woman presented to the emergency department with heavy vaginal bleeding and menstrual cramping lasting 7 days. Her menarche was at age 13 with regular light menstrual cycles that typically last 3–4 days. However, she skipped her menses during the previous month, and during this cycle, the patient experienced an episode of excessive bleeding with clots. Upon admission to the emergency department, a transabdominal pelvic sonogram was performed using a GE Logiq E9 with a C1–6 curved-array transducer to assess the uterus and ovaries. The sonogram revealed a heterogeneous fluid collection within the endometrial canal, a normal uterine myometrium, and a possible right hematosalpinx versus a right ovarian cyst. The attending obstetrics and gynecology physician, on call, was assigned to the case. He requested that the patient make an office appointment for a repeat pelvic sonogram, in a few weeks. A transabdominal pelvic sonogram was performed 3 weeks later using a GE Voluson E8 (Austria GmbH & Co OG) with a GE C1-5-RS curved-array transducer. This sonographic examination revealed what appeared to be complete uterine didelphys on transverse imaging with a separate right and left uterine horn (Figure 1). Longitudinal imaging of the right uterus revealed an extensive amount of heterogeneous material present within the right endocervical canal and vagina measuring 9.0 cm × 2.9 cm × 3.58 cm in size (Figure 2). The left uterus and cervix were grossly unremarkable. The right adnexa contained a tubular-shaped, complex mass adjacent to the right ovary. Magnetic resonance imaging (MRI) of the pelvis was then ordered to further evaluate the findings noted with sonography.
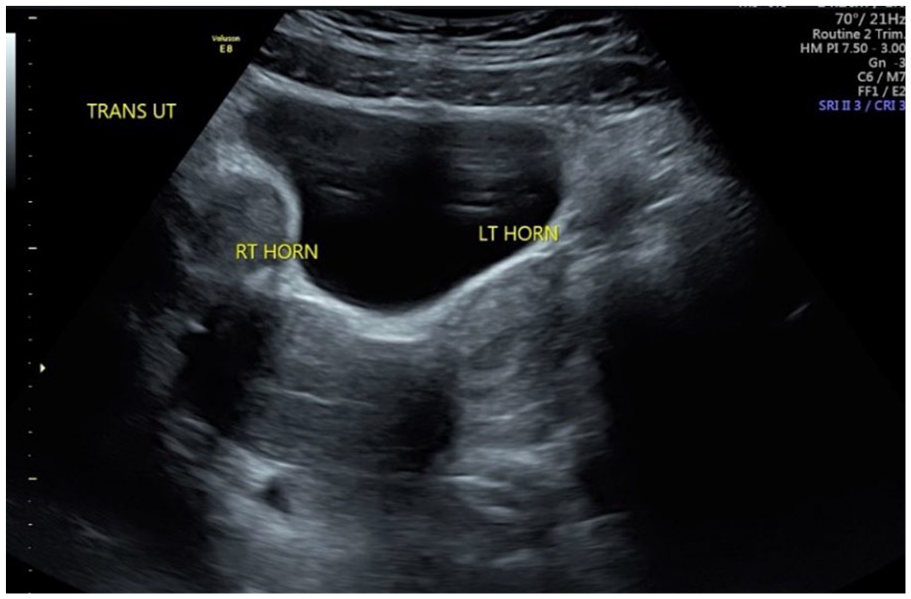
Transverse, transabdominal sonogram demonstrating a right uterine horn and a separate left uterine horn suspicious for uterine didelphys.
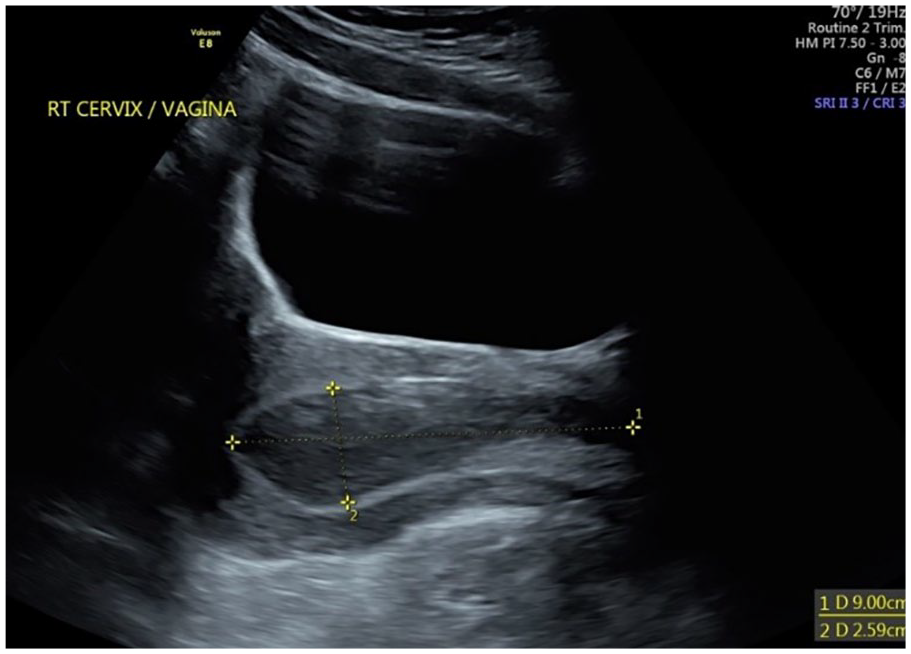
Longitudinal, transabdominal sonogram demonstrating the right cervix and vagina with hematocolpos measuring 9.0 cm × 2.9 cm.
The MRI demonstrated uterine didelphys with a vaginal duplication. The right hemi-vagina was obstructed causing hematometrocolpos. The right hemi-vagina was filled with debris and hemorrhage. There was a cystic mass adjacent to the right ovary measuring 2 cm in size, consistent with a dilated right fallopian tube and hydrosalpinx. The left uterus was not enlarged but did have fluid noted within the endometrial canal. The left cervix was patent and the left hemi-vagina was nonobstructed (Figures 3–6). The recommendation from the results of the MRI was to evaluate the renal tract to confirm a diagnosis of HWWS, which would also include ipsilateral renal agenesis.
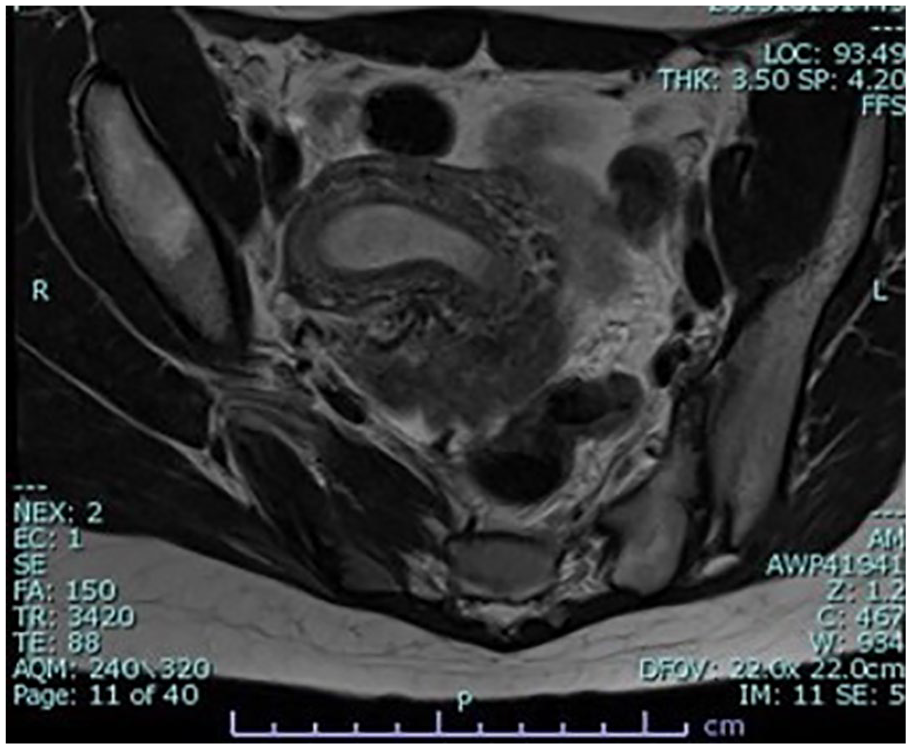
T2 magnetic resonance image demonstrates a right uterine fundus and endometrial stripe with hematometra.
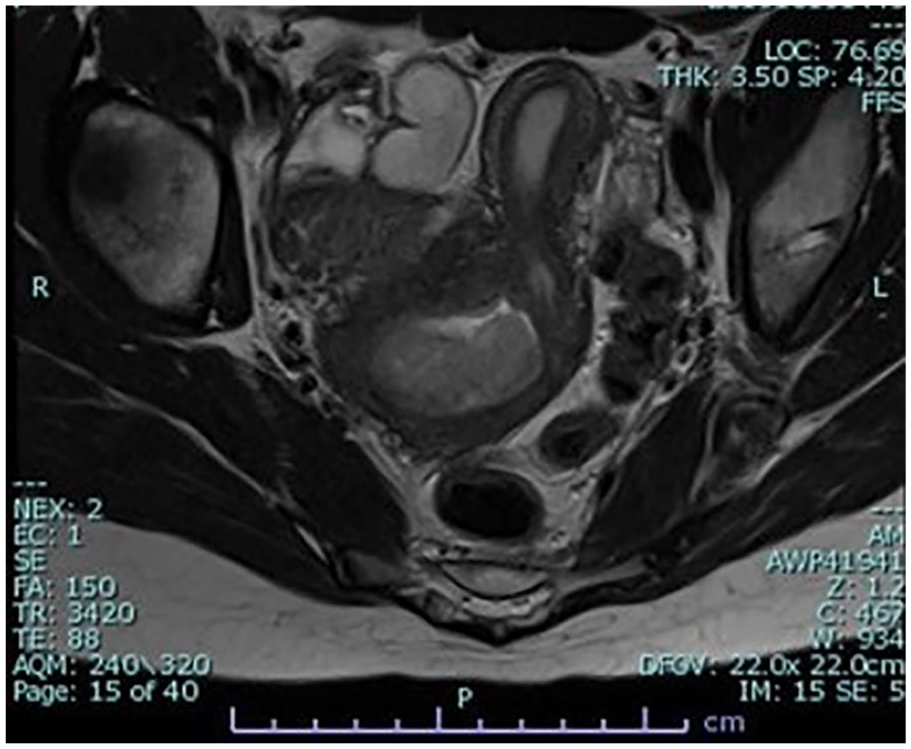
T2 magnetic resonance image demonstrates a dilated right cervix with hematocolpos and a left uterus deviating to the patient’s left.
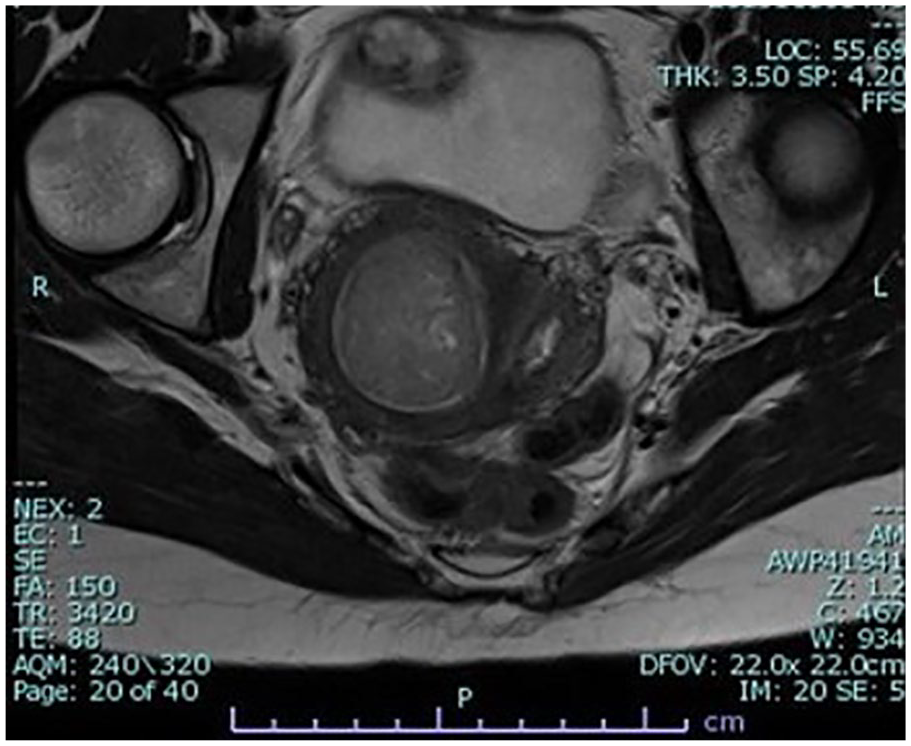
T2 magnetic resonance image demonstrates a side-by-side image of the dilated right cervix with hematocolpos and a normal left cervix.
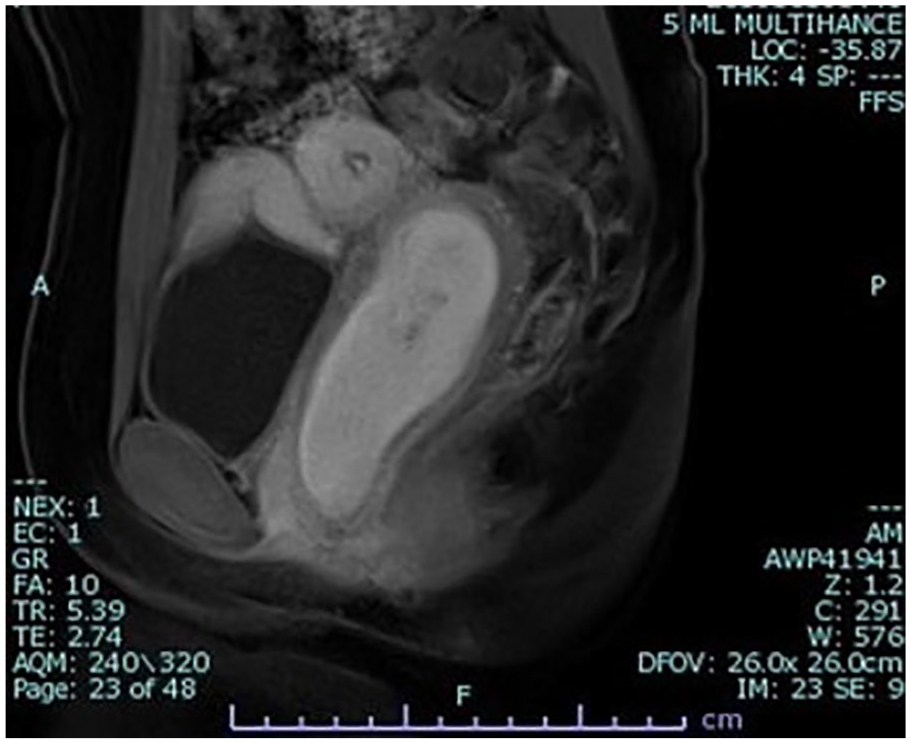
T2 magnetic resonance image demonstrates a sagittal image of the right uterus with hematometrocolpos and retrograde menstruation into the right fallopian tube.
The patient had a renal sonogram performed shortly after confirming ipsilateral renal agenesis of the right kidney with an empty right renal fossa. The left kidney was visualized in the correct location within the left renal fossa, inferior to the spleen (Figures 7 and 8).
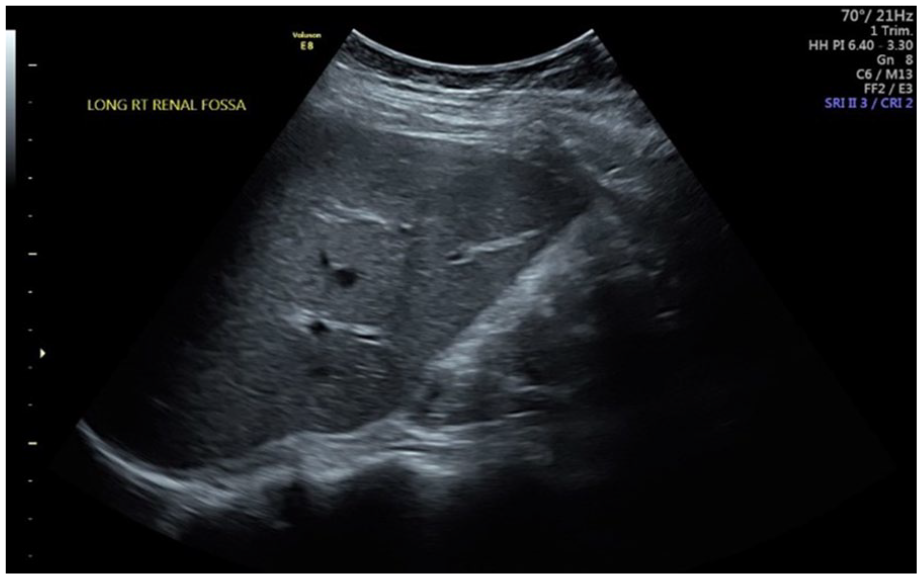
Longitudinal sonogram of the right renal fossa demonstrating a normal-appearing longitudinal right lobe of the liver. The right renal fossa is empty consistent with ipsilateral renal agenesis.
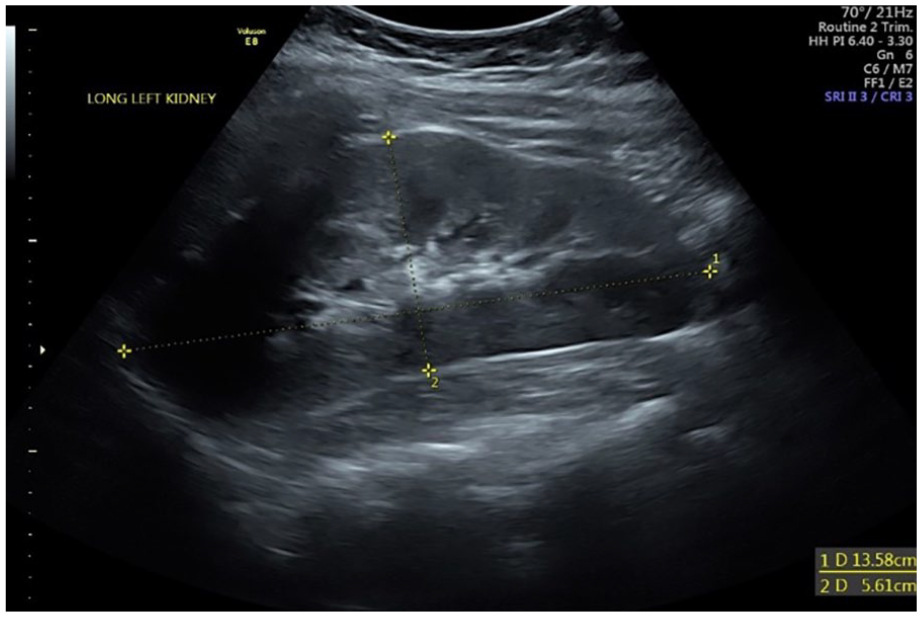
Longitudinal sonogram of the left renal located in the left renal fossa, inferior to the spleen.
With the clinical presentation, the patient was given continuous oral birth control for a year for menstrual suppression and to delay surgical intervention. Upon returning to the clinic 1 year later, a follow-up pelvic sonogram was performed using a GE Voluson E8 (Austria GmbH & Co OG) with a GE C1-5-RS curved-array transducer. On examination, uterine didelphys was again confirmed on transabdominal imaging, with the appearance of a worsening hematometrocolpos (Figures 9–13). The right uterus contained heterogeneous material within the vagina and cervix and extended into the endometrial cavity (Figure 14). The debris and hemorrhage noted within the endometrial canal measured 2.57 cm × 3.45 cm × 3.88 cm near the uterine fundus. The right adnexa demonstrated a hematosalpinx increasing in size from prior examinations measuring 8.0 cm × 1.95 cm × 2.2 cm (Figure 15).
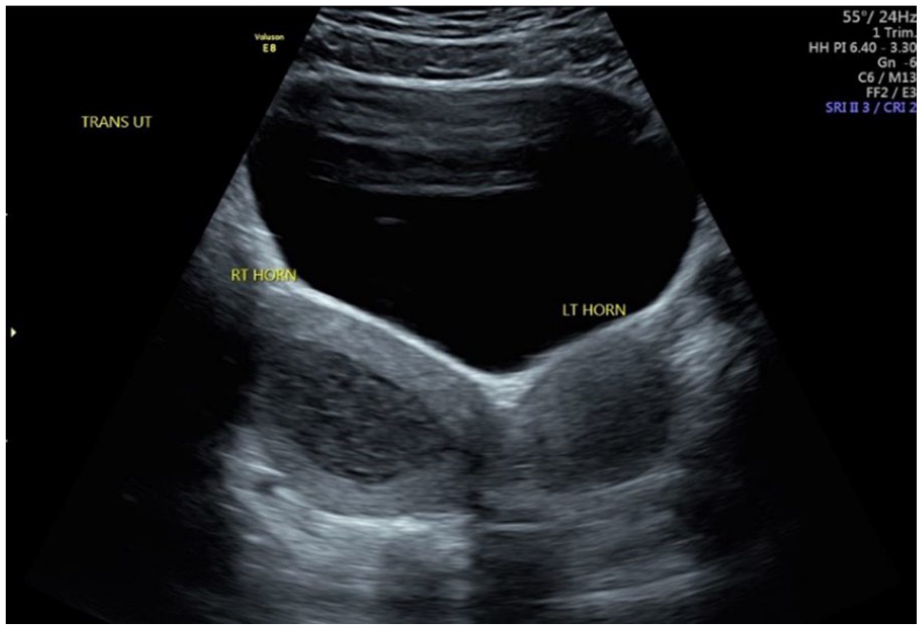
Transabdominal sonogram demonstrating uterine didelphys with an increase in amount of hematometra debris in the right endometrial canal.
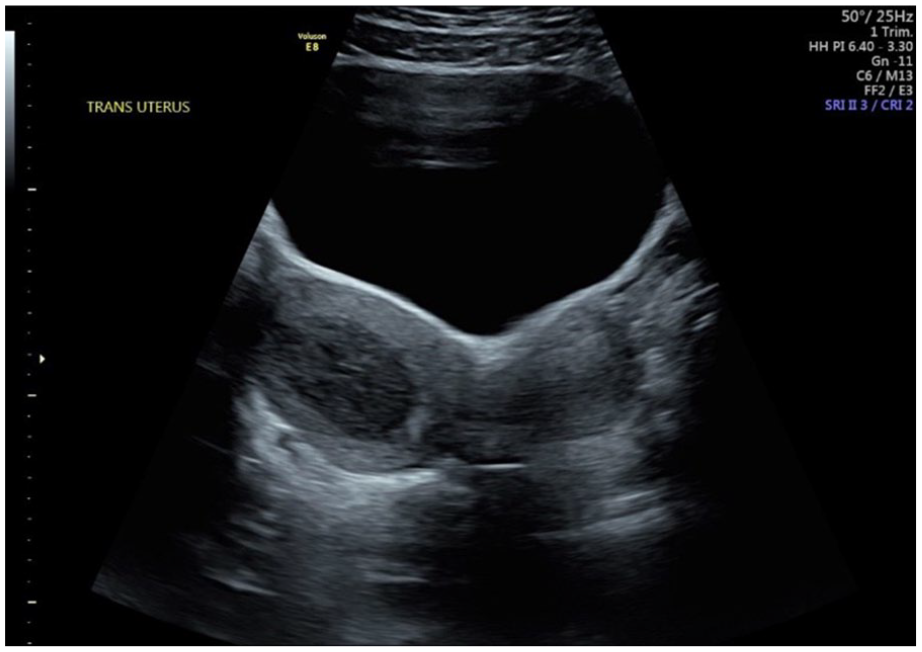
Transabdominal sonogram demonstrating uterine didelphys with separate right and left uterine horns and hematometra in the right endometrial canal.
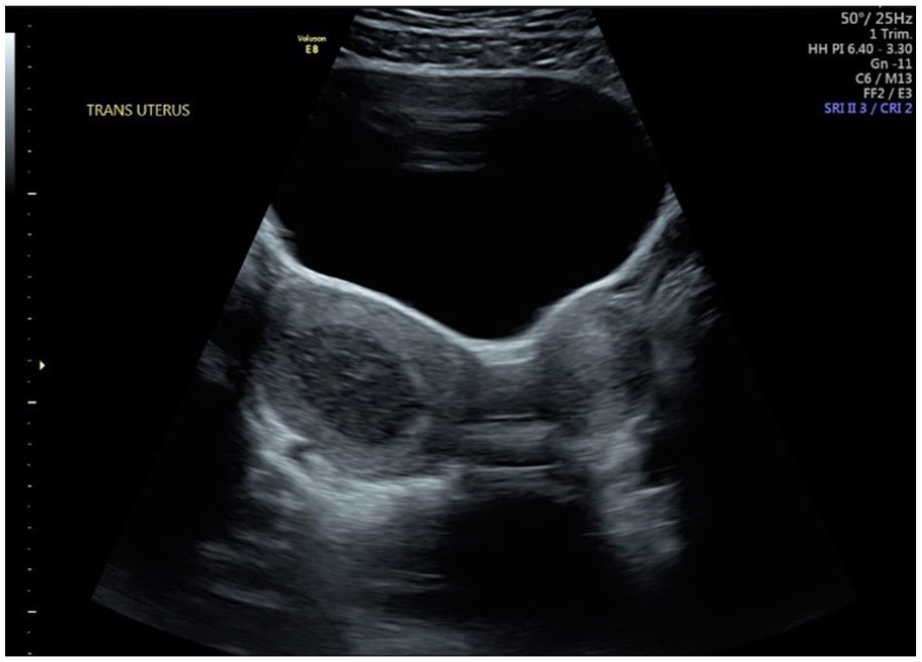
Transabdominal sonogram demonstrating uterine didelphys with separate right and left uterine horns and hematometra in the right endometrial canal.
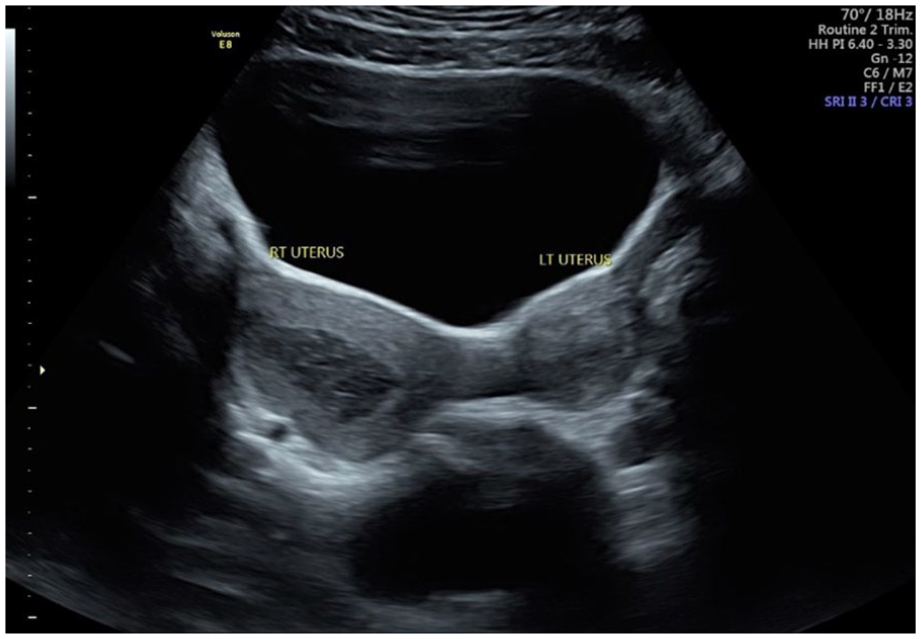
Transabdominal sonogram demonstrating uterine didelphys with separate right and left uterine horns and hematometra in the right endometrial canal.
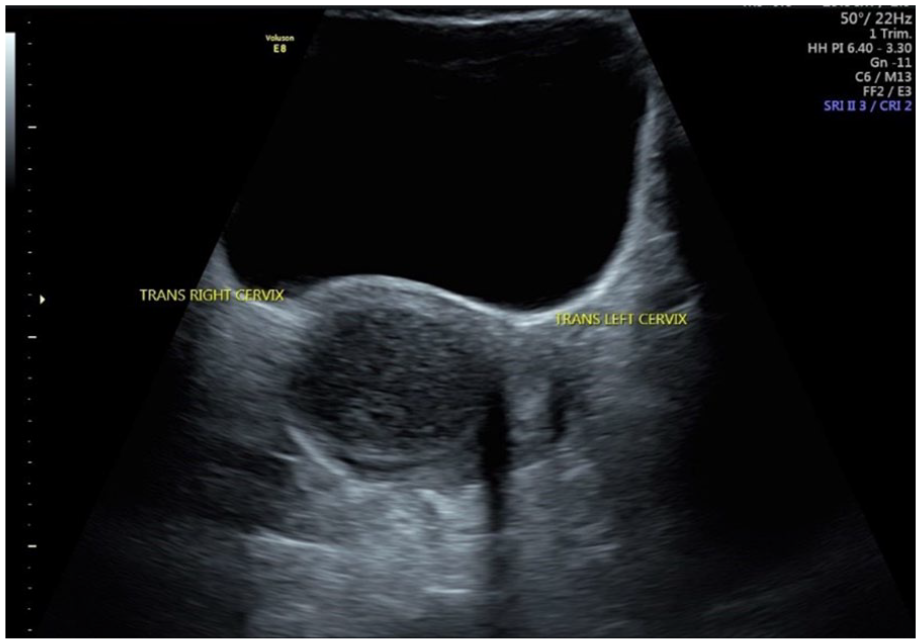
Transabdominal sonogram demonstrating hematometra within the right cervix and a normal left cervix.
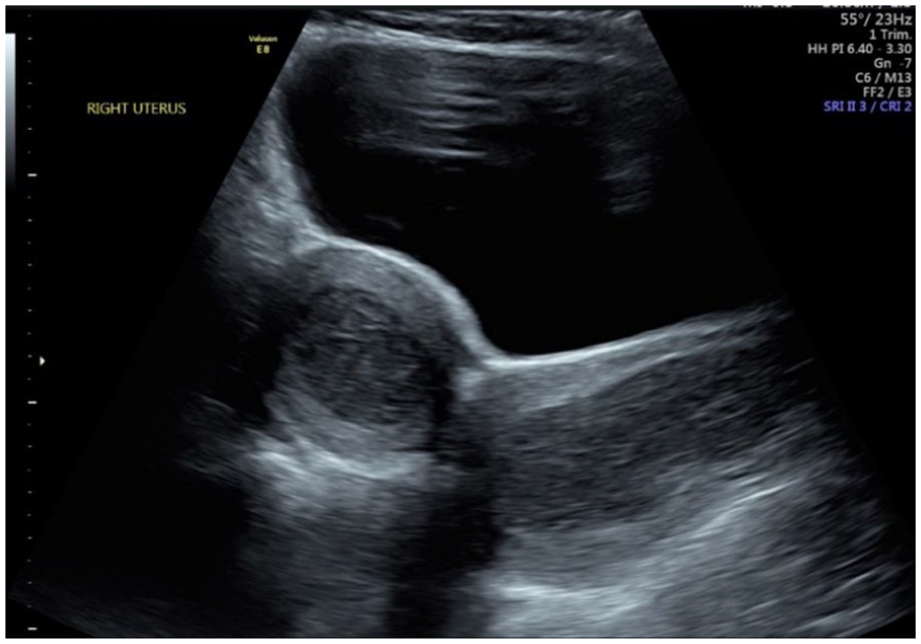
Longitudinal ultrasound of hematometrocolpos of the right uterus.
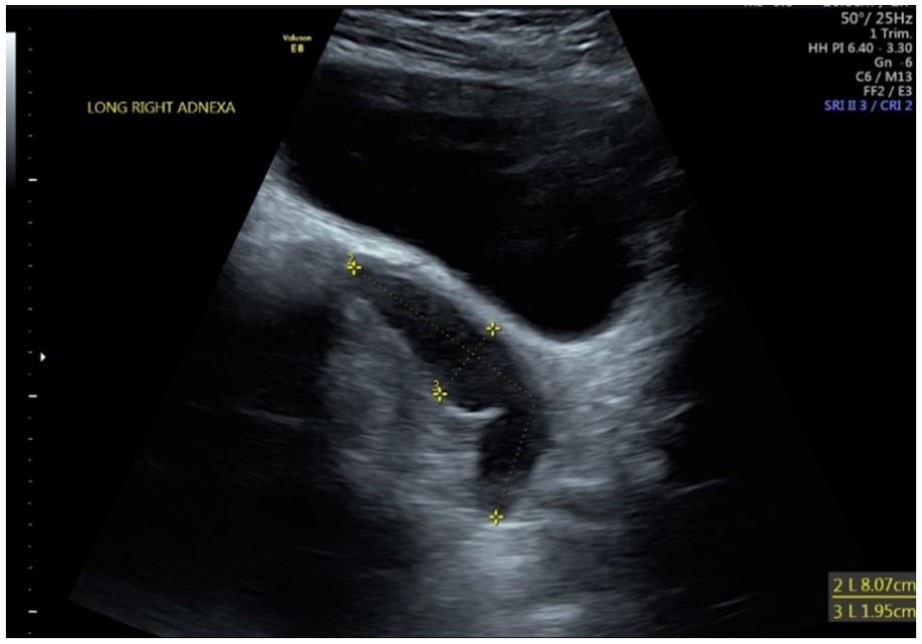
Longitudinal ultrasound demonstrating right hematosalpinx from retrograde menstruation into the right fallopian tube.
The patient underwent a colpotomy with decompression of the hematometra and hematocolpos. A 20-mm chevron incision was made at the dependent area of the right hemi-vagina. The septal tissue was resected. Approximately 100 mL of retained blood and menstrual fluid was expressed from the right uterus. The obstructed cervix was further dilated, and hysteroscopy was performed, which aided in the evacuation of menstrual fluid from the right uterus. The patient tolerated the procedure well. On clinical follow-up at 3 and 6 months after surgery, the patient had returned to normal menstrual cycles. She was put on oral contraceptives to aid in the regression of endometriosis. She reported minimal pelvic pain and had no signs of chronic infection.
Discussion
Different clinical presentations exist for those patients with HWWS.1,3,9–11 Zhu et al 12 developed a classification system for these clinical presentations and ease of reference. Class I consists of patients with complete hemi-vaginal obstruction with or without cervical atresia. Class II is characterized by a small communication between either the vaginas or uteri. 12 The sonographic features of the reproductive tract in both classes include uterine anomalies (didelphic/bicornuate uterus) and a hypoechoic area below one cervix. In the transverse imaging plane, widely divergent uterine horns can be seen. Because of the complete obstruction, class I is more likely to show an accumulation of menstrual fluid pooled in the uterus and an enlargement of the uterine cavity on a sonogram. 12 Patients initially present shortly after menarche with dysmenorrhea due to an obstructed hemi-vagina. In the case presented here, this patient would fall into class II—incompletely obstructed hemi-vagina. 12 Class II is often associated with delayed diagnosis and treatment. The delay in diagnosis in this case was likely due to a small communication between the two vaginas, which enabled regular menstrual flow for a period of 3 years after menarche. Over time, a clot or blockage likely formed and completely obstructed the patient’s hemi-vagina, causing the acute presentation.
Multiple anatomic variants of HWWS exist, necessitating the need for the sonogram to provide imaging evidence that leads to an accurate diagnosis. Fedele et al reviewed 87 HWWS cases and found 27.6% differed from the classical anatomic variant of uterine didelphys with OHVIRA. 13 These variants include a septate uterus where a membrane divides the inner portion of the uterus without external separation of the uterus, a bicornuate uterus where the external surface and the inner portions of the uterus are separated by varying lengths, and the more classic didelphys uterus which is basically two separate uteri with unilateral cervical atresia. The variants carry differing therapeutic implications. 13 In this case, the patient presented with the classic HWWS variant. However, without an understanding of these other variants, there may be a delay in diagnosis or inappropriate management approach.
Menstrual suppression is the preferred initial treatment according to the American College of Obstetrics and Gynecology (ACOG). 14 Menstrual suppression allows for relief of pelvic pain and provides time for an adolescent to mature and understand the diagnosis and the implications of surgical correction. It also promotes shared decision-making among family members. Surgical correction is the definitive treatment modality and is warranted with worsening symptoms despite conservative treatment with menstrual suppression.
During embryogenesis, the urinary and reproductive system development is closely related, which may explain the ipsilateral renal agenesis with the reproductive tract abnormalities of HWWS. 15 As such, suspicion should be heightened in patients with a single kidney. Tuna et al describes a case where an absent kidney was discovered on a 36-week prenatal sonogram. 16 A fetal MRI confirmed a cystic lesion behind the bladder and right renal agenesis. A vaginal septotomy was performed on the fourth day of life. 16 Earlier diagnosis and treatment helped avoid long-term complications and ultimately preserve future fertility.
Imaging is a critical step in diagnosis and is usually accomplished by sonography or MRI. Sonography is the first utility in real-time detection and is a low-cost option. High suspicion of uterine anomalies can be aided by the sonographic detection of hematocolpos, especially during adolescence. 15 According to a series of 40 cases reviewed by Zhang et al, sonographic findings were in accordance with MRI in 82.5% of the cases. 11 The remaining cases were diagnosed as uterine didelphys by sonography but were actually bicornuate or septate by MRI. 11 Sonography is important in the initial suspicion and diagnosis as early as prenatal imaging. It can be used to evaluate progression of HWWS as in the case reported.
MRI is considered the gold standard for definitive diagnosis of HWWS. 9 This imaging modality can provide enhanced detail and diagnostic accuracy to complement sonographic findings. Most importantly, it has utility in treatment planning as MRI can better assess the site of obstruction. 11 Ultimately, MRI is recommended to further characterize the uterine abnormality and site of obstruction, and determine approach to treatment. In addition, MRI is preferred for the investigation of pediatric Mullerian duct abnormalities. 16 The uterus in the neonatal period is marked by a prominent myometrium and echogenic endometrium due to maternal hormonal stimulation. 15 In infancy, the hormonal stimulation is no longer present, and thus sonographic uterine evaluation becomes much more challenging until menarche. This is on account of the prepubertal uterus having thin walls, which makes it difficult to distinguish from the cervix. With the hormonal stimulation that accompanies puberty, the uterus enlarges and the fundus becomes much more prominent. 17
Conclusion
The correct diagnosis of uterine anomalies requires familiarity with diagnostic criteria, recognition of corresponding imaging, and accuracy in obtaining/reading those images. 18 Accuracy is vital to determine proper treatment, which can vary not only between classes but also among anatomic variants of uterine anomalies.8,10 The sonographic diagnosis relies on being able to recognize two distinct uterine cavities that are widely separated in the transverse plane. If one of the cavities is noncommunicating, a hematometra or hematocolpos may result, which is evident by the presence of fluid distending either uterine or vaginal cavities. Sonography plays a vital role in diagnosis as the most common initial imaging performed.8–11 For this reason, it is important that the characteristics of uterine anomalies like HWWS be recognized with sonography so that the diagnosis is made early, treatment can be initiated, and complications avoided.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Permission Statement
Permission was granted by the patient to publish the case.
