Abstract
Objectives:
This study developed a musculoskeletal ultrasound (MSUS) protocol to evaluate rehabilitation outcomes in systemic sclerosis.
Materials and Methods:
Three MSUS methods (gray-scale, Doppler, strain elastography) and two acquisition techniques (long- vs short-axis; transducer on skin vs floating on gel) were examined in the forearm before and after rehabilitation treatment. For gray-scale, tissue thickness measures and intrarater and interrater reliability were calculated (intraclass correlation coefficients [ICCs]), and paired
Results:
Five people with diffuse cutaneous systemic sclerosis participated. The most valid and reliable gray-scale technique was with the transducer in long-axis, floating on gel. Doppler and strain elastography did not detect changes. Both dermal and subcutaneous thickness measurement error was small; intrarater and interrater reliability was good to excellent. Preliminary data indicate that treatment may lead to dermal thinning.
Conclusion:
A replicable protocol was established and may be an adjunct to rehabilitation outcome measurement in systemic sclerosis.
Systemic sclerosis (SSc) is a chronic autoimmune disease that results in excess collagen production and vascular changes.
1
Skin thickening is a key metric used by rheumatologists to track disease severity and progression. In rehabilitation, skin thickening is also important to occupational and physical therapists as they seek to improve functional consequences of skin thickening, notably, restriction of range of motion in fingers, hands, and arms; reduced strength; and associated pain. Therapists measure effectiveness of their treatments based on the ability to improve movement, function, and pain, but do not typically measure skin involvement as an outcome in rehabilitation. Even when function improves, therapists often do not have methods to understand
Clinical evaluation of skin involvement, particularly skin stiffness, is typically completed using the modified Rodnan Skin Score (mRSS), which involves applying pressure at various points on the body. 2 Although the mRSS is the gold standard in rheumatology practice and research trials, 3 there is high measurement variability among raters, 4 and it may not be sensitive in later-stage disease and in specific subgroups.4,5
Musculoskeletal ultrasound (MSUS) is increasingly used in rheumatology; it is relatively inexpensive, noninvasive, and well tolerated by patients. 6 Current applications of MSUS in rheumatology include diagnosis of inflammatory and noninflammatory rheumatic disease and assessment of treatment response.7-9 MSUS has the potential to add sensitivity and precision to measure changes in dermal and subcutaneous tissues,10-12 which would be important in rehabilitation in which these tissues are manipulated with the goal of improving movement. Existing MSUS protocols for evaluating the skin are highly heterogeneous, 13 and there are currently no widely adopted standardized protocols to measure skin involvement in SSc. Only one study could be found that used MSUS to examine the link between disease features and upper extremity function in SSc which focused only on the hand. 14
To address critical gaps, our team developed an MSUS protocol to be used to detect changes in skin with rehabilitation treatments such as manual therapy or negative pressure treatment which involved treatment of the forearm and wrist. Our interdisciplinary team consisted of a rheumatologist, radiologist specialized in musculoskeletal disorders, rehabilitation ultrasonographer, occupational therapy practitioners, and rehabilitation researchers. This study aimed to determine which MSUS image acquisition and analysis techniques were most valid and useful for examining morphology and pathophysiology of primary structures, of interest in SSc upper extremity rehabilitation. The potential for MSUS, to identify change in these outcome measures, was also explored.
Methods
A pre-post, multiple case series design was used to develop, refine, and evaluate an MSUS protocol for SSc patients receiving upper extremity rehabilitation. The study was approved by the university’s institutional review board. All participants provided written informed consent.
Participants
Five participants with SSc were recruited from the Scleroderma Center between August 2019 and January 2020. To be eligible, participants needed to be older than 18 years, have a diagnosis of diffuse cutaneous SSc, be in an early disease stage (<5 years from first non-Raynaud’s symptom), and be English speaking. Exclusion criteria were any issues that would preclude meaningful participation in study procedures (e.g., concurrent medical issues, inability to tolerate treatment, inability to provide consent, and active hand ulcers on both extremities).
After eligibility was confirmed, each participant was scheduled for a research visit at the radiology clinic. At the visit, demographic data and common SSc clinical measures were obtained. Demographic information included age, sex, ethnicity, education, and employment status. Self-reported symptoms of pain, fatigue, and stiffness were rated on a 0–10 rating scale of severity, and upper extremity disability was assessed, using a shortened version of the Disabilities of the Arm, Shoulder and Hand (QuickDASH). 15 The participant’s self-reported physical function was assessed with the Patient-Reported Outcomes Measurement Information System (PROMIS) Physical Function scale 8b. 16 Objective hand and arm measures were collected by the therapist clinician on the study team (M.B.). Total active motion (TAM) was calculated by summing total active range of motion (ROM) for each finger and thumb using a goniometer; 1175° is possible. 17 Grip strength was measured by Jamar dynamometer in pounds of pressure; 18 the mean value of three trials on the right upper extremity is reported. Skin hardness was assessed by a durometer; a higher score means more severe skin hardness. 19 After baseline measures were obtained at the visit, all participants underwent a pretreatment MSUS assessment, a negative pressure occupational therapy treatment, and a posttreatment MSUS assessment that was repeated immediately following treatment. All treatments and measures were obtained on the right forearm (except for participant 4 who had an active hand ulcer on the right forearm, so the left forearm was used). The visit lasted between 1 and 2 hours.
Treatment
Tissue mobilization was conducted using LymphaTouch (LymphaTouch LLC, Helsinki, Finland), an electronic method of mild tissue mobilization that lifts or stretches the skin using negative pressure to promote manual lymph drainage. 20 The device has been previously tested as part of a broader occupational therapy intervention for SSc. 21 Although the mechanism of action for SSc rehabilitation is not known, this treatment is hypothesized to break up skin adhesions, allowing freer movement and improving hand and arm function.
Each participant received treatment on dorsal and volar aspects of their forearm and wrist using a standardized protocol with the LymphaTouch device. Working from proximal to distal beginning at the lateral elbow, ten 2-second pulses were provided along the length of the dorsal forearm covering approximately eight areas using the 60-mm head with a gray ring. Next, using either the 50- or 60-mm head, ten pulses were used in two placements over the dorsal and volar hand. Finally, a 35-mm head was used to apply ten pulses to each finger volarly and dorsally. The same procedure was then completed distal to proximal from the dorsal wrist over the dorsal forearm and finally from the volar wrist to the medial elbow. The pressure used was the maximum that the patient could tolerate, around 200–250 mm Hg.
Sonographic Image Acquisition
Sonography was conducted using a GE LOGIQ 9 ultrasound system (GE Healthcare, Milwaukee, WI, USA) and a 12-mHz linear array transducer with frequency set at 15.0 Hz and gain between 41 and 43. As much as possible, all environmental conditions were standardized across participants and treatment and evaluations were conducted in the same room. A radiologist with training in musculoskeletal techniques acquired all images. To maintain quality control, the transducers were checked by the radiologist and ultrasonographer prior to imaging.
For the first two participants, a full regional scan of the upper extremity was conducted. 22 The scan began with examination of the anterior and posterior humeroradial and humeroulnar joints at the elbow in both longitudinal and transverse, with the participant positioned both in full elbow extension and in 90° of flexion. Longitudinal and transverse scans of both ulnar and radial sides of the elbow, forearm, and wrist were completed beginning on the dorsal side and followed by a full scan of the volar side. The study team used the images obtained from the comprehensive scans of these two participants to develop a standardized SSc protocol for the final three participants. Specifically, locations, images, and sonographic techniques were selected to best examine those tissues and sites primarily targeted by the rehabilitation treatment.
For the standardized SSc protocol, participants sat in a chair with the right upper extremity positioned palm down with the volar forearm and elbow supported on a table. The hand was positioned at the table’s edge, supported at the distal radial-ulnar joint so that the fingers dangled freely. The dorsal surface of the participant’s forearm was measured from the lateral epicondyle to the distal end of ulnar styloid, divided into thirds, and the midpoint of each third marked with an X using a surgical dermatological marker (see Figure 1). Sonographic images and skin hardness were obtained at each of the three sites marked with an X.
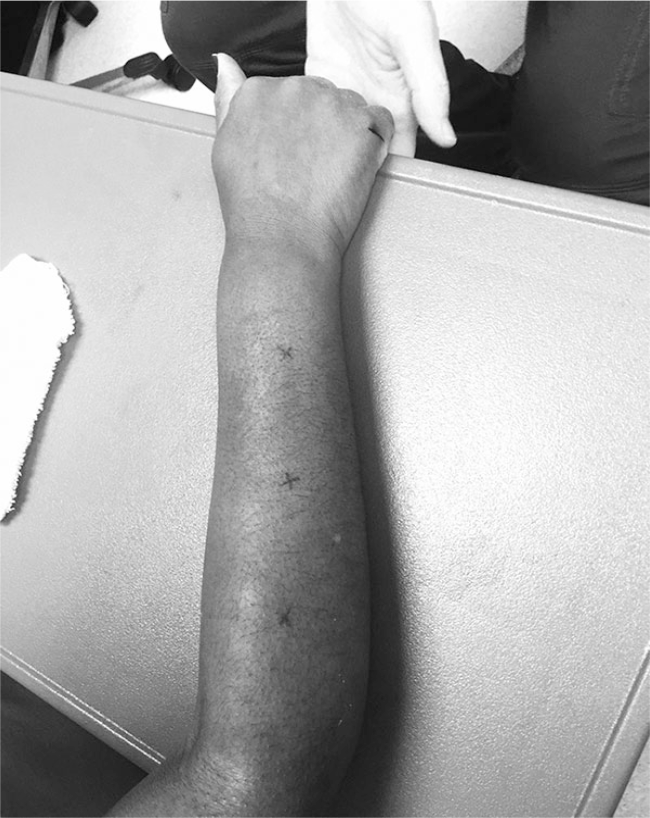
The position of the extremity and the imaging marks.
Image acquisition was conducted with the transducer in both transverse (short-axis) and longitudinal (long-axis) planes using gray-scale, color Doppler, and strain elastography at each of the three sites. Gray-scale images were obtained using two techniques to identify the most valid and useful acquisition protocol for examination of dermal and subcutaneous thickness (Figure 2). First, as noted in documented recommendations, 23 copious gel was used to allow the transducer to float without causing tissue compression. Second, the transducer was allowed to sit on the skin surface while the radiologist was cautious not to apply any additional pressure beyond weight of the transducer itself. Color Doppler was used to examine vascularity or blood flow changes within the subcutaneous soft tissue, muscles, or tendons at each treatment site; the number or intensity of colored pixels indicates the amount of active blood perfusion. 24 Strain elastography images were obtained using a free-hand technique by the radiologist in which a gradual, uniform, and repetitive manual compression was applied through the transducer; 25 tissues’ elastic properties are reflected in colors assigned to each pixel with a scale ranging from red—softest tissue with high elasticity—to blue—hardest, minimal elasticity tissue structure. 26
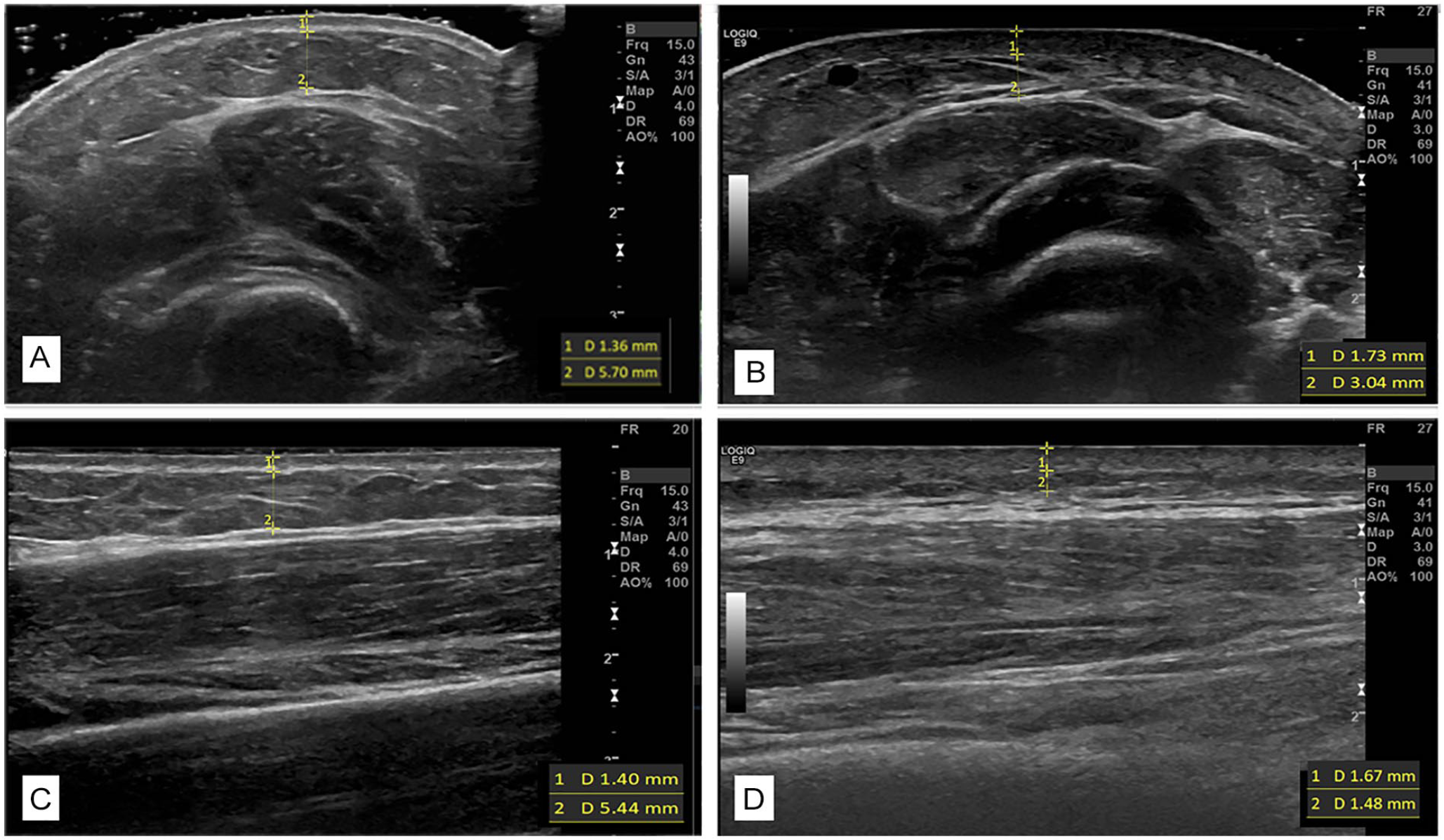
Gray-scale images of the forearm demonstrating (A) floating of transducer in the cross-sectional plane versus (B) transducer on the surface in the same place, as images in the longitudinal planes (C) floating and (D) on the surface. Borders and measures for dermal and subcutaneous images included.
Sonographic image analysis
All images were numbered, deidentified, and transferred into ViewPoint 6 software (GE Healthcare) for analysis. Two researchers neither obtained images nor provided treatments, one novice (D.M.K., rater 1) and one expert who was a registered musculoskeletal ultrasonographer with more than 10 years of experience (S.C.R., rater 2). Because images from the first two participants were used to develop a replicable protocol for the remaining participants, only images for participants 3, 4, and 5 were used in analysis. The raters reviewed all images from these last three participants and agreed upon the highest quality images to be analyzed. One gray-scale image was selected for each forearm location (sites 1, 2, 3 from proximal to distal), time (pre, post), axis (short, long), and transducer placement (floating, surface), a total of 24 images per participant. In the case that a high-quality gray-scale image was not available, Doppler or elastography images were interrogated by the researchers to identify possible replacement images. The process was completed for Doppler and elastography images.
The same two researchers (S.C.R., D.M.K.) independently measured dermal and subcutaneous thickness on each gray-scale image (Figure 2). Dermal thickness was identified as the distance between epidermal entrance echo and the subcutaneous fat from the deep edge of the epidermis to the deep edge of the dermal layer. 27 Typical dermal layer measurements in normal skin are 1.6 mm, but in SSc, it varies from 0.5 to 3 mm. 27 The subcutaneous layer was recognized as the distance between the deep edge of the dermal layer and superficial edge of the fascial layer of the muscle. Using digital calipers, measurements were taken in millimeters at a rater-selected point within the center one third of each image where the individual structural boundaries were the most echogenic. The average of three measurements of each layer was recorded, and each rater completed three trials for all measures.
For Doppler and elastography analysis, preintervention and postintervention images were placed side by side for each site and axis. Because no appreciable color was noted on any Doppler images, no further analyses were completed. A two-step process was used for elastography images. First, color transitions from blue (hard) to red (soft) within the dermal and subcutaneous layers in the region of interest (ROI) were subjectively examined using the modified lesion scale. 28 The amount of change in tissue hardness from pre to post was scored using a 4-point Likert scale: 0 = no difference noted, 1 = slight difference (25%–30% of the image color changed), 2 = moderate difference (50% of the image color changed), 3 = significant difference (>75% of colors in the ROI changed). Second, if more than 25% of image pairs were noted to have significant change in color (i.e., scoring 3 on the Likert scale), an objective quantification of magnitude of color change and color intensity within both the dermal and subcutaneous layers within the ROI was interrogated using ImageJ.
Statistical analyses
Descriptive statistics were calculated for demographic and clinical data. To evaluate measurement differences between raters, paired
Results
Descriptive information of participants, included in this case series study, is located in Table 1. Participants’ mean age was 48.2 years; four were female and one male. Mean baseline pain severity scores were 4 of 10. Participants reported below-average functional abilities on QuickDASH and PROMIS measures, had moderate limitations in total active ROM, and demonstrated below-average grip strengths. Skin hardness from the durometer assessment averaged 40.6 units across the landmark sites of the dorsal surface of the forearm.
The Study Participants’ Characteristics.
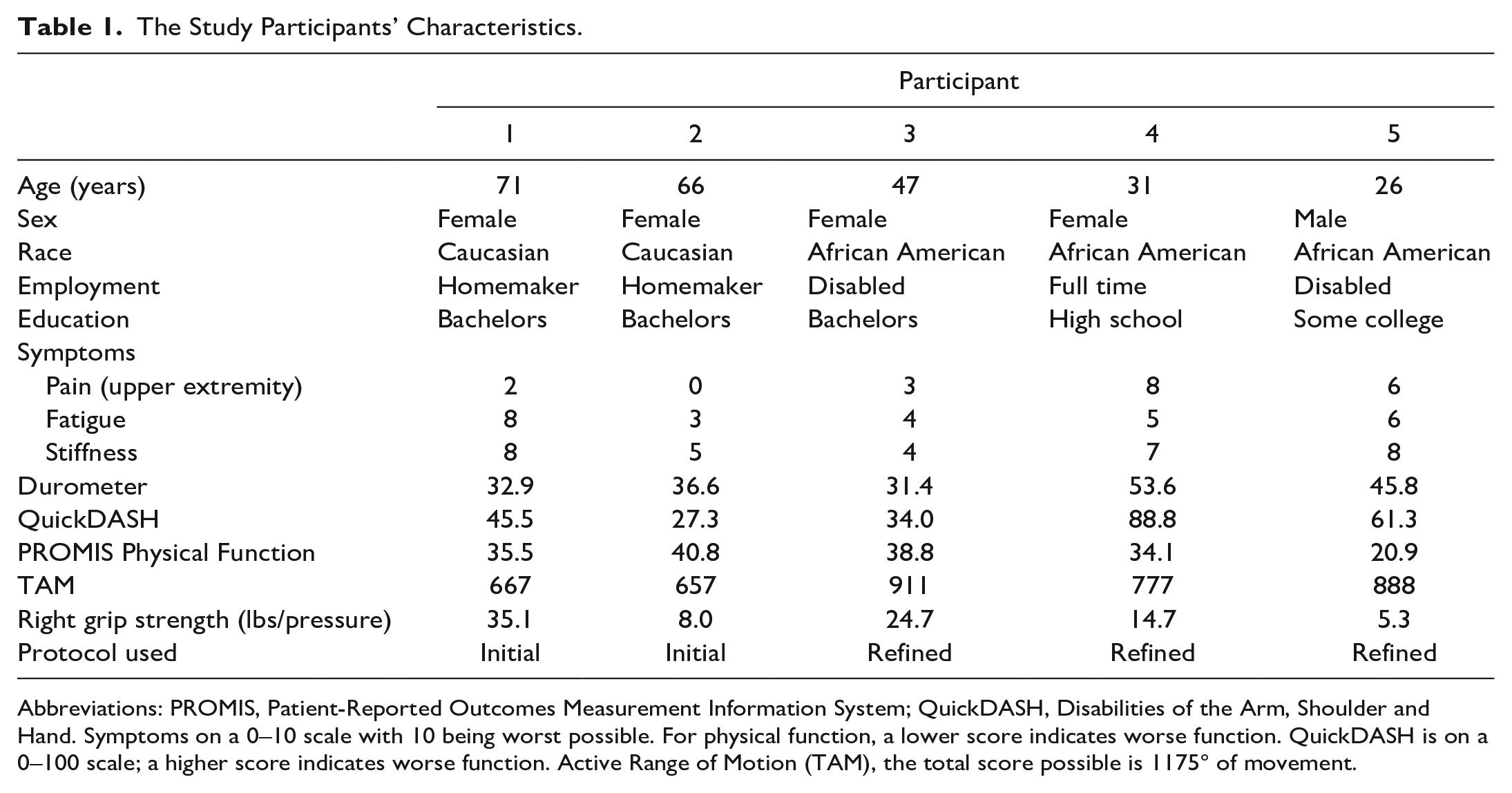
Abbreviations: PROMIS, Patient-Reported Outcomes Measurement Information System; QuickDASH, Disabilities of the Arm, Shoulder and Hand. Symptoms on a 0–10 scale with 10 being worst possible. For physical function, a lower score indicates worse function. QuickDASH is on a 0–100 scale; a higher score indicates worse function. Active Range of Motion (TAM), the total score possible is 1175° of movement.
Following the initial review, 63 images were identified as having adequate quality for measurement of dermal and subcutaneous thickness across the three participants. Based on expert rater’s measures (rater 2), the average dermal thickness was 1.74 mm (
Dermal Thickness Averages, Measurement Errors, and Reliability by Image Categorization.
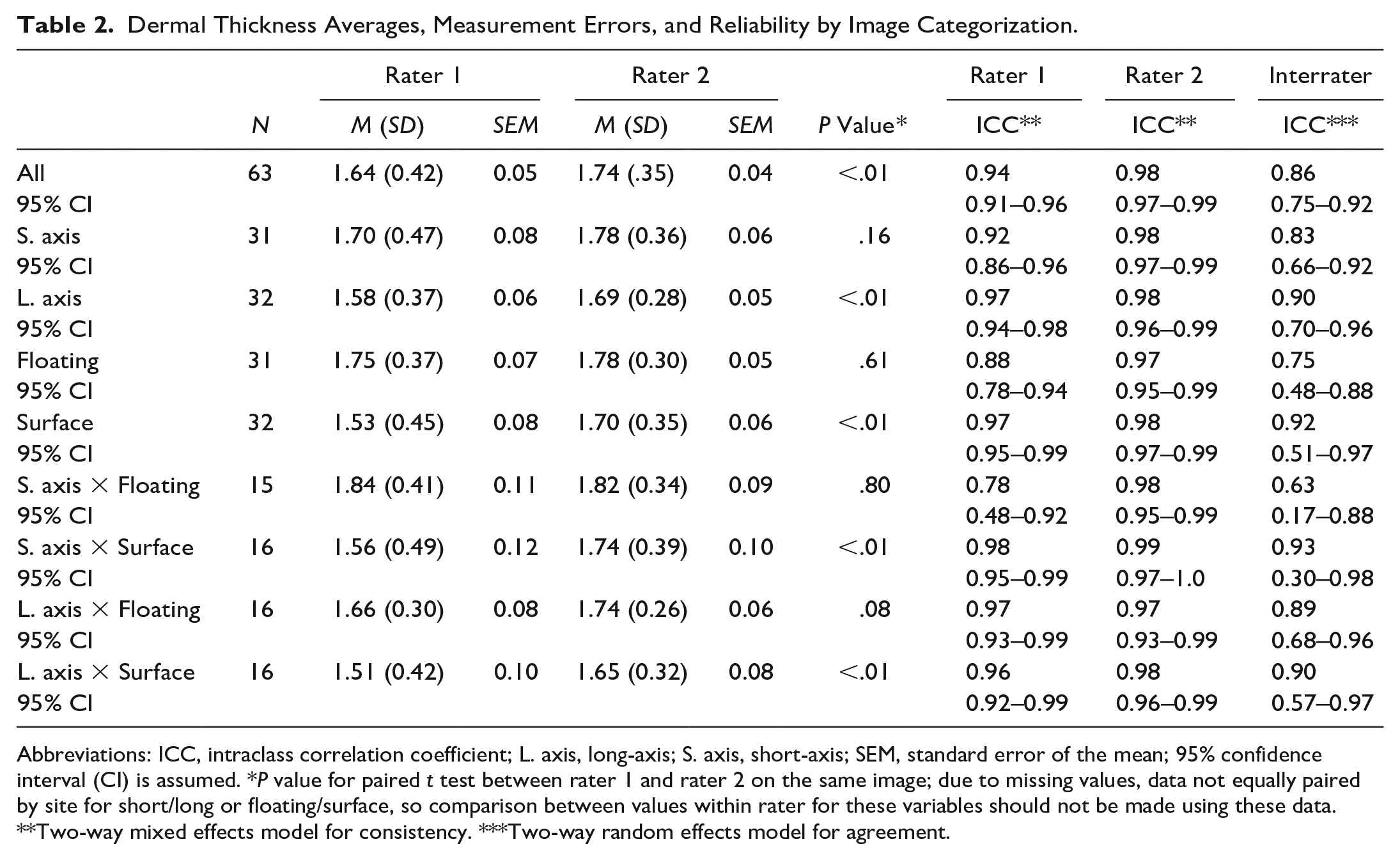
Abbreviations: ICC, intraclass correlation coefficient; L. axis, long-axis; S. axis, short-axis; SEM, standard error of the mean; 95% confidence interval (CI) is assumed. *
Subcutaneous Thickness Averages, Measurement Errors, and Reliability by Image Categorization.
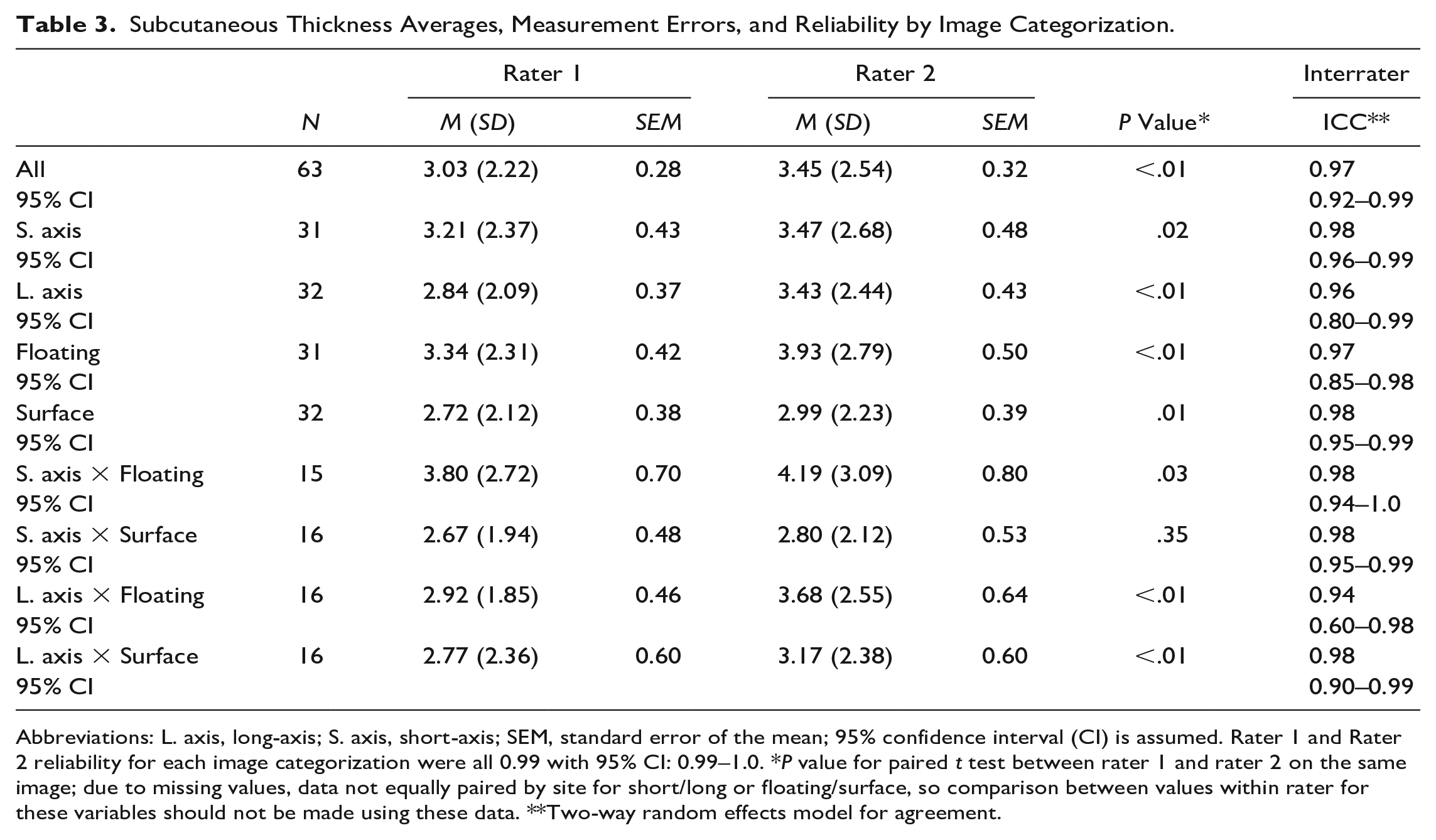
Abbreviations: L. axis, long-axis; S. axis, short-axis; SEM, standard error of the mean; 95% confidence interval (CI) is assumed. Rater 1 and Rater 2 reliability for each image categorization were all 0.99 with 95% CI: 0.99–1.0. *
Gray-Scale Measurement Reliability
Both raters, novice (rater 1) and expert (rater 2), obtained dermal thickness results with small overall measurement error, generally less than 0.10 mm. Error was noted to be slightly higher in short-axis than long-axis and higher in surface than floating conditions. With exceptions of short-axis, floating, and short-axis by floating measures, the expert rater’s dermal thickness measures were statistically larger than those of the novice rater; however, these differences were all less than 0.20 mm. The expert rater demonstrated excellent intrarater reliability across all categories of measurement (i.e., all ICCs >0.95), whereas the novice rater demonstrated good to excellent intrarater reliability with exception of measurements of images obtained in short-axis with the transducer floating (ICC: 0.78, [0.48–0.92]). Both raters were more reliable measuring in long-axis than in short-axis, and the expert rater was more reliable measuring floating than surface images. All interrater reliability measures for dermal thickness evaluation were good to excellent with the exception of the measurement short-axis by floating.
Similar to dermal measures, error in subcutaneous thickness measures was low, ranging from 0.28 to 0.80 mm, with slightly higher error in short-axis than long-axis and in floating than surface. Although the expert rater consistently measured significantly higher than the novice rater, none of the measurement differences between raters exceeded the error, indicating that differences between raters were likely not clinically meaningfully. All intrarater and interrater reliability outcomes for subcutaneous thickness were within excellent ranges (>0.90; Table 3).
Gray-Scale Technique Validation
Given the good to excellent reliability ratings for most measures, differences in values when the transducer was floating versus placed on the skin surface were examined. Dermal and subcutaneous thickness measures in both floating and surface by the expert rater were paired by location and time within participant. When taken in long-axis at the same location, dermal thickness measurements were statistically larger (
Doppler and Elastography Findings
No significant or interpretable signals were identified in any of the color Doppler images, and consequently, no further analysis was completed. Analysis of strain elastography in short-axis images was difficult as the rounded shape of the forearm led to artifacts. These artifacts were realized as different shades of blue and green within the dermal layer clearly affected differentially by the underlying bone as demonstrated in Figures 3A and 3B. Similar inconsistencies in color were noted in strain elastography images obtained with the transducer floating (see Figure 3C), likely due to uneven wave compression resulting from not pushing on the skin surface. Strain imaging in long-axis with the transducer placed on the surface was the only valid technique without artifacts (see Figure 3D). However, upon review, these strain images were considered to have low reliability as significant shifts in coloration were noted among repeated images obtained at the same location during the same time point (i.e., pretreatment or posttreatment). No further analysis of strain elastography images were conducted.
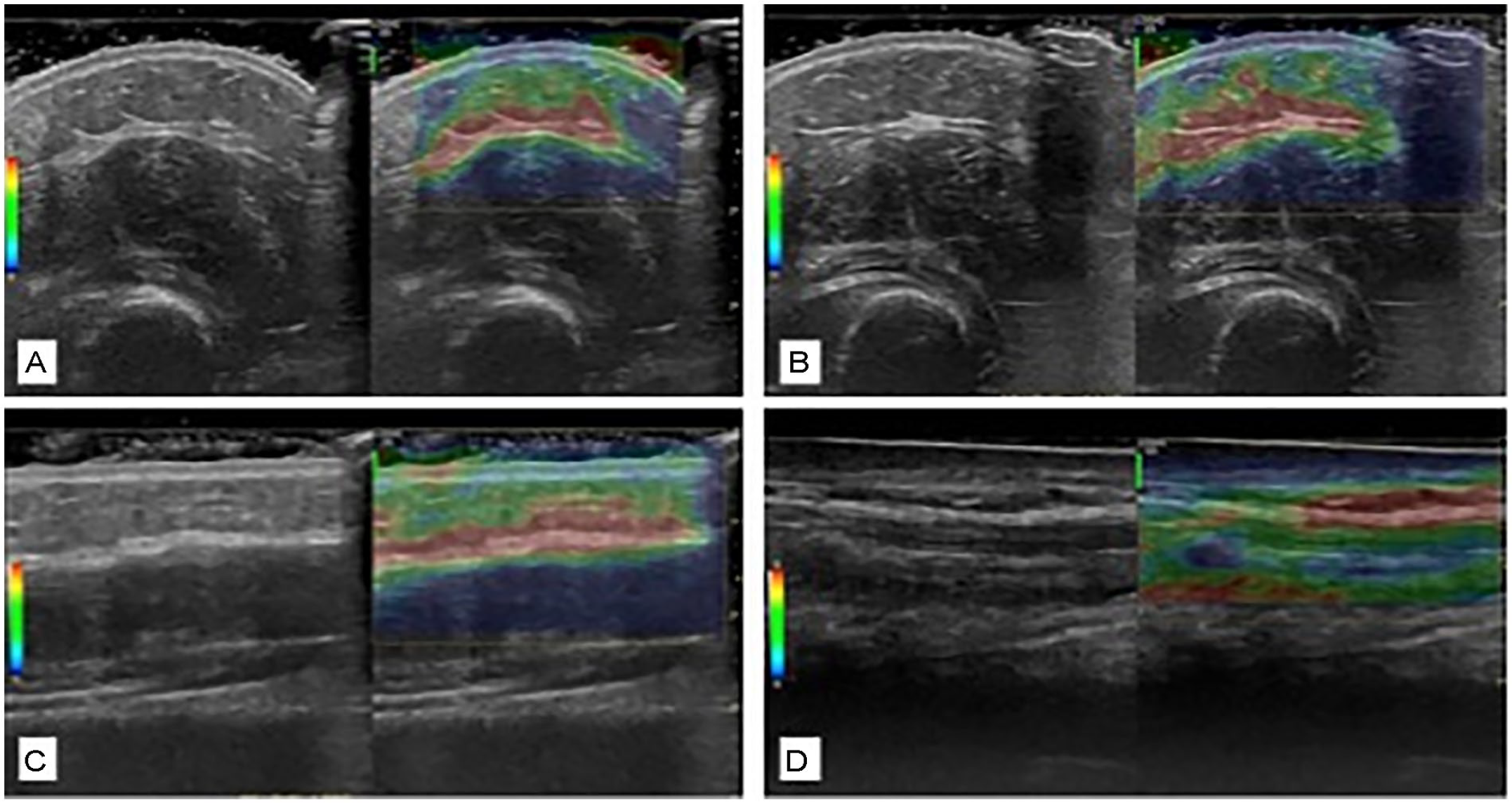
(A, B) Pre-post treatment elastography images in short-axis and elastography image longitudinal (C) floating versus (D) surface.
Exploration of treatment changes
Pretreatment and posttreatment values for dermal and subcutaneous thicknesses were identified by the location of paired images acquired using the most reliable and valid technique (i.e., gray-scale, long-axis, floating). Data for six pre-post treatment pairs across the three participants are reported in Table 4. In these case series data, posttreatment dermal thickness values were consistently smaller than pretreatment measures (
Case-Wise Analysis of Changes in Dermal and Subcutaneous Thickness Pretreatment and Posttreatment.
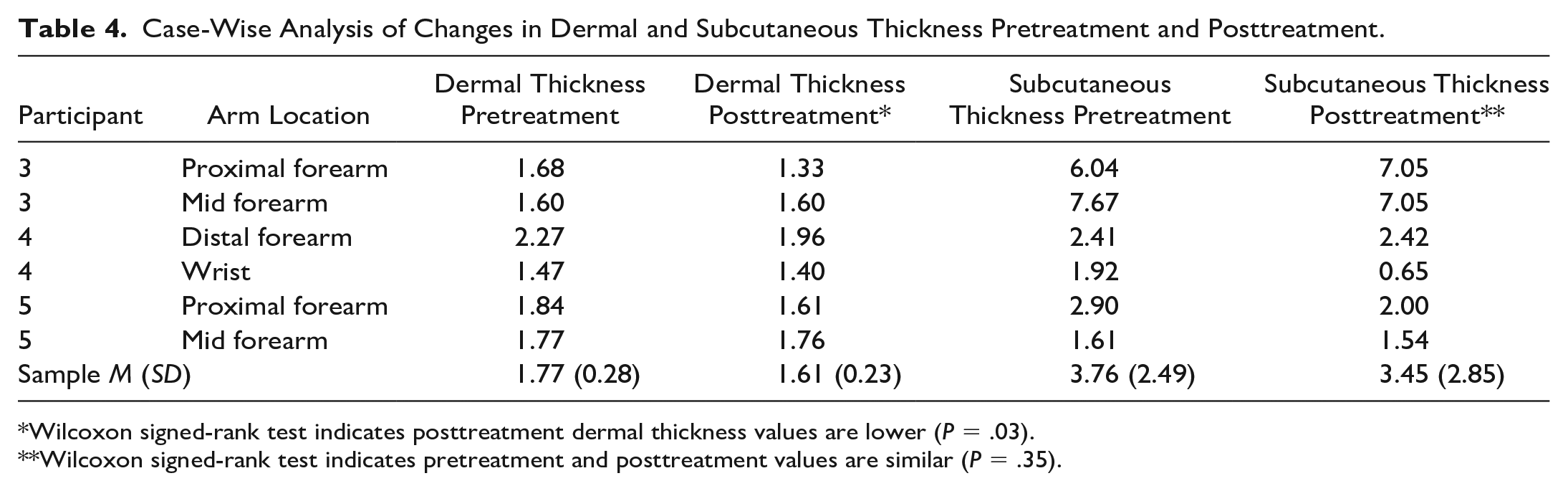
Wilcoxon signed-rank test indicates posttreatment dermal thickness values are lower (
Wilcoxon signed-rank test indicates pretreatment and posttreatment values are similar (
Discussion
This study aimed to determine an MSUS acquisition and analysis technique most appropriate for evaluating SSc rehabilitation outcomes and to explore which measures were most sensitive to change after a one-session rehabilitation treatment. We have three main findings. First, gray-scale imaging with traducer floated on gel in the longitudinal axis was the most valid and reliable MSUS acquisition technique to measure dermal and subcutaneous thickness. Second, color Doppler and strain elastography were not useful for examining superficial forearm structures in our SSc patient sample. Third, there are preliminary indications that dermal thickness may be a useful outcome measure in upper extremity rehabilitation, which can be explored in prospective studies along with other indicators such as tendon mobility.
In this study, transducer placement either on the surface or floating resulted in equally reliable measures for the expert rater, but images acquired in the long-axis were most reliable for obtaining measures such as dermal and subcutaneous thickness across both raters. Dermal and subcutaneous thickness measures were larger when the transducer was floating on gel compared to images acquired when the transducer was placed directly on the skin surface. These trends suggest that the weight of the transducer on the skin may compress dermal or subcutaneous layers, thereby decreasing the measurements. As such, floating the transducer is likely more valid for image acquisition. Moreover, demonstrating these trends in the long-axis images, but not in the short-axis, further supports the use of long-axis techniques.
Doppler is useful for assessing vascularization and tissue perfusion of musculoskeletal structures, to evaluate inflammation, and to monitor treatment response in joints and soft tissues. 24 Changes in Doppler may indicate neovascularization or increased blood flow, each could be indicative of a positive treatment outcome. One possible explanation, for the lack of Doppler findings from pre- to post-LymphaTouch treatment, was that the treatment might not be providing short-term changes in blood perfusion, into the treated region. While color Doppler was not found to be useful, other Doppler techniques may be appropriate for evaluating SSc patients. For example, changes in blood perfusion may be detectable as a long-term outcome as opposed to following one-session treatment. Moreover, rather than investigating changes in perfusion within tissues of SSc patients, power or spectral Doppler may be more useful for evaluating changes in magnitude or rate of blood flow through specific vessels in or around a treated region. For the upper extremity, changes in these measures of blood flow in small finger vessels could be highly useful.
Strain elastography is an effective sonographic technique to evaluate differences in tissue density; 26 however, this technology is best used with homogenous tissue that allows for equal compression (e.g., breast tissue) and is less reliable in superficial musculoskeletal structures, as is evident in our findings. In particular, varying densities and inconsistent shapes of the forearm and limitations in sizing the ROI in the small superficial region result in a higher probability of image artifacts, which can lead to erroneous or inconclusive evidence. 26 Although it was not possible to conduct a rigorous review of elastography data across all participants, at least one participant had valid pre-post images. These images seemed to indicate a moderate shift in tissue stiffness. Given these data, strain elastography is likely not recommended for use in evaluating the forearm, hands, or fingers of SSc patients.
Improvements in point-of-care technology, increased access, and specialized education have broadened the ability for use of sonography by rheumatologists and rheumatology professionals for purposes beyond diagnostics.30-32 For instance, rehabilitation treatments may be better targeted to specific subclinical SSc disease manifestations as detected by MSUS. Importantly, MSUS could help rheumatology professionals understand underlying mechanisms of action for their treatments. This type of information is critically important to the development and refinement of rehabilitation treatments. In addition, MSUS could offer more sensitive and accurate evaluation of disease changes in response to treatment, thereby improving outcome measurements that typically consist of self-reported function, assessment of movement, or therapist-observed disease features.
In addition to investigating other uses of Doppler and strain elastography, using dynamic sonographic techniques may also be worth further evaluation for use in examination of SSc treatment outcomes. Specifically, using sonographic imaging to capture tendon mobility may be a means of determining effective interventional methodology as identified by Korstanje and colleagues. 33 The operant theory of LymphaTouch is that the fibrotic plaques of SSc may be loosened by the negative pressure of the intervention, which may allow for increased movement of both skin and underlying structures. There is a need for further work to develop sonographic methods for measuring movement of the extensor digitorum communicus (EDC) and other tendons of the hand during the completion of functional tasks in patients with SSc to more fully evaluate treatment effects in this patient population.
Implications for Therapy
These study findings indicate that MSUS may be a useful rehabilitation tool for measuring treatment effectiveness as it allows opportunity to see structural effects on dermal and subcutaneous levels in individuals with SSc. In this sample, average dermal thickness values measured by the expert rater in long-axis, floating were slightly higher than these normative data at 1.74 mm. The mean dermal thickness values were noted to be higher in pretreatment measures (1.77 mm) with a maximum individual value of 2.27 mm, with consistently lower dermal thickness measures on posttreatment images. These data provide preliminary support for dermal thinning immediately following LymphaTouch treatment, indicating that this may be a useful measure and treatment combination for rehabilitation clinicians working with individuals with SSc. This preliminary effect in this small-sample, case series study should be further examined in a larger sample, over time, and compared with other functional outcomes to determine treatment impact. Studies should examine if this thinning is related to improvement in dermal pathology or momentary stretching of the skin following treatment, as well as evaluate how long the effects of the treatment last. Identifying the duration of increased dermal pliability could provide understanding of the best therapeutic window for providing other focused intense therapeutic interventions to maximize overall outcomes.
Limitations
Only images from participants three through five were included in analysis, and missing data points could have skewed analyses. Because the purpose of this study was to validate an imaging acquisition and analysis protocol, it was too preliminary to develop an a priori plan for comparison of sonographic changes to other patient outcomes. Moreover, given that data were collected following one brief treatment session, significant changes in functional outcome measures were not expected. However, having developed a valid technique and identified preliminary effect size on change in morphologic measures, future interventional pilot studies can further explore and compare intervention effects between these physiologic measures and functional outcomes.
Conclusions
MSUS can provide reproducible results of skin and subcutaneous tissue changes particularly in SSc. Imaging recommendations from this study indicate the most valid and reliable MSUS technique is using gray-scale imaging with the transducer floated on gel in the longitudinal plane. The developed protocol should be tested with larger samples and can be used as a measure to evaluate rehabilitation treatments.
Footnotes
Editor’s Note
The JDMS follows a scholarly, double-blinded peer review process. The peer review of this manuscript was managed by members of the JDMS Editorial Board and excluded the Editor listed as an author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported in part by a grant to Dr Murphy from LymphaTouch LLC. Dr Krause’s work was supported by an Advanced Rehabilitation Research Training grant from the National Institute on Disability, Independent Living, and Rehabilitation Research (90AR5020-04). Dr Khanna’s work was supported by the National Institutes of Health (NIH)/National Institute of Arthritis and Musculoskeletal and Skin Diseases (K24-AR-063129).
