Abstract
Fractures and suspected fractures are common causes for pediatric visits to the emergency department. Initial evaluation routinely includes imaging in the form of radiographs. Additional imaging modalities including sonography and magnetic resonance are used as problem-solving tools. Sonography has been reported to perform well when compared with radiography in multiple anatomies from the skull and nasal bones to the upper and lower extremities. In addition, sonography presents unique advantages including lack of ionizing radiation, cross-sectional coverage, evaluation of soft tissue injury, and the possibility of imaging the contralateral side. This article reviews the applications, advantages, disadvantages, technique, and performance of sonography in the imaging of fractures in children.
Introduction
Acute fractures are very common in children, with an annual occurrence rate of 9.47 per 1000 children and an overall risk of approximately 1 in every 5 children experiencing a fracture throughout childhood and adolescence. 1
Acute fractures are traditionally evaluated at presentation to the emergency department using radiography, which as an imaging modality is the gold standard for fractures.2–9 Lack of radiographic abnormality on the initial encounter at times results in inappropriate initial treatment, leading to missed fractures that are diagnosed upon follow-up or require confirmation by other imaging modalities.10,11
A fracture that is suspected upon physical examination but undetectable by plain radiography is deemed an “occult fracture.”10,11 Because occult fractures are commonly diagnosed days to weeks after onset of injury, complication rates, patient harm, and time away from work for patients and caretakers are higher but can be avoided with a prompt diagnosis.10,11 Delays in diagnosis also make the injury harder to manage if manipulation is necessary and can entail refracture if there is callous formation at the fracture site.10,12 Alternatively, the suspicion of fracture without confirmation might lead to overtreatment with unnecessary immobilization, decreased quality of life, and increased healthcare costs. 11 Occult fractures are not uncommon, with a pooled prevalence of 24% reported in a large meta-analysis of pediatric occult ankle fractures. 13 In the elbow, occult fractures are also common, with reported prevalence at 13%. 14
Sonography and magnetic resonance imaging (MRI) have been found to be valid alternatives to radiographs in several clinical scenarios.3,6,15–20 However, the use of sonography is not widespread. Lack of knowledge about the technique, applications, and advantages of sonography over other diagnostic approaches might be partially responsible. This article is a review of the available literature regarding sonographic evaluation of fractures in children.
Imaging Fractures
After acute injury, radiographic examination of the anatomic area is the preferred initial approach in children. Radiographic diagnosis is often straightforward and provides adequate diagnostic and treatment guidance. 21 The American College of Radiology Appropriateness Criteria,® 22 a set of evidence-based guidelines to assist referring physicians in appropriate imaging decision making, recommends radiography as the initial imaging modality in acute hand, wrist, elbow, ankle, and foot trauma. In spite of being the standard of care, no specific accuracy figures are reported for radiographs in any of the aforementioned anatomic regions. Radiographs are considered convenient, widely available, relatively easy to interpret, and less costly.
Limitations of radiographic examination include the use of x-radiation, which is the greatest contributor of manmade radiation to the general population. 5 In addition, radiography requires ossified bone to absorb x-rays, making unossified bone only partially evaluable in young children.23,24 Radiography is also less sensitive in detecting soft tissue abnormalities. 10
Sonographic Accuracy and Benefits
Many studies have evaluated the performance of sonography in the diagnosis of pediatric fractures with high reported accuracy. For example, in the evaluation of distal forearm fractures, which account for 20% to 36% of all fractures in children, Herren et al. 25 reported sensitivity and specificity of 99.5% when comparing sonography with standard two-view radiographs of the wrist. In this study, the one missed fracture was a torus fracture of the ulna, correctly identified in concurrent radiographs.
In elbow fractures, which account for 15% of all fractures in children, Rabiner et al. 9 reported a sensitivity of 98% for sonography when prospectively comparing it with standard radiographs. However, the specificity of sonography was determined at only 70%, mainly because effusions might happen in the absence of fracture. In this study, sonographic evaluation of the elbow was limited to the determination of an elevated posterior fat pad and/or lipohemarthrosis using both transverse and longitudinal views of the elbow flexed at a 90° angle.
In another large study comparing the accuracy of sonography performed by either radiologists or emergency department physicians for suspected hand fractures in children, Neri et al. 19 reported sensitivity at 91.1% for radiologists and 91.5% for emergency department physicians. The specificity was also very similar between both groups, reported at 97.6% and 96.8%, respectively. This study used radiographs as the reference gold standard.
Additional studies have reported similar results with accuracy at times approaching 100%, offering sonography as a feasible alternative to radiography in emergency departments. 12 Specific fracture sites in which ultrasound has been reported include the skull, 26 nasal bones, 15 facial bones, 27 clavicle, 23 elbow, 11 forearm,4,12,28 metacarpals, 18 upper extremities,3,29 hip,30,31 and foot and ankle.17,24,32,33 It has also been reported for both accidental and nonaccidental trauma.34,35 Reported test performance characteristics for different anatomies are summarized in Table 1.
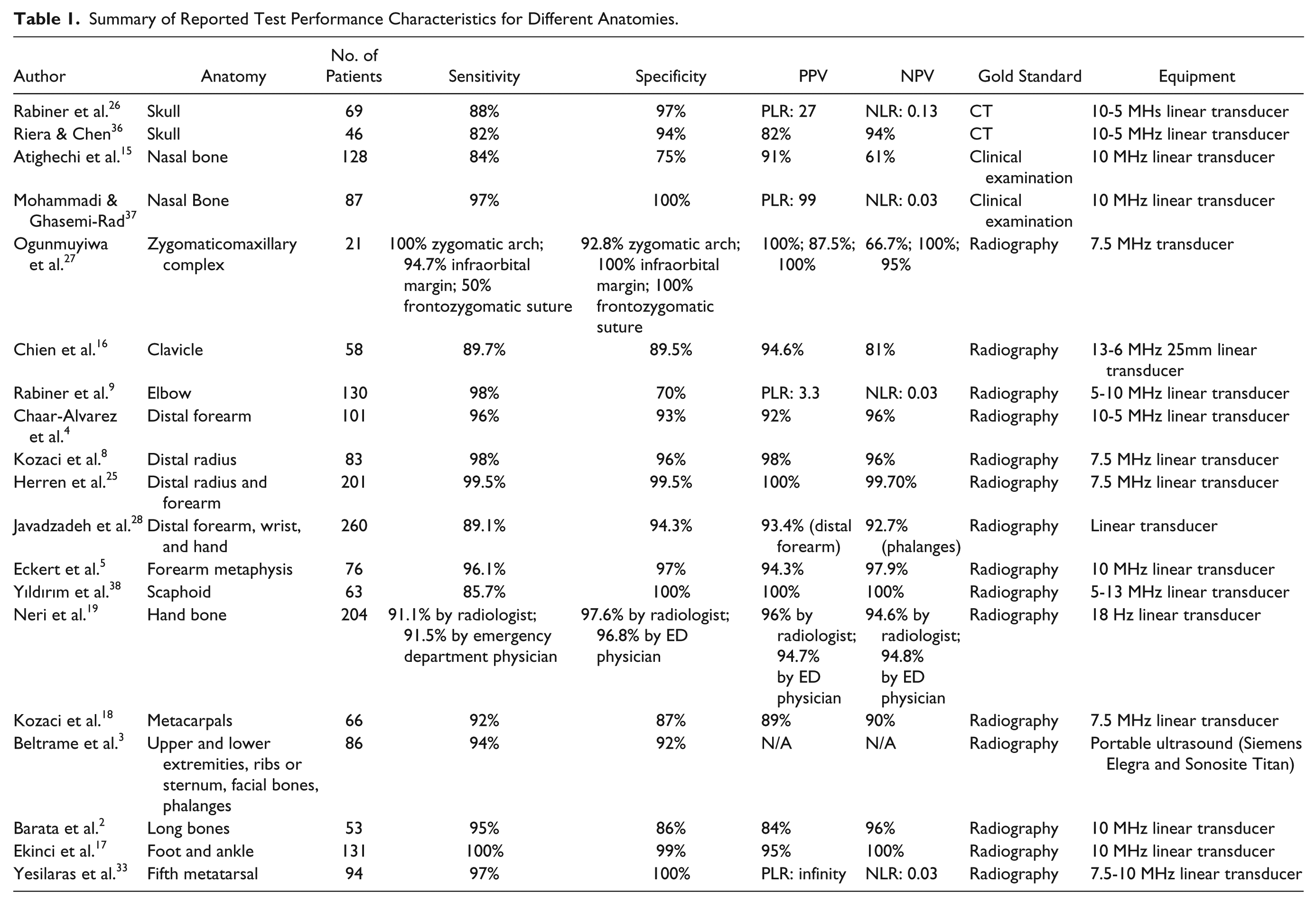
Summary of Reported Test Performance Characteristics for Different Anatomies.
These studies highlight some of the benefits of sonography. For example, in young children, ultrasound allows for the characterization of unossified bones and the periosteum, which are not seen in radiographs. The diagnosis of fractures in ultrasound can then be based either on indirect signs such as elbow effusions or on direct characterization of bone contour.23,24 Sonography also permits evaluation of the cortex of long bones along multiple planes (Figure 1). Areas of complex anatomy, including the foot, ankle, or pelvis, might also be approached by sonography as a noninvasive cross-sectional tool when conventional radiography does not provide conclusive results. 24
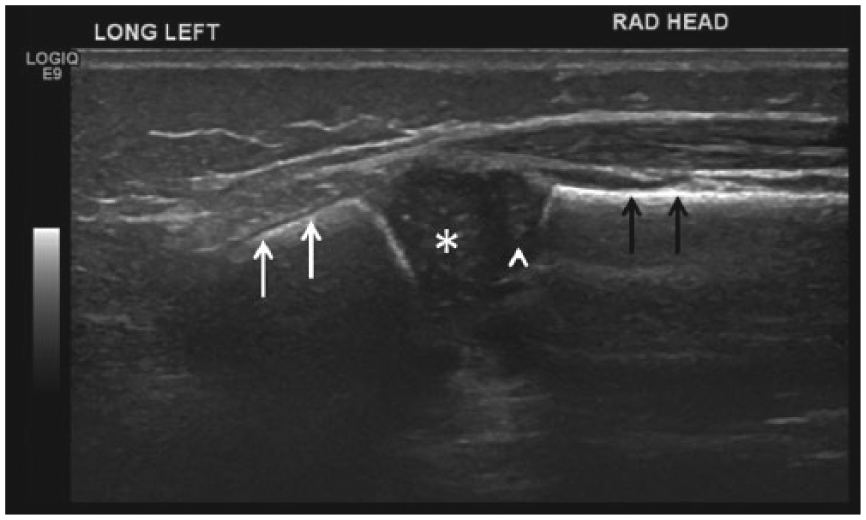
Normal left elbow ultrasound in a 2-month-old girl. Longitudinal view shows normal echogenic cortex with posterior shadowing in the humeral (white arrows) and radial (black arrows) diaphysis. Normal epiphyseal cartilage in the capitellum (asterisk) and radial head (arrowhead) as well as the normal radiocapitellar alignment are demonstrated. Soft tissues are also normal.
Sonography involves no radiation to the patient and often provides the same information as radiography.3,18,23,39 Musculoskeletal ultrasound is superior to radiography in diagnosing intra-articular bodies, physeal separation, subperiosteal hematomas, ligamentous injuries, and periosteum trapped between fracture fragments. 29
Radiography is less sensitive in detecting soft tissue abnormalities and unossified structures, for which ultrasound and MRI are better. 10 However, sonography requires no sedation, whereas MRI is less desirable because the lengthy process commonly necessitates anesthesia for young patients to prevent motion artifact.10,29 Sonographic evaluation is cheaper than MRI10,23 and can be completed quickly with little pain to the patient, with select studies reporting procedure times as low as 5 minutes. 11
A study of patient pain scores comparing radiography and sonography found the latter to be less painful, as repositioning the injured area to obtain several radiographic views causes greater discomfort than moving an ultrasound probe across the area. 23
Sonography allows for real-time comparison with the contralateral unaffected side within the same examination. 40 Evaluating the unaffected side might help differentiate fractures from normal anatomic variants that are common confounders in pediatric imaging. Evaluation of the unaffected side establishes the normal width of the growth plate, which adds certainty to the diagnosis of physeal separation. A previous study found that there is no significant difference and only minimal variation of the physeal plate widths in contralateral limbs in patients ages 0 to 12 years. 40
Sonography has also been reported to be a useful alternative to radiography in resource-limited settings.12,41 Particularly for nonurgent skeletal injuries, limited availability of radiographic equipment might result in significant wait times and delays in diagnosis and treatment. In such cases, sonography might allow for shorter length of stay and save time for the provider, patient, and caretakers.23,34 If the same physician performs both physical examination and sonographic evaluation, the patient’s clinical history and image findings can be quickly combined to reach a diagnosis. 17
Limitations of Sonography
A major disadvantage of sonography is its dependence on experienced operators, who may not be available in all emergency departments.12,42 In particular, sonographers need to be familiar with musculoskeletal and pediatric sonographic techniques, and it is common to find sonographers lacking both. A successful fracture sonography program might additionally require 24-hour coverage, which is not available in many emergency departments. Accuracy and competency in sonographic evaluation require comprehensive training, hands-on practice, and appropriate physician supervision. Several publications have described different options to overcome this barrier, including online instructional videos, demonstrations by orthopedic experts, didactic training sessions in osteoarticular sonography, and hands-on practice for a total training time of 2 hours to 1 full day.12,34,42,43 Many have advocated for a more active role of point-of-care sonography, which might obviate the need for dedicated sonographers by directly training emergency department physicians to perform the examinations.18,43
Another limitation of sonography is its inability to accurately image intra-osseous abnormalities and open fractures. Utility of sonography is also limited in patients who are unable to localize pain, in cases of polytrauma, and when a skeletal survey is indicated.10,42
Under current reimbursement schedules from payers in the United States, sonography is more expensive than radiography, which could represent a limitation to implementation. However, from an equipment and room-conditioning perspective, sonography might represent a cost-effective alternative, particularly in resource-constrained environments.12,41
Equipment and Technique
High-frequency sonographic transducers are highly sensitive to the region between cortical bone and periosseous soft tissue, making sonography ideal for identifying the cortical outline and any soft tissue changes at the fracture site.10,42
In pediatric patients, the depth of the bone is usually less than 4 or 5 cm, making linear-array, high-frequency transducers ranging from 9 MHz to 18 MHz preferable.11,12,18,24,31,34,42 Sonographic studies of pediatric fractures commonly use linear high-resolution probes, the frequency of which depends on the habitus of the patient, with higher frequencies used in smaller patients.11,24,31,34,42
Positioning and Planes
Best visualization of cortical disruption of fractures can be obtained when transducers are positioned orthogonally to the fracture line and focused at the region of interest, as fractures might be obscured when the transducer is parallel to the fracture line. 10 Imaging should be obtained in the sagittal and transverse planes extending superior and inferior to the area of concern.
Position of the patient will depend on the area of interest. If imaging for fracture along the diaphysis in long bones, no specific rotation or position is needed to obtain images. If trying to assess the epiphysis or metaphysis of the long bones, some rotation and specific positioning may be needed to access the area of interest. In cases where the formation of a fat layer is needed for better visualization, patients should be asked to keep the injured area stationary. 11 Standoff pads or “waterbeds” can be used when examining uneven surfaces. 24
Sonographic Diagnosis of Fracture
Normal Sonographic Appearance
The normal cortex produces a very bright or echogenic linear structure in the image representative of ossification, with hypoechoic shadowing posterior to the bone itself24,43 (Figure 2). The periosteum, which covers the external surface of bone, consists of two layers: an outer fibrous layer and an inner, more cellular and vascular layer. The inner osteogenic layer or cambium layer can form new bone, whereas the outer layer forms part of the insertions of tendons, ligaments, and muscles. 44 However, the periosteum forms part of the echogenic cortical line in normal bone and becomes visible only after injury.
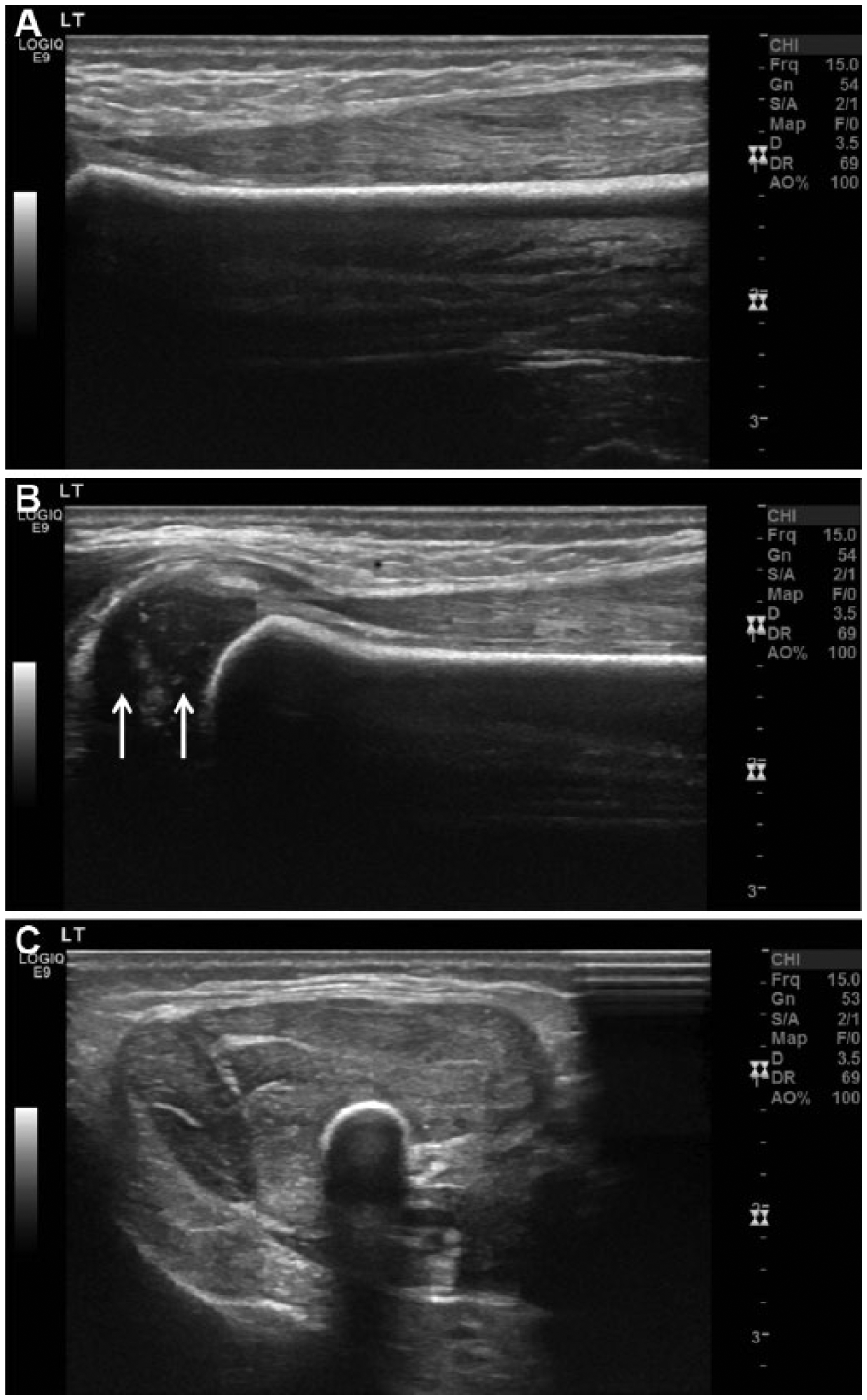
Normal left femur ultrasound in a 17-day-old girl. (A) Longitudinal view shows normal echogenic cortex with posterior shadowing in the femoral diaphysis. Note normal appearance of the overlying muscle and subcutaneous soft tissues. (B) Longitudinal view of normal epiphyseal cartilage (arrows) showing a homogeneously hypoechoic pattern with scattered hyperechoic foci of calcification throughout. The normal physeal width can also be seen. (C) Transverse view showing normal appearance of the femoral cortex and thigh soft tissues.
The appearance of unossified epiphysis is characteristic, with epiphyseal cartilage showing a homogeneously hypoechoic pattern with scattered hyperechoic foci of calcification throughout45,46 (Figure 2). The sonographic appearance of the ossified epiphyses is similar to the diaphysis with an echogenic continuous line.
The normal physeal width can be established by using the unaffected side for comparison in cases of suspected physeal plate separation 40 (Figures 1, 2B, and 3C).
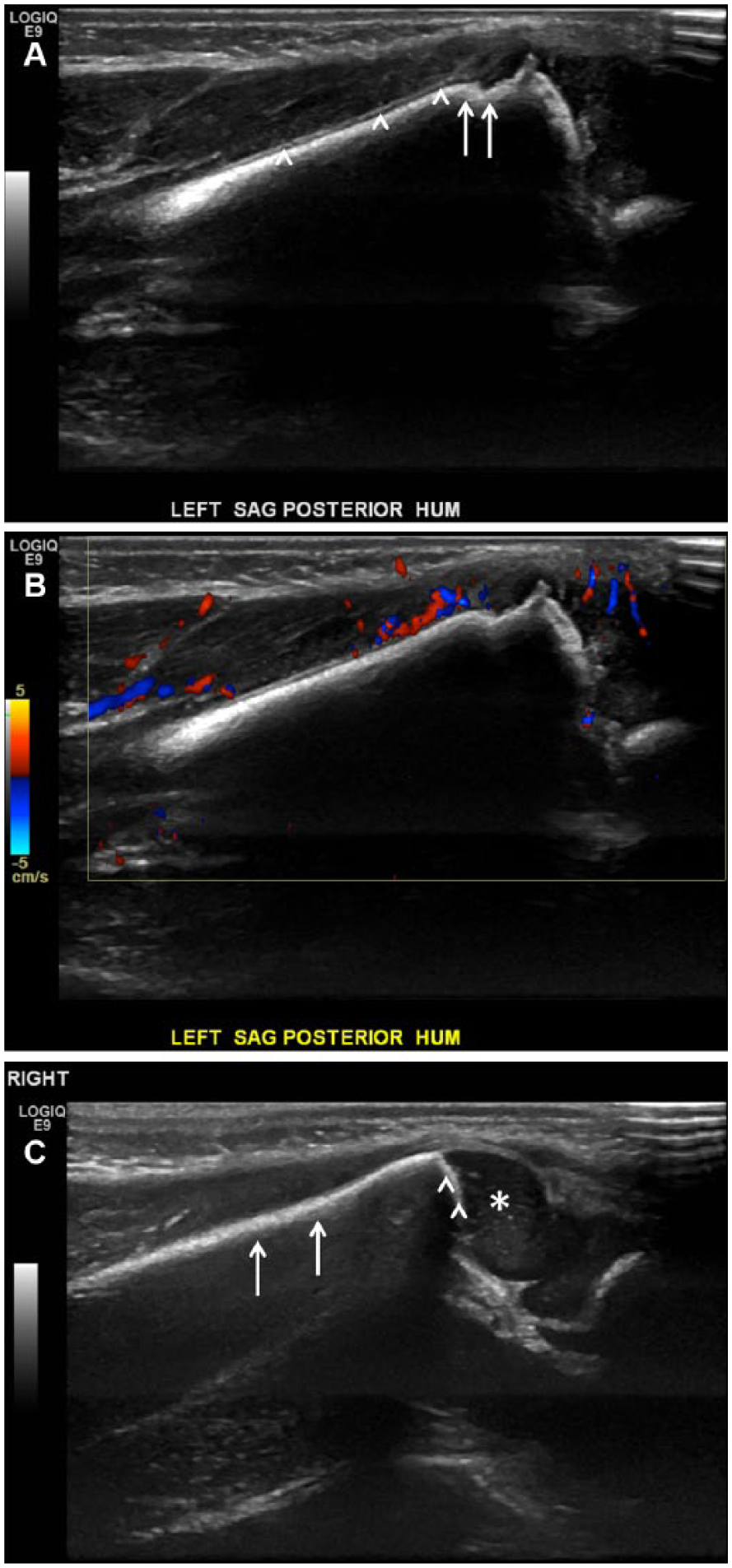
Left distal humeral fracture in a newborn boy. (A) Longitudinal ultrasound image of left humeral (supracondylar) fracture showing cortical step-off (arrows) and double line sign (arrowheads). (B) Longitudinal color Doppler image showing soft tissue hyperemia about the fracture site. (C) Longitudinal view showing normal right distal humerus images obtained for comparison. Note normal continuous echogenic cortex (arrows), normal epiphyseal cartilage (asterisk), and physeal width (arrowhead).
Diagnosing Fractures
A disruption in or discontinuity of the echogenic line that is the periosteum is consistent with a fracture 29 (Figure 3A). A commonly described indicator is the “double line sign,” which is simply the presence of two parallel hyperechoic reflections on long bones 47 (Figure 3A). Because the bony cortex appears brightly echogenic on ultrasound, it is possible to identify cortical disruption as small as 1 mm by high-resolution transducers. 43
Severity of step-off is identified by measuring the distance between two lines drawn along the edges of the fracture cortex and healthy cortex. 18 Other ways to describe the fracture relate to findings of a cortical gap, cortical bulging, a cortical deviation, or a circumferential hematoma covering the cortex (Figure 4).
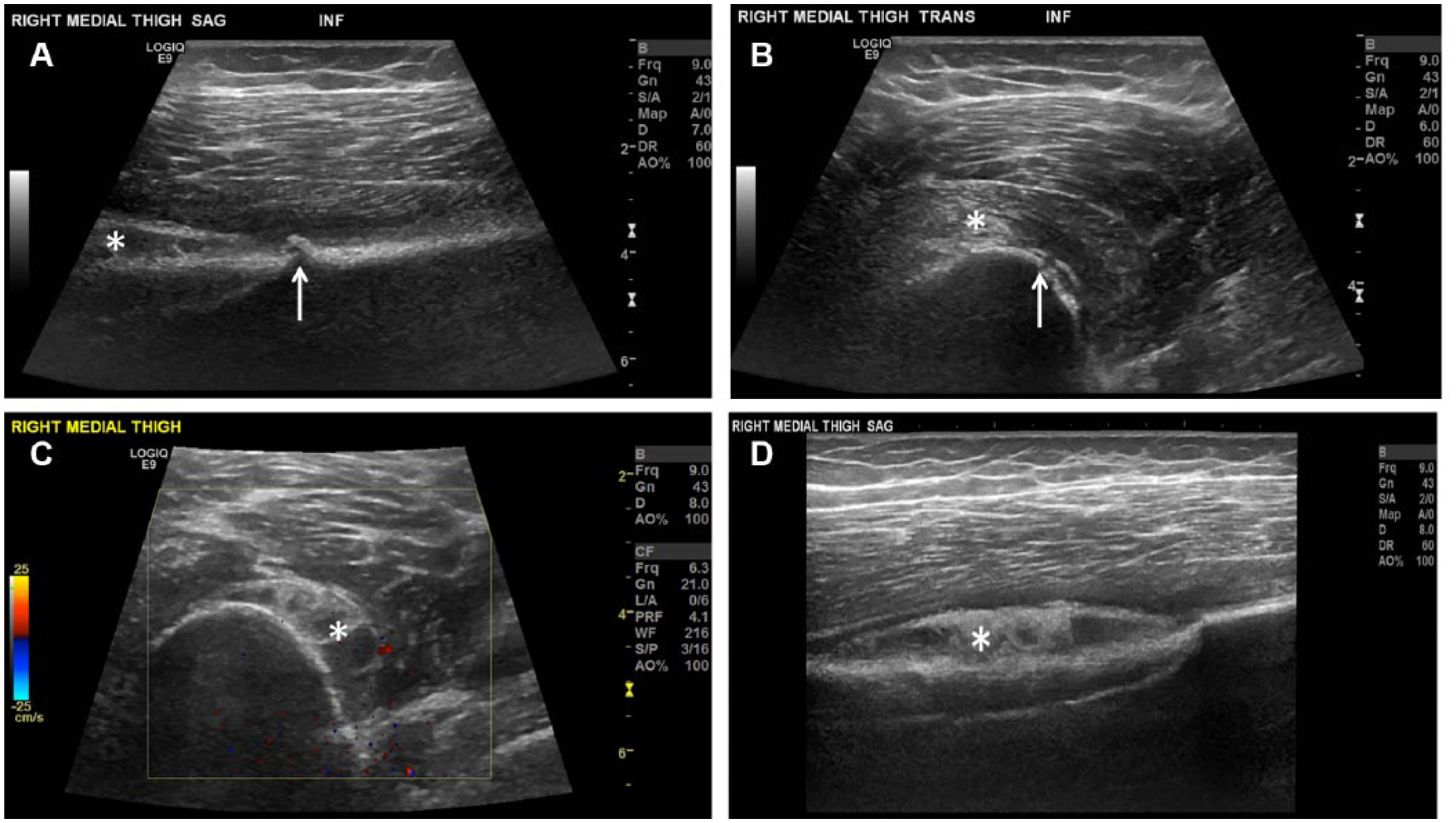
Nondisplaced right femoral fracture in a 16-year-old girl. (A) Longitudinal view of the mid femur shows a small step-off (arrow) and adjacent subperiosteal hematoma (asterisk). (B) Transverse view better depicts the cortical disruption (arrows) and overlying muscle edema (asterisk). (C) Transverse color Doppler view shows a larger component of the subperiosteal hematoma (asterisk), also seen in (D) longitudinal view (asterisk).
Comparison of the injured area with another section of intact extremity confirms the finding of fracture. 18 Associated findings are related to edema and/or hemorrhage of the adjacent muscles and soft tissues with mixed echogenicity and hyperemia (Figure 3B). Tendon tears are also apparent with sonography. 29
Additional Applications: Guided Reduction
In addition to aiding in the diagnosis of fractures, sonography has been proven an effective tool for guided realignment and reduction of fractures.12,24,39,43 Ultrasound-guided reduction of forearm fractures was successful in 82 out of 100 cases in one study, 39 whereas a smaller study including 47 children reported a success rate of 94%. 12 Instead of attempting reduction, reimaging to evaluate if the reduction is correct, and repeating until the reduction is adequate, 43 ultrasound-guided reduction allows for assessment of reduction quality in real time, potentially reducing the number of attempts down to one, decreasing patients’ time under anesthesia, and reducing radiation exposure.12,23,39,42 Ultrasound-guided fracture reduction can also save resources for the facility by avoiding the need for fluoroscopic and radiation protection equipment.12,39
The conventional practice of performing radiography after reduction may be rendered unnecessary if use of sonography can determine adequate alignment.23,39
Summary
There is increasing evidence that sonographic evaluation of suspected fractures in children is feasible and carries important advantages over radiography, including lack of radiation, improved visualization of complex anatomy, and evaluation of soft tissue complications. Its main disadvantages remain in the lack of local expertise and its dependence on operators, which can be overcome with education and increased visibility of sonography as a viable option. Sonographers and emergency department physicians should become familiar with this application as sonography continues to grow into the preferred imaging modality for children.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
