Abstract
Objective:
Sonography of the chest has traditionally been focused on chest wall and pleural space. Over time, clinicians have developed a practice of bedside lung ultrasonography (LUS) to supplement physical examination. In the wake of the SARS-COV-2 (COVID-19) pandemic, clinicians have increasingly turned to LUS as a tool to assess a patient’s condition.
Methods:
A literature search was performed using Google Scholar with access to articles through a major Midwestern university.
Results:
Computed tomography (CT) and radiographs are not always available in all treatment centers. While LUS may be limited, it is important to correlate findings with compatible imaging modalities. This review summarizes lung pathologies and the sonographic appearance of lung abnormalities.
Conclusions:
Radiology professionals have largely questioned the practice and findings of LUS. However, now, as the most qualified experts in the use of ultrasound, sonographers and radiologists have a duty to be educated in the use of LUS.
Sonographers and sonologists have long been taught that aerated structures, particularly lungs, cannot be evaluated with sonography. 1 Imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI) are favored for their ease and accuracy in interpreting findings in the lung, leading radiologists to dismiss the utility of lung ultrasonography (LUS). 2 Even chest radiography is preferred to LUS despite ionizing radiation, significant interobserver and intraobserver variability, and poor sensitivity and specificity. 3 The low cost and portability of basic ultrasound equipment have led clinicians without access to radiography or advanced cross-sectional imaging to pioneer the development of LUS. 2 In the setting of the SARS-CoV-2 (COVID-19) pandemic, this technique has become increasingly valid to clinical practice in treatment centers with limited resources. 4
Until recently, very few radiology-based publications have addressed the topic of LUS. There has, therefore, been a lack of collaboration between those practicing it and those best equipped to perform and interpret it. 5 This review is intended to bring practicing sonographers up-to-date with techniques for evaluating the lungs, potential findings, and their clinical implications. While the sonographic evaluation of an aerated organ is still limited, its utility is undeniable.
Methods
The literature search was performed using Google Scholar with access to articles through the University of Michigan. Search terms included chest, lung, radiology, pneumonia, interstitial lung disease, BLUE protocol, BLUE criteria, and COVID-19 combined with ultrasound or sonography. Articles published by radiology- and pulmonary-focused journals were emphasized for their level of expertise on the subject (Figure 1). Articles published by emergency medicine and other medical specialty journals were reviewed but not heavily weighted as they did not provide as thorough of a discussion as desired for this audience. Research published in the last 4 years was prioritized in compiling current recommendations, and earlier research was used to demonstrate findings that persisted over time (Figure 2). References for articles that were particularly informative were reviewed for additional sources. Twenty-two articles fit the inclusion criteria. Supplemental information was retrieved from textbooks.
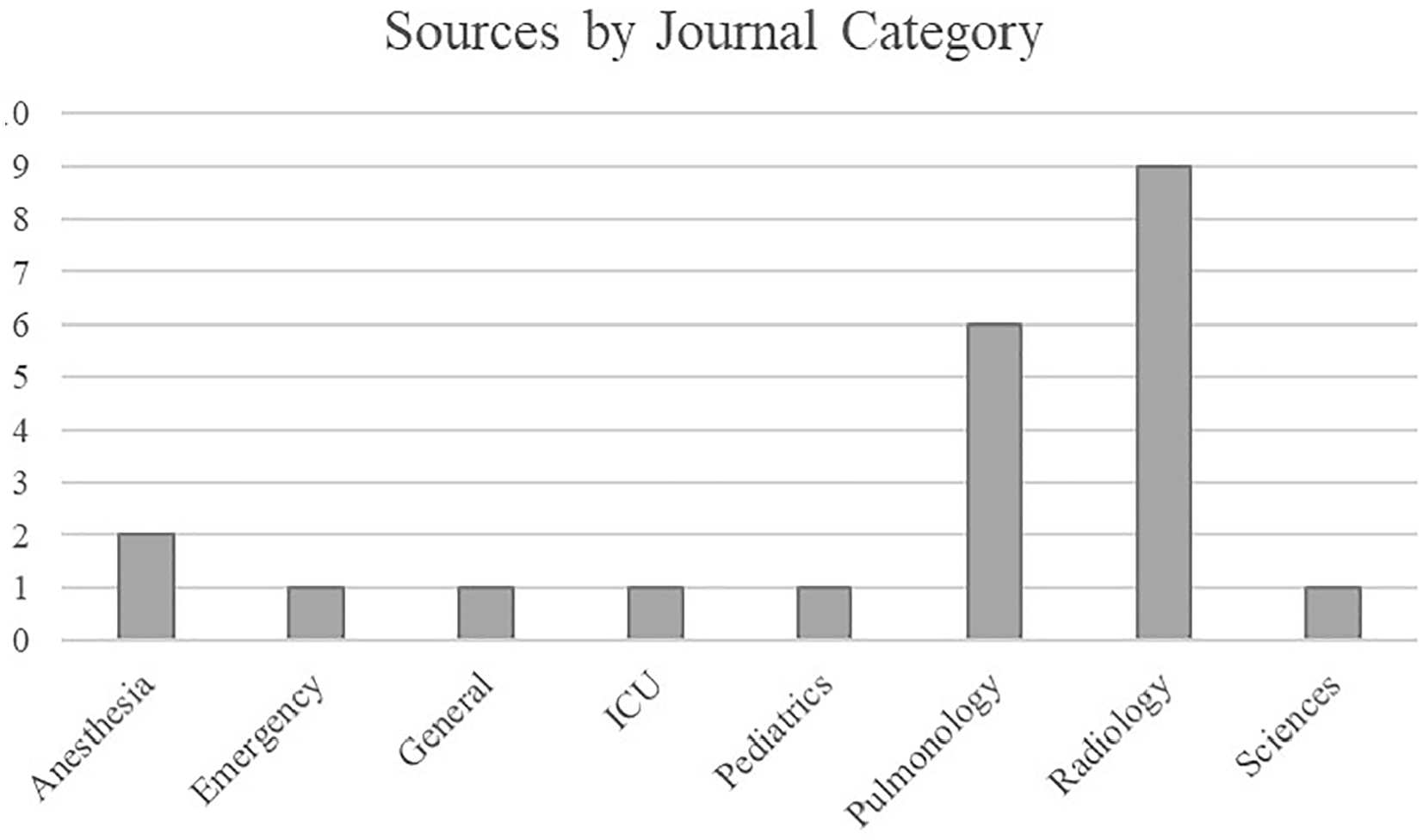
Emphasis was placed on sources from radiology and pulmonology medical journals. Sources from other assorted medical specialties and sciences were also consulted, demonstrating the widespread support for this practice. ICU, intensive care unit.
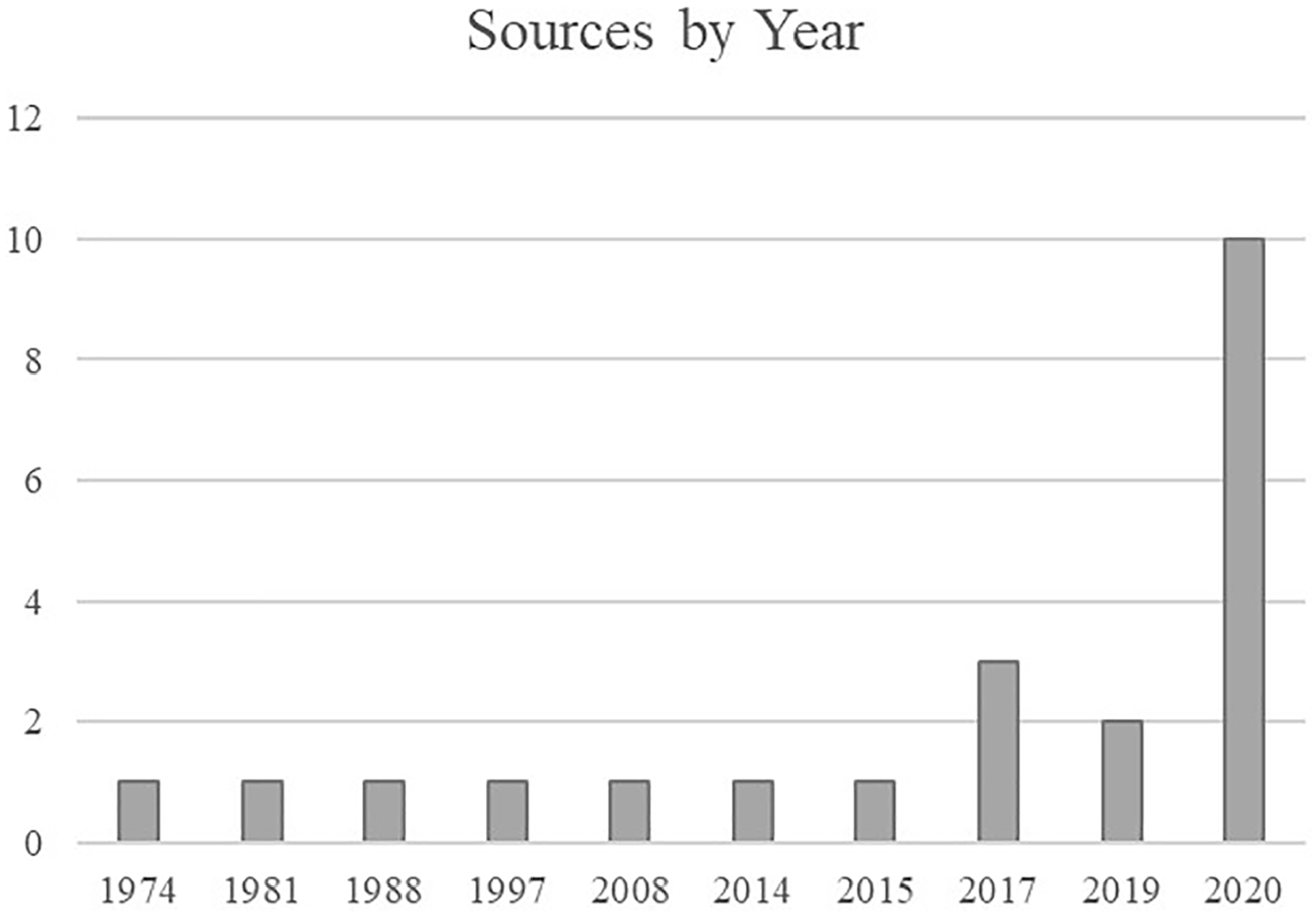
The literature search was focused on sources from the last 4 years, although sources from earlier years demonstrated the consistency of lung findings, as technology changed.
Background
For decades, researchers have experimented with using ultrasound to evaluate the lung. Dunn and Fry began evaluating the behavior of ultrasonic waves in canine lungs in the 1960s and noted a significantly higher attenuation of sound in the lung than in other tissues. 6 In 1981, Rantanen described the sonographic appearance of normal equine lung in evaluating pleural effusions, noting that the characteristic reverberation artifact is still seen today. 7 In 1988, as technology progressed, Birnholz considered the possibility of evaluating the lung’s surface with ultrasound, possibly for signs of interstitial lung disease. 8 From the 1990s until the present day, Lichtenstein has been at the forefront of this field, developing the bedside LUS in emergency (BLUE) protocol for evaluating critically ill patients and correlating findings with the underlying pathology.9–11
Discussion
Chest Imaging Modalities
Radiography
By far, the most common modality of chest imaging is the radiograph, but it is not the gold standard. While chest radiographs are both relatively inexpensive and portable, 12 studies have repeatedly reported wide interobserver and intraobserver variability in interpreting these images. 3 In addition, chest radiographic findings lack sensitivity and specificity. 3 Pneumonia, for example, cannot be ruled out with a negative chest radiograph. 13
Advanced cross-sectional imaging
CT is the most accurate diagnostic tool for imaging the chest. 12 CT provides a comprehensive and easily interpreted image of the chest 2 but cannot be used more frequently due to cost and exposure to ionizing radiation. 12 In addition, patients whose condition is critical enough to warrant a CT are often too unstable to travel to the department. 12 This limitation combined with excessive cost also minimizes the use of MRI. 12
Sonography
In contrast, LUS is economical, portable, and lacks ionizing radiation. 12 In settings without access to radiography, LUS has proven particularly useful. 14 It is more sensitive in detecting small pleural effusions than radiography and more accurate in characterizing effusions than CT. 15 Studies in both adult and pediatric populations have also demonstrated a high level of accuracy in detecting lung consolidation and interstitial lung disease. 12 House et al 14 found an interobserver agreement of 0.9 among expert practitioners. In a literature review, Pereda et al 13 found reasonable accuracy of LUS even in the hands of less experienced users. Lovrenski 5 used a more logic-based argument, writing that, in the absence of a gold standard for chest radiography, one should prefer chest imaging modality without exposure to ionizing radiation.
Indications
Because imaging the lungs with sonographyis a developing practice, the clinical indications for LUS are broad. It is often used when radiography is not available—such as in the office setting—or when it is not advisable—such as with pregnant or pediatric patients. 9 The most common indications include assessing patients with acute respiratory distress 5 and clarifying an opaque hemithorax on chest radiograph. 2 LUS is effective in detecting consolidation, characterizing effusions, differentiating between cystic and solid lesions, and following up prenatal malformations. 2 Intravenous contrast-enhanced ultrasound has also been used in complicated pneumonia to evaluate for necrosis. More specialized departments have explored the use of LUS in optimizing respiratory settings in ventilated patients 16 and quantifying fluid overload in dialysis patients. 5
Technique
The reason radiologists have largely abandoned LUS is the lengthy acquisition time compared with radiography. 5 The BLUE protocol, which is most frequently used by point-of-care clinicians, includes images from the anterior, lateral, and posterolateral approaches of each side of the chest. Each acoustic window is split into upper and lower halves for a total of six zones per hemithorax (Figure 3).10,11 The posterior views are particularly crucial as pathology tends to persist longer in the posterior lungs. 5 In pediatrics, subxiphoid views may allow a side-by-side comparison of hemithoraces; this approach is less useful as the patient grows. 2 Other protocols have further divided the imaging windows, creating additional views, but comparative studies have found no clinically significant change in results. The shorter protocol is, therefore, favored for timeliness. 17
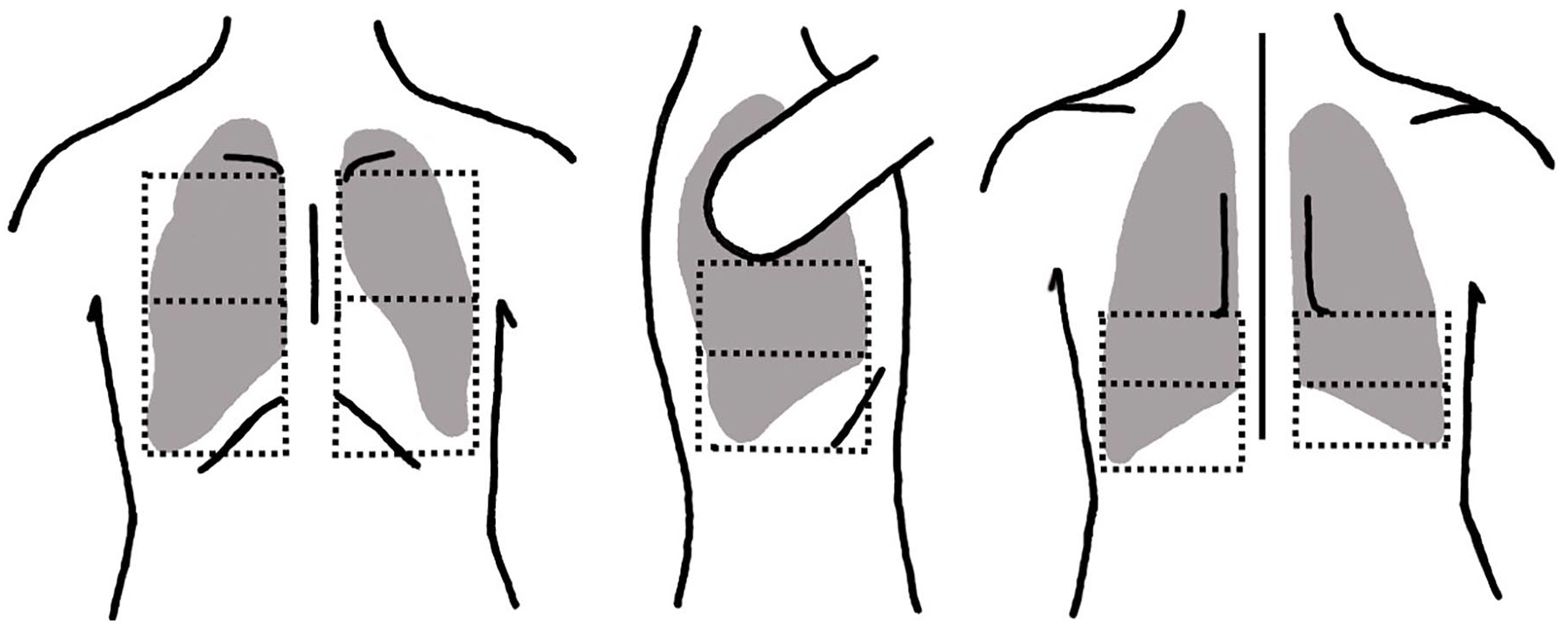
The BLUE protocol includes images from the anterior, lateral, and posterior regions of each hemithorax split into superior and inferior zones. BLUE, bedside lung ultrasonography in emergency.
Transducers should be chosen based on the patient’s body habitus. 4 Although nearly any transducer can be used, Gomond-Le Goff et al 18 found that the use of high-frequency linear transducers resulted in improved accuracy in evaluating lung signs among inexperienced users. In an aerated lung, any structure visualized deep to the pleural line is artifactual 19 ; therefore, a single focal zone should be positioned at the pleural line to maximize resolution. 4 Because sonographic evaluation of the lungs is largely based on artifacts, advanced imaging settings that can minimize artifacts, such as harmonics and compounding, are not only unnecessary but also discouraged.4,20
Findings
Normal
Each lung is composed of anatomical lobes. The right lung has three lobes—upper, middle, and lower, and the left lung has two lobes—upper and lower. A thin pleural layer lines the surface of the lung (visceral pleura) and the surface of the chest wall (parietal pleura). 15 The airways arise from the trachea, which branches into right and left main bronchi. The bronchi are composed of connective tissue (cartilage) and smooth muscle. They decrease in caliber and cartilaginous content as they branch peripherally. They eventually become bronchioles, which lack any cartilage. Individual lung lobules are formed mostly by the peripheral components of the bronchioles, including the terminal bronchiole, respiratory bronchioles, and alveoli. Each lobule is bound by fibrous septa. The alveoli are composed of elastic fibers interwoven with capillaries to facilitate oxygenation of the blood.21,22
These structures are not directly visualized with sonography of a normal lung, but knowledge of these structures is essential to understanding the sonographic appearance of both normal lung and pathology. Normal lung is implied when one demonstrates equidistant horizontal echoes arising from the pleural surface and sliding back and forth with respiration.2,3 The reverberation artifact is created by the difference in acoustic impedance between the chest wall and well-aerated alveoli.12,19 In point-of-care, the horizontal echoes are referred to as A-lines. 2 Soldati et al 19 prefer to refer to them as horizontal artifacts (Figure 4).
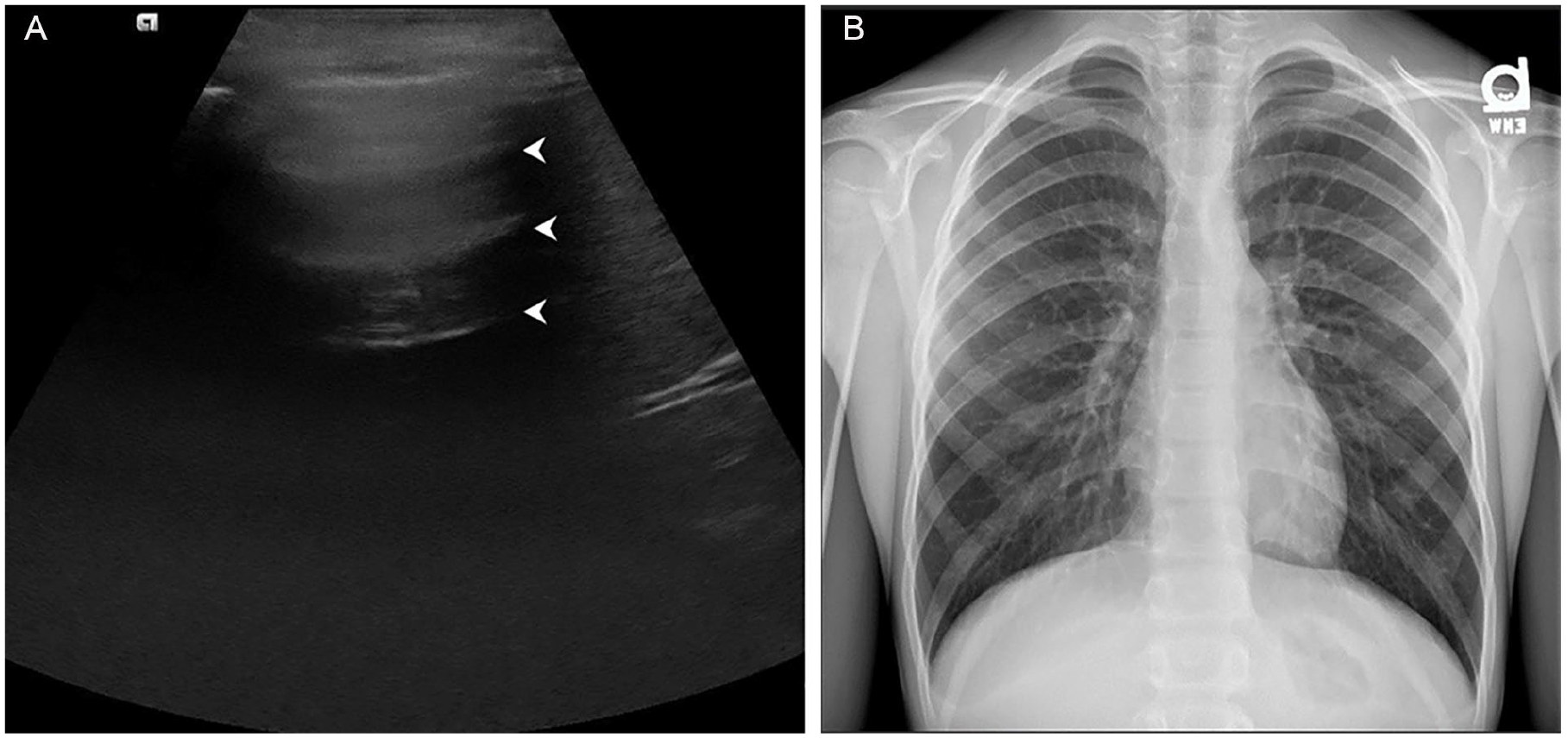
(A) Horizontal reverberation artifact (arrowheads) representing a normal, aerated lung on sonography and (B) the corresponding appearance on radiography.
For inexperienced practitioners, the use of motion mode (M-mode) can be a simple tool to evaluate lung sliding. 20 A normal M-mode tracing shows a stationary chest wall with a combination of referred cardiac and pleural motion creating a wave-like pattern in the deeper chest. This is referred to as the seashore sign.2,20
Pleural effusion
A pleural effusion occurs when the fluid accumulates between the parietal and visceral pleura. LUS is particularly effective at characterizing effusions and identifying debris, septations, loculations, and organized collections.3,5,12,20 The fluid that is purely anechoic is likely to represent transudative fluid, a result of homeostatic imbalance (Figure 5). 23 The fluid containing debris or septations is more likely to represent exudative fluid, which can be indicative of either inflammatory or neoplastic etiologies. 23 When infectious fluid organizes in the pleural space, an abscess will appear as a thick-walled complex collection which is displaced with inspiration. 15 The sonographic evaluation of effusions is well established and widely used to guide a patient’s treatment.
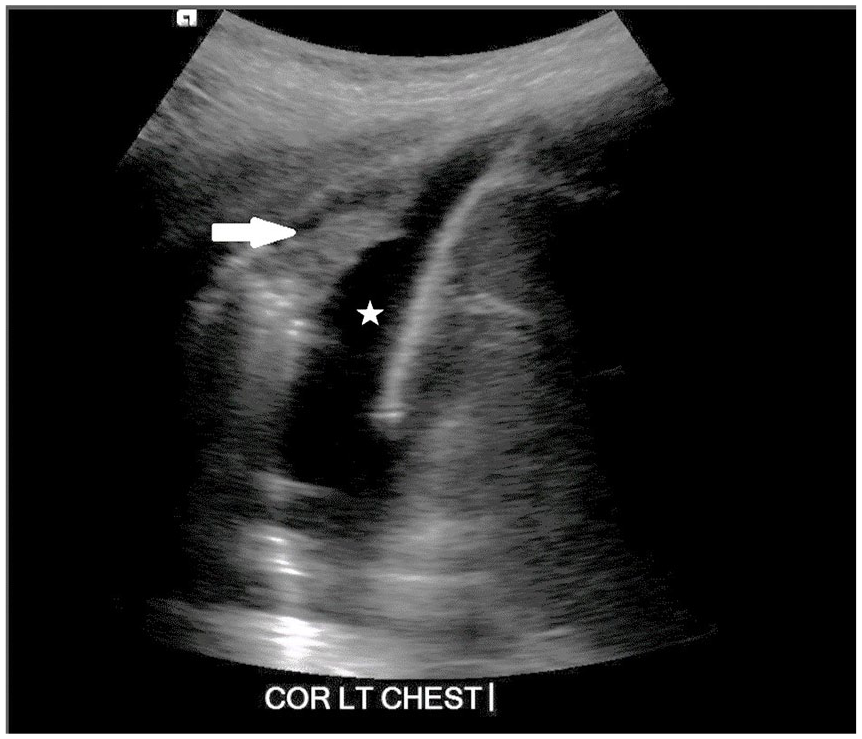
Coronal image of the left chest at the level of the diaphragm demonstrating an anechoic pleural effusion (star). A small area of the base of the lung (arrow) is hypoechoic, representing compressive atelectasis.
Pneumothorax
The absence of lung sliding with respiration has been used to indicate air in the pleural space, called a pneumothorax. 2 Clinicians have used the barcode sign to describe an M-mode tracing without motion in the deep chest. 2 This creates the appearance of fixed horizontal lines, reminiscent of a barcode. 20 Pneumothorax is not the only cause of lung sliding being absent. Pathologies that cause hyperinflation of the lungs, such as asthma and emphysema, can also cause diminished lung sliding. 5
Consolidation
The terms consolidation and hepatization are both used to describe the lung that is not aerated. 12 This loss of aeration is caused by an accumulation of fluid in the alveoli. 24 On LUS, consolidation presents as a homogeneously hypoechoic or echogenic lung with organized vasculature and branching echogenic air-filled bronchi (Figure 6). 24 On LUS, the air-filled bronchi are called bronchograms. 24 When bronchi are fluid-filled, bronchograms become indistinct, and the lung appears very similar to the liver in echotexture, hence the term hepatization. 5 An irregular line between nonaerated and aerated lung tissue should be demonstrated, when able, to differentiate the lung from the liver parenchyma. 12 LUS is more effective at finding small areas of consolidation than a chest radiograph. 24
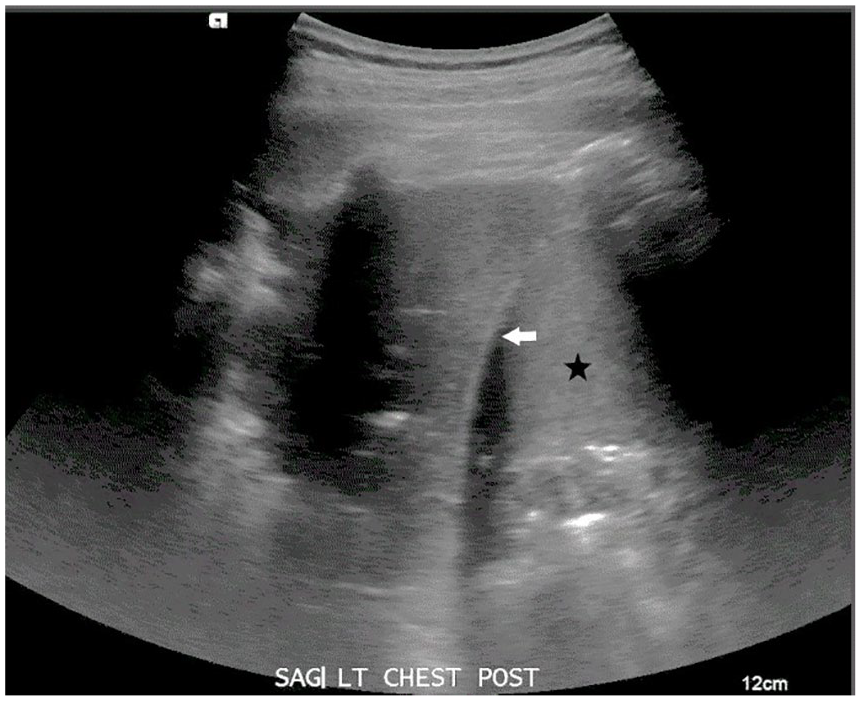
Posterior image at the level of the left diaphragm (arrow) demonstrating isoechoic lung parenchyma compared with the adjacent spleen (star).
Atelectasis
Subcentimeter hypoechoic areas within the lung may be confused with consolidation but are likely small areas of atelectasis. An example of this is demonstrated in Figure 5. 5 Atelectasis occurs when the lung loses aeration by collapsing, rather than being fluid-filled. 5 It can be caused by increased pressure on the lung due to large pleural effusion or by bronchial obstruction.15,23 Distinguishing atelectasis from consolidation using LUS has proven difficult. 5 Some have suggested that crowding of the bronchograms suggests atelectasis. 20 Authors have also reported that the absence of motion within the bronchi with inspiration implies atelectasis. 5 Unfortunately, proving crowding of the bronchograms or motion of the bronchograms with inspiration is difficult.5,20
Lung lesions
LUS is also useful for characterizing parenchymal lesions. 2 Lung necrosis is an extreme complication of respiratory infections, when there is compromised perfusion at the level of alveolar capillaries. 15 On LUS, lung necrosis presents as a heterogeneous area of lung parenchyma composed of both echogenic consolidated lung and hypoechoic necrotic tissue (Figure 7). 15 LUS is able to detect areas of necrosis earlier than other modalities. 5 Necrotic tissue can then progress to form an organized lung abscess with irregular walls, complex fluid, and possibly endoluminal gas. 15 Lung abscesses expand and contract with respiration, unlike pleural abscesses. 23 Other causes of lung abscesses include aspiration, septic emboli, chronic lung disease, lung cancer, and suprainfection of congenital malformations involving the lung.15,23 Solid lung lesions that contact the lung surface may also be detectable with sonography but can vary in echotexture, making them difficult to distinguish from lesions of infectious etiology. 15
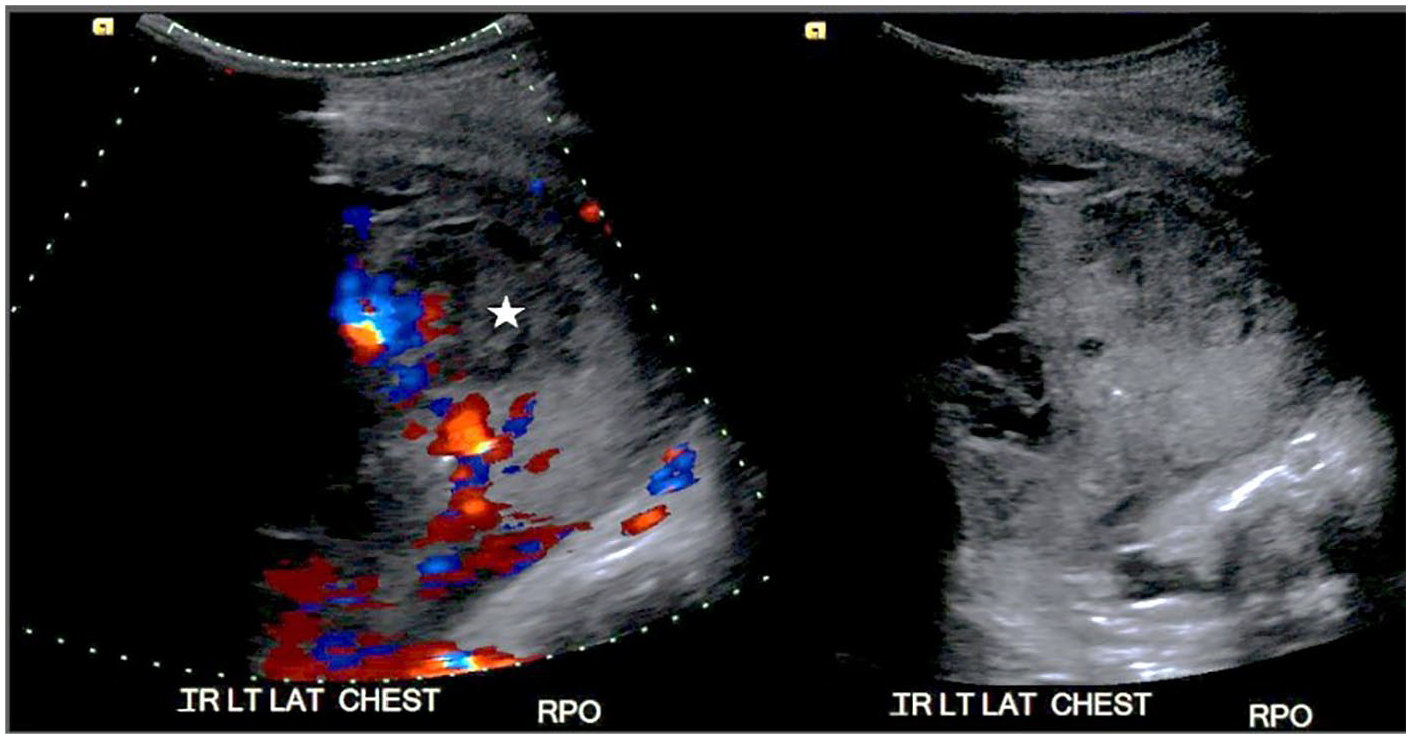
Heterogeneous areas (star) within consolidated lung without perfusion, consistent with areas of necrosis.
Interstitial disease
Subtle findings are present in the setting of interstitial lung disease. The lung may appear aerated at first glance, but the horizontal reverberation artifacts are not demonstrated. Instead, echogenic comet-tail artifacts are seen extending perpendicular from the pleura to the posterior edge of the screen (Figure 8). 12 Clinicians refer to these lines as B-lines.2,12,15 This vertical artifact is created when the acoustic beam becomes trapped in small fluid-filled spaces and echoes back and forth until it is reflected back to the transducer.9,19 This is called an acoustic trap 19 and indicates a microscopic loss of aeration or interstitial fluid.2,12 While few vertical artifacts may be seen in healthy patients, multiple (more than three) artifacts in a single intercostal space could indicate a broad list of parenchymal diseases, including pulmonary edema, interstitial pneumonia, lung contusions, asthma, and pulmonary fibrosis. 15
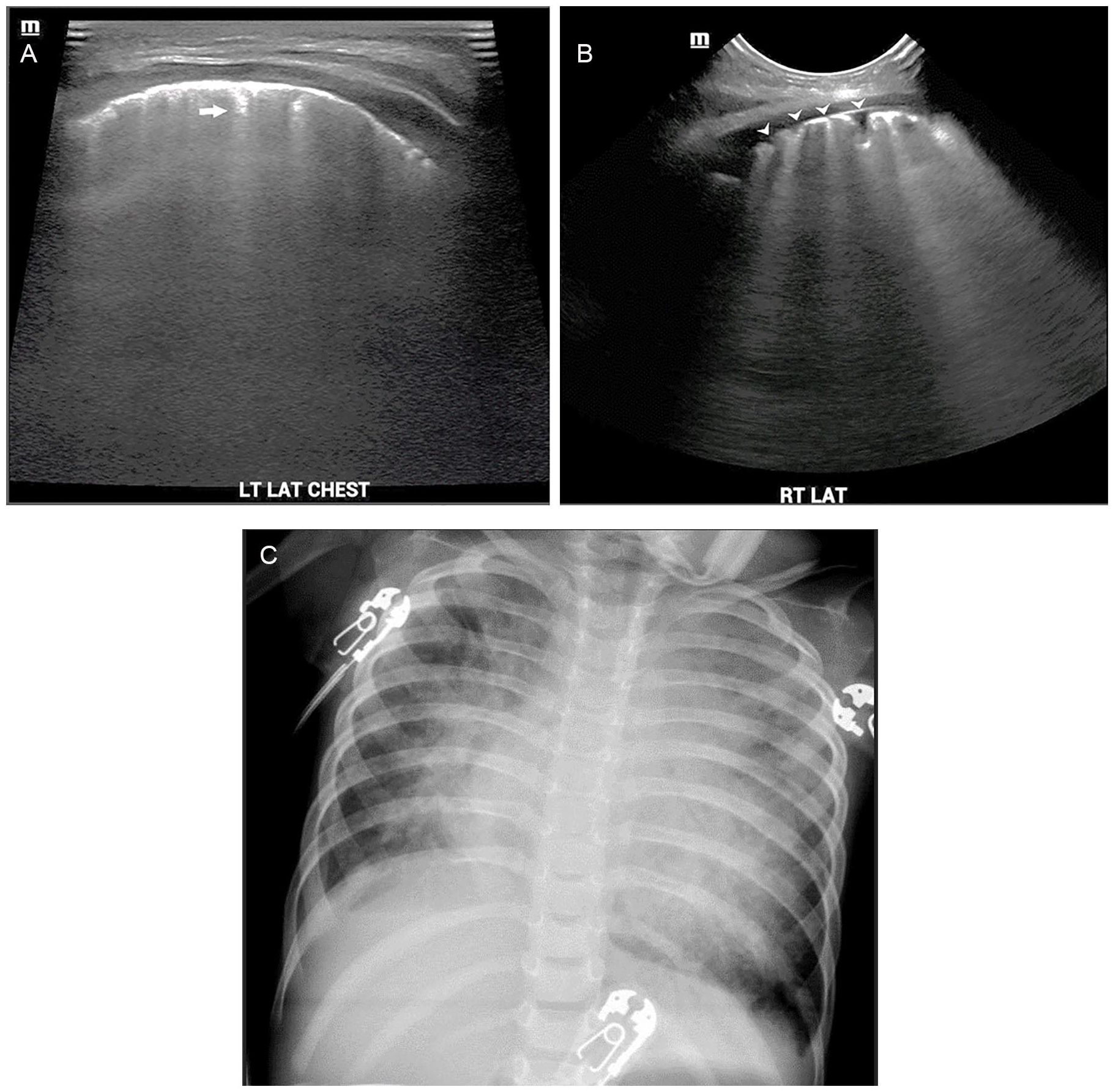
(A) Linear and (B) convex images demonstrating vertical artifacts (arrow), or B-lines, extending from an irregular pleural line (arrowheads) to the posterior edge of the screen and obscuring horizontal artifacts. (C) The corresponding radiograph shows diffuse hazy airspace opacities.
In the event of widespread disease, there may be multiple adjacent vertical artifacts noted within an intercostal space or innumerable confluent artifacts creating the sign known as the “white lung” as demonstrated in a COVID-19-positive patient in Figure 9. 19 With the white lung appearance, no horizontal reverberation artifact is seen, and the surface of the lung appears irregular. 19 This appearance has been correlated with the ground-glass appearance on CT, which indicates thickening of the septa surrounding the lung lobules.3,19
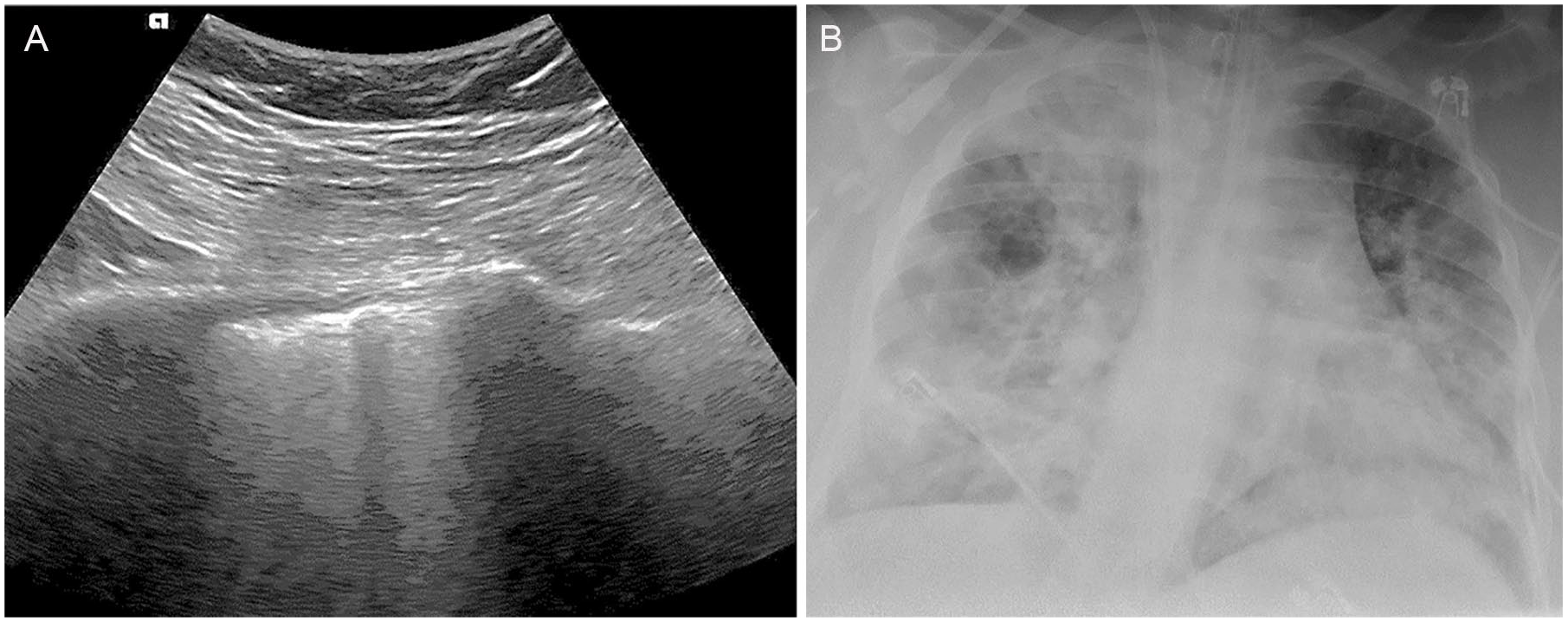
(A) Confluent vertical artifacts obscuring A-lines creating a white lung appearance in a COVID-19-positive patient. (B) Radiograph of a COVID-19-positive patient with severe diffuse bilateral airspace disease.
COVID-19
As COVID-19, the novel coronavirus, has spread across the globe, clinicians with experience in point-of-care LUS have begun describing the constellation of findings in COVID-infected patients. COVID-19 findings are consistent with the appearance of other viral pneumonias: the pleural line becomes irregular with multiple vertical artifacts. 25 Pleural thickening and small areas of consolidation (possibly atelectasis) have also been reported.20,26 Scoring models have been used for monitoring the trend of a patient’s condition. 25 The availability and portability of LUS make it amenable to all varieties of settings, including those with limited resources. Minimalist ultrasound equipment including wireless transducers and tablets can be covered to prevent contamination. 4 Even in these desperate times, recommendations include that one performing LUS for COVID-19 patients have prior experience with either LUS or at least general sonography. 25
Implications
Authors have described different scoring methods to quantify LUS findings. Images in each of the six zones are generally given a score from 0 to 3, and scores are combined for a cumulative score per lung. Authors agree that a score of 0 is considered normal and awarded to a zone in which only horizontal artifacts are visualized.4,16 A score of 1 is widely accepted to indicate an irregular pleural line combined with vertical artifacts.4,16,25 Both Smith et al 16 and Salem et al 25 have assigned zones, with largely extended white lung sign given a score of 2. A score of 3 is reserved for cases of consolidated lung.16,25 In evaluating COVID-19 patients, Soldati et al 4 gave zones with small areas of consolidation a score of 2 and gave the white lung appearance a score of 3 whether or not there is consolidation (Table 1).
Lung Ultrasonography Findings and Scores, as Generally Described.

Soldati et al gave COVID-19 patients a score of 2 for small areas of consolidation and 3 for white lung with or without consolidation.
Dr Lichtenstein further attempted to diagnose specific lung diseases by noting whether findings were anterior or posterior, or unilateral or bilateral.9–11 Toma argued against this practice, particularly in the bedside setting, saying that it “created a series of contradictions that have weakened the technique that had intrinsic validity of support.” 20 The practice of LUS is complicated, and standardizing it requires knowledge of ultrasound physics and correlation with other imaging modality findings. 20
General characterizations of lung findings, particularly in the setting of pneumonia, are recognized by the radiology community (Table 2). If a patient with a clinical diagnosis of respiratory infection presents with vertical artifacts alone, it is treated as viral pneumonia. 3 Findings of both lung consolidation and a pleural effusion are considered diagnostic of bacterial pneumonia. 3 Because LUS is so sensitive to small areas of unaerated lung, small hypoechoic areas (<5 mm) may be misinterpreted as signs of bacterial infection, especially in the hands of an inexperienced user. 20 The depth of suspected areas of consolidation should be measured perpendicular to the lung’s surface. Anything that measures less than a centimeter should not be an indication for antibiotic treatment. 24
Widely Accepted Sonographic Lung Findings and Their Implications.

Limitations
As with all sonograms, diagnostic accuracy is strongly dependent on the practitioner’s skills. 12 In the current setting, there are limited avenues for one to obtain appropriate training to perform accurate LUS. 1 Condensed training courses may not expose new practitioners to the broad possibilities associated with LUS findings. 1 False interpretations of data could lead to incorrect treatment, including overuse of antibiotics. 20
If pathology does not involve the pleural surface, it cannot be identified with LUS. 24 This technical limitation makes it impossible to completely eliminate radiography. 3 Correlation with physical examination findings is essential to a proper interpretation.5,24 For example, imaging alone cannot diagnose pneumonia but, when combined with laboratory and clinical data, can create a cohesive description of a patient’s condition. 20
Because LUS has been largely developed by clinicians, the physical mechanisms behind pulmonary artifacts are poorly understood. 19 Subsequently, ultrasound equipment does not have optimized settings for performing lung imaging. 19 For those who are not familiar with manipulating sonography equipment, optimizing lung images may be difficult.
Another limitation that could be addressed is the lack of imaging correlation between CT and radiography in patients who have a bedside LUS. 3 Studies of the correlation between LUS findings and clinical outcomes are also lacking. 12 There is little scientific data to determine whether LUS is sufficient to exclude bacterial pneumonia and forego antibiotic treatment. 3 This knowledge gap creates ambiguity in interpretive guidelines. 3
Recommendations
Those with extensive sonography experience are the best prepared to lead research in this field. While sonographers are unfamiliar with the specifics of lung imaging, their understanding of ultrasound techniques and physics has equipped them to contribute to this emerging practice. 25 Cooperative learning between those with formal sonography education and those with clinical training is essential to make this practice accurate and reliable. 5
Sonography is not a reasonable substitute for a high-quality chest CT. 19 Advanced techniques such as CT must be used in cases that are complex or not improving. 5 A goal of LUS should be to supplement a clinical examination, providing additional information for planning care. 19 As the most qualified experts in imaging, radiologists should help clinicians develop this practice, so it can be effectively used in point-of-care settings. Guidance should also include a clear explanation of the limitations of ultrasound 3 and warning signs that would indicate the need for formal imaging in a radiology department. 5 Correlation with the gold standard, CT, is essential to creating a foundation for interpreting ultrasound findings. To achieve this, Lovrenski 5 suggested performing research LUS following each clinically indicated CT and correlating findings.
Conclusion
While LUS is not widely used in imaging departments, it is becoming increasingly used in the clinical setting. The COVID-19 pandemic has propelled the use of LUS as a possible diagnostic tool. The artifacts created by the pleural surface may not represent anatomical structures, but they can demonstrate the condition of lung parenchyma. Limited-view protocols are proven to provide adequate diagnostic information and reduce overall examination time. Sonography of the chest is already widely performed in radiology departments to evaluate pleural effusion. Information about the condition of the lung is likely already in these images, but it is overlooked. Sonographers should become familiar with this specific anatomy.
The significant limitations of LUS are largely related to lack of knowledge and do not outweigh the real benefits it could offer. Chest radiography is widely used but far from perfect. With the information supplemented by LUS, some patients could avoid the cost and ionizing radiation associated with chest CT. Standardized scoring has the potential to quantitatively trend a patient’s condition. Sonographers and sonologists are encouraged to participate in the research necessary to delineate protocols, techniques, and interpretive guidelines that could standardize this practice for future use.
Participating in research is the only option to discredit the use of sonography in evaluating the lung. If it is truly unreliable, scientific evidence is needed to prevent clinicians from basing treatment on a nondiagnostic examination. Sonographers and radiologists have the technical expertise and knowledge of medical imaging needed to determine the reliability of the practice of LUS.
Footnotes
Acknowledgements
The author would like to thank Dr Aparna Joshi for guiding the structuring of this paper and Dr Erich Smith for providing correlating radiographic images.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
