Abstract
Prune belly syndrome is a rare disorder characterized by the absence of anterior abdominal wall muscles, bilateral cryptorchidism, and urinary tract malformations. The aim of this case study was to illustrate the developmental sequelae of prune belly syndrome, acquired through a series of antenatal sonography. A 20-year-old woman visited a diagnostic medical sonography center for an obstetrical sonogram. Her fetus was diagnosed with megacystis at 15 weeks’ gestation. The mother returned for repeated appointment and to observe fetal changes during the development of prune belly syndrome. First, the megacystis, hydroureter, and hydronephrosis were noted followed by hazy ascitic and amniotic fluid. The hemodynamics were altered in the umbilical artery, middle cerebral artery, and ductus venosus. Subsequently, the pressure was neutralized, but it was observed after birth that the newborn had cryptorchidism, a distended abdomen with wrinkled wall, and absent right kidney. In conclusion, prune belly syndrome could be caused by megacystis due to bladder outlet obstruction in otherwise normal karyotype fetuses. Megacystis leads to hydroureter, hydronephrosis, and abdominal distention. The persistent abdominal distention gives rise to the underdevelopment of anterior abdominal wall muscles and cryptorchidism.
Keywords
Prune belly syndrome is a rare, genetic birth defect affecting about 1 in 40,000 births. 1 About 97% of those affected are male.2,3 Prune belly syndrome is a congenital disorder characterized by a triad of symptoms, such as absence of abdominal wall muscles, cryptorchidism, and urinary tract defects. 4 This syndrome was named for the mass of wrinkled skin that is often (but not always) present on the abdomen of infants with this disorder. 5 The fetal anatomy as well as hemodynamic evaluation can be satisfactorily performed with ultrasonography.6,7 In the current case, the mechanism of the prune belly syndrome development was studied. Prune belly syndrome becomes a social and economic burden on families, but its timely antenatal sonographic diagnosis and treatment with vesicoamniotic shunting can reduce its development to a great extent. 8
Case Presentation
This case study was followed for two and a half years from March 2017 to September 2019. The patient was scanned with a Toshiba Xario (Japan) and a convex transducer (frequency 3–6 MHz) equipped with color, power, and spectral Doppler over the course of these examinations. 9 The sonographic procedure was explained to the patient and her husband. Written informed consent was obtained with the assurance that personal identifiers would be kept undisclosed. The studies were performed according to American Institute of Ultrasound in Medicine (AIUM) obstetrical ultrasound protocols, which are routinely used in the department.10,11 Color, power, and spectral Doppler were applied to the evaluation of umbilical and middle cerebral artery and the data were saved to the ultrasound equipment.
Patient Medical History
A 20-year-old pregnant woman (gravida 1, para 0) was sent by her gynecologist to the sonography department. Her chief complaint was intermittent hemorrhage for 26 days. She was educated, and her consent was easily gained for the purposes of this publication. The mother and father were not related genetically. There was no history of diabetes, hypertension, or any other known disease in either father or mother. No known anomalous fetal case was present in either the maternal or paternal family history. The detail of her serial sonographic visits is summarized in Table 1.
Sequelae of the Current Case (Prune Belly Syndrome).
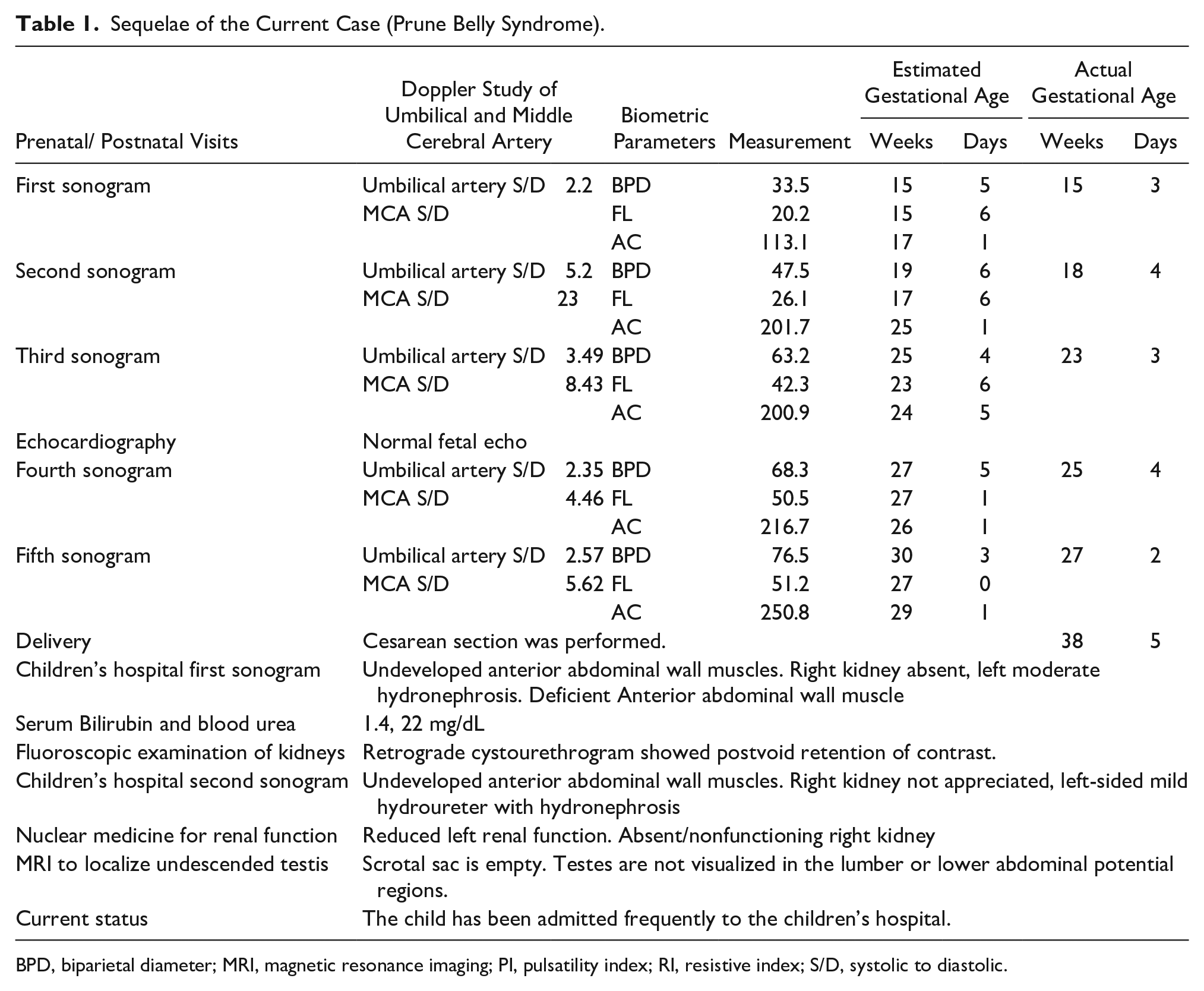
BPD, biparietal diameter; MRI, magnetic resonance imaging; PI, pulsatility index; RI, resistive index; S/D, systolic to diastolic.
Imaging Data
During the first visit, at 15 weeks and 3 days, the fetal skull was properly developed, and the hemispheres of the brain appeared normal in the transverse view (see Figure 1). Both fetal kidneys were sonographically appreciated, with mild hydronephrosis. The fetal foot (20.5 mm) and tibia (15.5 mm) appeared normal. The amniotic fluid was clear and appeared anechoic on the sonogram (see Figure 2). A large urinary bladder with thick walls and a “keyhole sign” (megacystis) was observed on gray-scale sonography. An excessive vascular perfusion, in the bladder wall, was observed on color Doppler (see Figure 3). Systolic to diastolic (S/D) ratio, resistive index (RI), and pulsatility index (PI) of the umbilical artery were 2.2, 0.54, and 0.91, respectively (see Figure 4).
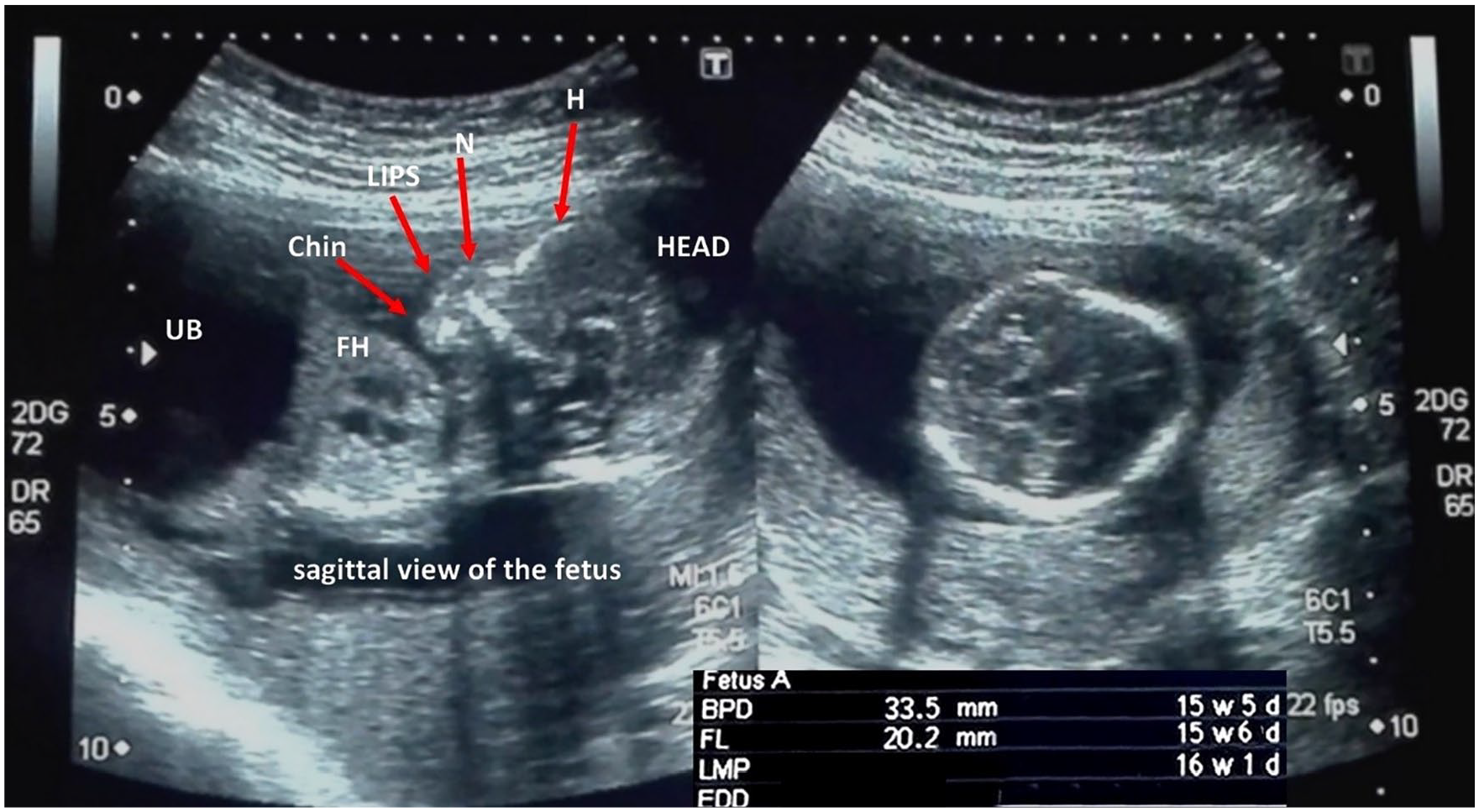
Fetal facial profile in sagittal view and biparietal diameter.
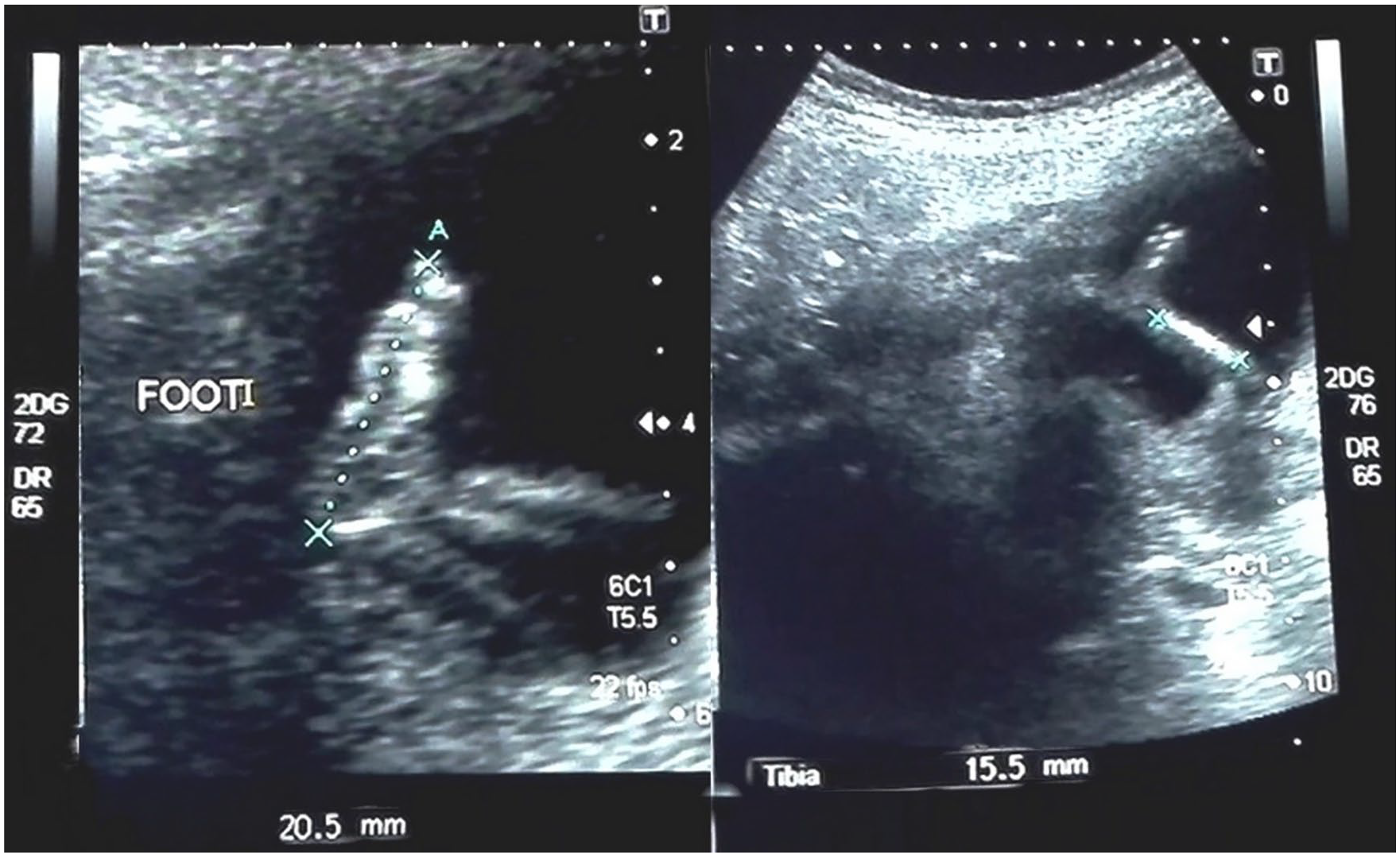
Foot and leg of the fetus measuring 20.5 mm and 15.5 mm, respectively.
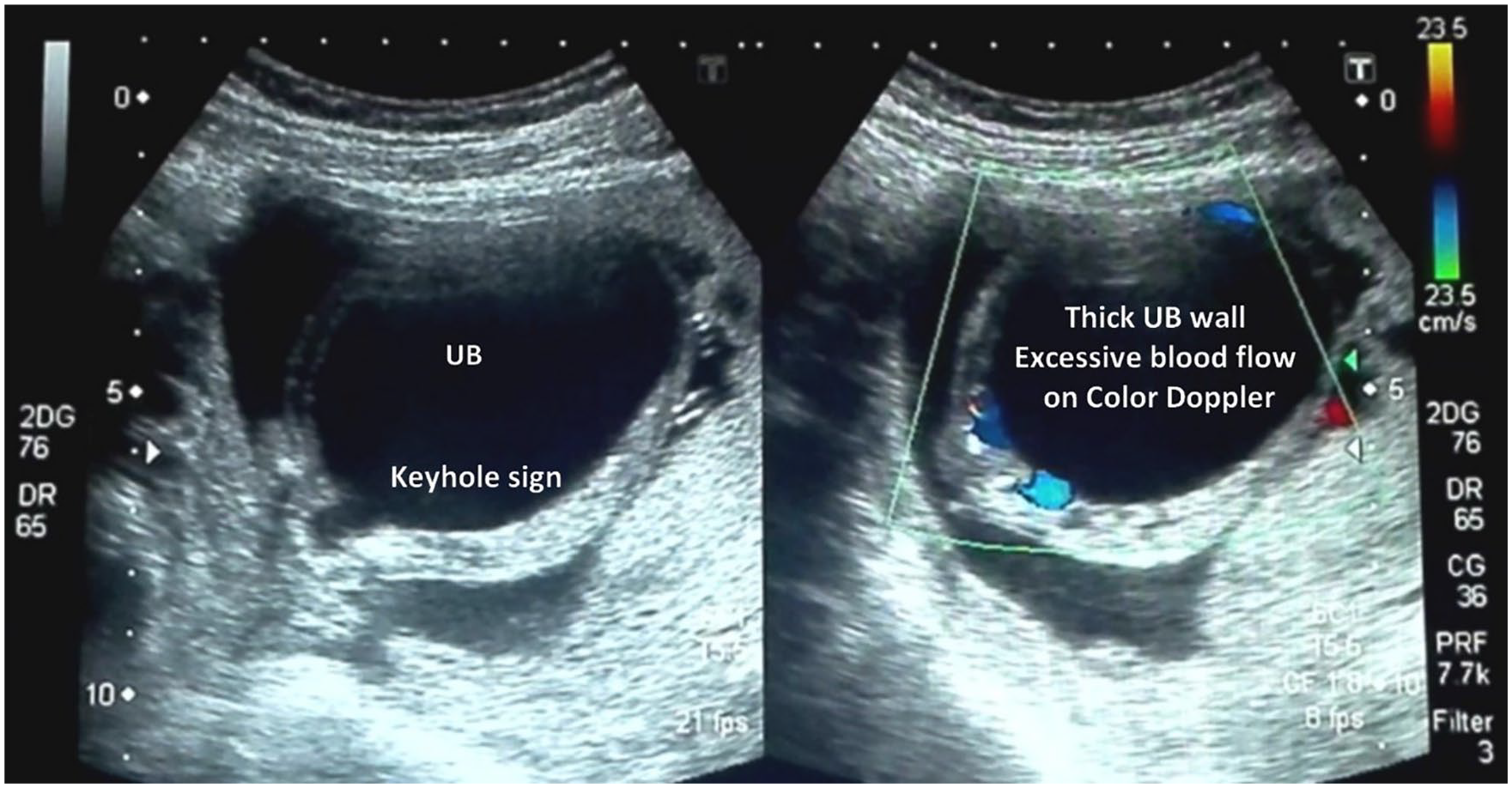
Thick-walled, hyperemic urinary bladder with keyhole sign.
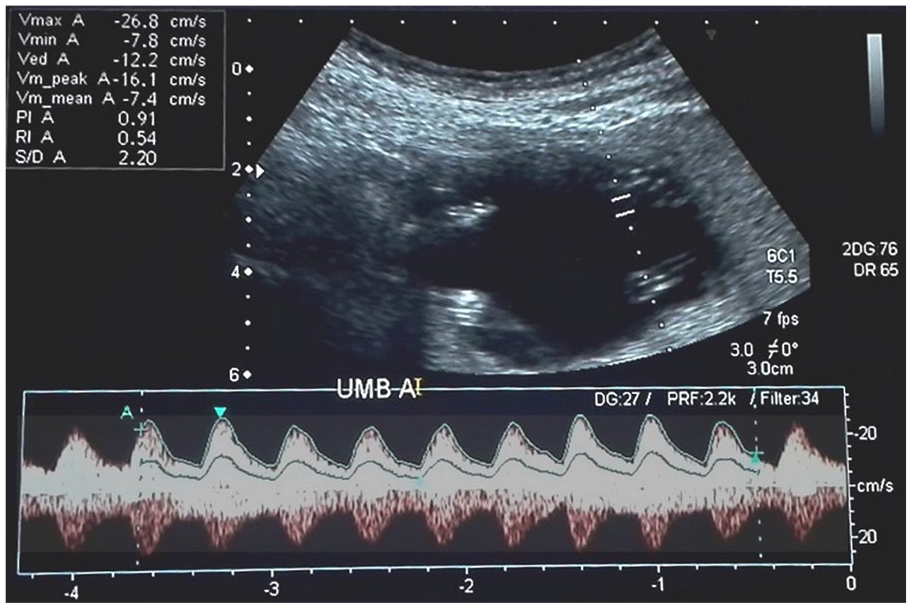
Low resistive spectral Doppler waveform of the umbilical artery.
Subsequent Sonographic Examinations
The second sonographic appointment occurred after a lapse of 22 days, but the actual fetal gestational age was 18 weeks and 4 days. This was noted as a discrepancy based on the fetal biometry. The biparietal diameter (BPD) was 47.5 mm (19 weeks, 6 days), the femur length (FL) was 26.1 mm (17 weeks, 6 days), and the abdominal circumference (AC) was 201.7 mm (25 weeks, 1 day). The urinary bladder was extremely distended, and on this examination, the fetus was noted to have severe ascites. The intestines were filled with echogenic contents. The liver and the echogenic intestines were freely floating in the ascitic fluid. The urine, ascitic fluid, and amniotic fluid were all slightly echogenic, with a noticeable “snowflake sign” during the dynamic gray-scale sonographic examination. Moderate renal hydronephrosis was seen bilaterally. The chest was shortened due to the compression of the ascitic fluid and megacystis. A four-chamber fetal heart, with intact interventricular septum, was appreciated (see Figure 5). On Doppler examination, the S/D ratio, RI, and PI of the umbilical artery were 5.29, 0.81, and 0.74, respectively (see Figure 6). Blood flow reversal was observed in the ductus venosus during the atrial contraction phase (nadir/lowest point), represented by the arrows in Figure 7. In the fetal middle cerebral artery, the S/D ratio, RI, and PI were 23.3, 1.0, and 2.31, respectively, as shown in Figure 8.
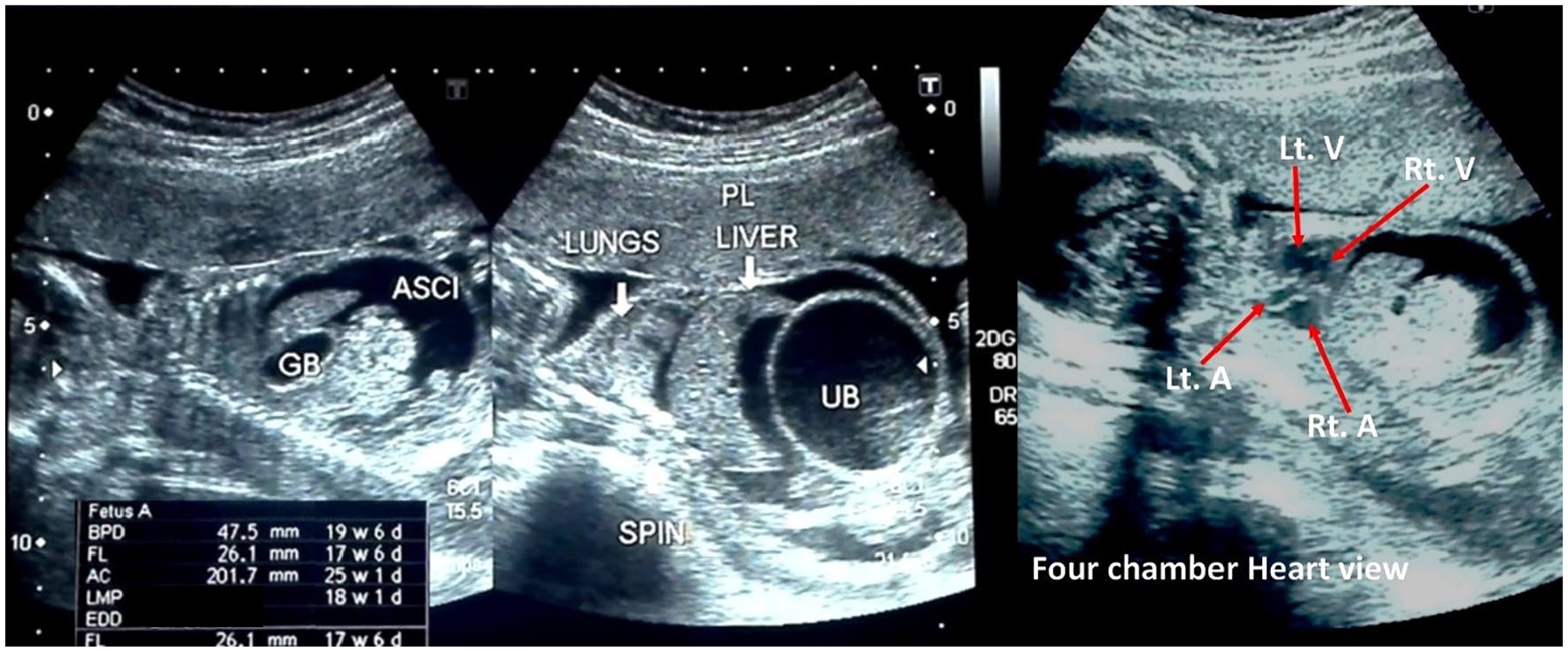
Short chest, hazy urine, ascitic and amniotic fluid, and four-chamber heart.
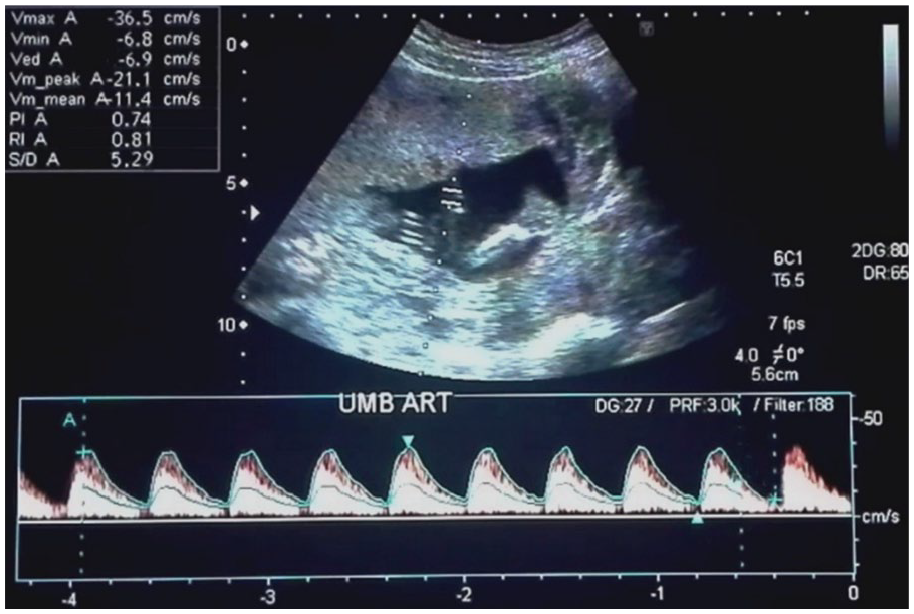
High resistive spectral Doppler waveform of the umbilical artery.
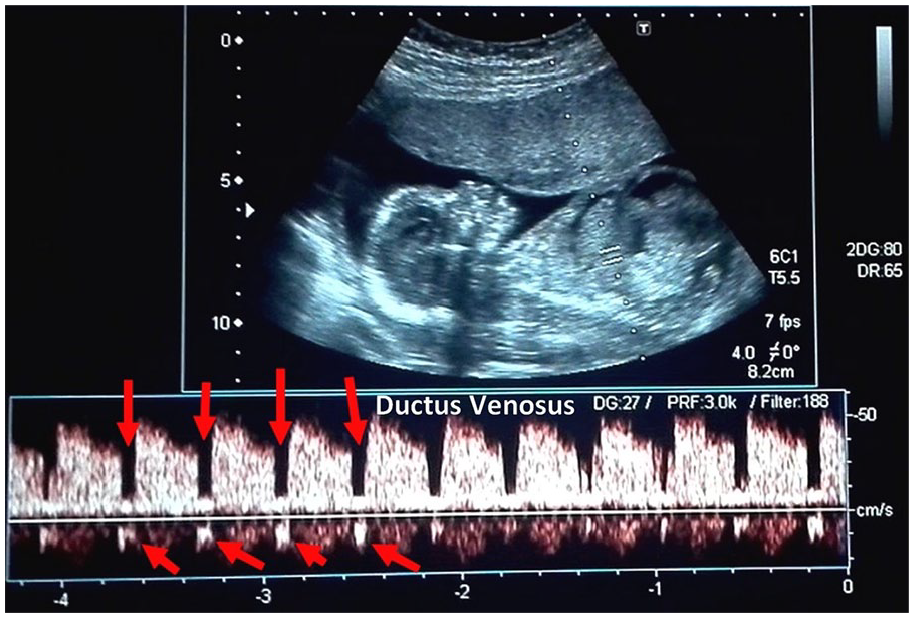
Spectral Doppler waveform of the ductus venosus with reversal of flow during atrial contraction.
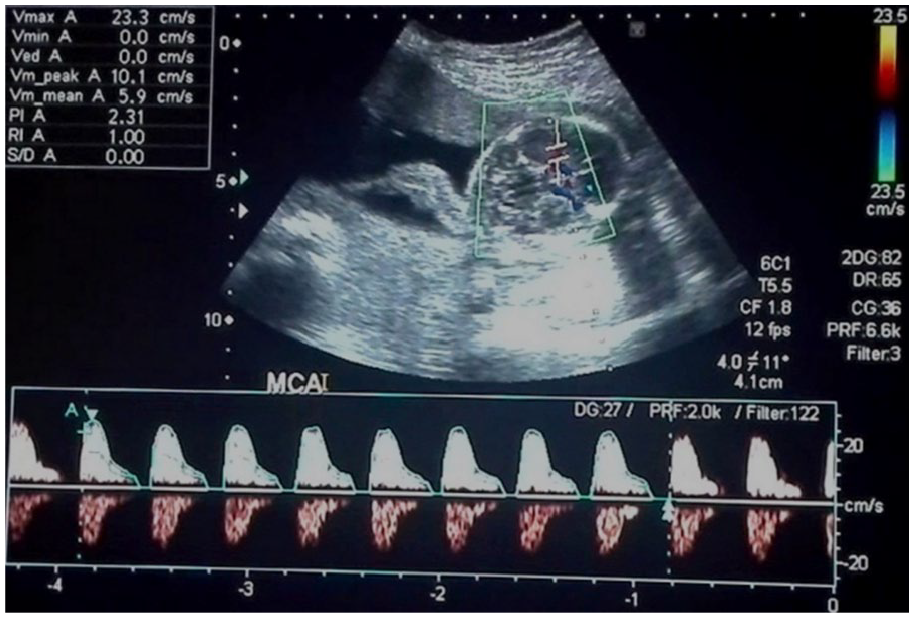
Spectral Doppler waveform of the fetal middle cerebral artery.
Thirty-one days after the second sonographic examination, the patient was reexamined, but now the actual gestational age was 23 weeks and 3 days. There was a minimal difference noted with the fetal biometry. The BPD was measured at 63.2 mm (25 weeks, 4 days), FL was 42.3 mm (23 weeks, 6 days), and the AC was measured at 200.9 mm (25 weeks, 1 day). The ascites previously seen had subsided and the urinary bladder was thick-walled (5 mm) but normal in size. A bilateral moderate hydronephrosis and mild hydrocephalus were also noted on this sonographic study (see Figure 9). A Doppler study was performed to determine the status of fetal vascular remodeling. The S/D ratio, RI, and PI of the umbilical artery were 3.49, 0.71, and 1.21, and the middle cerebral artery measurements were 8.43, 0.88, and 2.06, respectively. A fetal echocardiogram was performed, but the diagnostic findings were normal. A lapse of 15 days occurred before the patient returned for her fourth examination. On this sonographic study, the actual gestational age of the fetus was 25 weeks and 4 days, but a very small difference was found across the biometric parameters. The BPD was 68.3 mm (27 weeks, 5 days), FL was 50.5 mm (27 weeks, 1 day), and AC was 216.7 mm (26 weeks, 1 day). The urinary bladder was thick-walled (7.9 mm) but normal in size. The bilateral mild hydronephrosis and resultant enlarged kidneys (left, 46 × 26 mm; right, 47 × 23 mm) were noted on this examination. No fetal ascites, pleural effusion, or hydrocephalus was noted. The Doppler study of the umbilical and middle cerebral artery was completed. The S/D ratio, RI, and PI of the umbilical artery were 2.35, 0.57, and 0.83 and middle cerebral artery were 4.46, 0.78, and 1.51, respectively.
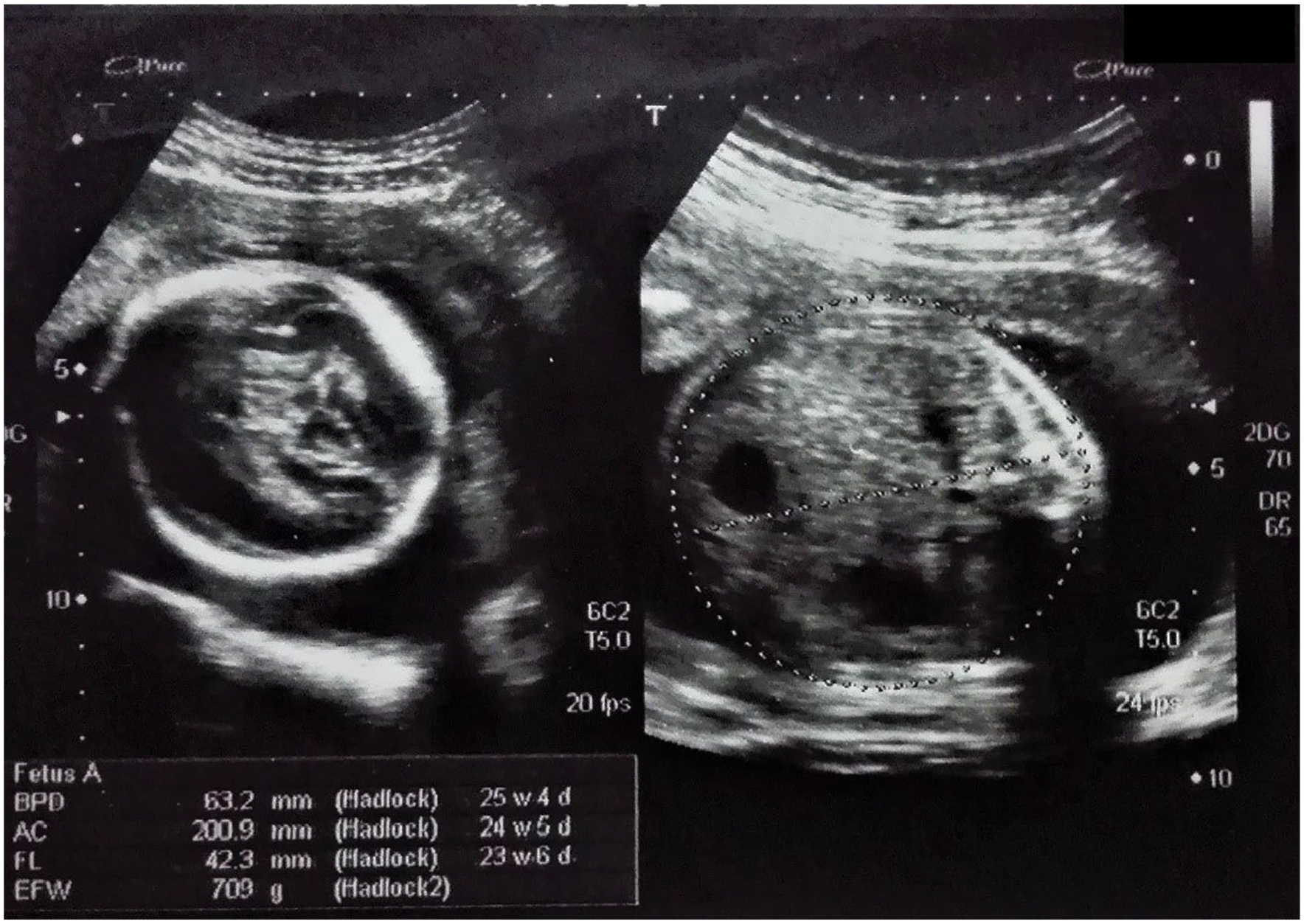
Fetal biometry at 23 weeks and 3 days (actual) of gestational age.
At the fifth sonographic visit, the fetus appeared to have survived the fetal complications, and most of the biometric parameters were normalized, with little discrepancy. The BPD was 76.5 mm (29 weeks, 1 day), FL was 51.1 mm (27 weeks, 0 days), and AC was 250.8 mm (29 weeks, 1 days). The actual fetal weight, which was calculated from the last menstrual period was 27 weeks and 2 days (see Figure 10). The amniotic fluid was normal in volume but remained hazy in its sonographic appearance. The Doppler parameters such as the S/D ratio, RI, and PI of the umbilical artery were 2.57, 0.61, and 1.00 respectively (see Figure 11). The S/D ratio, RI, and PI of the middle cerebral artery were 5.62, 0.82, and 1.88 respectively (see Figure 12). The ductus venosus had a normal blood flow pattern, as there was forward flow during atrial contraction (see Figure 13). The right kidney, along with its artery, was not visualized on gray scale or during Doppler evaluation. However, highly resistive blood flow was detected in the left renal artery on spectral Doppler with RI of 0.92, as shown in Figure 14.
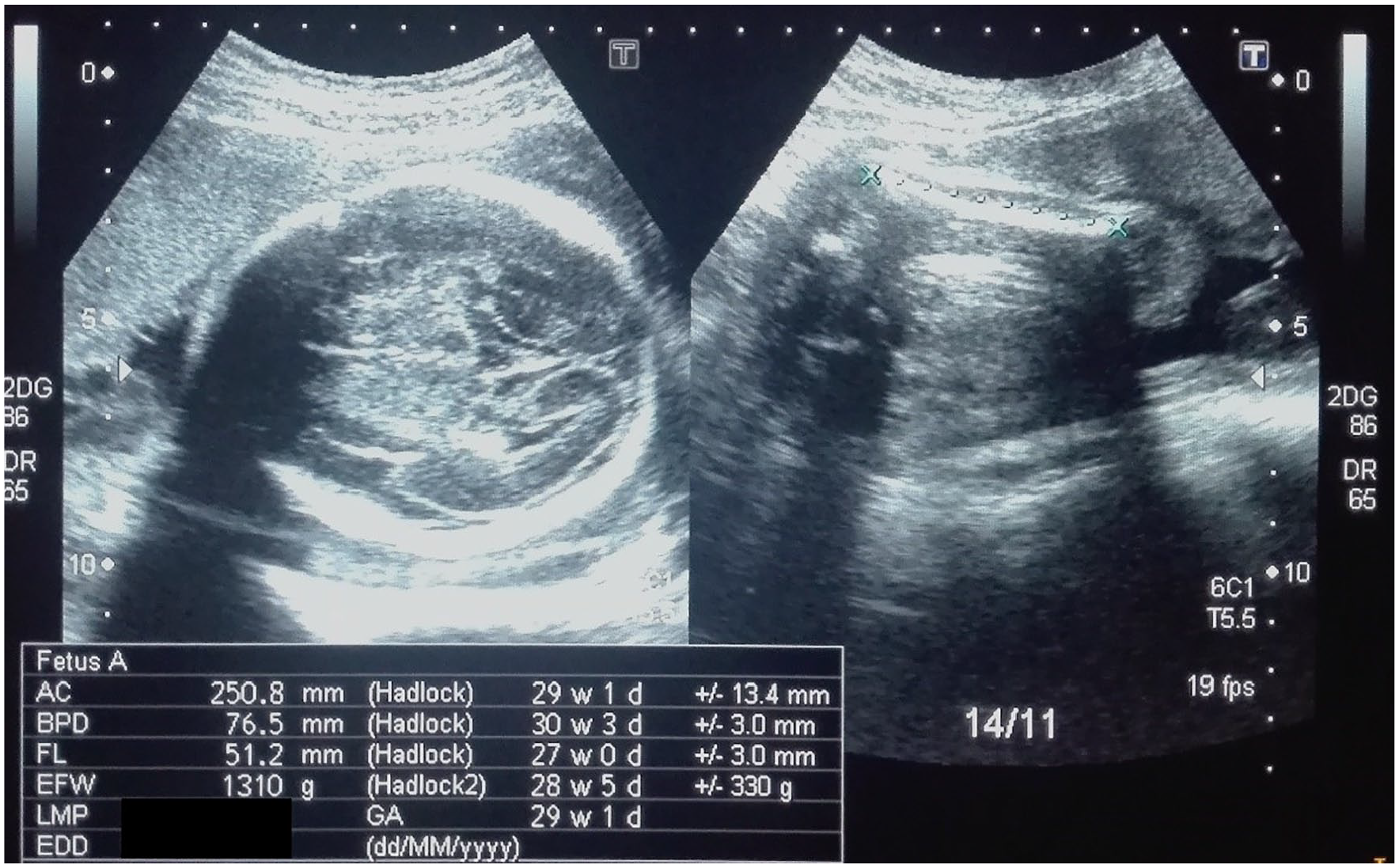
Fetal biometry at 27 weeks and 2 days (actual) of gestational age.
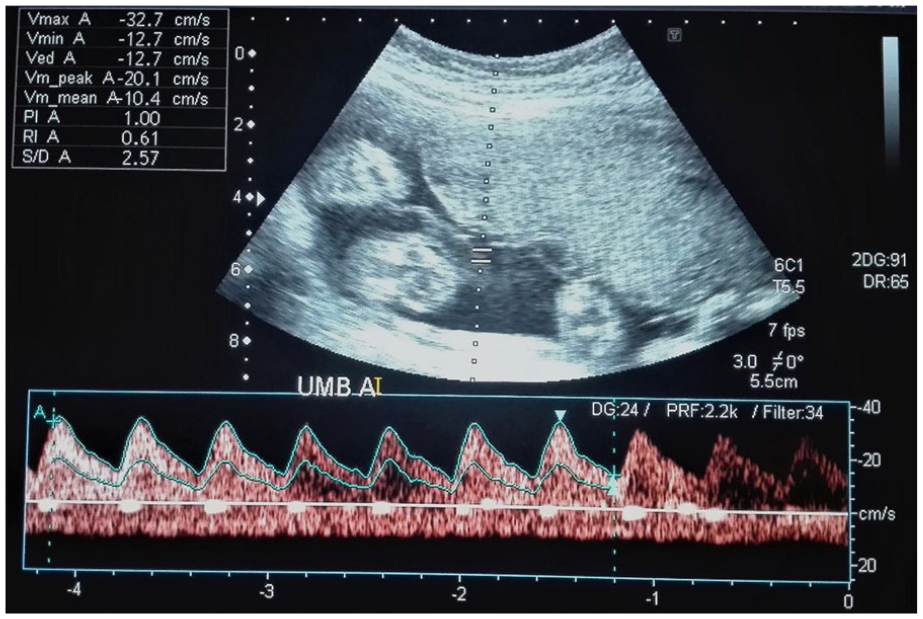
Normalized, low resistive spectral Doppler waveform of the umbilical artery after retrieval from a severe insult.
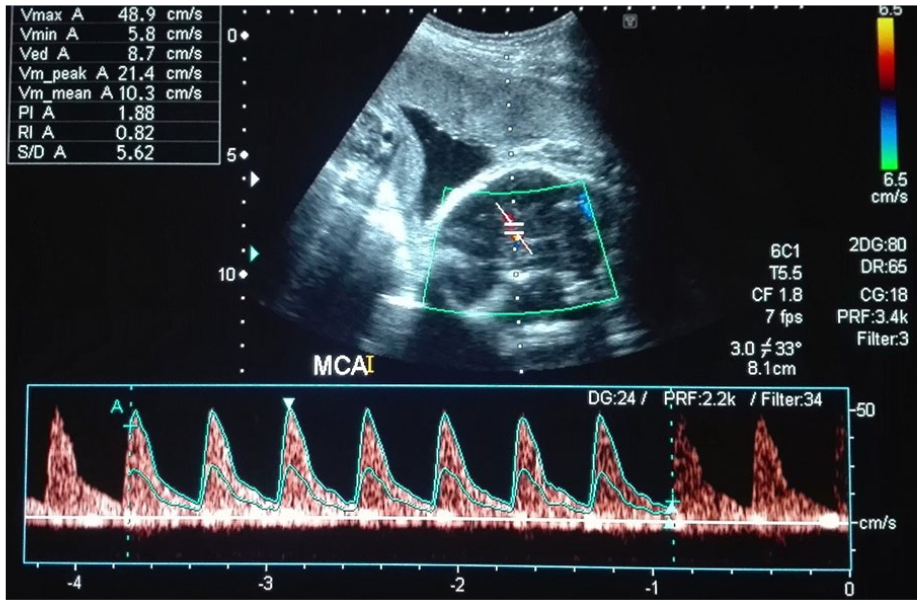
Spectral Doppler waveform of the fetal middle cerebral artery after normalization.
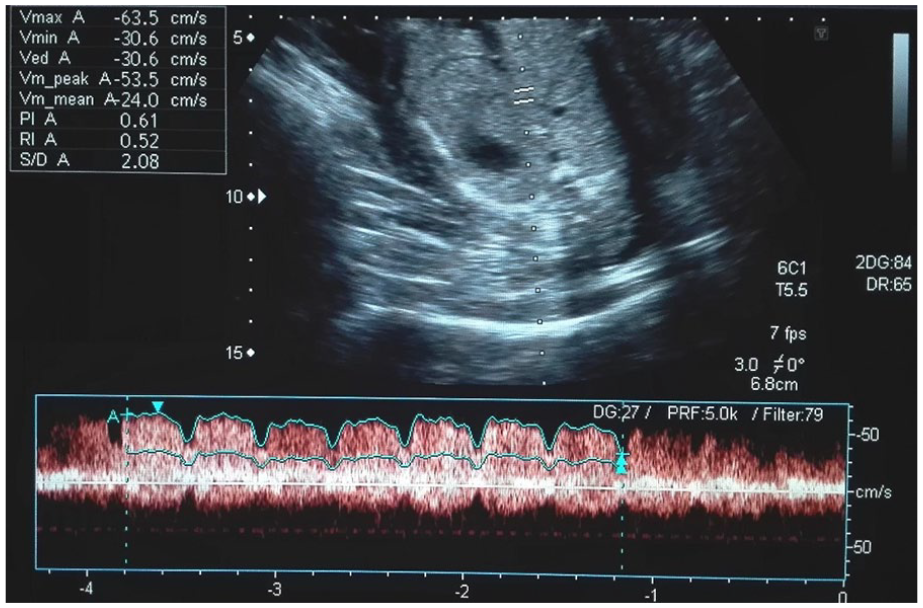
Spectral Doppler waveform of the ductus venosus with no reversal of flow during atrial contraction after retrieval from a severe insult.
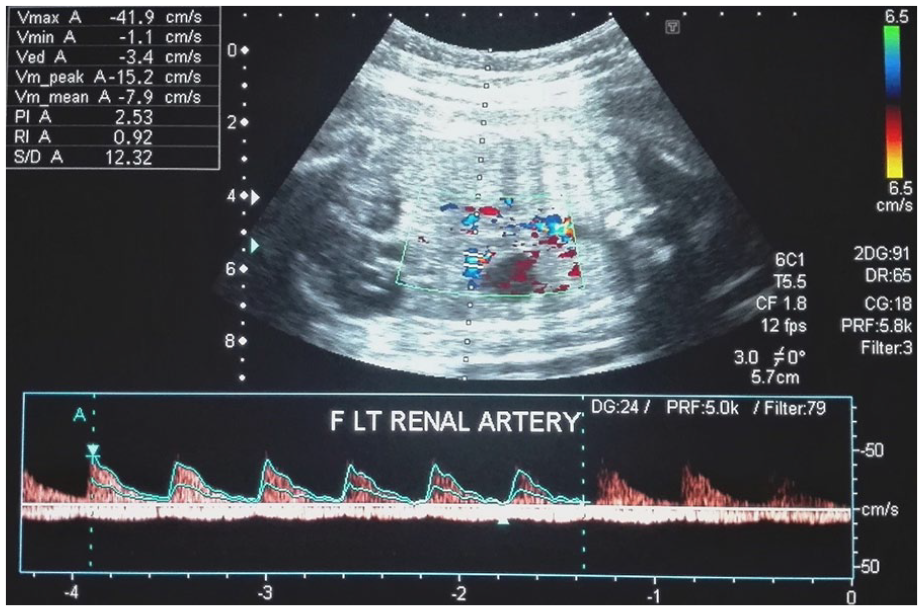
High resistive fetal left renal artery but right renal artery and right kidney were not visualized.
A caesarian section was performed at 38 weeks and 5 days of gestation. The abdomen of the neonate was distended, and the skin was wrinkled on physical examination (see Figure 15). But with a pediatric sonogram, undeveloped anterior abdominal wall muscles were noted, as well as cryptorchidism and the lack of a right kidney. Sonographically, moderate hydronephrosis was observed in the left kidney. Based on the pediatric abdominal sonographic examination and physical appearance, the infant was diagnosed with prune belly syndrome. At the age of 42 days, the infant underwent a retrograde cystourethrogram. Reportedly, a postvoid retention of contrast was observed that indirectly reflected urinary outlet stenosis, although no filling defect or stricture was seen in the micturating urethrogram. A subsequent pediatric sonogram was performed at the age of 80 days, and it again revealed undeveloped anterior abdominal wall muscles, an absent right kidney, and left-sided mild hydroureter with hydronephrosis. A nuclear medicine scan was performed for the evaluation of renal function. It was reported that the function of the left kidney was reduced and the right kidney was either nonfunctional or absent due to the lack of radionuclide uptake. At the age of one year and three months, a magnetic resonance imaging (MRI) examination was performed to locate the testes, but no evidence of testicular tissues was seen in the potential abdominal regions.
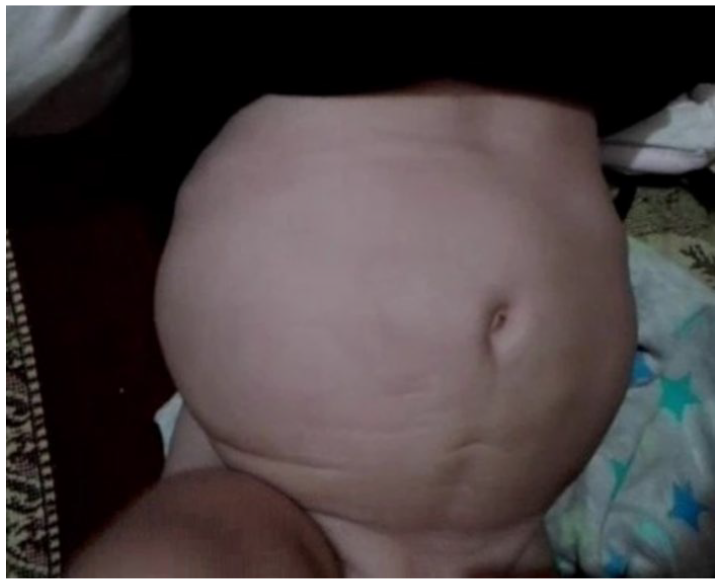
Abdominal distention of the baby after one year of the delivery through caesarian section.
Discussion
Prune belly syndrome (Eagle-Barrett syndrome) is a rare clinical entity, characterized by partial or complete abdominal muscle absence, cryptorchidism, and urinary disorder.2,12 Prune belly syndrome is often diagnosed prenatally by sonography or postnatally by physical examination. Children who are diagnosed with prune belly syndrome often have urinary tract infections.13,14 A previously published case of a 36-year-old diabetic primigravida woman was examined for having a baby with prune belly syndrome. In that case, an ultrasound-guided vesicocentesis was performed as an antenatal treatment to reduce the effect of insult. Although the treatment proved effective, the parents decided to terminate the pregnancy. The autopsy of the fetus revealed that it was a male with cleft lip, cleft palate, bilateral club feet, and polydactyly. 15 The current case study differs by comparison with no additional anomalies detected in the fetal facial profile and no signs of polydactyly.
Alkhamis et al. 16 presented an antenatal diagnosis of prune belly syndrome, as a conference case study challenge, in Saudi Arabia. A 29-year-old Arab pregnant woman (G4P3A1) was followed while having a fetus with prune belly syndrome. The case, diagnosed with sonography, had a large bladder, distended abdomen, bilateral small hypoechoic kidneys with hydronephrosis, and bilateral hydroureters. This case was suggested for prune belly syndrome based on the sonogram. At 32 weeks of gestation, a vesicocenthesis of the fetus was performed, but the parents decided to terminate the pregnancy. The delivery was induced and the infant was delivered vaginally with an Apgar score of 5 in 5 minutes. The sonographic findings of prune belly syndrome (distended abdomen and thin wrinkled skin, retracted chest, cryptorchidism, and clubbed feet) were confirmed. This also is quite different from the current case study as there was no evidence of a club foot. In the current case, the fetus was delivered by caesarian section and the child progressed. Nevertheless, the child continues to need ongoing medication for his recurrent urinary infection. Potential social, clinical, and economic challenges are likely for the child and his family due to his cryptorchidism and abdominal distention.
Another case was reviewed on the sonographic findings of prune belly syndrome that involved a 20-year-old woman (G1P0) who presented at 21 weeks of gestation. The patient was evaluated, with fetal ascites, normal fetal long bones, and cephalic biometry reported on ultrasonography. However, the AC measurement was more than the 95th percentile of the normal growth curve. Fetal defects were found in both kidneys, demonstrating dysplasia, hydronephrosis, and hydroureters. During the sonogram, the fetal bladder was empty and had thick echogenic walls. The fetal abdominal wall was distended and associated with oligohydramnios. Fetal thoracic hypoplasia and bilateral club foot were also noted on the fetal sonographic examination. The abovementioned sonographic criteria were suggestive of prune belly syndrome, although there was no megacystis. It was hypothesized that the concomitant presence of ascites and a persistent empty bladder was due to the rupture of the fetal right kidney. The urethral obstruction was suggested due to the presence of thick echogenic bladder wall. A normal male karyotype was the result from the fluid analysis taken at amniocentesis. After counseling, it was decided by the patient to terminate the pregnancy. It was concluded that the three concomitant sonographic signs (ascites, bilateral renal dysplasia, and a thick urinary bladder wall) are key features that suggest prune belly syndrome. 17 In the current study, the primigravida female having a fetus with prune belly syndrome was examined, but she decided to continue the pregnancy. As earlier reported, the infant was delivered alive and at this time is two years old. As was hypothesized in this case, the urinary bladder was empty and was probably due to possible renal rupture. This would be suggested based on this case’s initial sonogram of the urinary bladder that demonstrated a severely distended, bilateral hydronephrosis and hydroureter. This finding was left uninterrupted, which may have resulted in the right kidney rupturing. This would explain the subsequent diminished effect of the insult and how the urinary pressure was minimized. The rupture of the kidney is also a favored result due to the sonographic haziness (low-level echoes) of the ascitic fluid and urine, likely due to blood in the ascitic fluid.
A case was reported with prune belly syndrome, having vesicocutaneous fistula at the subumbilical region. The fetus of a G1P0 female was diagnosed for prune belly syndrome during the 16th week of gestation. 5 The amniotic fluid began to reduce in the last trimester and was greatly diminished by 36 weeks. The fetus was delivered through cesarean section due to abdominal dystocia during prevaginal delivery. There was no family history of a congenital disease. The infant’s physical examination demonstrated abdominal distention, with thin wrinkled skin, and the kidneys and bladder were not palpable. An abdominal examination discovered a small opening in the skin below the umbilicus that allowed for urine outlet. 5 Additional physical findings were bilateral undescended testes and an empty wrinkled scrotum. The baby was diagnosed with prune belly syndrome, but the stress was neutralized with the subumbilical vesicocutaneous fistula. 5 An additional case of prune belly syndrome was reported that was diagnosed antenatally with sonography. Those diagnostic findings were bilateral hydronephrosis, megacystis, and oligohydramnios. 18 The baby was delivered with a cesarean section at 35 weeks of gestation. Postnatally, the infant was found to be unable to void and had a distended abdomen and undescended testes. As documented in the previous case, this infant also had a small opening below the umbilicus with urine, which discharged through it. A postoperative voiding cysourethrogram showed bilateral high-grade vesicoureteric reflux, with stenosis in the anterior part of the urethra as well as a dilated posterior portion of the urethra. 18 In an additional case of prune belly syndrome, the infant was delivered vaginally at full term but had a vasicocolonic fistula. 19 In the current case, both kidneys were initially hydronephrotic, but on serial sonograms, only the left kidney and its vasculature were appreciated, and the right kidney was not visualized. This indirectly reflects that the right kidney may have ruptured, with urinary tract pressure reduced (see Figures 5 and 14).
The effect of prenatal amniotic shunting was studied to determine if it improved the outcomes of infants with prune belly syndrome. A study of 45 patients, over an 18-year period, demonstrated an infant mortality rate from prune belly syndrome of 27%. 20 Prenatal vesicoamniotic shunting was seen to improve its outcomes to a great extent. 20 Two published theories regarding the development of prune belly syndrome were reviewed. 14 The first of these theories poses that a defect in the mesenchymal development is an underlying cause of prune belly syndrome. In the first trimester, a defect in the mesodermal lateral plate gives rise to the maldevelopment of the urinary system and fetal abdominal wall musculature. The second theory postulates that an antenatal fetal bladder outlet obstruction prevents the outflow of urine and ultimately results in megacystis, hydroureter, and hydronephrosis. Intra-abdominal pressure increases, which results in poor development of the fetal abdominal musculature. The case presented would support the second theory for the development of prune belly syndrome. Fetal bladder outlet obstruction, particularly in cases of a posterior urethral valve, blocked the urethra during fetal development. The urethral blockage prevented the flow of urine through the urinary tract. The reflux of urine and hydrostatic pressure caused the bladder to become large (megacystis). The enormous enlargement of the bladder gives rise to abdominal distention and in turn affects the development of the anterior abdominal wall muscle. The overdistended fetal bladder compresses the inguinal canal, which can cause obstruction and prevent the testes to descend into the scrotal sac (cryptorchidism). In some fetal cases, the excessive urinary pressure builds due to megacystis, resulting in intrauterine death. It is possible that a fetus might survive this hostile condition, but it likely will come at the expense of the kidneys and ureter, with bladder rupture due to the release of bladder outlet obstruction. This mechanism of prune belly syndrome development is summarized in Figure 16.

Flowchart of the prune belly syndrome development.
Conclusion
Prune belly syndrome could be caused by megacystis, due to a bladder outlet obstruction, in an otherwise normal karyotype fetus. Megacystis leads to hydroureter, hydronephrosis, and abdominal distention. Severe intraurinary pressure can rupture and/or release the obstruction within the urinary system. The persistent abdominal distention can give rise to the underdevelopment of anterior abdominal wall muscles and cryptorchidism.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
