Abstract
Hepatoblastoma (HB) is a common primary pediatric liver tumor, and its typical presentation is a palpable, nontender abdominal mass that is detected through sonographic imaging. The current treatment standard for HB includes chemotherapy followed by liver resection or transplantation. Throughout the management of HB, high-quality sonographic images inform diagnosis, treatment interventions, and detection of surgical treatment complications. It is important that sonographers be familiar with the presentation and complications of HB on imaging examinations. The authors review sonographic detection, evaluation of HB at presentation, and common complications after liver resection and transplantation.
Although the overall incidence of pediatric hepatoblastoma (HB) is low (about 1% of all pediatric cancers in the United States), it is the most common solid tumor of the liver in children. 1 The typical presentation of HB is a palpable, nontender abdominal mass, for which children are generally referred for sonography, followed by a biopsy of the mass. 2 Diagnosis and treatment of HB requires a multidisciplinary care team, which includes pediatric radiologists, pathologists, oncologists, and surgeons, among others. Sonography is often the initial imaging modality in a patient with a suspected abdominal mass, as well as computed tomography (CT), magnetic resonance imaging, and follow-up sonographic examinations. These imaging tests have crucial roles in diagnosis, localization, staging of tumors, and detecting surgical treatment complications. Liver resection is the preferred surgical treatment for HB, preceded by chemotherapy to decrease the tumor’s size. Liver transplantation recommended if tumor location or size prevents resection.
It is important that sonographers be familiar with the presentation of HB tumors on imaging, even if there is a relatively low incidence of these tumors. This case series demonstrates common HB presentations on sonography for the purposes of educating sonographers. We review the role of sonography in the diagnostic evaluation of pediatric HB tumors and in the detection of complications after liver resection or transplantation.
Case Series
We review cases from a pediatric quaternary care academic medical center in a large, urban area that performs pediatric liver transplantation. The hospital has subspecialty expertise in pediatric radiology, oncology, and liver transplantation and serves a regional and national patient population, with increasingly complex medical presentations.
Patients of the hospital who underwent sonography for suspected HB and were subsequently diagnosed were retrospectively reviewed. Illustrative educational sonographic examples of HB and postoperative complications were identified. Because sonography was a primary imaging modality, used for initial tumor detection, after surgical treatment, and after transplantation, selected imaging studies were reviewed. These studies highlighted the presentation of HB tumors at diagnosis and demonstrated the detection of surgical complications after liver resection or transplantation. Because this was an educational case series, for the purposes of medical education, it was deemed exempt from the requirement for approval by the hospital’s institutional review board.
Case 1
A 12-month-old male infant with a history of prematurity, elevated α-fetoprotein (AFP), and a right upper quadrant mass was found to have a right lobe hepatic mass, extending to the medial aspect of the left lobe. On sonography, HB masses often appear as solid, heterogeneous masses (Figure 1A). Color Doppler evaluation of the mass showed diffuse vascularity with a disorganized pattern and both arterial and venous waveforms (Figure 1B). The patient subsequently underwent CT and needle biopsy, which confirmed Pre-Treatment Extent of Disease (PRETEXT) stage III HB. The infant underwent chemotherapy and tumor resection that was complicated by positive margins. The patient received additional cycles of chemotherapy and ultimately underwent liver transplantation.
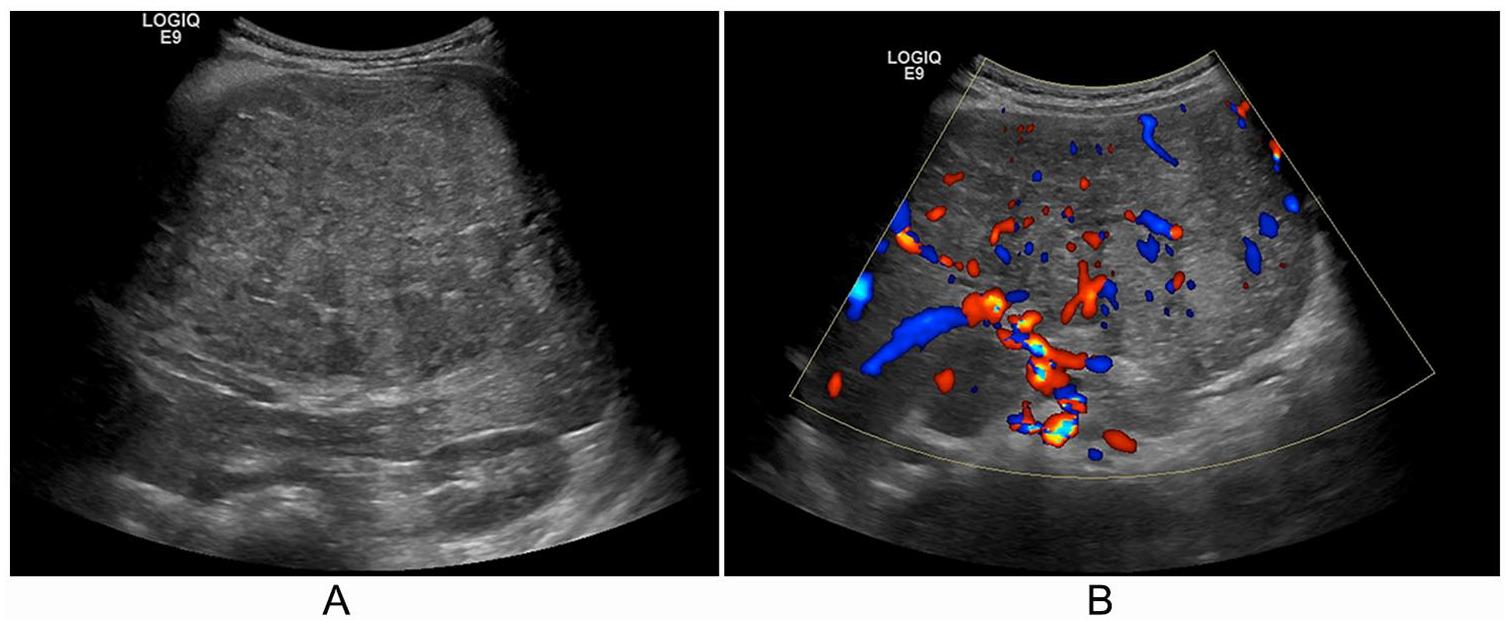
A 12-month-old infant with a history of prematurity, elevated α-fetoprotein, and a right upper quadrant mass, found to have a right lobe hepatic mass extending to the medial left lobe. (A) Sagittal right lobe with a solid heterogenous mass. (B) Color Doppler evaluation of the mass showing diffuse vascularity of the mass with disorganized pattern and the suggestion of both arterial and venous waveforms.
Case 2
A 3-year-old male child with PRETEXT stage III HB underwent chemotherapy followed by liver resection. On the first day after liver resection, sonography demonstrated a multiseptated fluid collection and adjacent bowel wall thickening (Figure 2A). Electronic calipers can be used to help identify and document the fluid collection size. The type and source of fluid collection needed to be identified to determine the appropriate intervention and to reduce the patient’s risk for hemodynamic instability. On day 7 after liver resection, oblique views of the fluid collection, on the inferior aspect of the liver (Figure 2B), were concerning for hematoma or biloma. Further evaluation with color Doppler showed an absence of blood flow to the complex fluid collection, in the inferior posterior aspect, of the remaining liver parenchyma. The patient was ultimately taken to the operating room and found to have an intra-abdominal bleed, confirming the suspected hematoma. The hematoma removal was well tolerated by the patient, and he was discharged from the hospital 1 month later.
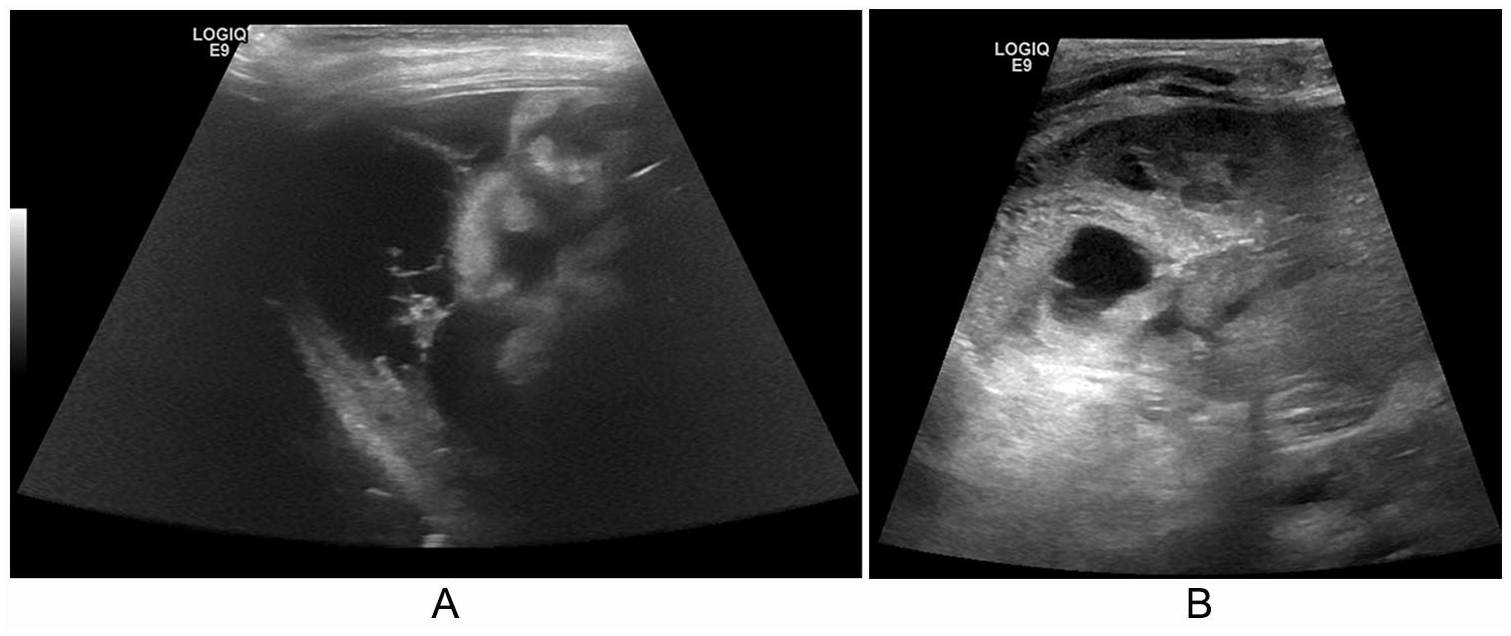
A 3-year-old patient with Pre-Treatment Extent of Disease stage III hepatoblastoma status post chemotherapy and liver resection. Day 1 postoperative trisegmentectomy. (A) Multiseptated fluid collection with adjacent loops of bowel. (B) Oblique view of a fluid collection at inferior posterior aspect of remaining liver concerning for hematoma or biloma.
Case 3
A 4-year-old male patient with refractory metastatic HB underwent routine CT to reevaluate disease status, which was concerning for a suspected right portal vein thrombus. The patient was initially diagnosed with stage IV HB with pulmonary metastases. The patient’s treatment course included chemotherapy, followed by partial liver resection and removal of pulmonary nodules. Figure 3 demonstrates a follow-up sonogram after routine CT, to visualize the portal vein. Initial sonographic images demonstrated color and flow in the main portal vein but suggested absence of flow in the right portal branch. In Figure 3A, echogenic material is seen filling the lumen of the right portal vein and a portion of the main portal vein, suggesting thrombus. The color Doppler image (Figure 3B) demonstrated echogenic material filling the lumen of the portal vein. The thrombus was treated with anticoagulants, but because of disease recurrence, the patient was started on chemotherapy.
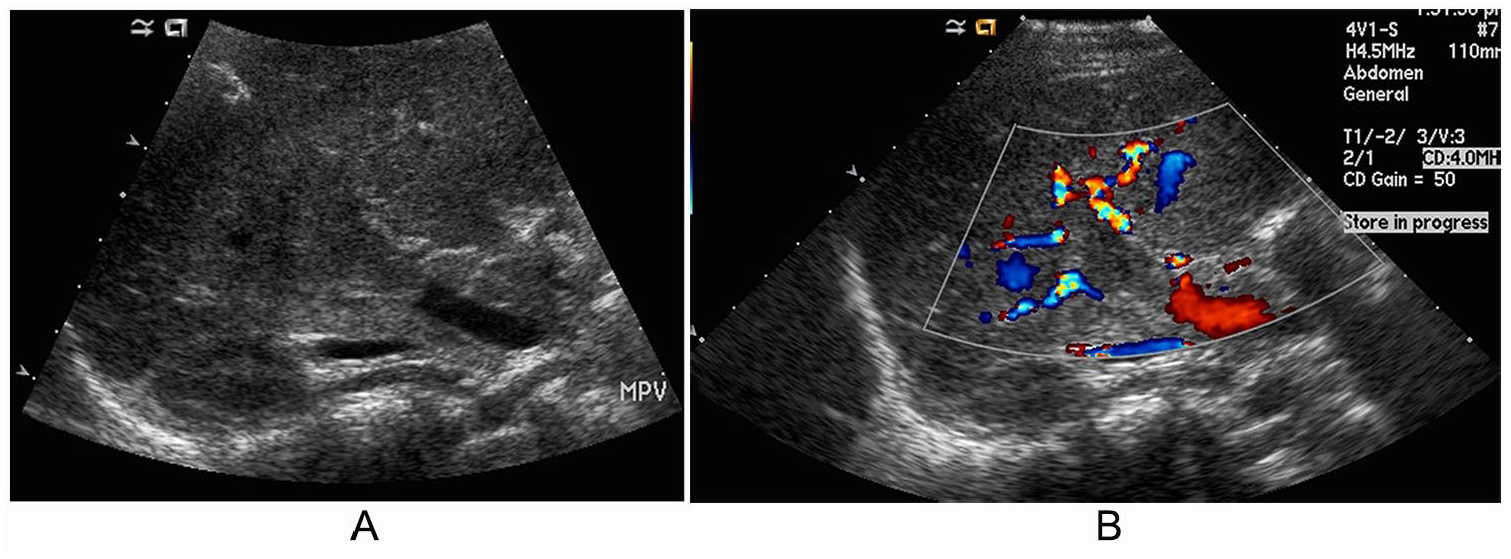
A 4-year-old patient with refractory metastatic hepatoblastoma status post chemotherapy and liver resection with a suspected right portal vein thrombus. (A) Echogenic material filling the lumen of the right portal vein and a portion of the main portal vein, suggesting thrombus. (B) Color Doppler filling around the echogenic material in the lumen of the portal vein.
Case 4
A 3-year-old female patient with PRETEXT stage IV HB and a history of prematurity and elevated AFP underwent neoadjuvant chemotherapy, followed by left lateral segment split liver transplantation. Sonography at 2 days postoperatively demonstrated no portal venous flow in the extrahepatic portion of the main portal vein, concerning for thrombus and/or occlusion. Figure 4A demonstrates an absence of flow in the portal vein with visualized flow in the adjacent vessel on color Doppler. The vessel adjacent to the portal vein showed a faint arterial waveform. A sonogram obtained on postoperative day 5 demonstrated potential areas of ischemia within the transplanted liver, another risk for graft failure. A sagittal view of the transplanted split liver in Figure 4B shows areas of heterogeneity, which are concerning for potential ischemia. On postoperative day 6, the patient returned to the operating room for portal thrombectomy, after which there was redemonstration of portal venous flow. The patient did not experience any further complications in the postoperative course.
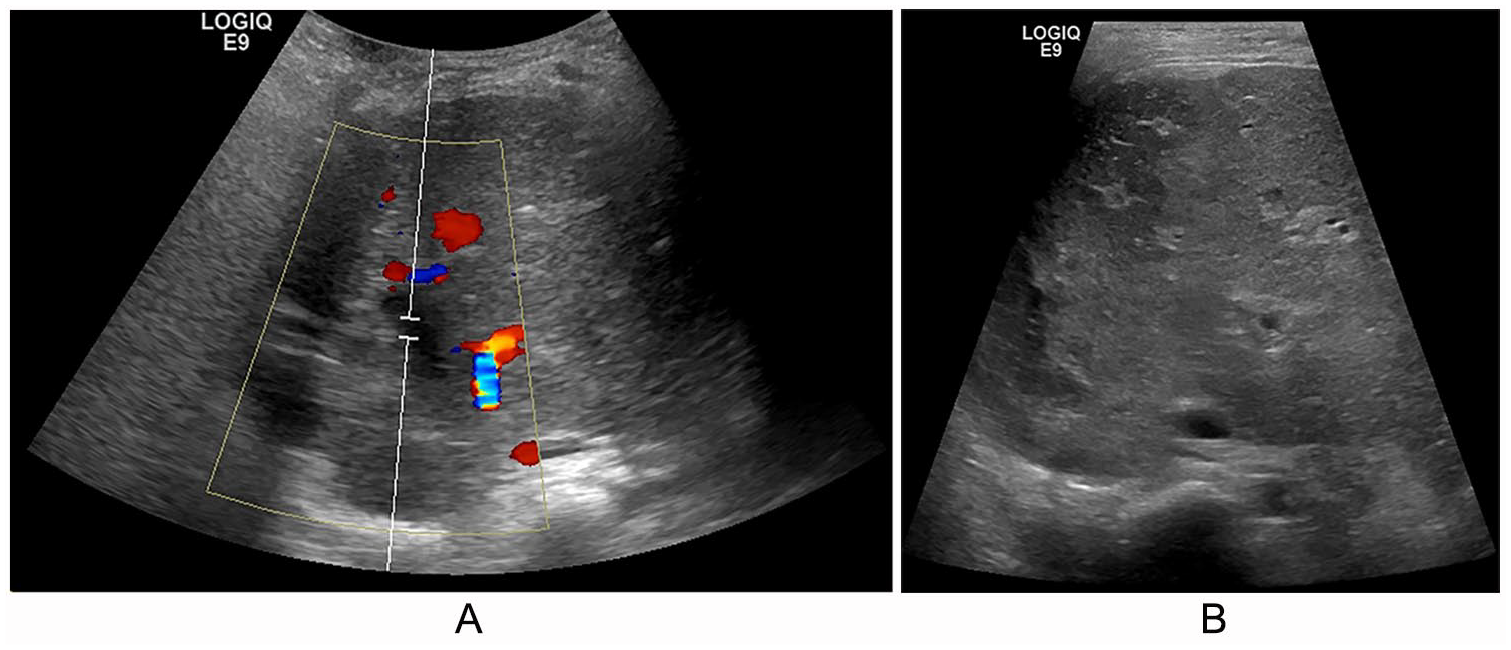
A 3-year-old patient with Pre-Treatment Extent of Disease stage IV hepatoblastoma and a history of prematurity and elevated α-fetoprotein, status post left lateral segment split liver transplantation, before and after thrombectomy of portal vein. (A) Color and pulsed-wave Doppler evaluation of the portal vein 2 days postoperatively from left lateral segment split liver transplantation showing absence of flow in the portal vein with visualized flow in the adjacent vessel. (B) Sagittal view of the transplanted split liver with areas of heterogeneity concerning for potential ischemia.
Case 5
A 3-year-old male patient had originally been diagnosed with PRETEXT stage IV HB and underwent chemotherapy followed by liver resection. This was complicated by a portal vein thrombosis and adhesions. Ten months after resection, the patient experienced multiple disease relapses in the remnant liver and underwent adjuvant chemotherapy before liver transplantation. On day 1 after liver transplantation, color Doppler (Figure 5A) demonstrated hepatofugal flow in the portal vein, despite appropriate direction of flow and pulsatility in the hepatic artery. When the patient returned to the operating room the same day, a partial stenosis of the portal vein was confirmed and remedied.
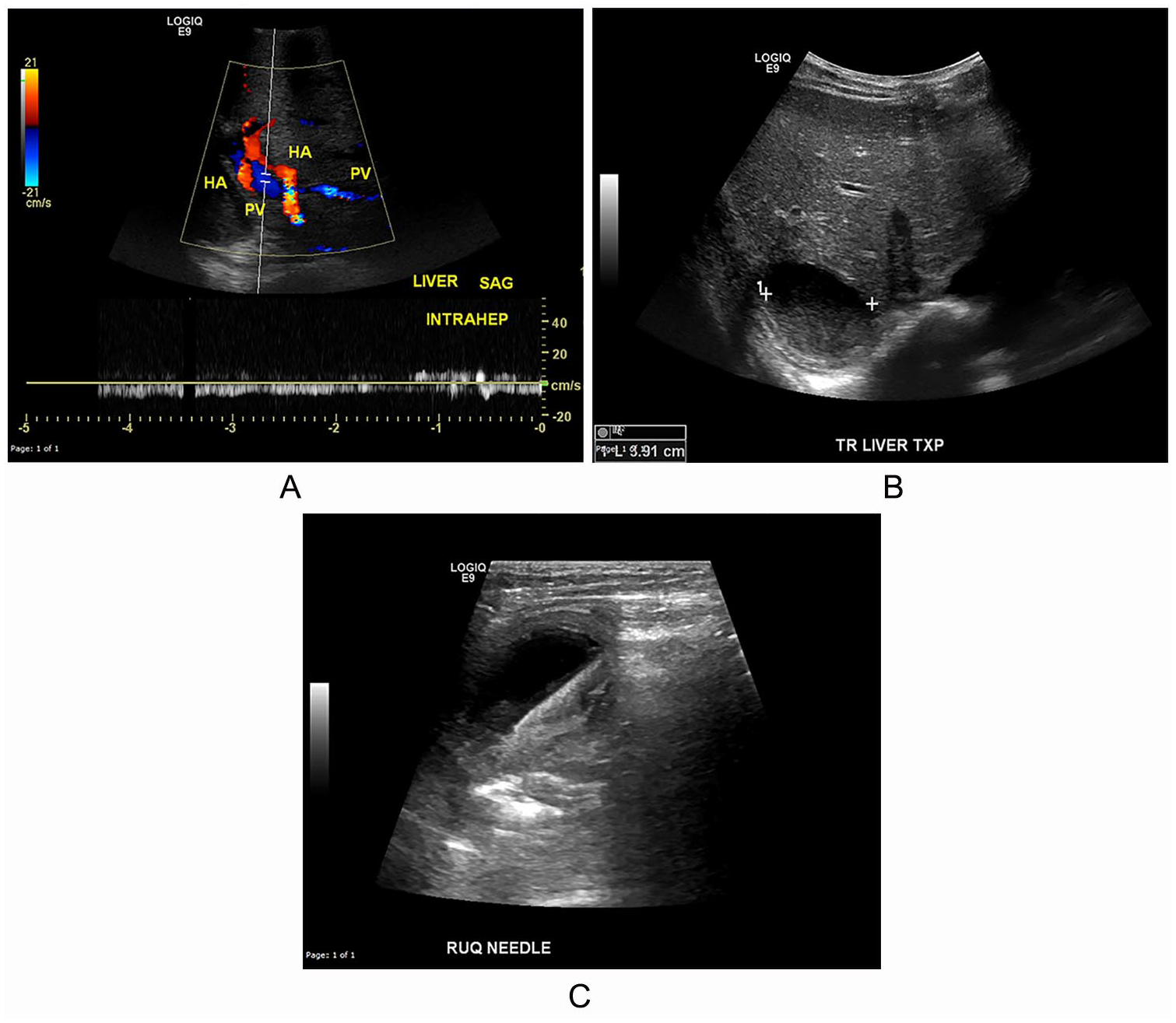
A 3-year-old patient with Pre-Treatment Extent of Disease stage IV hepatoblastoma status post chemotherapy, resection, and liver transplantation. (A) Pulsed-wave Doppler of the intrahepatic portal vein (PV) documenting hepatofugal flow. HA, hepatic artery. (B) Transverse image of the anechoic circular structure in the posterior liver with some internal low-level echoes, with calipers to measure the size of the structure. (C) Obtained during drainage of the intrahepatic abscess, this image displays the abscess with the needle in the center.
Three months after transplantation, the patient presented to the hospital with fevers of unknown origin and elevated liver function test results. The patient was referred for sonography, which revealed an intrahepatic abscess. Sonography showed a circular anechoic structure in the posterior portion of the transplanted liver. A transverse image of the anechoic circular structure (Figure 5B) includes caliper measurements of the structure. The patient went to the operating room for excision of the intrahepatic abscess and drainage of the intraperitoneal fluid collection (Figure 5C).
Case 6
An 8-month-old female infant with a history of prematurity and PRETEXT stage III unresectable HB underwent chemotherapy at an outside hospital. She was referred to the hospital for surgical evaluation. Sonography performed as part of her surgical evaluation identified a calcified mass on her liver (Figure 6A). The localization of the mass prevented liver resection, and the patient was placed on the transplantation list. Five months later, the patient underwent liver transplantation. On postoperative day 7, the patient experienced multiple episodes of bilious emesis and was referred for sonography. The sonographic study demonstrated multiple thickened bowel loops (Figure 6B) that were concerning for a small bowel obstruction. Follow-up CT confirmed a distal partial small bowel obstruction. The bowel obstruction was managed nonoperatively but failed to resolve. The patient returned to the operating room on postoperative day 13 and underwent lysis of an adhesion at the distal ileum.
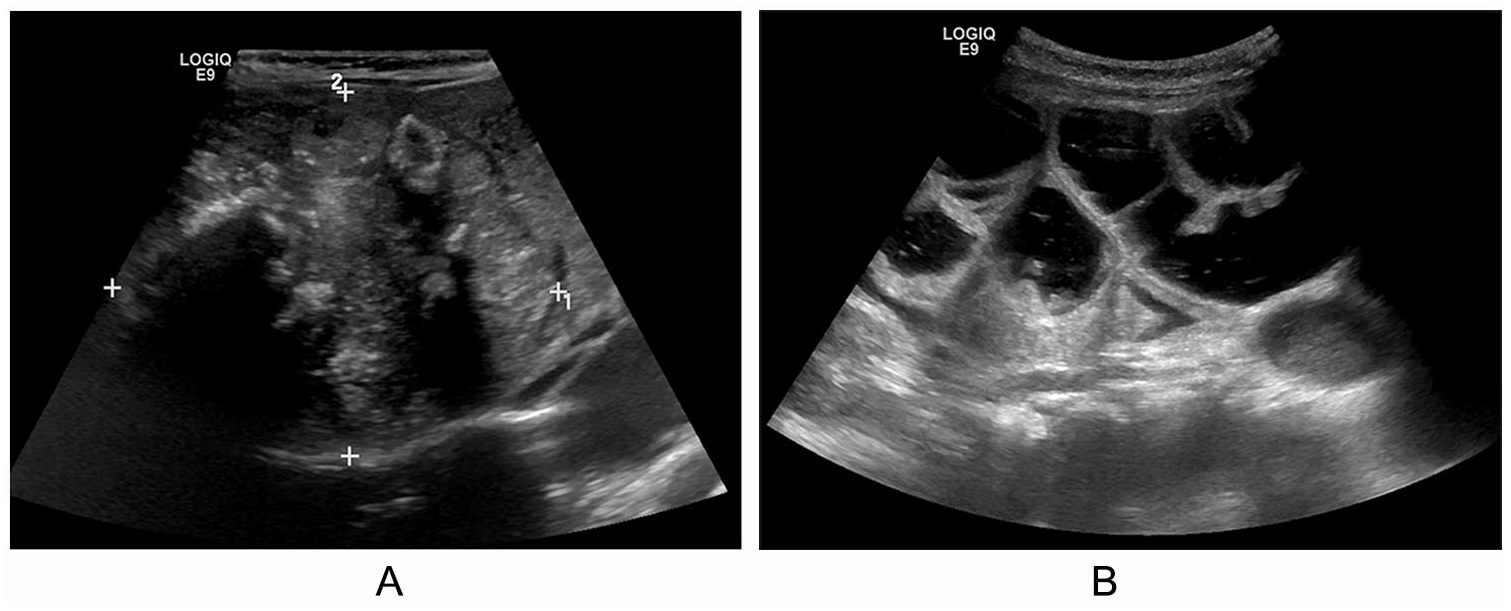
An 8-month-old infant with Pre-Treatment Extent of Disease stage III hepatoblastoma, before and after liver transplantation, with subsequent bowel obstruction. (A) Transverse image of the heterogeneous mass occupying the majority of the right lobe of the liver with minimal liver parenchyma appreciated medially to the mass. (B) Sagittal image displaying multiple large dilated thick-walled fluid-filled bowel loops in the left abdomen.
Discussion
This case series depicts an array of tumor presentations and surgical complications diagnosed in children with HB. Chemotherapy is the most common treatment for HB, followed by liver resection or transplantation depending on tumor size, location, and chemotherapy response.
HB is usually suspected after detecting a liver mass in children, as it is the most common pediatric primary liver tumor. 1 At initial detection on sonography, HB often presents as a singular solid, heterogeneous mass such as that illustrated in Figure 1. Color Doppler evaluation can show vascularity of the mass with both arterial and venous waveforms. Initial sonographic imaging should identify the tumor size and location. Elevated serum levels of the tumor marker AFP, prematurity, very low birth weight, and a familial history of adenomatous polyposis are all associated with HB.1,3 Three patients in this case series had histories of prematurity, and 2 of those patients also had histories of elevated AFP markers.
Throughout the management of HB, high-quality radiologic images inform the treatment course. The PRETEXT and Post-Treatment Extent of Disease staging system allows pediatric radiologists to assist in locating the extent of tumor involvement in the liver and surrounding organs before and after chemotherapy administration. 3 PRETEXT staging identifies the number of continuous liver sections that must be removed to excise the tumor completely4,5 and is summarized in Table 1. Staging has important treatment implications, as previous studies have found an association between initial response to chemotherapy and long-term risk for relapse.6,7 After diagnosis, staging is usually made with CT, tumor surveillance, and magnetic resonance imaging because of the sensitivity of these imaging modalities.8,9 Collaboratively, pediatric oncologists and surgeons determine a plan for neoadjuvant chemotherapy and liver tumor resection on the basis of the staging information and imaging examination results. 1
The PRETEXT Staging System. a

Abbreviation: PRETEXT, Pre-Treatment Extent of Disease.
Over the past 2 decades, long-term (5-year) overall survival of patients with HB has improved to <80%, which may be due to the increased use of neoadjuvant chemotherapy to reduce tumor size prior to resection.10,11 Tumor resection continues to be the preferred standard of care. 12 For unresectable tumors, often because of tumor location, vascular involvement, or poor chemotherapy response, liver transplantation is recommended, which has demonstrated good long-term survival. 13 Liver transplantation is also indicated after failed liver resection. 11 However, there is a risk for surgical complications after liver resection and transplantation. Therefore, frequent sonographic examinations are performed to monitor for any complications. The postoperative complication rate for patients after liver transplantation for HB has been reported to be as high as 20%. 14 After tumor resection or liver transplantation, sonography is a primary imaging modality for the detection of postoperative complications. This is because sonography is noninvasive and can easily be performed intraoperatively, at the bedside, or as needed.
Surgical complications after liver resection or transplantation for HB have been well documented.14–17 Common complications include biloma, biliary stricture, abdominal abscesses, fluid collection, intestinal obstruction, cholestasis, bleeding, and liver failure.14,17 Patients described in this case series also experienced intra-abdominal bleeding (Figure 2) and bowel obstruction (Figure 6). Many of these complications are associated with subsequent interventional and surgical procedures to drain fluid or lyse adhesions. 14 Liver transplantation for HB has also been found to confer an increased risk for hepatic artery thrombosis complications compared with transplantation for nonmalignant diagnoses.15,16 Furthermore, thrombus of the hepatic artery, portal vein, or vessels has been found to be associated with greater risk for graft failure, which could necessitate retransplantation.11,15,16 This case series illustrates thrombus in a patient after liver resection (Figure 3) and in another patient after liver transplantation (Figure 4).
Sonographers must be knowledgeable about surgical complications that may present on sonography. As demonstrated in the first patient (Figure 5), hepatofugal flow in the portal vein, despite appropriate direction of flow and pulsatility in the hepatic artery, can suggest of a portal vein thrombus. Inadequate flow in the portal vein can increase the risk for graft failure and contribute to a need for retransplantation, requiring immediate intervention. In this case series, a liver abscess and subsequent fluid draining are illustrated in Figure 5. Electronic calipers can help identify and document the size of fluid collections or abscesses. As previously noted, fluid collections often require repeat interventions or operations for drainage.14,18
Common postresection and post-transplantation complications reviewed in this case series are fluid collections, thrombosis of the portal vein and/or hepatic artery, bowel obstruction, and ischemia. Many of these complications delayed chemotherapy cycles or required surgical intervention. As the aim of this case series was medical education, it does not include statistics on overall complication rates or long-term survival for liver transplantation at this center. Frequent sonographic examinations in the postoperative period have been recommended for early detection and intervention for surgical complications.14,16 As sonography is often used as the primary initial imaging modality after surgery, it is valuable for sonographers to be aware of these important surgical complications.
Conclusion
The diagnosis of HB and recognition of surgical complications after liver resection and transplantation are essential for the care of patients with HB. Although the incidence of HB is low, it is important that medical sonographers be educated about its presentation on imaging examinations, as sonography is often the initial imaging modality used for tumor detection. We have reviewed an array of common surgical complications after liver resection and transplantation for which sonography was used as the imaging modality of choice.
Footnotes
Acknowledgements
We would like to acknowledge Brian Schott for his assistance with this project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
