Abstract
Transverse testicular ectopia (TTE) is an extremely rare congenital anomaly of the testis, characterized by migration of one testis toward the contrary hemiscrotum. TTE is usually associated with other testicular abnormalities such as persistent Mullerian duct syndrome, hypospadias, true hermaphroditism, and scrotal anomalies. Testicular sonography is the main initial diagnostic modality, followed by magnetic resonance imaging of the scrotum. These imaging modalities are important for determination of TTE and characterization of the testicular lesions. This case report provides the high-resolution ultrasonography, Doppler ultrasonography, magnetic resonance imaging, and diffusion-weighted imaging findings of a young man with seminoma in the transverse testicular ectopia. In addition, data on tumor stiffness, obtained with point shear-wave sonoelastography, are presented.
Key Takeaways
Transverse testicular ectopia is a rare cause of the unilateral scrotal swelling.
On sonography, both testes are found in the same hemiscrotum in patients with transverse testicular ectopia (TTE).
Seminoma and nonseminomatous germ cell tumors can occur in patients with TTE.
Transverse testicular ectopia (TTE), or crossed testicular ectopia, is a rare congenital anomaly in which both testes fuse in the same hemiscrotum and usually fail to descend properly during development. 1 TTE occurs in approximately one-quarter million men worldwide. Seminomas are the most common malignant tumors of the testis. 2 However, there have been few reports of imaging findings of seminoma originating in the TTE, and existing reports have not fully described the imaging findings. 3 This is a case report of high-resolution scrotal ultrasonography (US), Doppler sonography, magnetic resonance imaging (MRI), and diffusion-weighted imaging (DWI) findings of the seminoma in a patient with TTE. A selected review of the literature is provided.
Case Report
A 37-year-old man presented to a hospital with a long-term right-sided scrotal mass. According to the patient, this swelling of the right hemiscrotum had been present since childhood and stable over time. He had no weight loss, fevers, or other symptoms. On physical examination, a large, well-defined, soft tissue mass in the right hemiscrotum was present. The left testis could not be palpated in the left hemiscrotum or left inguinal canal. There was no pain on mobilization of the mass during physical examination. Blood tests were normal and biochemistry tumor markers were within normal limits. High-resolution scrotal US revealed an ectopic left testis in the right hemiscrotum. There was a 10-mm × 12-mm mass that was uniformly hypoechoic, was well defined, and had a smooth margin within the ectopic testis (Figure 1a). A few microcalcification foci were also seen in both testes, and there was a small hydrocele in the left hemiscrotum. Color Doppler US showed weak vascularity in the mass (Figure 1b). Point shear-wave elastography (pSWE) was performed with a 12- to 8-MHz linear probe (Acuson S3000; Siemens, Erlangen, Germany). The median pSWE value was 2.3 m/s (range, 1.9–2.8 m/s) of the testis mass (Figure 1c), which was much higher than for normal testis parenchyma (0.56–0.73 m/s).
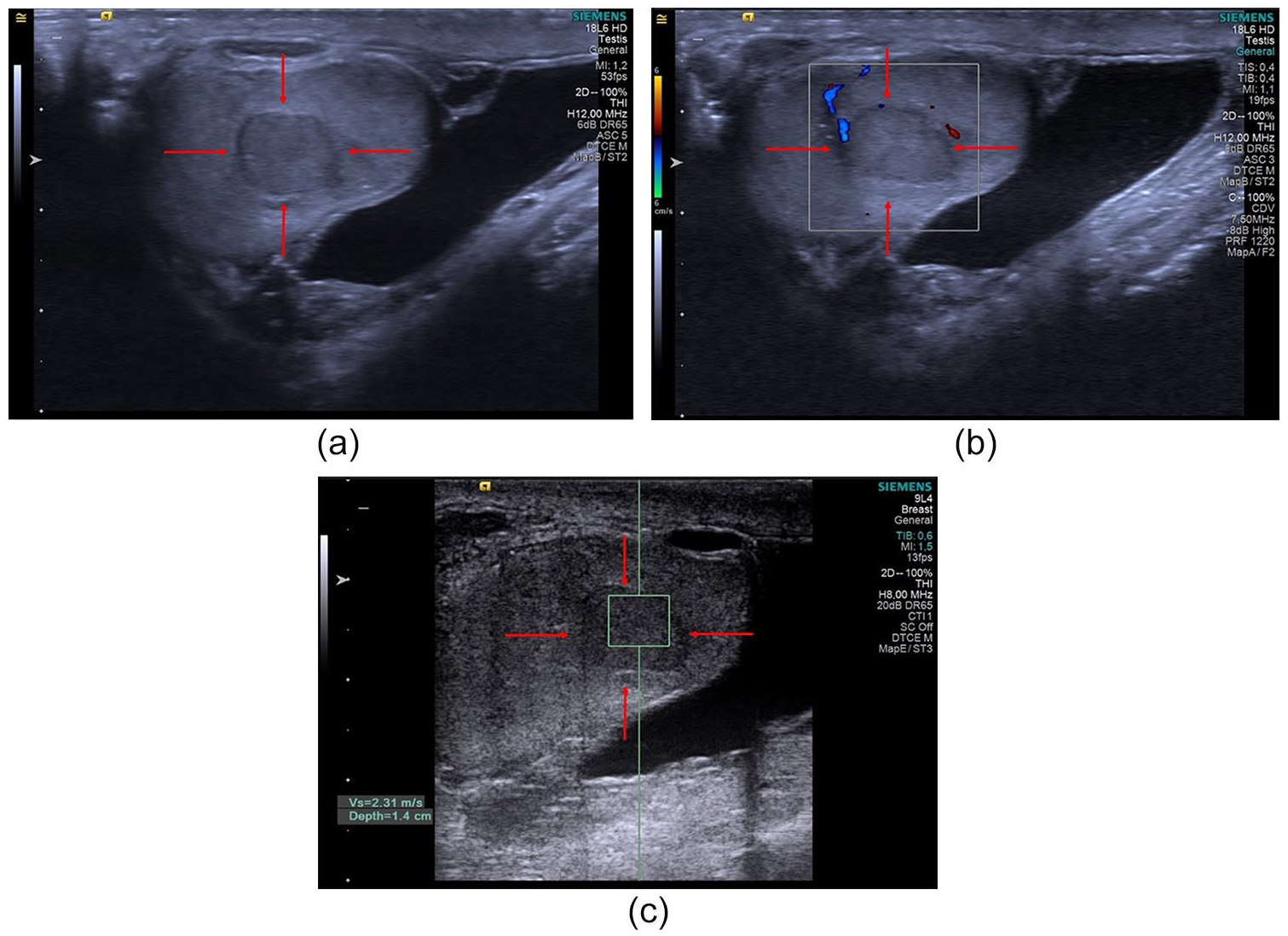
High-resolution ultrasonography (US) of the scrotal mass in the testis. (a) An ovoid, 10-mm × 12-mm homogeneous, hypoechoic, well-bordered lesion is present in the transverse ectopic testis (red arrow). Color Doppler shows mild vascularity in the testicular mass (red arrows) (b). Point shear-wave elastography demonstrates increased stiffness of the testicular mass (red arrows) (c).
After three days of sonography, the patient underwent a scrotal MRI examination with intravenous administration of gadolinium chelates. Axial and sagittal turbo spin-echo (TSE) T2-weighted imaging (WI), axial TSE T1-WI, DWI (b-values of 0, 50, and 1000), and 3D T1-weighted fat-suppressed gradient-echo axial and sagittal sequences were performed before and after contrast media administration.
The MRI revealed a 1-cm solid lesion between the ectopic left testis and the compressed and medially situated right testis in the right hemiscrotum. The lesion had a well-defined capsule that was mildly hypointense on T2-WI, slightly hyperintense on T1-WI, and slightly hyperintense on DWI (Figure 2a–e). Prominent signal loss was apparent on the apparent diffusion coefficient (ADC) map (Figure 2f), which had values in the range of 0.85 × 10−3 mm2/s. The lesion showed significant enhancement on postcontrast T1-WI (Figure 2d). The right spermatic cord showed diffuse wall thickening (Figure 2b), and the right seminal vesicle was tortoise and dilated. The left seminal vesicle and the left spermatic cord were not in their normal positions, but the bladder and the prostate appeared normal (Figure 2b). Clinically, the lesion was suspected as a germ cell tumor within the TTE.
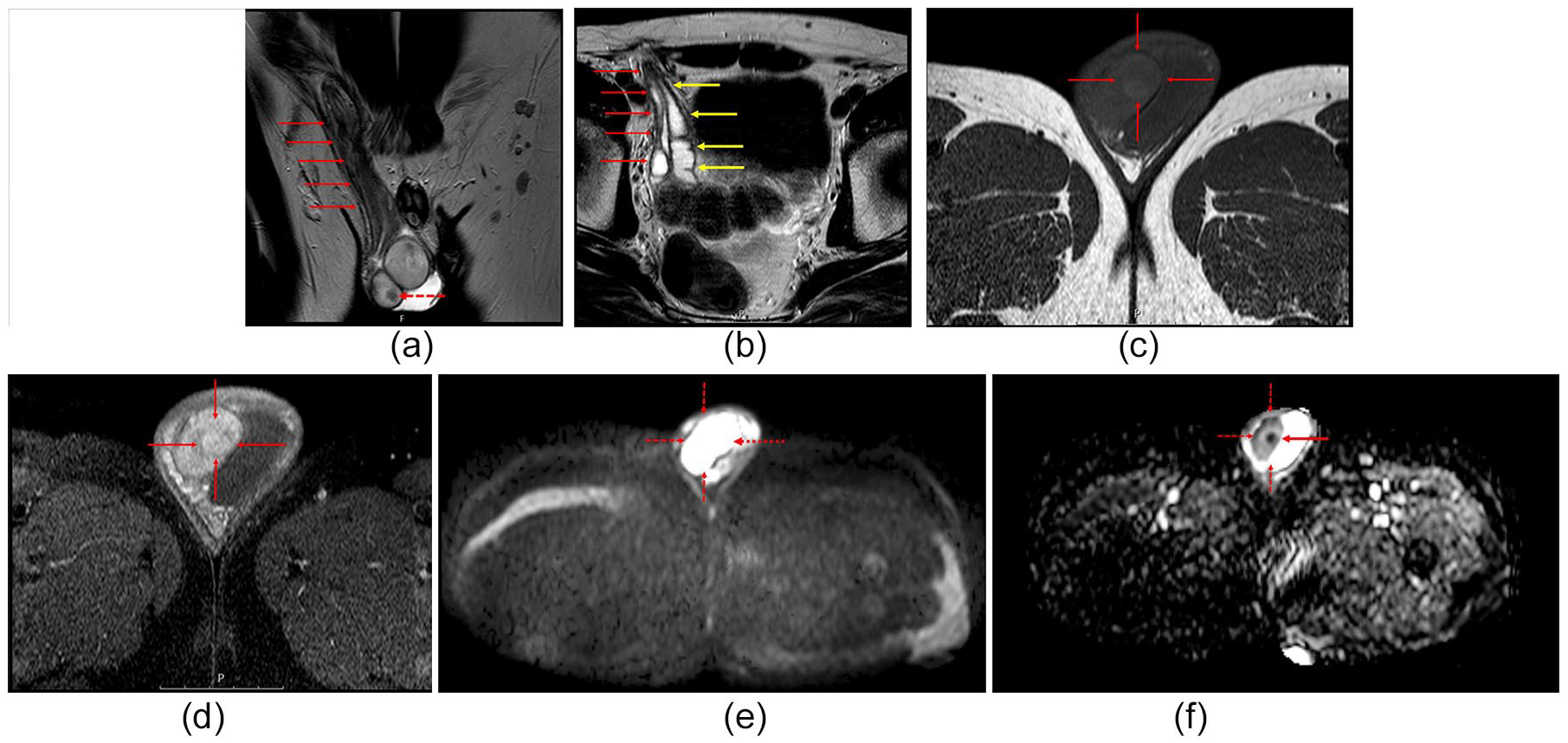
Contrast-enhanced magnetic resonance imaging (MRI) of the scrotum. Coronal turbo spin-echo (TSE) T2-weighted (a) and axial TSE T2-weighted images (b) revealed a well-defined, round lesion with a peripherally hypointense halo located in the transverse ectopic testis (stippled red arrow). The right spermatic cord was thick and contained both dilated vas deferens (red and yellow arrows) (a, b). The peripheral halo was hyperintense on T1-weighted imaging (WI) (c) and hypointense on T2-WI (red arrows). The axial contrast-enhanced fat-saturated T1-WI shows heterogeneous and significant enhancement of the lesion (red arrows) (d). The axial high b-value diffusion-weighted imaging (DWI) trace image shows hyperintensity of the lesion (red arrows) (e). Diffusion restriction is noted in the mass on the apparent diffusion coefficient (ADC) map (red arrows) (f).
The patient immediately underwent complete surgical excision of the mass via a left orchiectomy. Histopathological examination revealed a seminoma. The postoperative period was uneventful, and the patient was free from symptoms at the 19-month follow-up.
Discussion
Seminomas are the most common testicular neoplasms arising from the germ cells of the testis, and they account for 56% of all germ cell tumors of the testis. They may be associated with testicular dysgenesis, microlithiasis, and undescended testis. 4
TTE is a rare congenital condition that can affect the superficial inguinal pouch; suprapubic, femoral, and perineal areas; and the base of the penis. 5 Since the first case was first described in 1886 by von Lenhossék, fewer than 100 additional cases that have had radiologic imaging have been reported in the English literature. 5 The etiology of TTE remains unknown, although a failure of synthesis or release of Mullerian inhibitory factor (MIF) is known to be involved. 5 MIF, also known as the anti-Mullerian hormone, exhibits critical status with testosterone in genital differentiation in male fetuses. In TTE, the testicles descend toward the same hemiscrotum.
Patients with TTE have a high risk of testicular malignancies; an incidence rate of malignant transformation of 18% was reported in such patients. 6 There have been reports of embryonal carcinoma, seminoma, yolk sac tumor, and teratoma in TTE patients. 6 Clinical and radiologic findings in TTE depend on tumor size, but small testicular masses are usually asymptomatic. These tumors can be detected on physical examinations and/or scrotal US as an incidental testicular mass.
Gkekas et al. 7 have reported a classification system of TTE. These classifications are as follows: type I, associated with inguinal hernia; type II, accompanied by Mullerian duct remnants; and type III, associated with other genitourinary anomalies other than Mullerian duct remnants. In the case presented, type II TTE and testicular swelling were clinically nonspecific. According to the literature, Mullerian duct remnants and other genitourinary anomalies must be excluded in a patient with TTE after surgical orchiopexy as well as long-term follow-up. 7
The differential diagnosis of TTE includes inguinal hernias, hydrocele, testicular torsion, and polyorchidism.2,6 Polyorchidism clinically can be confused with TTE. Polyorchidism is a congenital and benign condition of the scrotum. On US, polyorchidism is similar in echotexture to the adjacent testis and lacks a mass effect. 2 Testicular torsion is an acute and painful condition and seen usually in the adolescent. The definitive diagnosis of the testicular torsion can be made with color Doppler. A hydrocele is also a well-known entity of the scrotum and is a consistent finding in cases of scrotal swelling, without pain. 7 US can reveal excess fluid in the hemiscrotum coexistent with a normal testis. Inguinal hernia is the most common cause of groin swelling in the population. Inguinal hernias clinically present with a sudden onset of groin pain and swelling. The history of groin swelling is not expected in patients with inguinal hernias. The diagnosis can be made easily with US.
TTE is a complex congenital disorder. US is generally the preferred modality used to diagnose scrotal pathologies due to it being noninvasive, fast, and easily demonstrating scrotal anatomy. 3 On US, both testes can be found in the same hemiscrotum in patients with TTE. In the patient presented, a complex hypoechoic mass was also observed on the level of the right spermatic cord due to the double spermatic cord. MRI confirmed a TTE on the right side of this patient. On the right side, the coronal T2-weighted images detected two separated spermatic cords. In the pelvic region, the left seminal vesicle could not be visualized clearly on MRI. The kidneys might appear in the normal abdominal position in patients with TTE.
Testicular tumors in patients with TTE usually arise from the ectopic testis. 7 In this patient, the testicular lesion originated in the ectopic testis and was sonographically well defined and hypoechoic; Doppler also demonstrated increased vascularity. Sonoelastography of the tumor suggested a hard, solid lesion indicative of malignancy. MRI revealed a solid and well-defined mass that was slightly hyperintense on T2-WI and showed prominent heterogeneous enhancement on postcontrast T1-WI. The differential diagnosis included germ cell tumors of the testis, fibrous pseudotumor of the testis, and sex cord stromal tumors.4,7 Usually, hypercellular malignant tumors show limited diffusion on DWI. The addition of DWI to conventional MRI sequences has been shown to be helpful in differentiating malignant urogenital tumors from benign neoplasms. In the present case, there was a prominent diffusion restriction on DWI. Based on the US, Doppler, sonoelastography, and MRI findings, it was concluded that the solid malignant tumor originated from TTE. According this literature search, there were only a limited number of cases with TTE. The present case is quite different from published cases with high-resolution radiologic images of the seminoma for a patient with TTE.
The sonographic appearance of seminoma is often characterized by well-defined, lobulated margins that are hypoechoic and homogeneous in the testis. 4 Seminomas usually contain structurally thin or thick fibrous septa. The cystic component and intratumoral calcification are rare in seminomas. Color Doppler can be used to detect the internal vascularity of the seminoma. Two-dimensional shear-wave sonoelastography revealed stiffness in the seminoma due to its cellularity and fibrous content. MRI with contrast is also useful in the characterization of testicular mass. A seminoma usually demonstrates strong enhancement and restricted diffusion on MRI.
The standard treatment method for testicular tumors is surgery. Radical inguinal orchiectomy is the standard global treatment method for a testis tumor. Testicle-sparing surgery also can be used for small testis tumors. 5 In the current patient, a standard surgical procedure was used because he also had complex testis anatomy due to TTE.
Conclusion
Although rare, TTE should be considered when examining a congenital unilateral testicular mass. Seminoma and other germ cell testicular malignancies can be present in TTE. It would appear that this is the first report of a seminoma in a patient with TTE and for which US, Doppler, sonoelastography, and MRI findings are described.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
