Abstract
A luteoma is a benign ovarian mass that can have malignant characteristics on sonography. Pelvic sonography is used to help distinguish benign from malignant masses. The workup of adnexal masses during pregnancy may involve the use of both transabdominal and transvaginal sonography as well as Doppler imaging, magnetic resonance imaging, and serum tumor markers to identify the risk of malignancy. This study presents a case of a benign ovarian luteoma of pregnancy detected during sonography. The mass was removed during the first trimester due to malignant sonographic features. Counseling and treatment regarding ovarian masses should consist of a multidisciplinary team made up of physicians in maternal-fetal medicine, radiology, neonatology, and sonographers.
Timely management of ovarian masses during pregnancy is crucial for the health of the mother and the fetus. 1 Historically, pelvic examinations were the only method to detect adnexal masses during pregnancy. One major limitation of pelvic examinations is the decreased sensitivity in overweight and obese patients. 2 Over the years, pelvic examinations have been supplemented with transvaginal and abdominal sonography due to their high sensitivity and specificity for identifying adnexal masses as well as reliability for surgical planning. 3 The increased use of routine sonography in prenatal care has led to the increased detection of adnexal masses during pregnancy. 1 Two-thirds of adnexal masses found in pregnancy are found within the first two trimesters. 3 Due to the risk of rupture, torsion, and malignancy, accurate assessment of adnexal masses is essential. 2 Transabdominal and transvaginal sonography including color and pulse wave Doppler imaging, magnetic resonance imaging (MRI), and tumor markers all assist in the discrimination between benign and malignant masses. 4 Treatment plans are based upon the gestational age of the pregnancy, type of ovarian mass, and malignant components. 2
Pelvic sonography has been determined by the American College of Obstetrics & Gynecology (ACOG) as the first-line modality to evaluate intrapartum adnexae. 3 On sonography, ovarian malignancies tend to have solid components, thick septations, mural nodules, or papillary excrescences. 3 Blood flow to the mass can frequently be noted with Doppler, and free fluid in the pelvis can raise suspicion for metastatic disease. 1 These findings with sonography can be used to stratify masses from very low risk to very high risk. 3
Unfortunately, sonography does not allow for sufficient visualization to stratify risk in up to 20% of ovarian in masses. 3 MRI can assist in the diagnosis of ovarian tumors during pregnancy and is considered a second-line imaging modality. 3 MRI is a useful adjunct when sonography is inconclusive or when the patient’s body habitus complicates the study. 4 However, the gadolinium dye used in contrast MRIs has been shown to be associated with skeletal malformations in animal studies, which is why MRI is a second-line imaging technique during pregnancy. 3
The management of adnexal masses is primarily based on the morphology seen on a sonogram. 3 Masses with low risk are treated conservatively and observed throughout pregnancy. 3 Fifty to 70 percent of adnexal masses identified in pregnancy will resolve without future complications. 3 Surgery is often reserved for masses that are highly suspicious for malignancy on imaging or in symptomatic patients. 3 Since surgical resection of ovarian masses during pregnancy poses increased risks to the mother and the fetus, intrapartum surgical resection is controversial. 3 Surgical resection is usually recommended in cases of mass enlargement, rupture, torsion, hemorrhage, or high suspicion of malignancy. 2 Due to the risk of preterm labor or fetal demise, it is safer for the mass to be removed during the second trimester. 3 However, strong suspicion for malignancy may prompt earlier removal. 3
The clinical value of each pelvic sonogram depends heavily on the operator’s knowledge and expertise, especially when obtaining and analyzing images. 3 It is important to recognize the normal morphological changes of pregnancy to precisely identify masses suspicious for malignancy. 1 A case of an ovarian mass is presented with high suspicion of malignancy during a pregnancy. The ovarian mass was surgically removed during the first trimester with final pathology consistent with a benign ovarian luteoma. Comparison of the present case with the International Ovarian Tumor Analysis (IOTA) system for ovarian mass classification is provided in the discussion. Using the IOTA system can aid clinicians in the diagnosis and treatment of intrapartum adnexal masses. 5
Case Report
A 33-year-old G2P1 at approximately five weeks’ gestation presented to the emergency department with complaints of vaginal bleeding and abdominal cramping. A sonogram was performed that revealed a single, live intrauterine pregnancy at five weeks, two days of gestation, a left ovarian mass, and a moderate amount of free fluid. She then presented to the clinic for follow-up evaluation and obstetrical care. A sonogram was performed at seven weeks, four days with a GE Voluson E8, Morgantown, WV, USA. The examination was performed transvaginally with a RIC 5-9 D transducer, GE, Morgantown, WV, USA. The sonogram confirmed a viable intrauterine pregnancy (IUP) with fetal heart tones of 162 beats per minute and a crown-rump length consistent with seven weeks, three days (Figure 1). A large amount of pelvic ascites was noted extending into both adnexae (Figures 2 and 3). A mass containing both solid and cystic components was seen on the left ovary measuring 3.9 × 3.7 × 4.1 cm (Figure 4). Color and spectral Doppler confirmed blood flow surrounding and within the mass (Figure 5). The internal vascularity resistance was low, measuring 0.34 to 0.38 (Figure 6).
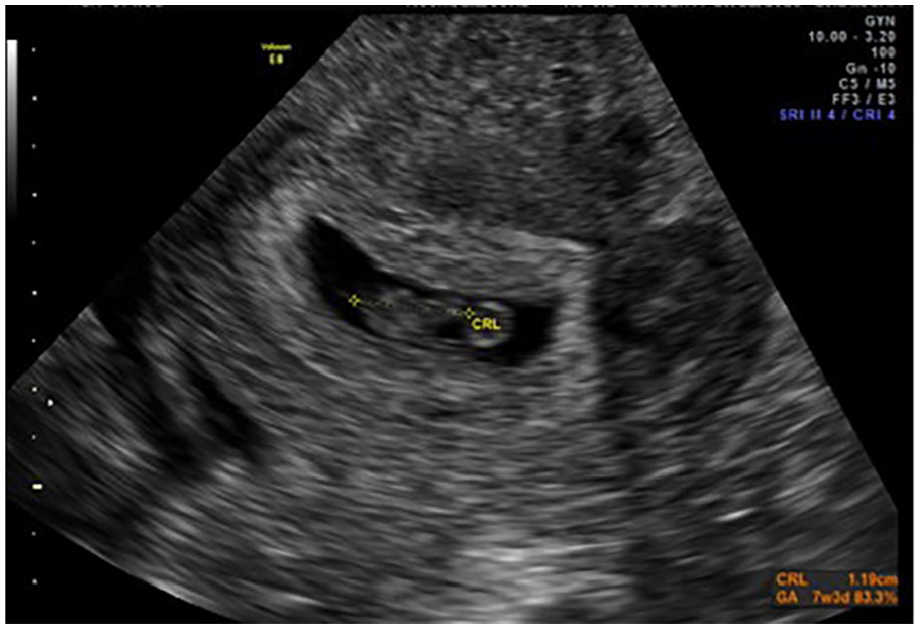
Transvaginal sonogram of the fetal pole with a crown-rump length of 1.19 cm, seven weeks, three days.
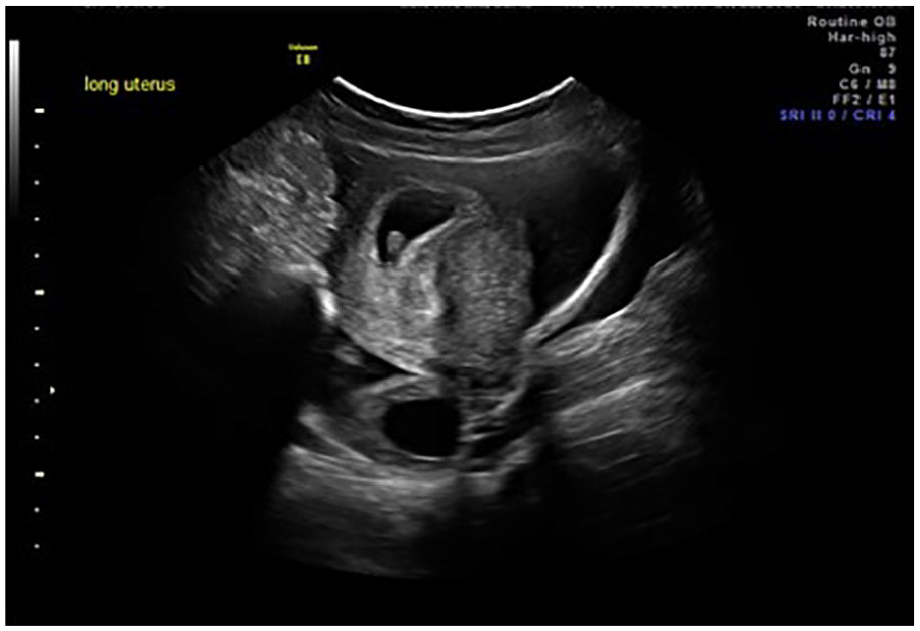
Transabdominal sonogram of the uterus and right ovary posteriorly. An intrauterine pregnancy is noted within the uterus. Pelvic ascites is also noted in the image.
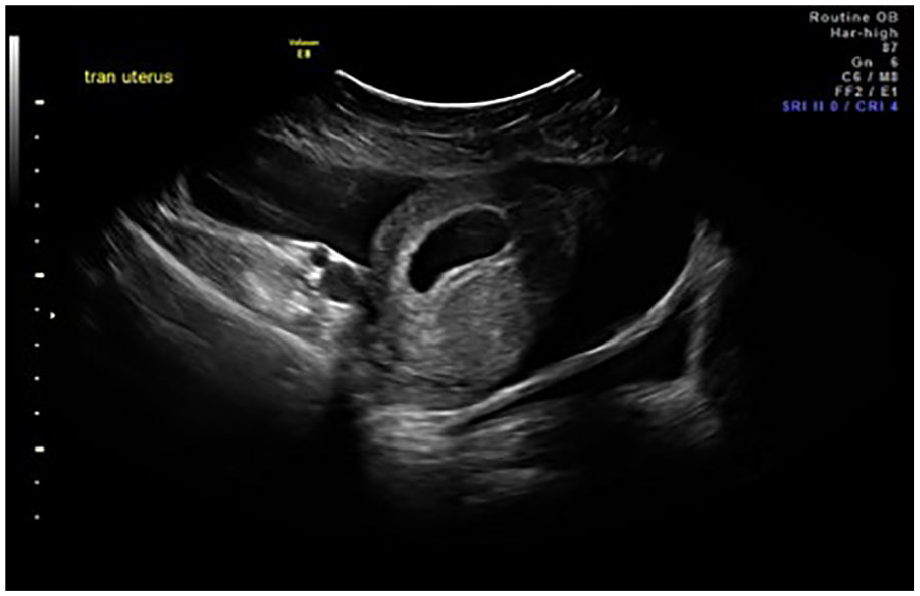
Transabdominal sonogram demonstrates an intrauterine pregnancy and extensive free fluid.
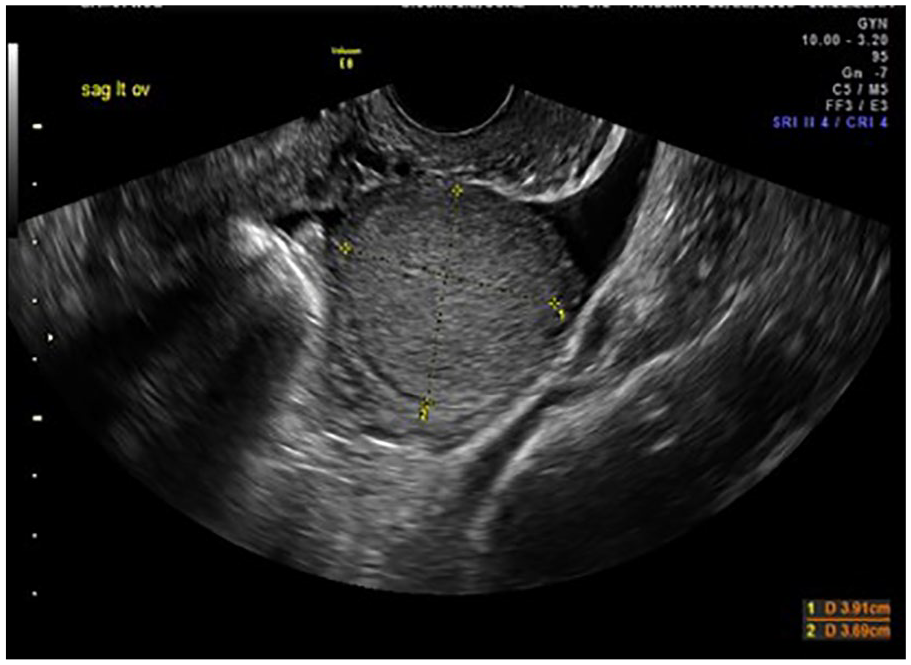
Transvaginal sonogram that depicts a solid left ovarian mass. The mass appears well circumscribed and homogeneous.
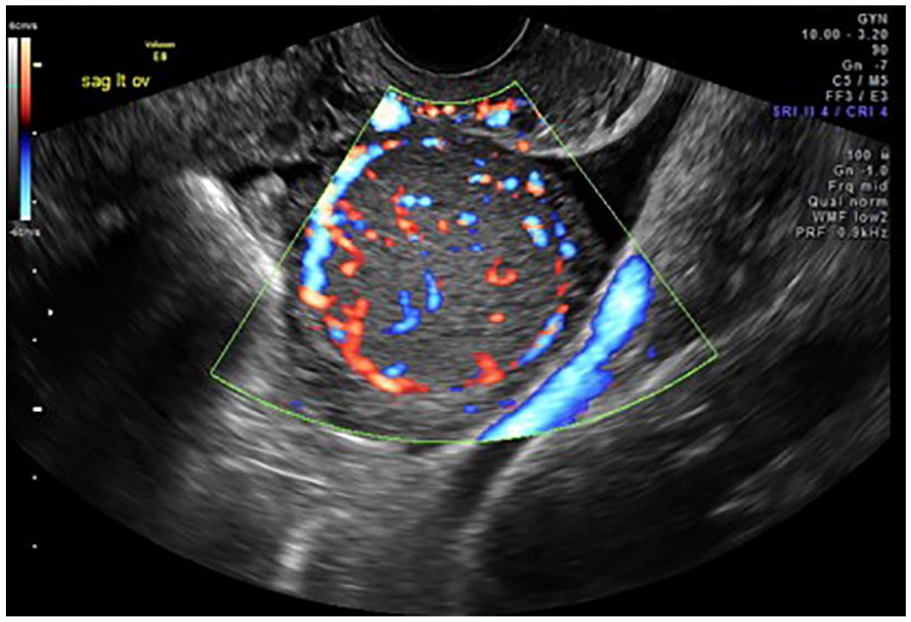
Transvaginal image of the internal blood flow of the mass. Vascularity is noted in and around the mass.
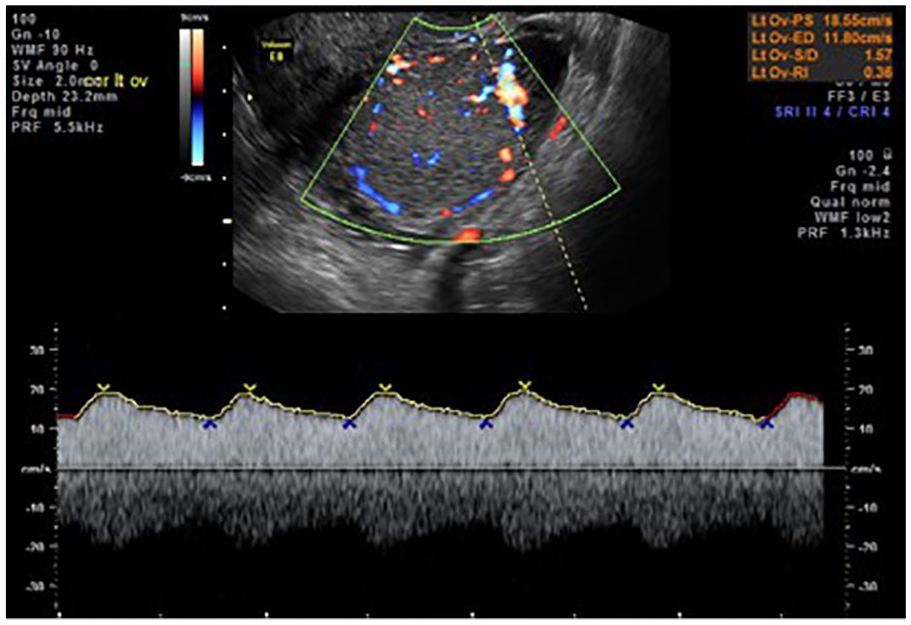
Doppler waveform of arterial flow within the mass that demonstrates low resistance with an Resistive Index (RI) of 0.36.
Due to concern for malignancy, an MRI of the abdomen and pelvis was obtained. It revealed a solid ovarian mass with associated free fluid in the pelvis. The left ovarian mass measured approximately 4.1 × 4.0 × 5.1 cm and had numerous T2 hypointense vascular flow voids along the periphery as well as within the mass (Figure 7). The mass was isointense on T1 images and had a homogeneous increase signaling on T2 images (Figure 8).
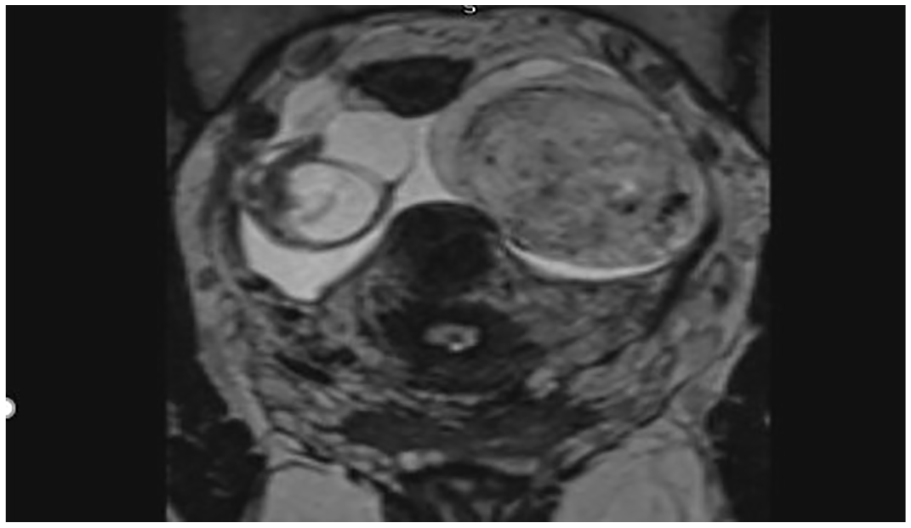
A T2 magnetic resonance image demonstrates a 4.1 × 4.0 × 5.1-cm solid mass within the left ovary. T2 hypointense vascular flow voids correlate to vascular flow on sonography.
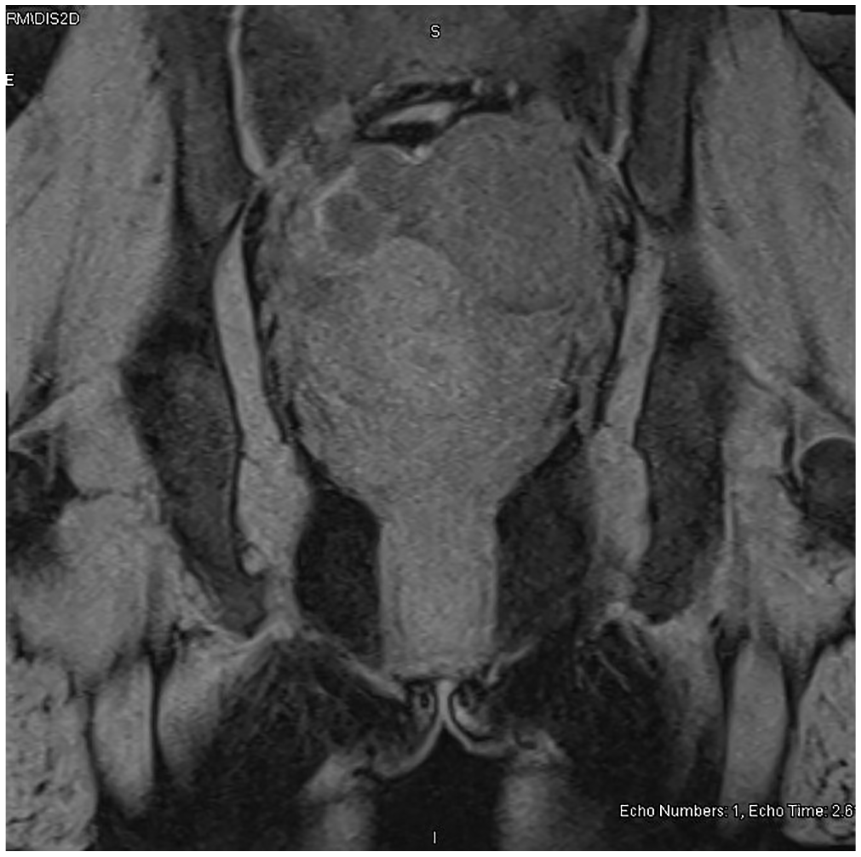
A T1-weighted magnetic resonance image shows an isointense left ovarian mass.
Serum tumor markers revealed a CA-125 level that was 2.5 times greater than the upper limits of normal. Other serum markers for malignancy obtained, including a Thyroid-Stimulating Hormone (TSH), Lactate Dehydrogenase (LDH), Alpha-Fetoprotein (AFP), and Human Epididymis Protein 4 (HE-4), were all within normal limits.
Treatment options were discussed with the patient, which included expectant management with surgery delayed until after pregnancy versus immediate surgical excision. The patient was counseled that if she delayed surgery until after pregnancy, she would run the risk of malignant metastasis, worsening her prognosis. She ultimately decided on the removal of the ovarian mass during the first trimester of pregnancy. At 10 weeks’ gestation, the patient underwent laparoscopic salpingo-oophorectomy with pelvic washings. The pathology specimen showed a solid and circumscribed nodule confirming the mass as a benign luteoma. The tumor was sent for pathologic evaluation and stained positive for SF1, calretinin, and inhibin, all of which are markers for sex cord–stromal tumors. 2 The patient tolerated the procedure well and vaginally delivered a healthy baby girl at 39 weeks, 5 days of gestation.
Discussion
Although ovarian carcinoma during pregnancy is infrequent (1:8000–1:20,000), patients are usually asymptomatic, leaving sonography as one of the only methods of detection. 6 The gravid uterus presents a challenge when viewing the ovaries on sonography. 1 During the second trimester, most ovaries are found along the uterine wall at the level of the anterior iliac spine, but some can be found posterior or anterior to the uterus. 6 Normal ovaries measure around 2 to 5 cm. 6 Transvaginal sonography usually allows for the best view of the uterus and ovaries, but later in gestation, a transabdominal approach may be helpful in getting the best view of the ovaries. 1 A full bladder can also help visualize the uterus and ovaries on transabdominal sonography. 1
In the gravid patient, malignant ovarian masses can be found outside of the pelvis due to the mass effect of the gravid uterus. Besides location, numerous sonographic characteristics of adnexal masses are also associated with increased risk of malignancy during pregnancy. These include solid components or heterogeneous/complex appearance, excrescences/papillary structures, internal septations, bilaterality, irregular borders, increased vascularity, low resistance blood flow, and presence of ascites. 7 Adnexal masses that do not have concerning features are likely to be physiologic cysts and can be managed expectantly, often resolving during the course pregnancy. 8
Luteomas are solid ovarian tumors filled with lipid-laden stromal cells. 9 Because of their solid appearance and increased vascularity on sonography, they are often mistaken for having a high malignant potential. 7 They comprise 0.5% to 1% of all ovarian tumors, and only around 200 cases have been documented. 10 Although benign, they can be hormonally active by producing androgens, and thus maternal virilization during pregnancy should raise suspicion for a luteoma. 11 Most luteomas, however, have little or no androgenic effect because serum concentrations of androstenedione, testosterone, and dihydrotestosterone are only increased in about 30% of patients. 11 Also, any increase in serum free testosterone is offset by the large increase in sex hormone–binding globulin (SHBG) levels that occurs during pregnancy. 11 Multiple gestation, advanced maternal age, and maternal history of polycystic ovary syndrome can increase the risk for the development of luteomas. 12
Luteomas may also result in virilization of female fetuses. 13 Although the fetus in this case was a female, evidence of virilization was not present likely on account of removal of the luteoma during the first trimester. Luteomas are usually diagnosed incidentally at the time of cesarean section. 8 With sonography, luteomas are typically hypoechoic, vascular, thick-walled tumors ranging between 6 and 10 cm in size. 14 In approximately half of cases, they are bilateral. 12 Pregnancy luteomas typically regress promptly after delivery, suggesting that human chorionic gonadotropin (hCG) plays a role in stimulating or perpetuating their androgen production. 14 Due to the misleading malignant appearance of luteomas on ultrasound, many luteomas are unnecessarily removed during pregnancy. 5
The IOTA group has developed the Simple Rules (Table 1), which are a classification system to predict benign versus malignant ovarian tumors. 5 This classification system uses specific sonographic features of adnexal masses to determine the risk of malignancy and facilitate better treatment of adnexal masses. 5 The scoring system is based on a set of “M” or malignant features and another set of “B” or benign features. 5 Risk stratification is based on the overall size, the diameter of the largest solid component, tumor architecture, wall contour, presence of ascites or acoustic shadows, and Doppler flow findings. 5 Using the algorithm, one can determine the likelihood of malignancy (predicting the risk of malignancy).
Simple Rules from International Ovarian Tumor Analysis for Predicting Malignant Potential of an Ovarian Mass.
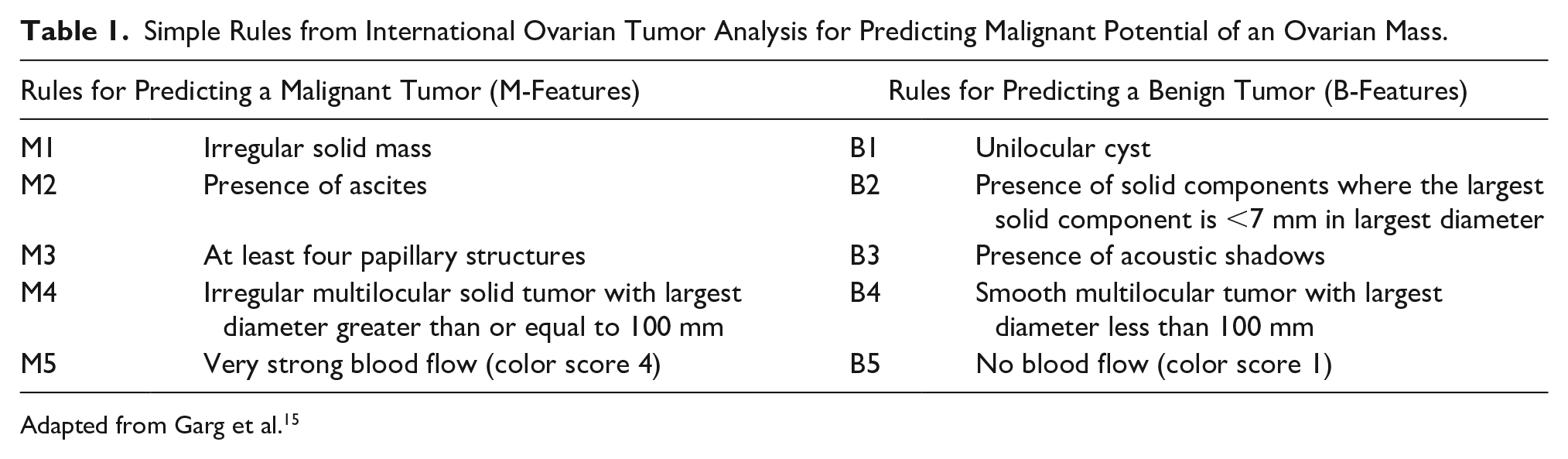
Adapted from Garg et al. 15
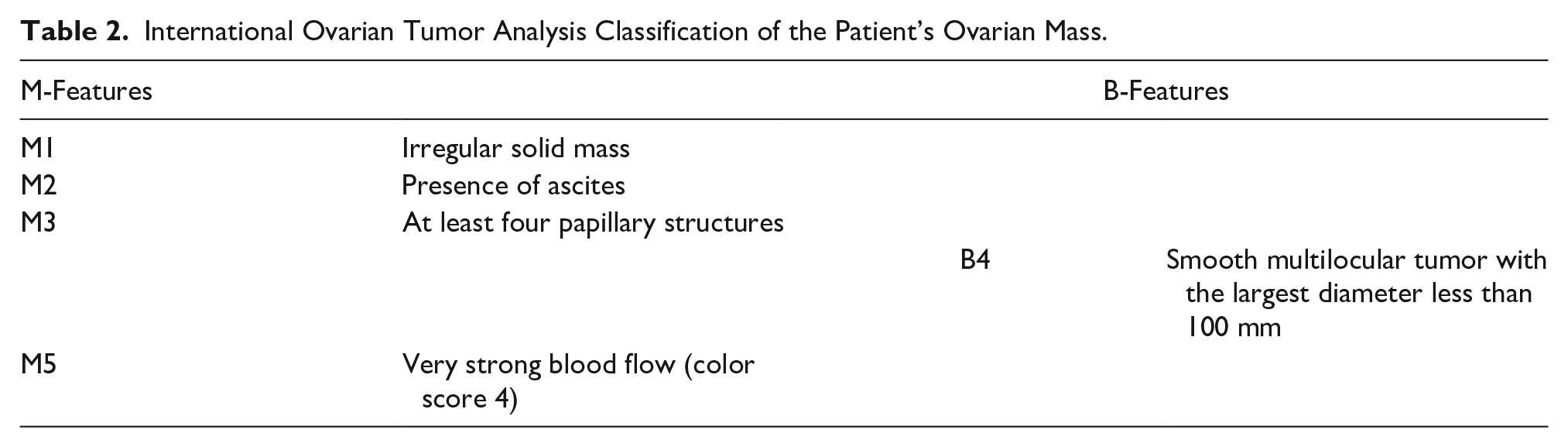
With sonography becoming more routinely used, this case illustrates the need to reevaluate the diagnostic criteria with ultrasound and MRI. Currently, the IOTA Simple Rules are the best way to stratify sonographic findings into masses concerning for malignancy, benign appearing or inconclusive. Sonographic findings of this mass showed the following findings and risk assessment (Tables 2 and 3).
International Ovarian Tumor Analysis Classification of the Patient’s Ovarian Mass.
Risk Stratification of Malignant Potential Using the International Ovarian Tumor Analysis Classification.

Timmerman et al. 5
Based on the sonographic findings of this patient’s mass, there were more M-features compared to B-features, indicating the mass had a very high risk for malignancy with an estimated individual risk of malignancy of 87.5%. 15
Due to the limitations of sonographic examination of ovarian masses during pregnancy, MRI can be used as a supplemental imaging modality. 4 The MRI findings supported the concern for malignancy found on sonography. The mass had numerous T2 hypointense vascular flow voids surrounding and within the mass, which confirmed the Doppler findings of a solid mass. A fibrous tumor was not suspected based on the findings of an isointense signal on T1 with the homogeneous hyperintense signal of T2.
In additionally, tumor markers are used to evaluate adnexal masses, but caution must be taken because many serum tumor markers such as CA-125 are physiologically elevated in pregnancy. 1 The CA-125 peaks between 30 and 60 days after conception and can reach levels as high as 1250 U/mL. 1 This patient’s CA-125 was 2.5 times the upper limit of normal, which is approximately equivalent to the peak physiologic level during pregnancy. HE4 is a tumor marker overexpressed in patients with serous and endometrioid ovarian carcinoma. 1 HE4 was also drawn on this patient and was within normal limits, but its value is unclear during pregnancy. 1
Many factors, including the skill of the sonographer, patient’s body habitus, and available technology, are imperative in the workup of adnexal masses during pregnancy. 3
Although the mass discovered in our case was benign, the risk for malignancy was high, and there was no definitive way to diagnose the mass without excision. Timely imaging, diagnosis, and intervention are all crucial in the treatment of adnexal masses during pregnancy.
The workup of an adnexal mass during pregnancy involves the synthesis of several imaging modalities as well as the integration of laboratory values and clinical symptoms. 2 Counseling and treatment regarding ovarian masses should consist of a multidisciplinary team made up of maternal-fetal medicine, radiology, sonographers, and neonatology.2,4 The prognosis for the mother and fetus relies heavily on the accurate diagnosis and potential removal of ovarian masses during pregnancy. 1
Conclusion
Although an uncommon finding, a luteoma in pregnancy can be evaluated using color Doppler with transvaginal and transabdominal sonography. 3 The sonographer can provide useful clues when imaging these masses during pregnancy. If an ovarian mass is identified, it is essential for the sonographer to capture the overall size, tumor architecture, wall contour, presence of ascites or acoustic shadows, and Doppler flow velocity. 15 The mass can then be stratified based on malignant risk according to the IOTA Simple Rules. 15 MRI is a useful adjunct imaging modality and aids in proper surgical planning during pregnancy. 4 Accurate sonographic images assisted in the care of this patient and unborn fetus and provided useful information to give the patient and the fetus the best possible treatment outcome.
Footnotes
Authors’ Contributions
Kelly Devlin, Amber Payton, Jessica Rubino, and Robert Shapiro carried out conception and design, acquisition and interpretation of data, and drafting. They reviewed the paper and gave final approval.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
