Abstract
Objective:
Congenital heart defects (CHDs) are the most common cause of infant death in the United States. Severe, complex cardiac anomalies make up approximately half of the cases and carry significant morbidity and mortality. The purpose of this literature review was to define a CHD screening protocol to be used in the first trimester.
Methods:
Literature was pulled that discussed the impact of color Doppler and spatiotemportal image correlation on image quality, explored the benefits and limitations of early screening, and analyzed the risk of bioeffects to the developing fetus.
Results:
A simplified cardiac screening protocol has been shown to be a feasible addition to the first trimester nuchal translucency scan.
Conclusion:
Color Doppler and three-dimensional sonography can be utilized during the first trimester to evaluate the fetal heart for major anomalies between 11 weeks and 13 weeks 6 days. Recommendations for future research and guidance from accrediting bodies are provided.
Keywords
Congenital heart defects (CHDs) are the most common congenital abnormality and the most common cause of infant death in the United States.1,2 Fifty percent of CHDs are considered severe and involve consequential long-term morbidity and mortality. 2 Between five and nine neonates in every 1000 births are impacted, and the majority occur in the low-risk population. Sonography is considered the gold standard for diagnosing and evaluating CHDs prenatally. 3 Most CHDs are already established by the late first trimester, but the fetal heart is not routinely analyzed until 18 to 22 weeks. 3
Advancements in sonographic imaging have led to higher-resolution and better-quality images, making cardiac evaluation in the first trimester feasible. This has led to the implementation of first trimester fetal heart evaluation in conjunction with the nuchal translucency (NT) scan in both high- and low-risk populations. Risk factors for fetal CHDs include a relevant obstetric or familial history, an abnormal basic cardiac evaluation, the presence of indirect markers for CHD, a chromosomal anomaly, an extracardiac defect, a monochorionic twin pregnancy, maternal diabetes mellitus or gestational diabetes, obesity (body mass index [BMI] greater than 30), teratogen exposure, or the use of assisted reproductive technology (ART) 4 (Table 1).
Risk Factors for Fetal CHD.
There is a 2% to 4% risk of CHD if only one of the fetus’s siblings is affected, but this number rises to 10% for more than one sibling. A 12% risk is present if the mother of the baby has a congenital cardiac anomaly. 2
This literature review examines the recommended protocols for late first trimester cardiac evaluation, discusses indirect markers for CHD that may be noted during the NT scan, and describes three-dimensional (3D) rendering of the fetal heart. It also addresses the benefits and limitations that may be encountered by the sonographer, along with the potential for bioeffects or fetal harm due to Doppler utilization.
Methods
Sources for this literature review were obtained from multiple databases including PubMed, Medline, Google Scholar, and CINAHL. The search terms used include “first trimester fetal heart,” “first trimester cardiac ultrasound,” “first trimester fetal echocardiography,” “4D fetal echo,” “first trimester cardiac screening,” “spatiotemporal imaging correlation,” “termination of pregnancy and stress,” and “ultrasound bioeffects in the first trimester.” The articles that resulted from the search were evaluated for eligibility using predetermined inclusion criteria. These criteria included articles that were released from 2014 to 2019, full articles, and articles that were in English. Articles that were not sufficiently focused on first trimester fetal echocardiography were excluded from this review. Relevant textbook chapters from A Practical Guide to Fetal Echocardiography: Normal and Abnormal Hearts by Abuhamad and Chaoui were included in the literature review, as these offered a relatively succinct distillation of essential information. The websites of organizations were searched for current sonographic recommendations. These organizations included the American Institute of Ultrasound in Medicine (AIUM), the American College of Obstetrics and Gynecology (ACOG), the International Society of Ultrasound in Obstetrics and Gynecology (ISUOG), the Perinatal Quality Foundation, and the Fetal Medicine Foundation. These organizational guidelines inform the standard of care in current practice and establish the status quo.
Results
Recommended Protocols and Guidelines
Evaluation of cardiac anatomy in the late first trimester increases the detection rate of CHD by 60% to 80%. 5 The development of the fetal heart is mostly complete by 7 to 8 weeks’ gestation, and most major forms of cardiac anomalies are present by this time. 1 Therefore, the ideal time to incorporate early cardiac screening is between 11 weeks’ and 13 weeks 6 days’ gestation, when the crown-rump length (CRL) is between 45 and 84 mm. 1 This CRL correlates with the recommended time to perform the NT scan. 3 Cardiac images can be obtained both transabdominally and transvaginally, with each method having its own advantages and disadvantages.
For the transabdominal approach, a higher-frequency transducer should be initially utilized before switching to a lower frequency. A curvilinear 9-MHz transducer or linear abdominal transducer can be helpful in obtaining high-quality images.6,7 Transabdominal imaging should always be attempted first, as 93.1% of cardiac anomalies can be detected using this method. It has the advantage of flexibility in acquiring various planes of cardiac anatomy and can be accomplished on most patients after 12 weeks’ gestation. 4
Transvaginal sonography (TVS) should only be attempted if transabdominal imaging is unsuccessful in a patient who is at high risk for a fetal CHD or if a CHD is suspected. 8 A high-frequency transvaginal transducer with a bandwidth of 9 to 12 MHz should be utilized. 6 Some reasons TVS may be necessary include obesity, a small CRL, or difficult fetal position. 4 Transvaginal insonation of the first trimester fetal heart allows for higher-resolution imaging in early gestations (i.e., 11 weeks). A disadvantage to this technique is the reduced flexibility in obtaining imaging planes. 6 TVS also adds time to the study for patient preparation and transducer preparation/disinfection. The heart may be difficult to visualize if the fetus is in any other position besides low transverse. 7 Additional costs to the patient and organization should also be taken into consideration when adding a transvaginal scan to an examination (i.e., sonographer/physician time, disinfection supplies, transducer supplies, etc.).
Optimizing gray-scale images is essential for obtaining high-quality cardiac images in the first trimester (Table 2). A fetus in the dorsoposterior position (NT position) is best. The image should be magnified so that the fetal thorax occupies one-third of the screen. The sector width should be narrowed to increase the frame rate. A low-dynamic-range setting should be utilized for high-contrast imaging, and the highest-resolution transducer possible should be applied. 7
Steps to Optimize Gray-scale and Color Doppler Images of the Fetal Heart.
Color Doppler plays an important role in the interrogation of cardiac anatomy in the first trimester. A study performed by Wiechec et al. showed that a combined approach of color Doppler and gray-scale imaging had a sensitivity of 88.57% and a specificity of 100% in the identification of CHDs. This same study also showed that the use of color mapping over the four-chamber view (4CV) and three-vessel trachea view (3VTV) was more effective than gray-scale imaging of cardiac structures. 5 These views can be difficult to interpret in early gestation because of small CRL. Images of cardiac anatomy utilizing color mapping are essential to diagnose CHD. To optimize color Doppler, the sonographer must first optimize the gray-scale image (Table 2). A narrow color box should be used along with a midwall filter, midvelocity color Doppler range, and mid to high color persistence. Low color Doppler gain and low power output should be applied. Bidirectional Doppler will yield the most information regarding cardiac flow directions. 7 Pulsed wave Doppler can be utilized to evaluate the peak systolic velocity of the tricuspid valve or to evaluate the a-wave in the ductus venosus.
Protocols for first trimester fetal heart evaluation may differ between facilities, but most studies have agreed that images should be taken from the 4CV sweeping cephalad through the crossing of the outflow tracts to the 3VTV. The ideal fetal position for transabdominal imaging is with the fetal spine situated at either the 6-o’clock or 12-o’clock position. This helps avoid oblique sections through the heart. 5 First, cardiac position and axis should be determined. The location of the heart and stomach in relation to the fetal position should be assessed to be sure that both are on the left 9 (Figure 1). The cardiac axis is determined by the orientation of the interventricular septum (IVS) of the heart relative to the anteroposterior axis of the chest. Between 11 weeks and 12 weeks, the cardiac axis typically appears more midline. The fetal cardiac axis is established by 12 weeks’ gestation and usually ranges from 25° to 65°. If abnormal, this is likely to be related to conotruncal anomalies or complex CHDs. Thus, the cardiac axis should be included as part of the screening if an abnormality is suspected. 10 The heart to chest area ratio can also be evaluated at this time and should be measured if the heart appears to be subjectively large. A ratio greater than 0.28 is suggestive of cardiomegaly. 4
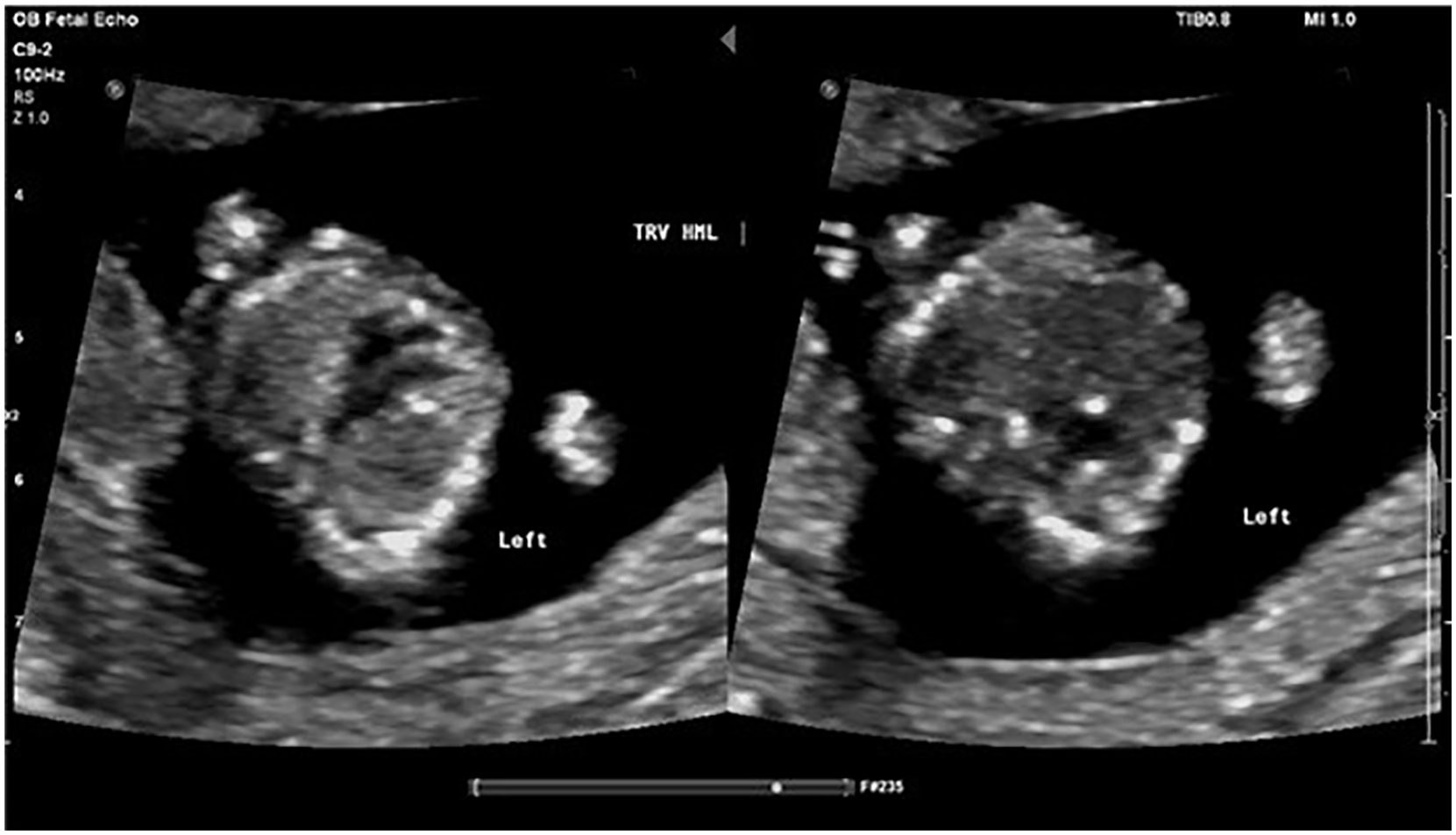
A transverse image of the fetal thorax (left) and fetal abdomen (right) depicting situs solitus of a fetus in the transverse head maternal left position.
The next image that should be taken is the 4CV. Guidelines released by ISUOG recommend that evaluation of the transverse fetal chest at the level of the 4CV is required to document normal position and development of cardiac anatomy 11 (Figure 2). Color mapping should be utilized to view the number of “colored stripes” (color Doppler filling the ventricles) in the 4CV to determine the presence of a univentricular heart or similar anomalies (Figures 3 and 4). A subjective impression of the “stripes” within the 4CV should be evaluated to determine how many ventricles are present and whether they are concordant in size. The IVS should be intact separating the stripes. The ideal insonation angle for evaluation of the IVS is around 45°, allowing for better access to the 4CV and 3VTV. 5 The presence of two atrioventricular (AV) valves should also be assessed from the 4CV for proper opening and closure. 12 This view of the heart has around a 50% detection rate for CHDs. 4 Some of the anomalies that are associated with an abnormal 4CV are atypical cardiac axis, tricuspid valve insufficiency, univentricular heart, complete atrioventricular septal defect (AVSD), ventricle discordance due to valve abnormalities, hypoplastic left heart syndrome (HLHS), and mitral or tricuspid valve atresia. 7

An illustration of the transverse fetal chest demonstrating the apical 4CV.
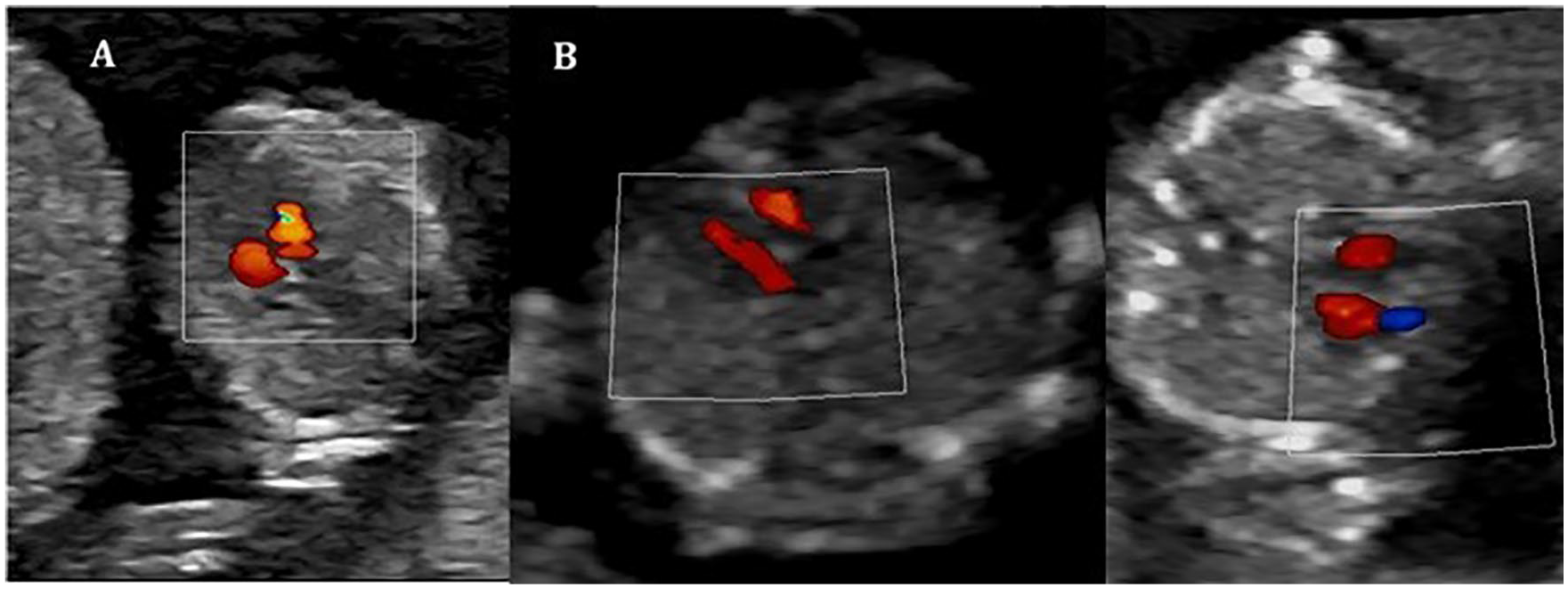
Transverse images of the fetal thorax demonstrating the 4CV using color mapping. Ventricle filling is displayed here via color “stripes.” (A) 58.6-mm CRL. (B) 69.7-mm CRL. (C) 76.7-mm CRL.
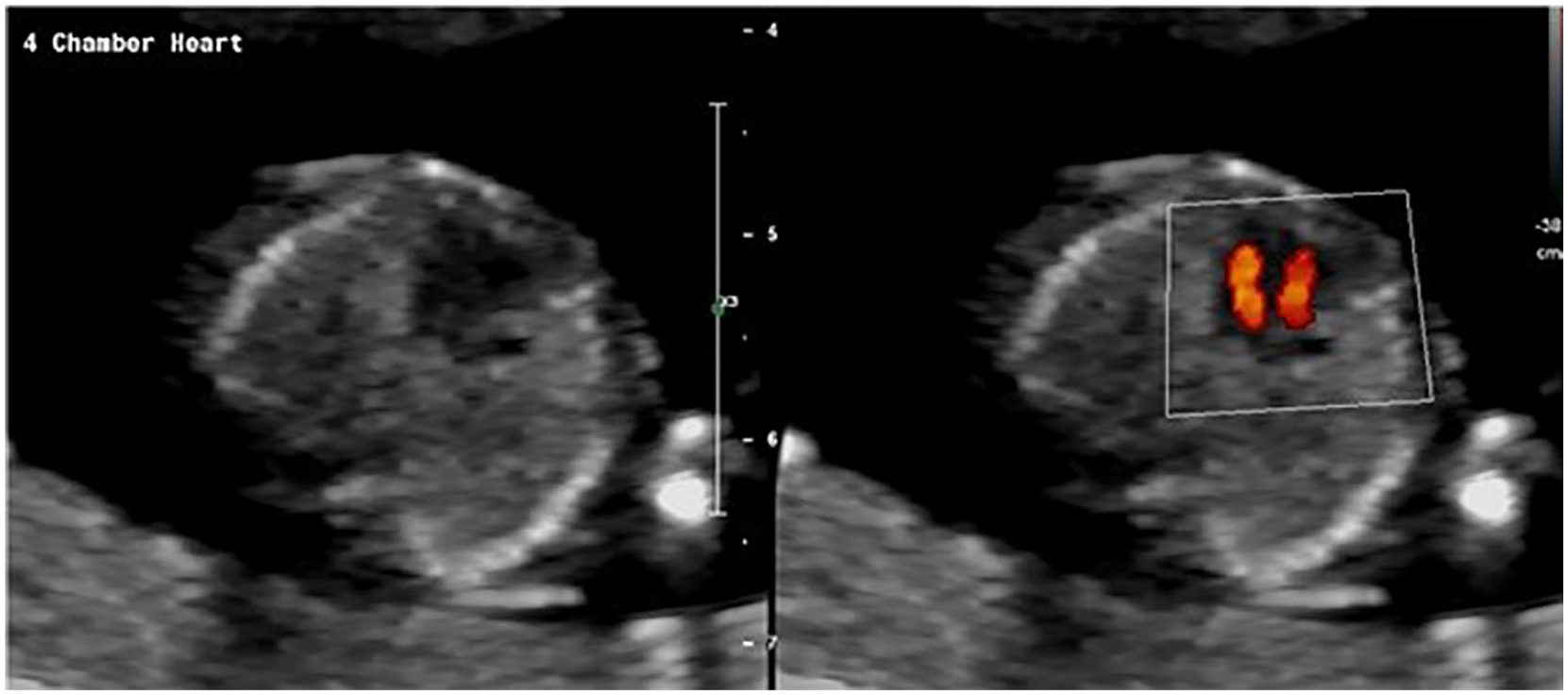
Representation of how the use of color mapping increases visualization of the ventricles.
After evaluating the 4CV, the outflow tracts should be insonated as they exit the heart. Both the origin of the aorta from the left ventricle (left ventricular outflow tract [LVOT]) and the origin of the main pulmonary artery from the right ventricle (right ventricular outflow tract [RVOT]) should be identified. The crossing of these vessels should be seen as they exit the heart. The accuracy in identifying the outflow tracts is increased when color mapping is applied. 12 According to Nemescu and Onofriescu, the LVOT is one of the more difficult structures to visualize in the first trimester. 13
The 3VTV should be evaluated once the outflow tracts are identified (Figure 5). It is best to use color mapping on these vessels because of their small size at this gestation. It is important to identify that two arteries are present in this view (aorta and main pulmonary artery), and they should be subjectively evaluated for their size ratio. The main pulmonary artery should only appear slightly larger than the aorta. The arterial flow in the 3VTV should course in the same direction without evidence of turbulent flow. The normal path of the aorta and main pulmonary artery creates the “V-sign” (Figure 6). This sign allows for the identification of conflicting artery sizes, reversed flow in one artery, a large aorta (common arterial trunk or double-outlet right ventricle), one vessel (transposition of the great vessels [TGA] or double-outlet right ventricle), to-and-fro arterial flow, and an interrupted aortic arch. 5 The 3VTV is particularly sensitive in identifying conotruncal abnormalities when only one artery is visualized. 5 Anomalies that may be associated with an abnormal 3VTV are coarctation of the aorta, pulmonary stenosis, pulmonary atresia with an intact IVS, HLHS, right aortic arch, conotruncal abnormalities, TGA in double-inlet left ventricle, and tricuspid atresia type 2A. 5 In addition to still images, a cine clip should be obtained in an inferior to superior sweep from the 4CV through the crossing of the outflow tracts up to the 3VTV utilizing color mapping. 8
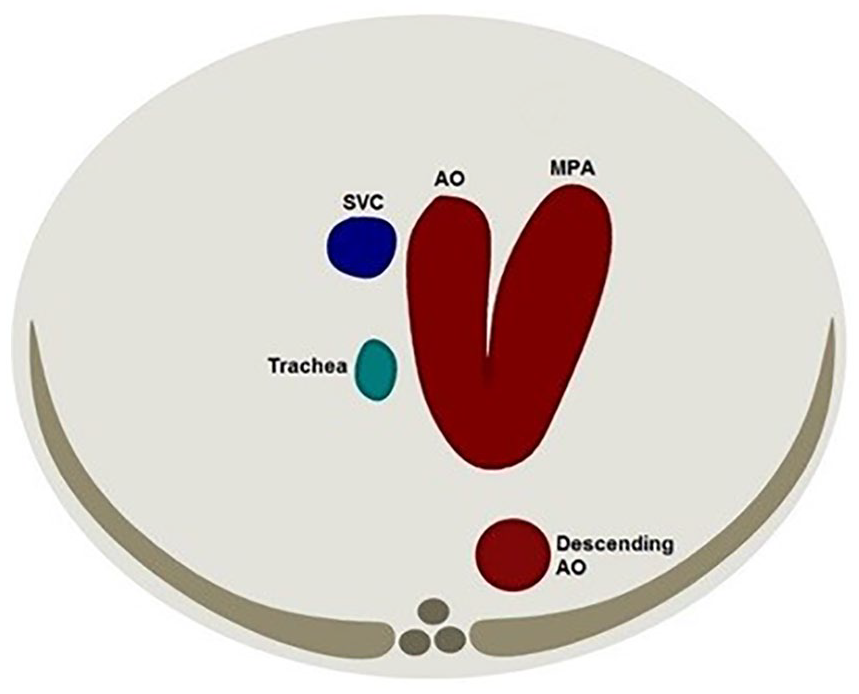
An illustration of the transverse fetal chest demonstrating the 3VTV.
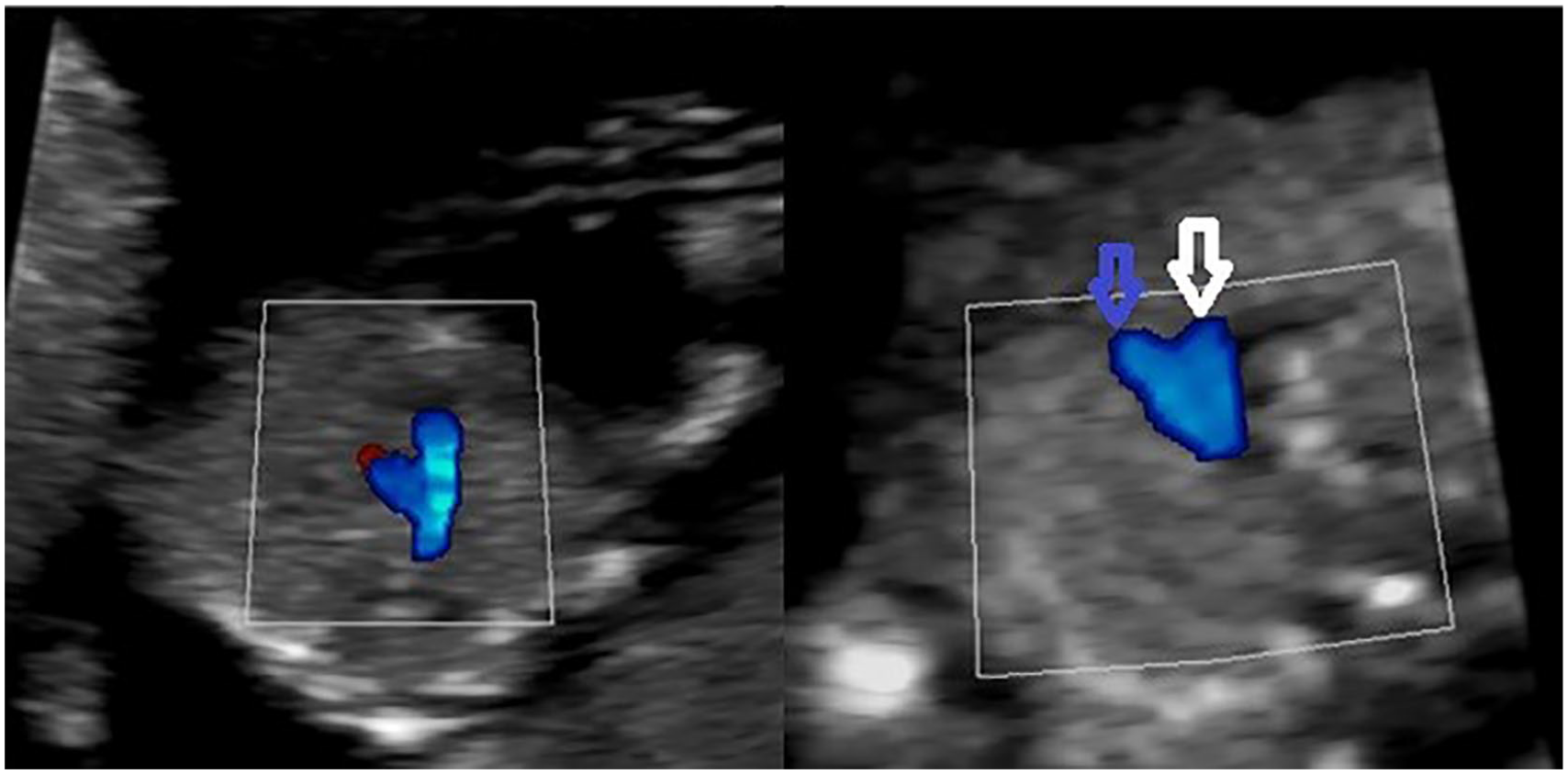
Images of the transverse fetal thorax superior to the 4CV displaying the 3VTV. The “V-sign” is shown in these images of the aorta and main pulmonary coming together on the left side of the fetus at the ductus arteriosus. The white arrow points to the transverse aorta. The blue arrow points to the main pulmonary artery. (A) 58.6 mm CRL. (B) 69.7 mm CRL.
The 4CV, outflow tracts, and 3VTV are considered a simplified protocol that should be primarily used on low-risk patients (Table 3). A study has shown that this shorter method can be utilized during the transabdominal NT scan to identify CHDs. 12 Additional images can be taken in patients who are considered at high risk for fetal cardiac anomalies or in patients where CHD is suspected. These images include the aortic arch and ductal arch, but they are only obtainable in 70% to 80% of fetuses in the first trimester 4 (Figure 7).
Simplified First Trimester Cardiac Screen Recommended Protocol.

Abbreviations: AV, atrioventricular; LVOT, left ventricular outflow tract; RVOT, right ventricular outflow tract; 3VTV, three-vessel trachea view; 4CV, four-chamber view.
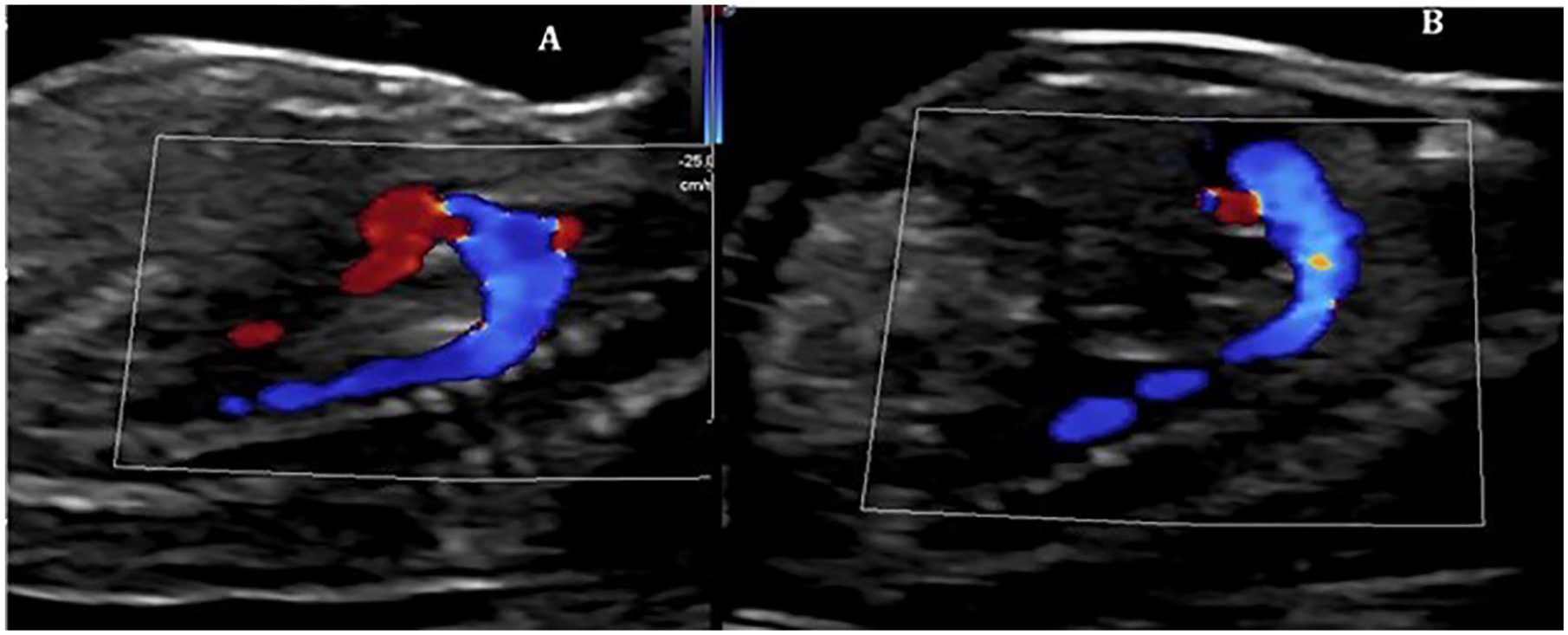
(A) Sagittal image of the aortic arch. (B) Sagittal image of the fetal arch in a fetus with a CRL of 69.7 mm.
Indirect Markers for Congenital Heart Defects in the First Trimester
In addition to evaluating the fetal heart objectively during the NT scan, indirect markers for CHDs may be present at this time, including a thickened NT, tricuspid regurgitation (TR), and an absent or reversed a-wave in the ductus venosus.2,5,10 These markers are often evaluated routinely during the first trimester NT scan because they are also associated with abnormal karyotype. 14 The Fetal Medicine Foundation offers an online 11- to 13-week scan course that includes information on how to properly image these areas. 15 A thickened NT is present in 56% of fetuses with an associated CHD. When the NT is greater than the 99th percentile, there is a sensitivity of 21% and a specificity of 98.7% for identifying severe CHDs. A direct relationship is present between NT and the risk for cardiac anomalies.6,7
When a thickened NT is identified in conjunction with TR and/or abnormal ductus venosus, there is a higher risk for CHD. Both are more likely to be associated with cardiac dysfunction. The risk for CHD increases eightfold when TR is identified between 11 weeks and 13 weeks 6 days and may be related to diastolic dysfunction in the heart. A study showed that structural cardiac defects were identified in 59% of fetuses with TR. 2 A normal ductus venosus waveform appears biphasic 7 (Figure 8). An abnormal appearance will present as either a reversed or absent a-wave during atrial contraction 6 (Figure 9). When this type of flow is present in conjunction with a thickened NT, the risk of CHD is increased threefold. Reversed or absent flow in ductus venosus is often related to right-sided cardiac lesions, an AVSD with a single AV valve, or HLHS due to increased right heart volume and/or pressure overload. Diastolic dysfunction may also be present. 6
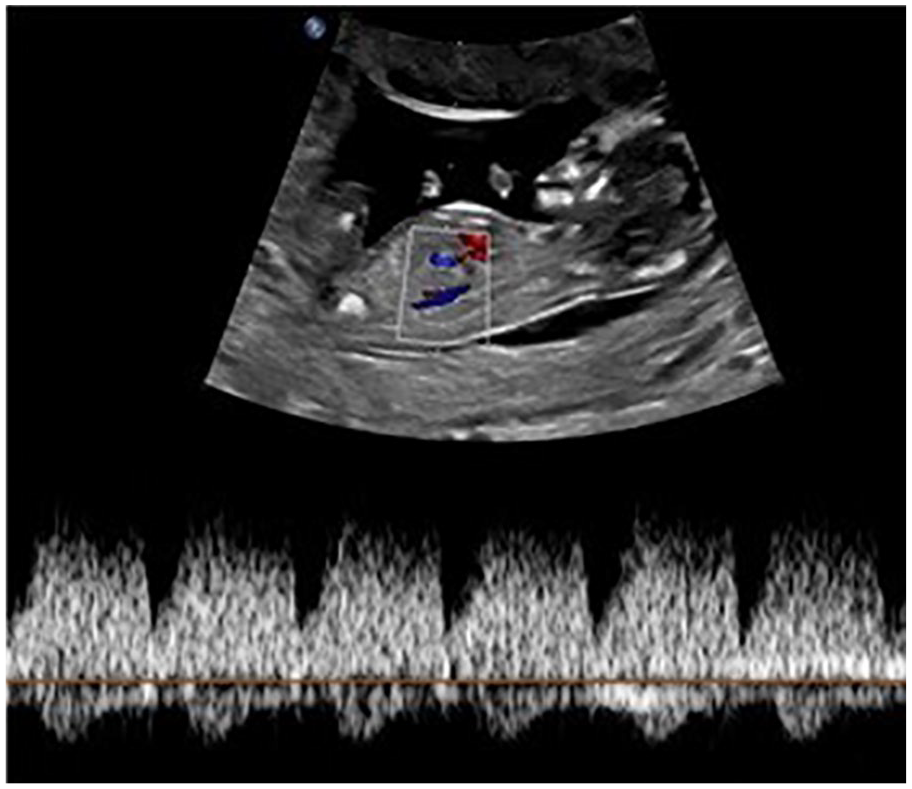
A normal ductus venosus at 12 weeks and 6 days.
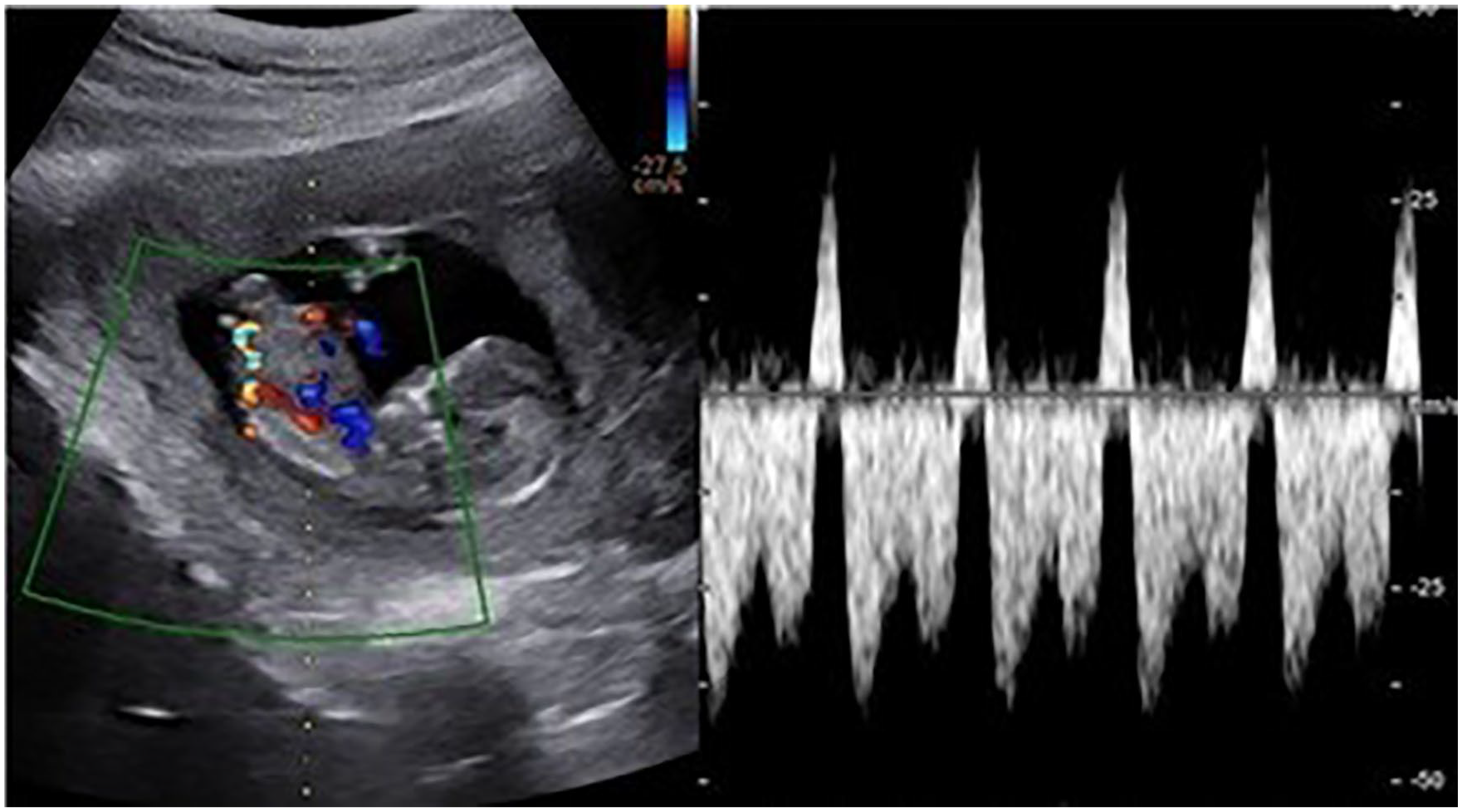
An example of a reversed a-wave in the ductus venosus. (Case courtesy of Dr. Ayush Goel, rID 54485, radiopedia.org.)
Four-Dimensional Cardiac Imaging
Four-dimensional (4D) cardiac imaging can be accomplished through spatiotemporal imaging correlation (STIC). According to A Practical Guide to Fetal Echocardio-graphy, STIC is an “indirect, motion-gated, offline mode based on the concept of using tissue excursion concurrent with cardiac motion to extract the temporal information regarding the cardiac cycle.” First, a volume image is acquired and processed. The systolic peaks are used to calculate fetal heart rate, and the images are arranged in the order that they transpire. A cine loop is created of a single cardiac cycle. 16 Images are best when obtained from an apical 4CV with the fetal spine between five and seven o’clock, without shadows or fetal/maternal motion. The angle of insonation is best between 20° and 35°, and the acquisition time should be set around 10 seconds.4,14 This method can adequately show the 4CV, crossing outflow tracts, LVOT, RVOT, and 3VTV 4 (Figure 10).
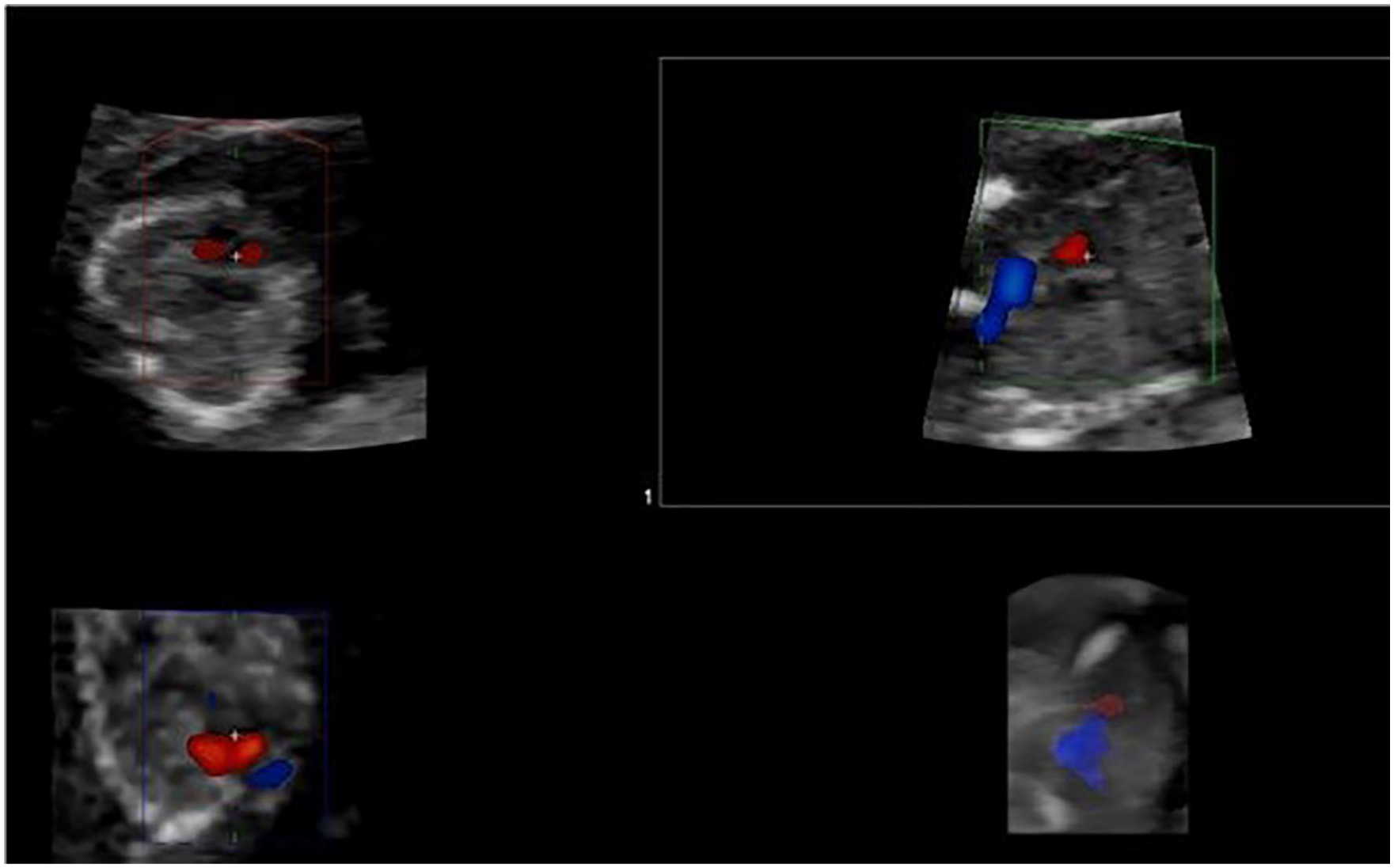
(A) A STIC image taken at the level of the 4CV with the spine at seven o’clock. (B) When manipulated, the 3VTV can be visualized with color Doppler.
One of the advantages to STIC is that it can evaluate atrial/ventricular wall motion and valve motion. Another benefit is that a volume (3D image) can be produced within seconds after the acquisition is completed. It can also be used in conjunction with other sonographic technologies, such as color, power, and high-definition flow Dopplers. 16 STIC can be performed using either a transabdominal or transvaginal method. 17 One limitation is that there is a delayed volume acquisition time that may be impacted by fetal or maternal movement, resulting in artifact. 16 This is the most common reason for inadequate STIC images. 14 Despite this pitfall, enough data can be collected from these images to determine a normal heart from an anomalous heart. STIC can identify cardiac anomalies with 95.3% accuracy. 17
Tomographic ultrasound imaging (TUI), also known as multislice analysis, can be applied to the STIC technique. This method displays multiple two-dimensional (2D) planes at the same time to provide a consecutive, anatomic view of the fetal heart. 16 It requires postprocessing on a system separate from the ultrasound system. In a study performed by Turan et al., the results showed that STIC-TUI could reproduce images commonly used in fetal echocardiography between 11 and 14 weeks and was able to diagnose complex CHDs in a significant proportion of cases. 14 An advantage of TUI is that it can display multiple planes (i.e., sagittal, transverse, coronal) from one acquisition. These slices allow for the assessment of CHDs that impact multiple regions of the heart. This technique could lessen operator dependency, but a skilled sonographer is still necessary to obtain an appropriate STIC acquisition. 16 STIC-TUI displays all acquired planes in one image in sequence. 14 One of the limitations of TUI is that the reproduced planes are reconstructed and not taken in real-time. 16
Discussion
Based on the information discussed above, fetal cardiac screening in the first trimester is feasible. By the 11- to 14-week gestation mark, the fetal heart is almost fully developed, and any major cardiac anomalies will be visible. This basic screening technique yields high sensitivity and specificity when paired with color Doppler. There are endless benefits to the early identification of major CHDs. Parents and physicians will both have more time to consider the options available to them. 18 Families will be able to mentally prepare for the surgeries and potential comorbidities that face their unborn child. A plan can be made so that the patient can be transferred to a tertiary hospital for additional testing, delivery, and proceeding surgeries. Studies have shown that neonatal morbidity and mortality decreases with the prenatal diagnosis of CHD. 9 An abnormal cardiac finding in the first trimester could trigger a second trimester fetal echocardiogram or a detailed anatomic assessment to evaluate for additional extracardiac defects. If a minor cardiac anomaly is identified early, it can be followed for progression. This opens the opportunity for early intervention if necessary. 1 Studies have shown that echocardiographic screening examinations in the first trimester are effective for reassuring couples because of the very high negative predictive value of 98.9%.3,12 Many major CHDs are associated with an abnormal karyotype, and if identified early, genetic counseling and testing could be initiated earlier. In addition, counseling with appropriate specialists could begin.
An early positive finding of CHD in the first trimester could be a source of stress and anxiety for parents. In the event of a suspected genetic anomaly in addition to a cardiac anomaly, genetic testing such a chorionic villus sampling or an amniocentesis can be performed. 19 If only a cardiac abnormality is suspected, the patient can be counseled on prospective outcomes, but further ultrasound evaluation would not occur until 18 to 22 weeks’ gestation per current ISUOG guidelines. 11 Thus, this period of 4 to 11 weeks between the first trimester cardiac screening and the second trimester could cause mental unrest for parents. Despite this, many parents would prefer to know about any major cardiac anomalies at an earlier stage. 11 In the event of severe, complex cardiac anomalies, a patient may choose to terminate the pregnancy. When defects are identified earlier, it allows for an earlier termination of pregnancy (TOP) or gives the patient more time to consider this option. Some states will not allow medical abortions after 22 weeks’ gestation, and others will not allow them after the point of fetal viability. 18 Around 64.5% of patients that undergo a medical TOP tend to have higher reports of posttraumatic stress (PTS). If the termination is performed in the second trimester, the patient is more likely to suffer from PTS 6 weeks after the procedure than if it were done in the first trimester. PTS can be associated with perinatal grief, depression, and anxiety. Gestational age has been shown to directly correlate with an increased probability of a diagnosis of PTS or posttraumatic stress disorder. 20 An earlier TOP also presents less of a medical risk for the mother. 1
While there is a vast array of benefits to early cardiac screening, there are also some limitations. False-positive and false-negative examinations may occur due to a variety of technical constraints.4,21 A small CRL could potentially limit the resolution of fetal heart images, especially during the 11th week. The structural complexity of the heart and the extensive operator training that is necessary could be obstacles. Maternal or fetal motion could result in artifact. Maternal obesity could potentially limit the penetration of the sound beam transabdominally, causing inadequate imaging. Unfavorable fetal position could also make it difficult to visualize cardiac structures. 21 Despite these technical limitations, one study showed that a high BMI, small CRL, increased distance between the transducer and the heart, interposition of the placenta, and difficult fetal position were insignificant contributing factors when compared with the level of training the sonographer possesses. 13
Operator experience is an important factor when evaluating the fetal cardiac anatomy in the first trimester for anomalies. Appropriate training is necessary to perform these examinations precisely. One study showed that when a trained sonographer was imaging the early fetal heart there was a 90% sensitivity and 99.5% specificity for the identification of CHDs. 9 Another study performed looked at the feasibility of late first trimester cardiac screening. The frequency of successful examinations was found to be directly related to the number of previous scans the sonographer performed. The results showed that the learning phase for this type of examination was approximately 180 cases. For every 20 examinations the sonographers completed, the likelihood of a successful evaluation of the first trimester fetal heart increased by 1.2%. By the end of the learning curve, images could be successfully obtained within 3 minutes in 98% of fetuses. 13 In the study performed by Turan et al., it was concluded that the ability to obtain a highly optimized 2D image, as required to create a proper STIC volume acquisition, increased with operator experience. 14 Limited experience results in a low detection rate of CHDs. The lack of funding to train sonographers in this advanced sonographic technique is a major barrier in some facilities. Proper instruction and hands-on training are essential to yield a diagnostic study.
Another limitation of the early fetal heart evaluation is that some cardiac lesions may be missed. Septal defects are notoriously difficult to identify in the second trimester, let alone the first trimester.4,8 The pulmonary veins are also difficult to see in early gestations and can only be obtained in 25% to 55% of fetuses in the late first trimester. 6 Some CHDs can progress throughout pregnancy and may be normal at the time of the first trimester ultrasound. This may lead to false reassurance of anxious parents. 4 Other commonly missed lesions during early cardiac evaluation are coarctation of the aorta, branching anomalies, Tetralogy of Fallot, total anomalous pulmonary venous return, partial/small AVSD, valvular pulmonary stenosis, absent pulmonary valve, TGA, and structural anomalies of the AV valves. Despite this, a normal heart can be confirmed with a specificity of 80%. 4
Many find the use of Doppler on the fetal heart controversial because of the potential for fetal harm or bioeffects. It has been well documented that ultrasound has the possibility of tissue heating and cavitation at certain levels, but these levels are not reached for the duration of the first trimester fetal cardiac screening.1,2,22 The thermal index (TI) describes the likelihood for a rise in temperature along the sound beam. Some sources recommend that this level be kept below or equal to 1.0, and others recommend it stay less than or equal to 0.7. 22 A study by Nemescu and Berescu showed that B-mode, color Doppler, pulsed Doppler of the tricuspid valve, and pulsed Doppler of the ductus venosus resulted in the following median TI levels, respectively: 0.0, 0.2, 0.4, and 0.1. All of these numbers are well below even the most conservative recommendations. TI exposure should follow the principles of ALARA (as low as reasonably achievable), and studies should last no more than 5 to 10 minutes, with the maximum amount of time being 60 minutes in the first trimester.21,22
Both the AIUM and ISUOG have official statements regarding the safe use of Doppler ultrasound during the 11- to 14-week scan. The AIUM recommends that TI ideally be kept below 0.7 with a maximum of 1.0. A TI of 1.0 should only be reached if it is necessary to obtain sufficient images, and exposure times should be kept as short as possible. Pulsed Doppler should only be used to “refine risks for trisomies” and should not be used routinely. 23 ISUOG is in agreement with the AIUM in all of the above-mentioned areas. 24 In relation to TI, a study was conducted that showed that measured temperature rise and TI are not directly related. TI only allows for an estimation of temperature increase. This is why exposure time and ALARA are important in the first trimester, because an ultrasound operator may have a difficult time estimating where the highest temperature changes could be along the sound beam. 25 STIC is considered a form of 4D imaging, and it has been recommended to keep the TI less than 1.0. In addition, the scan time during real-time volume acquisition should be kept under 30 minutes. This is because often STIC is combined with color Doppler to increase visualization. Therefore, exposure should be limited. 26
The mechanical index (MI) measures the potential that an ultrasound will create inertial cavitation. Cavitation is not possible in utero because of the absence of a gas-liquid interface. 22 However, it is recommended that the MI stay less than or equal to 0.7. According to Nemescu and Berescu, the MI values for B-mode, color Doppler, pulsed Doppler of the tricuspid valve, and pulsed Doppler of the ductus venosus were medians of 0.6, 0.7, 0.39, and 0.4, respectively. Currently, there is no known reason to restrict scanning times for the sake of reducing the risk the inertial cavitation. 22
CHD screening in the late first trimester during the NT scan is feasible. Proper training and education is required for the operator. Credentialing in OB/GYN or fetal echocardiography by the American Registry of Diagnostic Medical Sonography (or equivalent) will help in acquiring the necessary skills. 6 For early diagnosis of CHD to be considered effective, it is essential that the study be performed by someone who has extensive experience in fetal echocardiography. 6 As stated before, the learning curve is about 180 scans before the sonographer will be competent. A study from Wiechec et al. has shown that a simple first trimester screening protocol for the fetal heart is an effective method for identifying CHD earlier in gestation. 5 Obtaining the 4CV is no more difficult than obtaining images of indirect CHD markers such as NT, tricuspid flow, and flow in the ductus venosus. 12 Once an operator has surpassed the 180-scan learning curve, 98% of images of the early fetal heart could be successfully obtained in 3 minutes. 13 However, it is unclear how much extra time may be necessary for sonographers during the initial training period before they become proficient in imaging early cardiac anatomy.
The first trimester screening for CHDs does not replace a routine heart evaluation in a targeted anatomy scan or fetal echocardiogram in the second trimester. Some CHDs are considered progressive and may appear normal in the first trimester.1,4 In addition, more information can be obtained from these examinations in the second trimester, such as Doppler analysis of all valves and vessels in systole and diastole. 5 Discrepancies between great artery sizes and ventricular septal defects may not be apparent in the first trimester. 14 A second trimester fetal echocardiogram is the current gold standard for the identification of CHD. 14 Earlier diagnosis in the first trimester just allows for earlier counseling of patients and is only meant as a tool to identify major defects earlier in gestation. 14
A few suggestions can be made regarding the future of cardiac screening in the first trimester. It will be important, as more facilities take this approach, that a more standardized protocol be developed. A collaborative creation of guidelines by a combination of professional sonographic organizations could better standardize and improve the quality of cardiac screenings. Further research into the feasibility and efficacy of first trimester fetal heart evaluation could aid in the creation of these guidelines. In addition, the mean BMI for many studies trended below 30. Additional research in this area could better analyze the potential technical limitations that obesity may have on this type of sonogram. This could be accomplished by recruiting obese participants for this type of imaging research or by comparing image quality in patients with a BMI less than 30 versus more than 30. Furthermore, additional research should be conducted to determine if earlier detection of CHD improves overall outcomes for the mother and the fetus.
Conclusion
Congenital cardiac anomalies can be devastating if not identified prenatally. Research suggests that early screening, between 11 weeks and 13 weeks 6 days, is effective and feasible in discovering major CHDs. A simplified protocol can be utilized to make these diagnoses, and color flow mapping should be applied during this time period due to technical limitations associated with early gestational age. 12 Early identification allows physicians more time to create a plan and screen for other anomalies (extracardiac or chromosomal) while giving patients more time to cope with the diagnosis and make well-informed decisions involving their care.1,18 While this examination is operator dependent, early first trimester cardiac screening is associated with high sensitivity and specificity for CHD when performed by a trained sonographer. 9 The identification of cardiac anomalies in the first trimester yields many benefits and is a feasible addition to the late first trimester sonographic protocol.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
