Abstract
A tumor is an excessive growth of cells that results from the body’s inability to balance the growth of new cells and the destruction of old cells. Tumors can occur throughout the body and are classified as either benign or malignant. However, cardiac tumors are a rare occurrence. When present, several imaging modalities are available to illustrate their presence and characteristics. Not all cardiac masses look similar and, depending on their size and location, may pose different health risks to the patient. This case series introduces six left atrial myxomas with dissimilar appearances initially detected by transthoracic echocardiography, along with cross-correlation by transesophageal echocardiography, computed tomography, and mechanical resonance imaging.
An atrial myxoma is a benign tumor that can develop in either the right or left atria. 1 Typically, these tumors are attached on the atrial septum via a stalk, which is classified as pedunculated. This case series includes atrial myxomas that were discovered locally. Each case provides a distinctive presentation of these benign tumors and illustrates to sonologists and sonographers alike that although each case represents the same disease, atrial myxomas have the potential to exist in different patterns, have different characteristics, and have the capability of affecting hemodynamics differently in certain presentations. This intent of this case series is to highlight the possible differences in myxoma presentation with the addition of different imaging modalities.
Case 1
A 79-year-old man with a history of coronary artery disease (CAD), pacemaker support, and chronic renal disease was referred to the echocardiography laboratory for evaluation of ischemic cardiomyopathy. Upon examination, a I/VI systolic murmur to the left sternal border was detected along with a I/VI systolic murmur to the base. Due to the patient’s cardiac history, a routine transthoracic echocardiogram (TTE) was ordered to further assess the patient’s cardiac anatomy. The TTE demonstrated a large mass located in the left atrium (LA) measuring 2.4 × 2.6 cm from the apical four-chamber view (Figure 1A). It is important that the sonographer delineate a suspected mass from multiple views to exclude adjacent mediastinal structures or artifact that is often seen in the far-field of the ultrasound beam. In this case, the apical two-chamber view demonstrated a large echogenic mass in the LA as seen in the apical four-chamber view (Figure 1B). In addition, cross-correlation by transesophageal echocardiography (TEE) and computed tomography (CT) angiography confirmed the size and location of the mass. The TEE demonstrated a 3.0 × 2.15-cm mass located in the LA with its attachment on the interatrial septum (Figure 1C). The CT demonstrated a soft tissue mass measuring 2.2 × 1.9 × 2.3 cm in the LA abutting the interatrial septum (Figure 1D). Because of the classic location and appearance of the mass, all findings were consistent with a large myxoma.
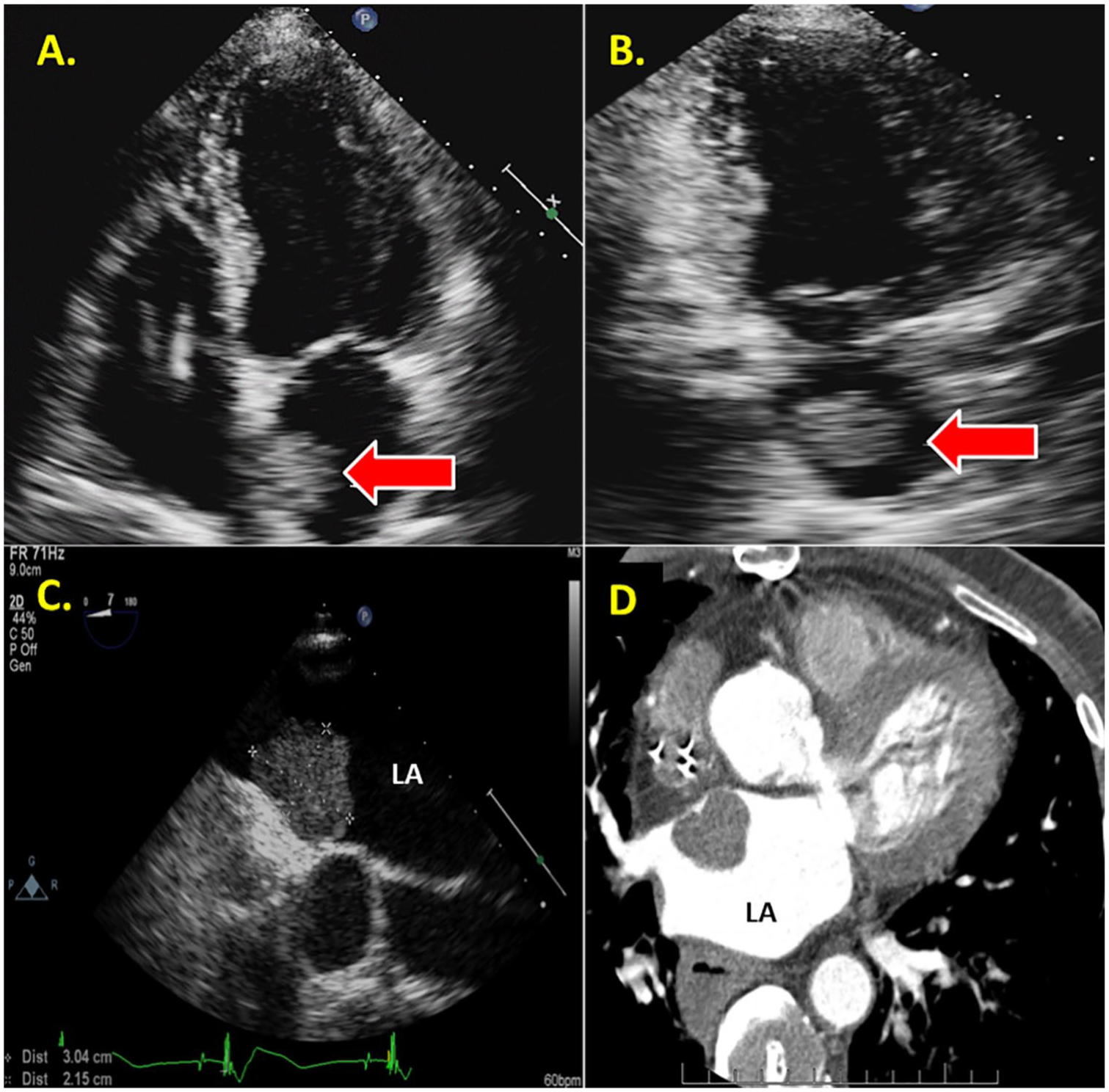
(A) TTE apical four-chamber view demonstrating a large echogenic mass (arrow) in the LA. (B) TTE apical two-chamber view confirming a large echogenic mass (arrow) in the LA. (C) TEE confirming a large echogenic mass in the LA (arrow) attached to the IAS. (D) CT confirming a large soft tissue mass (arrow) attached to the atrial septal wall. Given the location and site of attachment of the mass to the IAS, findings are most consistent with a myxoma. CT, computed tomography; IAS, interatrial septum; LA, left atrium; TEE, transesophageal echocardiography; TTE, transthoracic echocardiography.
Case 2
A 62-year-old female patient was referred for precordial chest pain, hyperlipidemia, hypertension, and fatigue. The pain was located in the midsternal area and radiated to the left and right shoulder. A TTE was ordered to assess the chest pain. The TTE findings demonstrated a 2.9 × 4-cm echogenic mass attached to the left atrial side of the interatrial septum (Figure 2A,B). An additional two-chamber view confirmed the existence of the mass to prove that it was not artifact (Figure 2C). A TEE confirmed the mass, which exuded a cystic, fixed appearance and maintained a broad-based attachment to the atrial septum (Figure 2D). Fluid-filled cavities were evident in the mass, and there was increased vascularity by color flow Doppler. A CT confirmed a 3.3 × 3.6 × 2.9-cm heterogeneous mass in the left atrium (Figure 3, upper left).
(A) A TTE apical four-chamber view demonstrating a large mass (arrow) attached to the IAS just below the PV. (B) A zoomed-in TTE apical four-view chamber providing evidence of the heterogeneous makeup of the myxoma. (C) A TTE apical two chamber demonstrating a highly heterogeneous atrial mass (arrow) filling the majority of the LA. (D) A TEE mirroring the findings of a heterogeneous mass (arrow) in the left atrium. IAS, interatrial septum; LA, left atrium; PV, pulmonary veins; TEE, transesophageal echocardiography; TTE, transthoracic echocardiography.
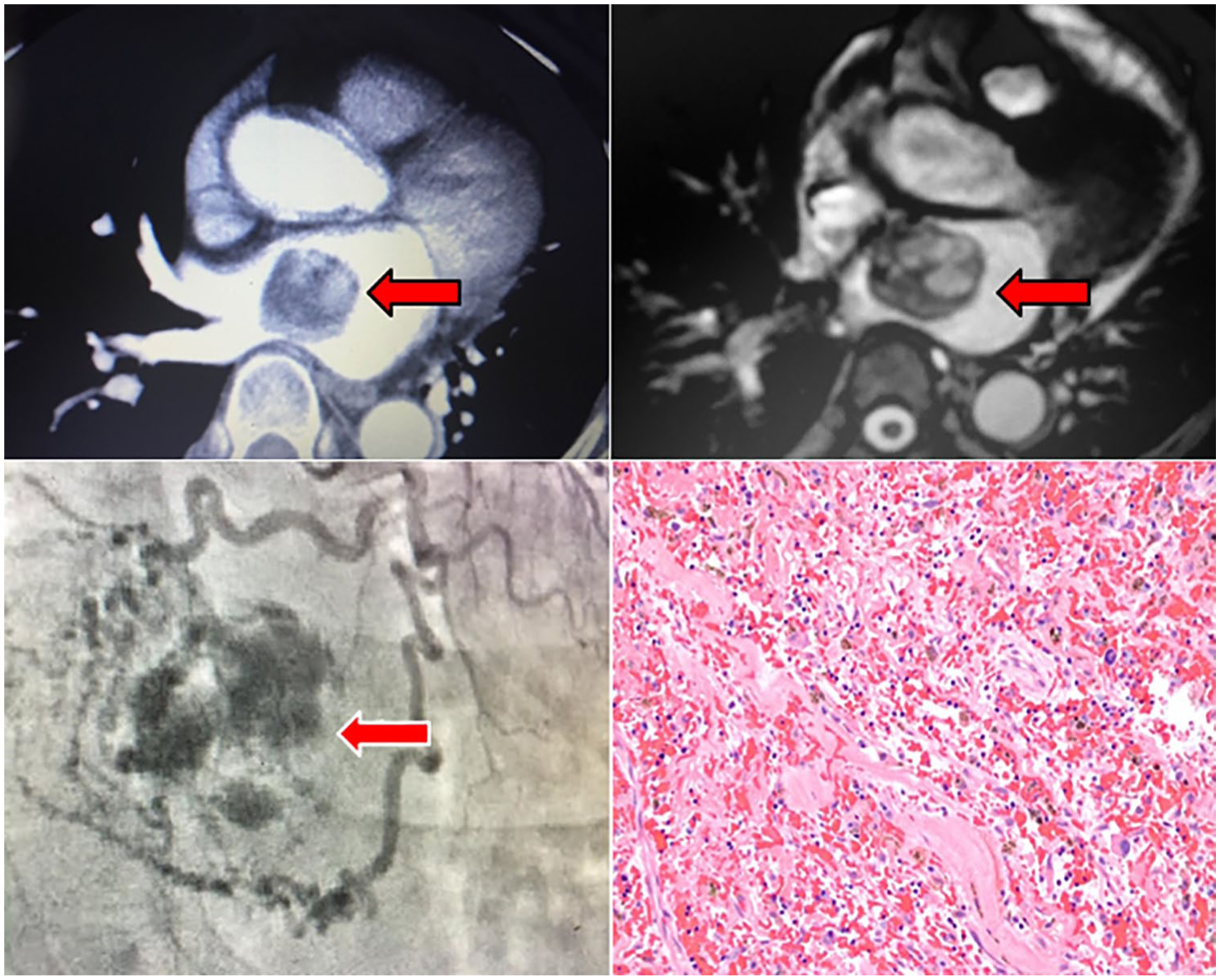
A CT showing the heterogeneous mass in the LA (top left). A cardiac MRI demonstrating a broad-based mass attached to the fossa ovalis (top right). A coronary angiogram illustrating the vascularized myxoma with the coronary circulation (lower left). Stellate cells typical of myxoma in a background of hemorrhage and hemosiderin-laden macrophages. Hematoxylin and eosin. Original magnification: 200× (lower right). CT, computed tomography; LA, left atrium; MRI, magnetic resonance imaging.
Cardiac magnetic resonance imaging (MRI) revealed a broad-based, well-circumscribed mass attached to the fossa ovalis. The mass measured 3.8 × 3.2 cm in the coronal plane and 3.6 × 2.8 cm in the axial plane (Figure 3, upper right). A late enhancement scan revealed thrombus on the surface of the mass, which put the patient at an even greater neurological embolization risk. Furthermore, during the postcontrast weighted images, the mass enhanced with contrast, proving neovascularization. A presurgical coronary angiogram discovered that the mass was filling from the left atrial branch of the left circumflex (Figure 3, bottom left). During surgery, the mass was visualized as deeply rooted into the atrial septum. Because of this, a right atriotomy was performed and the atrial septum was reconstructed with a bovine pericardial patch. The final diagnosis of an atrial myxoma was correlated with pathologic investigation of the mass (Figure 3, bottom right).
Case 3
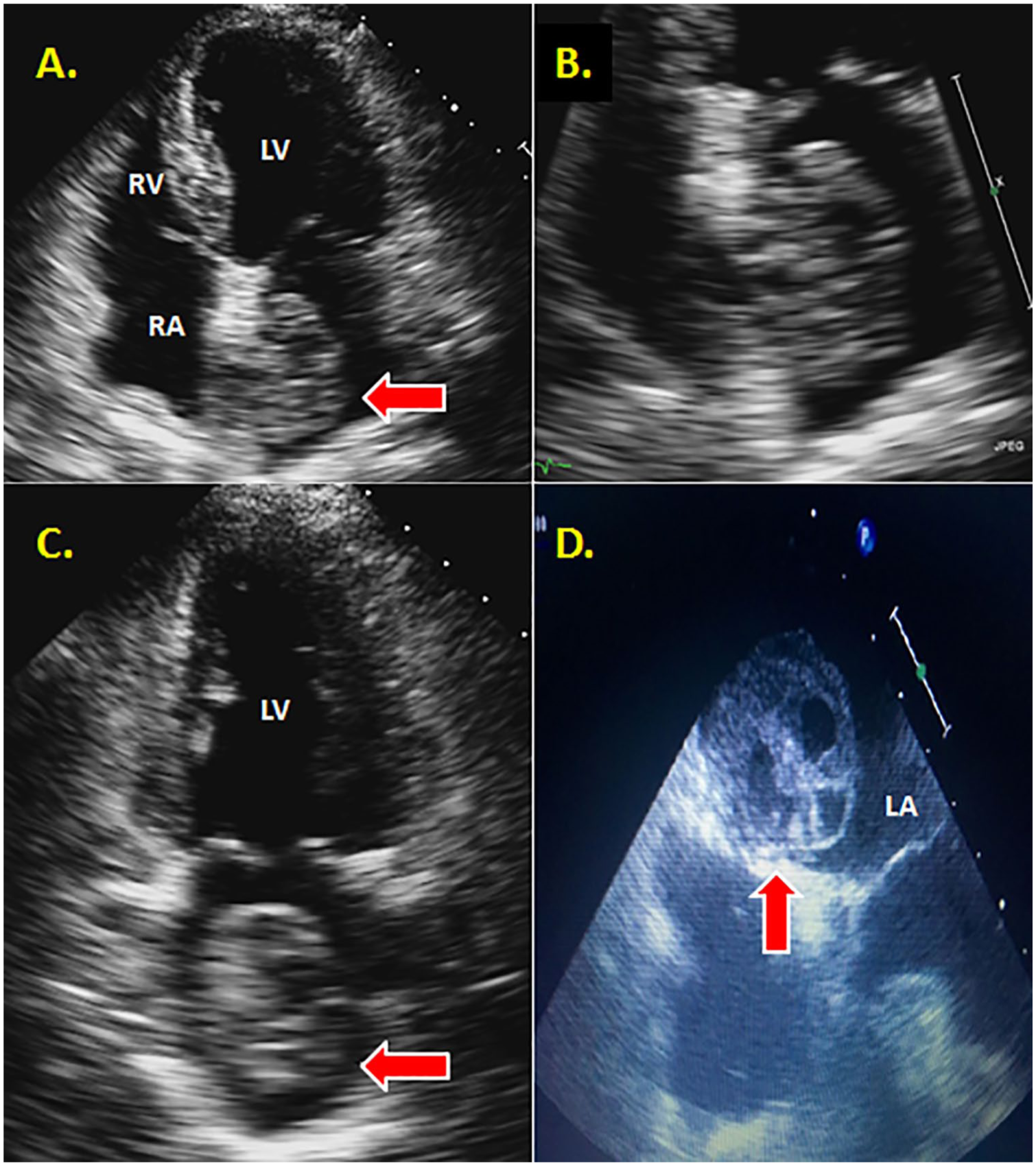
A 44-year-old man with a history of dyspnea on exertion and fatigue with exercise presented to his primary care physician. A CT with contrast was ordered that demonstrated a filling defect and a soft tissue mass that measured 3.5 × 4.7 cm and extended from the left atrium into the left ventricle. A TTE was ordered to further delineate the cardiac structures and mass. Upon initial investigation, the parasternal long axis presented a large 4.6 × 6-cm pedunculated mass in the left atrium (Figure 4A). The mobility of the mass was causing an obstruction in diastole and mimicking mitral stenosis (Figure 4B–D). Continuous-wave Doppler produced a mean gradient of 11 mm Hg (Figure 5A). Furthermore, this patient was at risk not only due to the size and valvular involvement of the mass alone but also because of the potential of gelatinous material embolizing from the mass, putting the patient at risk for stroke and transient ischemic attack (TIA) (as seen in Figure 4B,C). An apical two-chamber view once again demonstrated the sheer size of the mass in the left atrium (Figure 5B).
(A) A TTE parasternal long-axis view indicating the left atrial myxoma filling the majority of the LA in systole. (B) A TTE parasternal long-axis view demonstrating the mobility of the myxoma during diastole. (C) A TTE apical four-chamber view during diastole illustrating the mobility of the mass. (D) Color Doppler on TTE showing turbulent blood flow around the myxomas during diastole. LA, left atrium; TTE, transthoracic echocardiography.
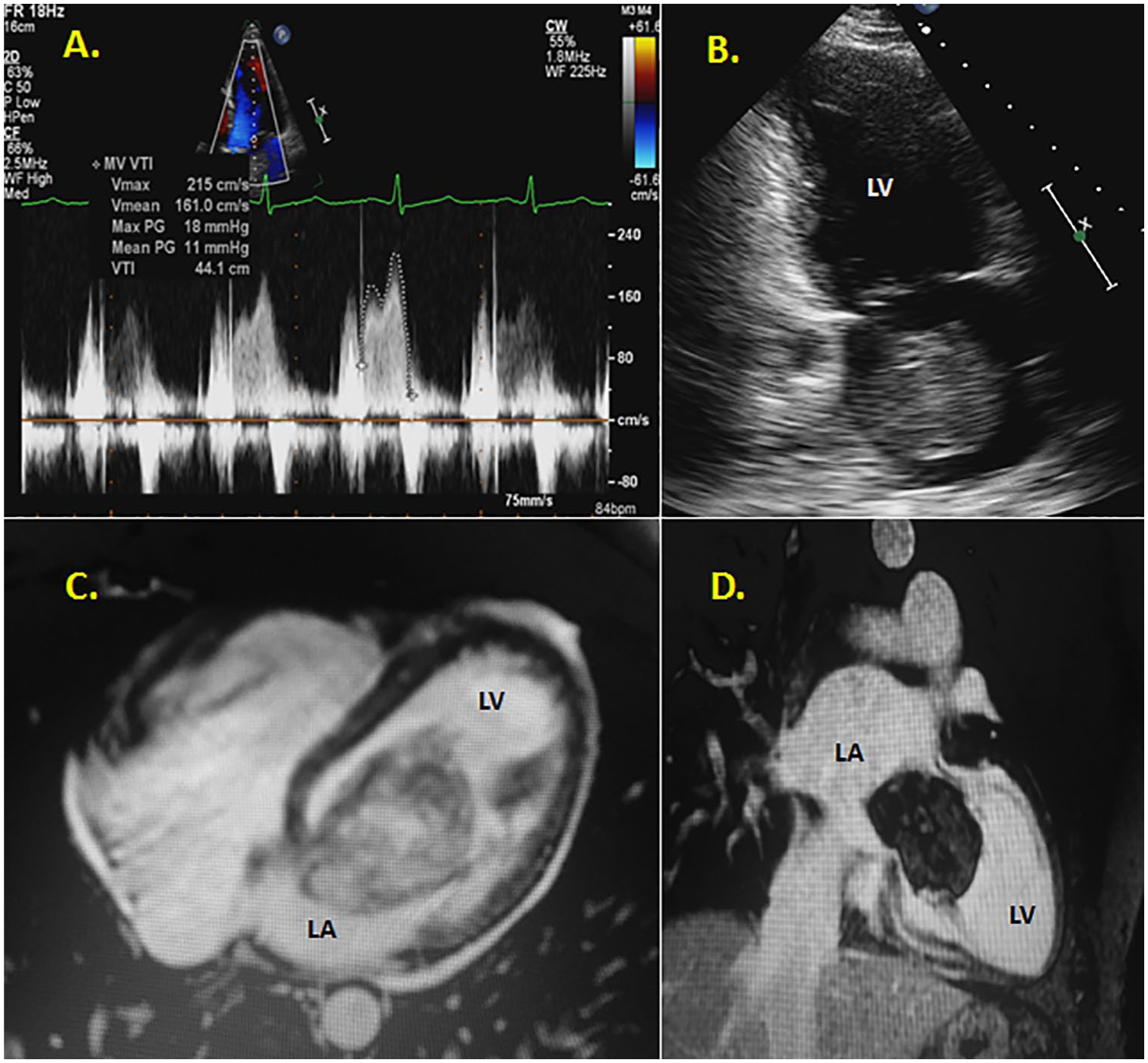
(A) TTE continuous-wave Doppler through the MV mimicking mitral stenosis (mean pressure gradient of 11 mm Hg) caused from the myxoma obstructing the MV orifice. (B) A TTE apical two-chamber view demonstrating the size of the mass in relation to the LA. (C) A cardiac MRI demonstrating the size of the myxoma in diastole. (D) An additional MRI frame showing the size of the myxoma in relation to the heart. LA, left atrium; MRI, magnetic resonance imaging; MV mitral valve; TTE, transthoracic echocardiography.
A cardiac MRI validated the TTE and CT findings, further detailing the mass with an attachment point at the fossa ovalis. On MRI, the mass was measured larger at 6.9 × 5 cm (Figure 5C,D and Figure 6, upper left). In the operating theater, a TEE was performed for intraoperative monitoring (Figure 6, upper right). The atrial mass was again visualized prolapsing into the left ventricle, causing 1 to 2+ mitral regurgitation. The mass was excised using a biatrial approach to resect the mass in its entirety (Figure 6, bottom left). The resulting atrial septal defect was repaired with a bovine pericardial patch. The final pathology report corroborated the initial suspicion of left atrial myxoma (Figure 6, bottom right).
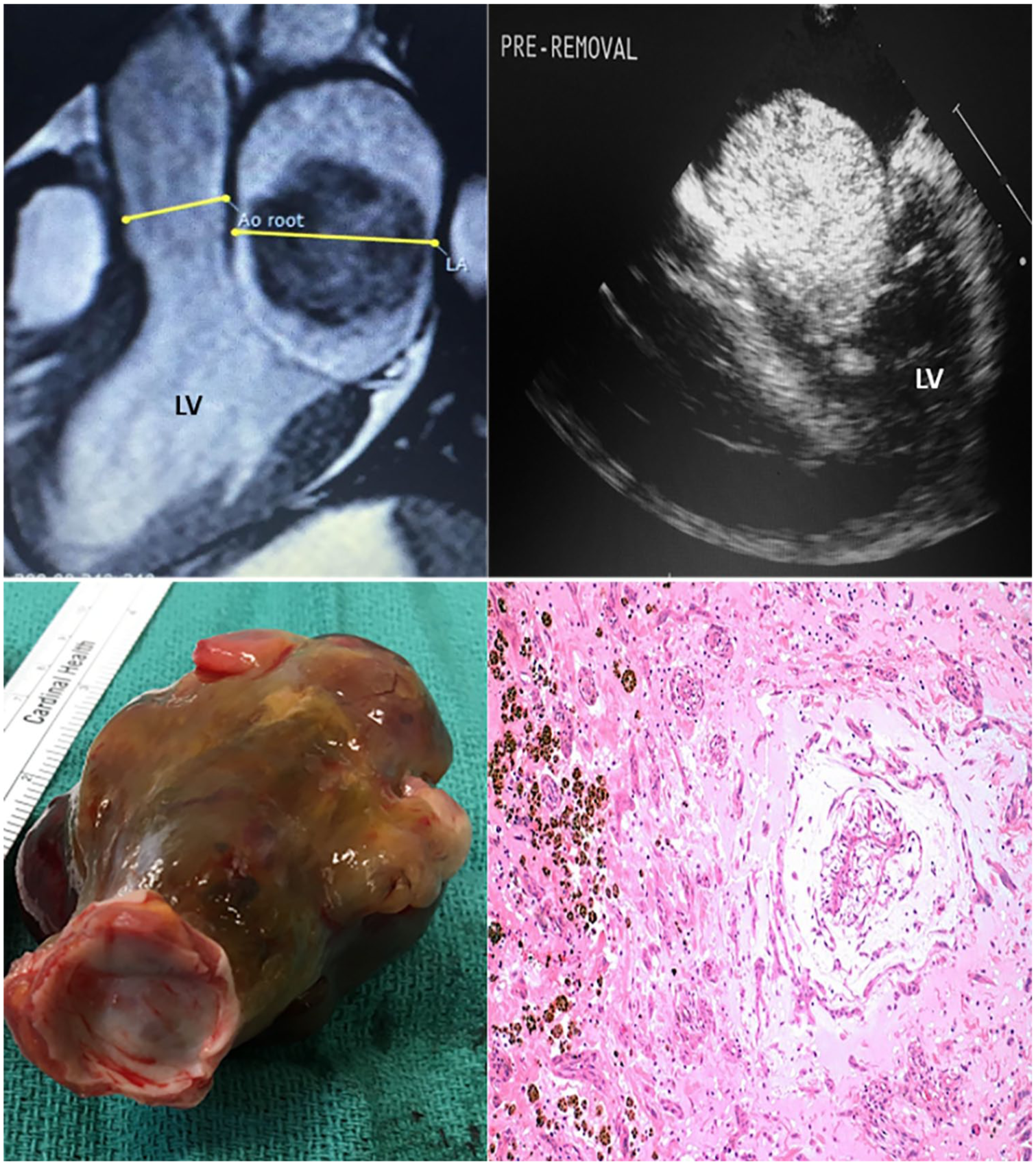
An MRI detailing the size of the myxoma adjacent to the aortic root and LA (top left). A preoperative TEE illustrating the myxoma in diastole (top right). The excised atrial myxoma was sent to pathology. The circular end closest to the reader was the section of the peduncle attached to the fossa ovalis (lower left). Cords of stellate cells appear in a myxoid background typical of cardiac myxoma. Hematoxylin and eosin. Original magnification: 100× (lower right). LA, left atrium; MRI, magnetic resonance imaging; TEE, transesophageal echocardiography.
Case 4
A 65-year-old Indian woman with a history of hypertension, hyperlipidemia, an abnormal electrocardiogram (EKG), and a family history of cancer was referred to her cardiologist to establish cardiovascular care. A routine TTE was ordered due to hypertension and the abnormal EKG. Upon interrogation of the apical window, a 2.0 × 1.5-cm heterogeneous mass was visualized in the left atrium in close apposition with the interatrial septum. Filamentous strands were visualized on the mass and appeared to be mobile (Figure 7A).
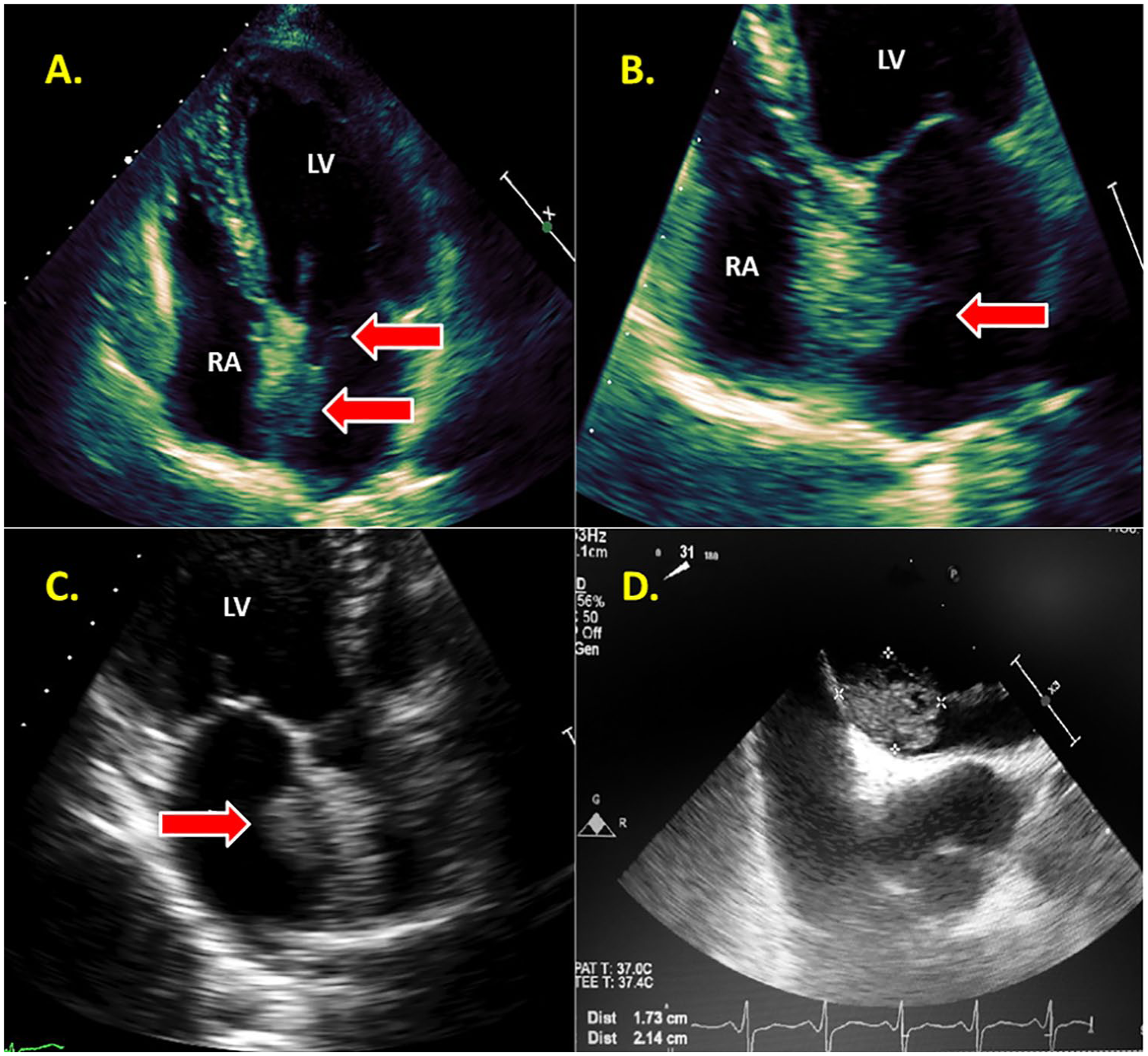
(A) A TTE apical four chamber focused on the left atrial mass with filamentous strands (arrows). (B) An atrial focused apical four chamber demonstrating growth of the left atrial myxoma into the RA. (C) As with any complicated pathology, multiple views and angles are necessary, as seen with this zoomed apical long-axis view. (D) A TEE showing the measurement of the mass connected to the IAS. IAS, interatrial septum; RA, right atrium; TEE, transesophageal echocardiography; TTE, transthoracic echocardiography.
The mass measured 2.6 × 2.0 cm and crossed the atrial septum and protruded 8 mm into the right atrium (Figure 7B). As with any advanced pathology, multiple transthoracic echocardiogram views should be obtained to visualize the lesion from all possible angles. An apical long axis demonstrated the mass in relation to the atrial septum from a third window (Figure 7C). A 2D and 3D TEE detailed the mass as spherical, echogenic, and pedunculated at the limbus of the fossa ovalis (Figures 7D and 8A,B). On CT, a filling defect was visualized in the left atrium attached to the interatrial septum, which was consistent with a myxoma (Figure 8C). Because the myxoma involved a major portion of the left atrial septum and portions of the right atrium, the section of the septum containing the base of the mass was carefully dissected circumferentially. The subsequent defect was repaired with a bovine pericardial patch. The perioperative TEE confirmed the successful placement of the septal patch. The final pathologic diagnosis supported the initial suspicion of a left atrial myxoma (Figure 8D).
(A) Measurement of the filamentous strands protruding off the atrial mass. (B) A 3D TEE illustrating the filamentous strands of the atrial mass, putting the patient at an even greater risk of stroke. (C) A chest CT showing the filling defect in the LA in relation to the IAS. (D) Stellate cells and myxoid stroma with hemosiderin-laden macrophages. Hematoxylin and eosin. Original magnification: 200×. CT, computed tomography; IAS, interatrial septum; LA, left atrium; TEE, transesophageal echocardiogram.
Case 5
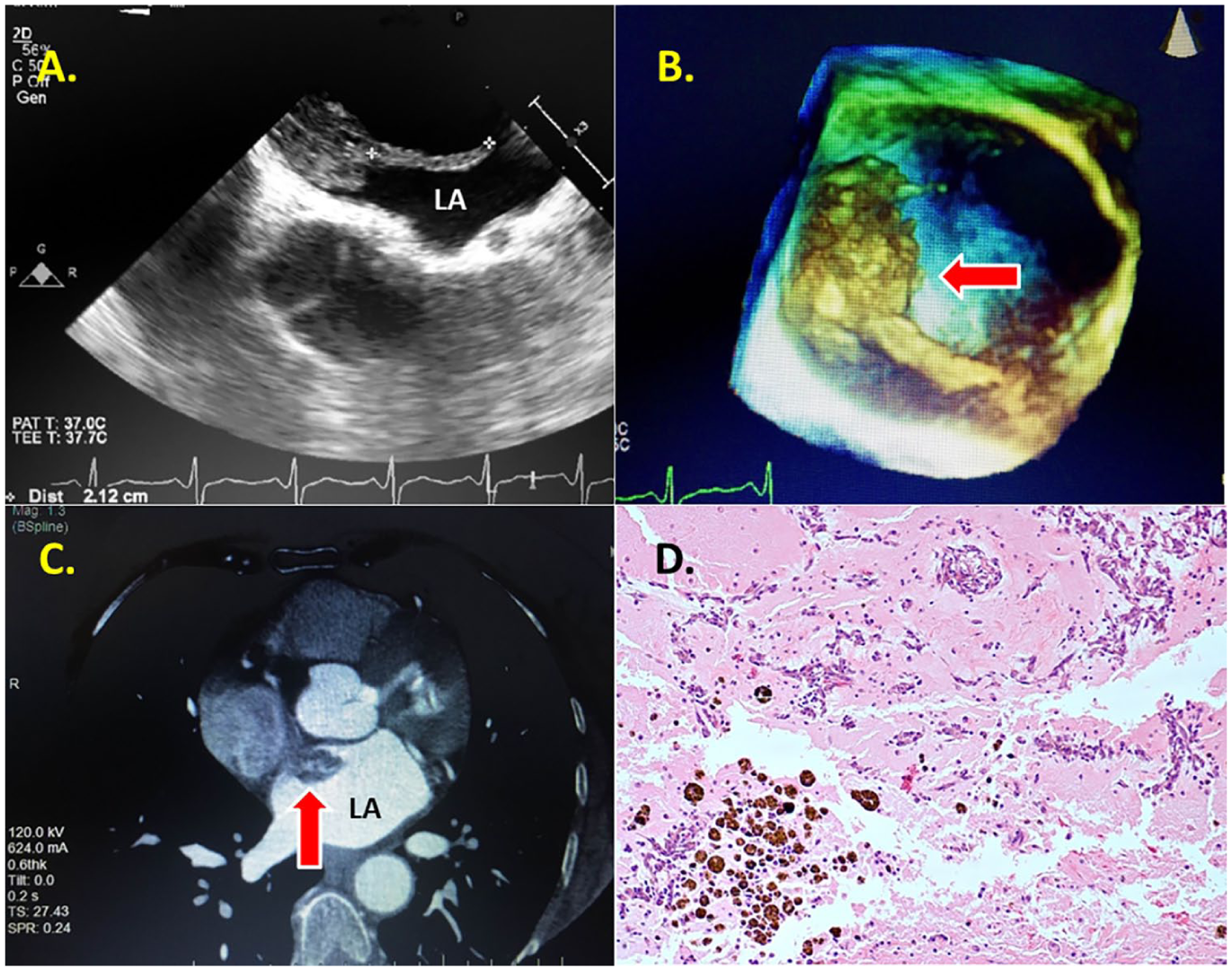
In this case presentation, a 57-year-old woman with a history of takotsubo cardiomyopathy and nonobstructive coronary artery disease presented to the echocardiography laboratory for a resting TTE. A suspicious echogenic structure was located in the left atrium in close association with the interatrial septum visualized from all atrial-focused windows (Figure 9A–C). Due to the technically difficult nature of this study, a TEE was performed to rule out an atrial tumor/mass. The 2D and 3D TEE confirmed the presence of a large, partially heterogeneous, mobile mass with multiple prolonged extensions, occupying much of the left atrium and protruding into the left ventricle, which appeared to emanate from the interatrial septum (Figure 9D and Figure 10, upper left and upper right). A chest CT validated the presence of a left atrial-filling defect, which measured 4.9 × 2.4 cm, consistent with a mass/tumor (Figure 10, bottom left). After surgical excision, the specimen was sent to pathology, where pathologic diagnosis confirmed a benign atrial myxoma (Figure 10, bottom right).
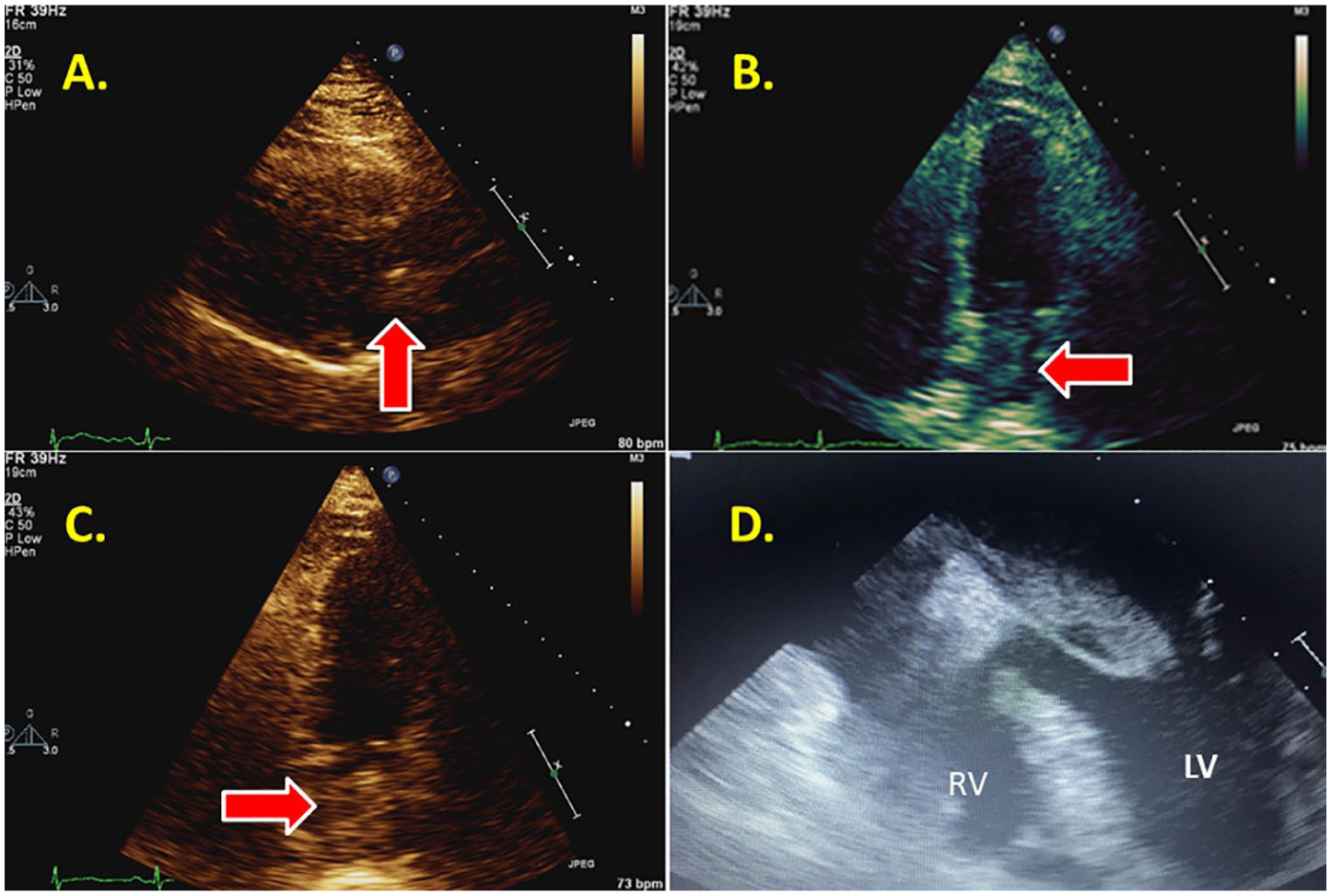
(A) A technically difficult parasternal long axis in a patient with a suspected mass in the LA. (B) A technically difficult apical four chamber of the same patient with a suspected left atrial mass. (C) An apical two chamber demonstrating a suspected mass in the LA of a technically difficult patient. (D) A TEE view illustrating the suspected mass seen prolapsing into the left ventricle. LA, left atrium; TEE, transesophageal echocardiogram.
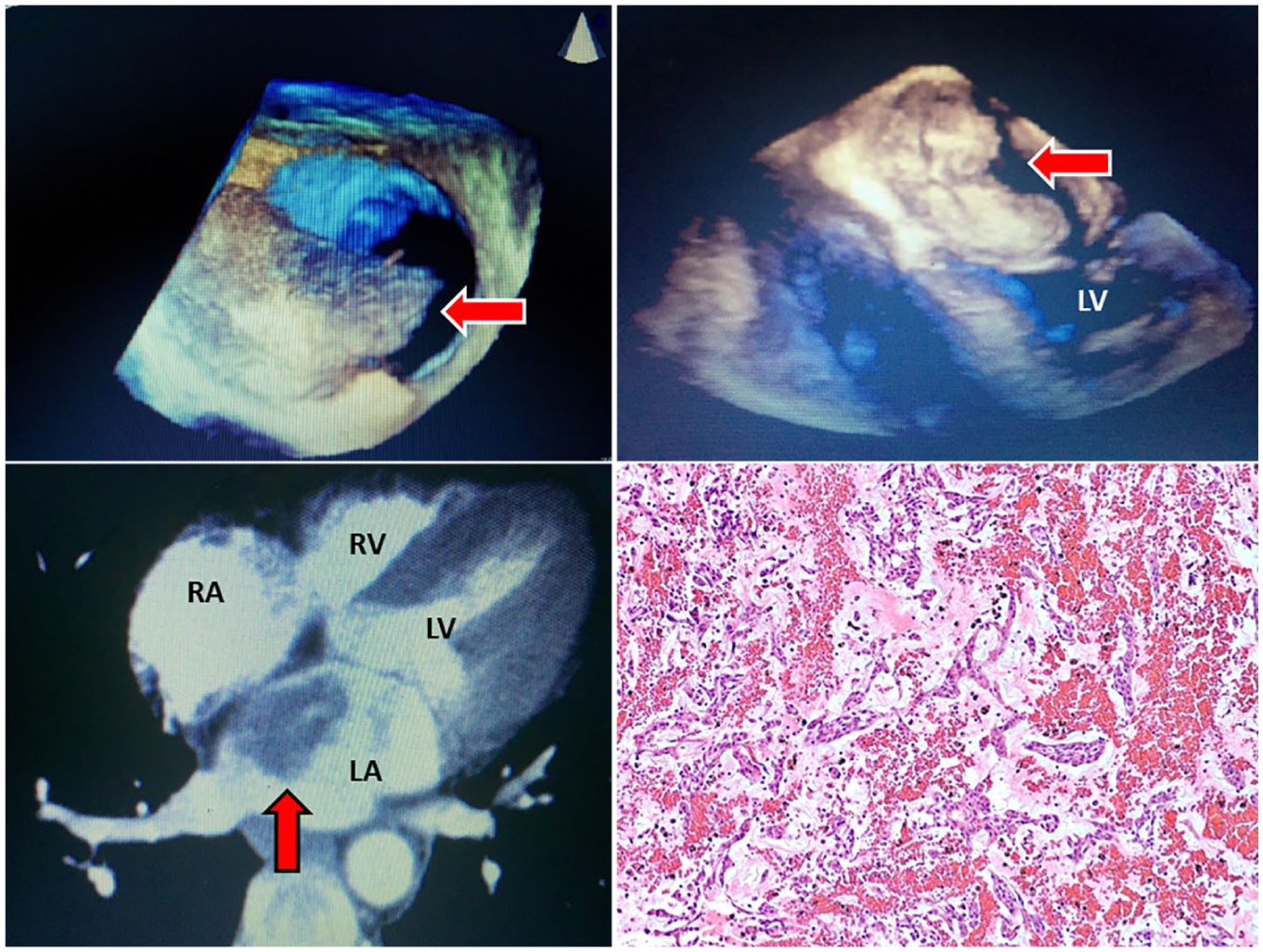
A 3D TEE showing the heterogeneity of the mobile mass (top left). A 3D TEE showing the atrial mass prolapsing into the LV in diastole (top right). A chest CT showing the location and attachment point of the atrial mass (lower left). Stellate myxoma cells and myxoid to slightly hyalinized parenchyma with hemosiderin-laden macrophages and hemorrhage. Hematoxylin and eosin. Original magnification: 200× (lower right). CT, computed tomography; LV, left ventricle; TEE, transesophageal echocardiogram.
Case 6
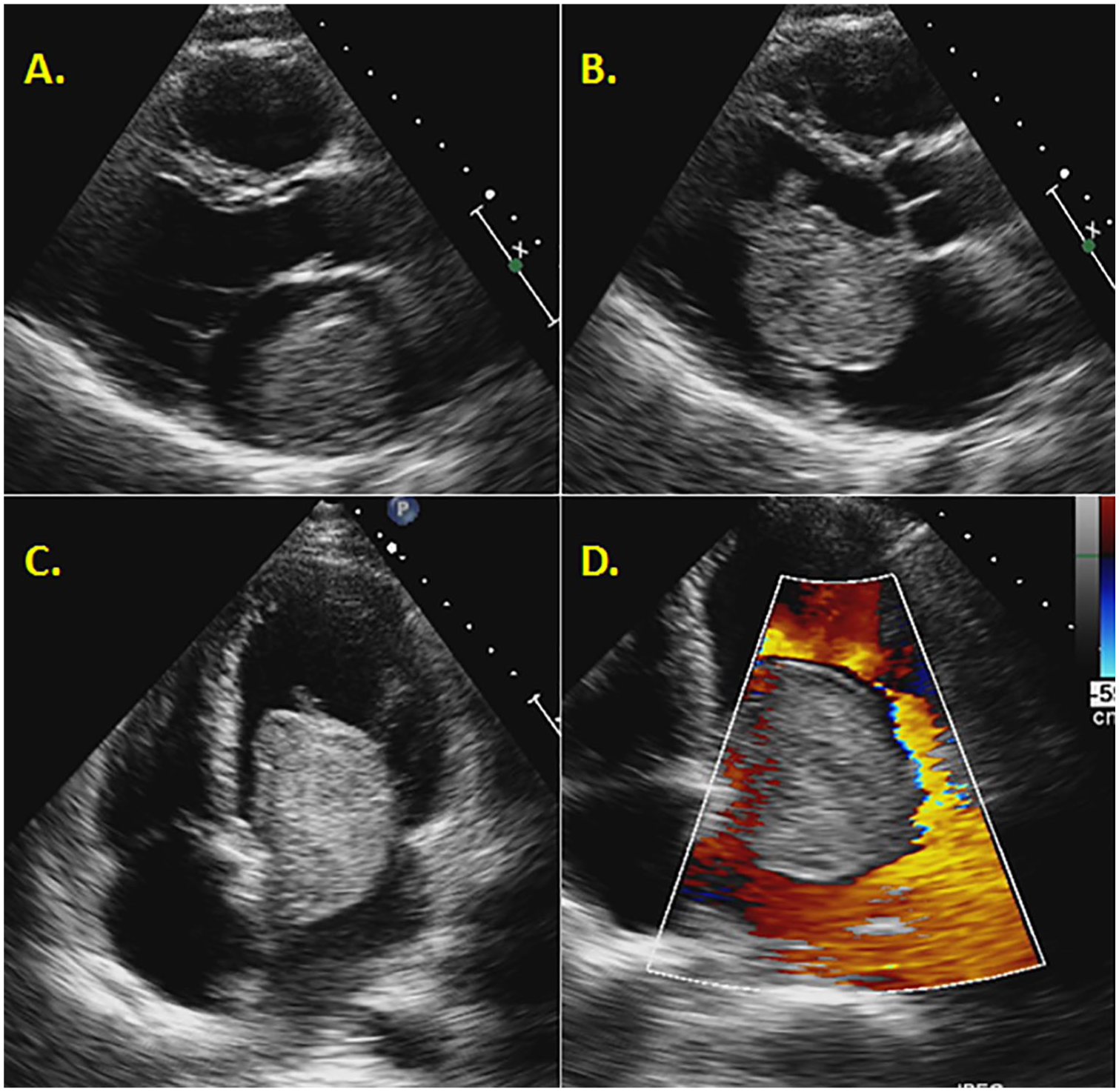
A 68-year-old woman with no history of cardiovascular disease was referred by a primary care physician for cardiac evaluation. A coronary calcium score (CCS) was performed and reported unremarkable; however, an incidental finding of an enlarged thoracic aorta prompted additional testing, including a CT angiography (CTA) of the chest and an echocardiogram. Prior to the second round of cardiac testing, the patient presented to the emergency room with symptoms of severe fatigue and low blood pressure. The patient was diagnosed with dehydration and orthostatic hypotension and responded to intravenous fluids. Following the emergency room visit, testing was expedited and the CTA of the chest confirmed a mild dilatation of the ascending thoracic aorta but also showed a large mass in the left atrium (Figure 11). The echocardiogram was performed and confirmed a left atrial mass emanating from the atrial septum and prolapsing across the mitral valve into the left ventricle. No significant mitral regurgitation or mitral stenosis was associated, and the systolic function was within normal limits. The patient did not report any symptoms of chest pain or any unexplained constitutional symptoms such as fever, chills, or weight loss.
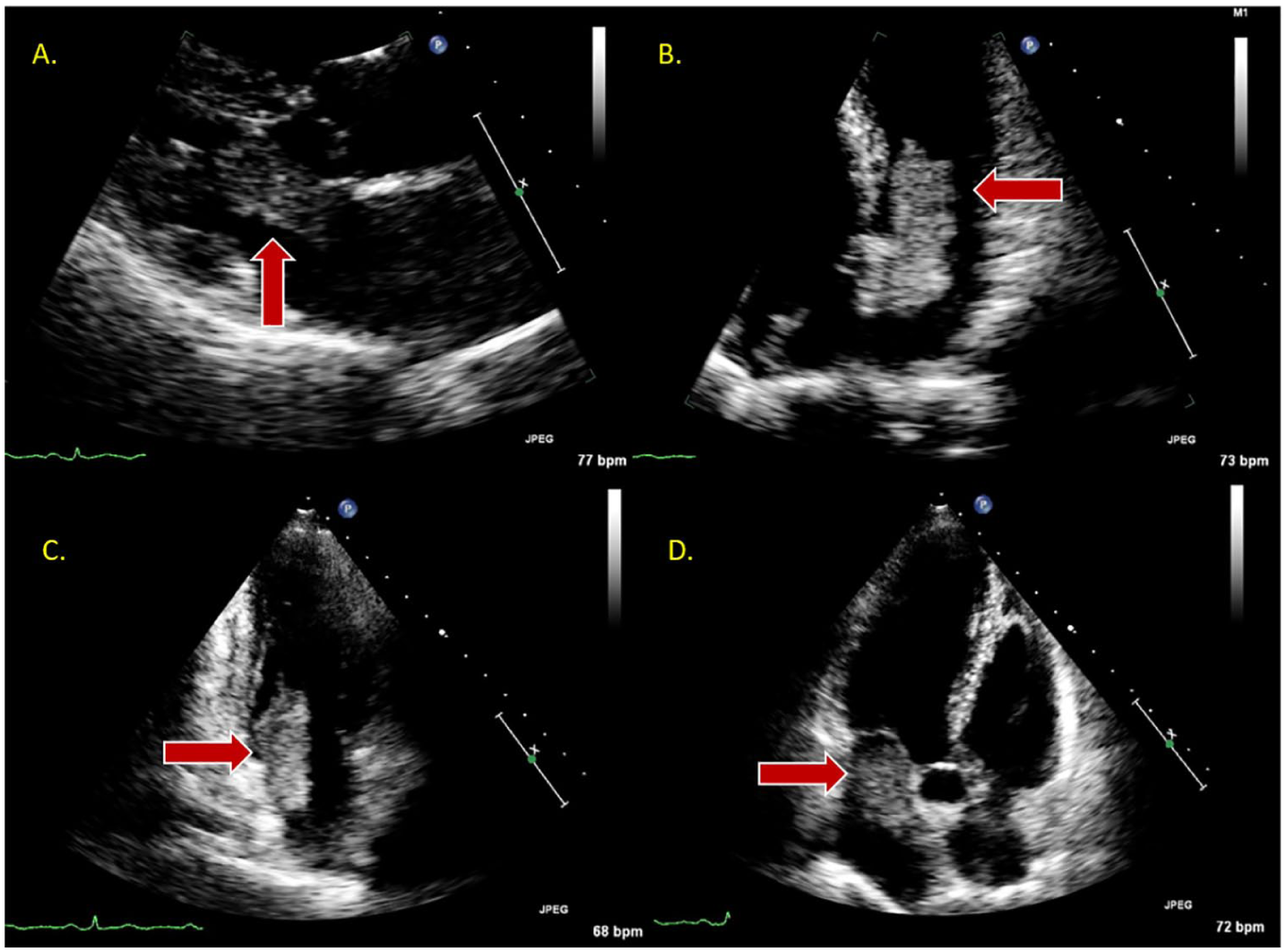
(A) A 2D TTE parasternal long axis in a patient with a suspected mass in the left atrium protruding into the LA in diastole. (B) A 2D TTE apical four-chamber view demonstrating the mobility of the myxoma in diastole. (C) A 2D TTE apical two-chamber view further indicating the location and involvement of both the LA and LV. (D) A 2D TTE apical long-axis view illustrating the myxoma in end diastole. LA, left atrium; LV, left ventricle; TTE, transthoracic echocardiography.
From the initial parasternal long-axis view (Figure 11A), the mass was highly mobile and vivid without any artifact disruption. Views from the apical four chamber showed features consistent with a large left atrial myxoma and measured 4.6 × 1.7 cm (Figure 11B–D). A TEE was performed as a final step before surgical intervention. The TEE confirmed a large left atrial myxoma stemming from the atrial septum and crossing over into the left ventricle during diastole. The patient underwent a robotic-assisted resection of a left atrial myxoma and patent foramen ovale (PFO) closure. The minimally invasive approach included a pericardiotomy and a left atriotomy to remove the mass through the atrial septum. After removal of the myxoma, the large PFO was sutured and the surgery was complete.
Discussion
Cardiac masses visualized with TTE can include foreign bodies, thrombi, and tumors. 1 Intracardiac tumors are classified and described as benign or malignant, primary or secondary, atrial, ventricular, or valvular. 1 Primary benign tumors include myxomas (50%), papillary fibroelastomas (10%), lipomas (10%), rhabdomyomas (8.5%), fibromas (4%), hemangiomas (2%), and teratomas (1%). Of all primary cardiac tumors, myxomas are the most common tumor at 50%. 2 Myxomas are most frequently detected in the left atrium and are most commonly found in women (65%).1,2 However, a small percentage can occur in the right atrium or both atria, as seen in the fourth case study. Other cardiac tumors can be found on valves (papillary fibroelastoma), on the epi/subendocardium (lipoma), and in the myocardium of the left ventricle (fibroma and rhabdomyoma). 2
Myxomas are initially diagnosed by cardiac auscultation or incidentally by TTE.3,4 Many can also go undetected during a postmortem examination. During auscultation, the heart sound results in a “plopping” noise due to movement of the myxoma, as the mitral valve opens during ventricular diastole. 3 In this case, the patient is usually symptomatic with shortness of breath or dyspnea on exertion as the most common symptoms. Although these neoplasms are not malignant, they still pose a threat to the patient’s health. After surgical resection, the recurrence rate of these neoplasms is 3%, requiring serial imaging to evaluate regrowth by echocardiography. 5
Myxomas are typically pedunculated with a stalk, most commonly connected to the interatrial septum. 3 Neurological complications such as TIA and/or cerebrovascular accident can occur in 20% to 35% of patients due to the mobility of the mass and potential embolization of fragments and filamentous strands, as visualized in case 4. 6 Furthermore, due to significant obstruction across the mitral valve that can occur with large myxomas, syncope and, in a rare event, sudden death can occur.7,8 On EKG, more than half of these patients present with atrial flutter or atrial fibrillation because of left atrial dilatation.
The six myxomas described above are significant because although they are all the same tumor, they are remarkably different in size, hemodynamic significance, and appearance. Each has their own differing characteristics in regards to filamentous strands (case 4), multiple-chamber involvement (case 4), and heterogeneity (case 2). It is important to identify the size and, particularly, location of the mass for aid in surgical excision.
Not all myxoma cases will be of excellent echocardiographic quality. When scanning a technically difficult patient (as seen in case 5), myxomas visualized within the left atrium will not always be easy to assess, thus making it challenging to evaluate unique characteristics. While interrogating these types of cases, it is important for sonographers to differentiate between what is artifact caused by cardiac structures or other structures within the mediastinum from what is a true tumor within the heart. Some artifacts associated with TTE move with the motion of a beating heart. When a myxoma is suspected but not clearly discerned, we must look to the context clues of a beating heart.
Since myxomas do not necessarily move in the same fashion with every heartbeat, like an artifact would, sonographers should look for unsystematic myxoma movement that would distinguish it from artifact in challenging case studies. In case 5, the area of concern is demonstrating a mirror-like artifact from neighboring structures. To further investigate this area, multiple windows should be used along with off-axis, nontraditional imaging to prove that any suspicious-appearing structures are true tumors within the heart that are seen from several different views.
Besides traditional views and windows to assess atrial myxomas, off-axis echocardiographic imaging may be helpful in delineating atrial myxomas that are not at expected locations within the heart. As previously mentioned, moving up or down a rib space and visualizing a myxoma from more than one imaging window are important given the nature of different types of artifacts visualized with the use of TTE. As sonographers, when a clear view of the myxoma presents itself, we should implement our calipers and measure the length and diameter of the tumor from multiple angles as well as any filamentous strands protruding off the tumor (as seen in case 4). Measuring these masses and any strands in echocardiography will give the surgeon a better plan with regard to excising the tumor.
Furthermore, in the presence of an atrial myxoma obstructing the mitral valve orifice during diastole (as seen in cases 3 and 6), it is paramount that cardiac sonographers use a continuous-wave spectral Doppler, in addition to color Doppler, across the mitral valve from the apical windows to investigate high-pressure gradients that may mimic mitral stenosis. A velocity time integral and mean pressure gradient will provide cardiologists with the necessary hemodynamic information on the severity of valvular obstruction. In case 3, the mean gradient was 11 mm Hg. From guidelines written by the American Society of Echocardiography, a supportive mean gradient of 11 mm Hg is considered severe mitral stenosis.
The infrequency of myxomas visualized by TTE may result in misdiagnosis, as small masses may not be seen by 2D TTE. A common differential diagnosis of cardiac myxoma is atrial thrombosis.9,10 If a cardiac myxoma is suspected, the use of 3D TTE (when available) and TEE should be used to better delineate the size, location, and point of attachment because of its higher sensitivity in diagnosing cardiac myxomas, as seen in a few of these cases. 1 Supplementary real-time TEE investigation of myxomas is crucial with a technically difficult TTE and when it is challenging to differentiate between a tumor and a thrombus. 8 TEE has better specificity and sensitivity when likened with TTE. 8 For visualization of attachment points, cardiac CT and MRI are helpful in the diagnosis of myxomas because of their larger regions of interest. Furthermore, CT and MRI are not restricted to acoustic limitations that are common with TTE. 1 Although CT and MRI allow for better overall visualization of myxomas, TTE is superior in detecting masses less than 1 mm because of its better spatial and temporal resolution. 1
Microscopically, these tumors are consistent with cardiac myxomas. Under the microscope, myxoma cells are typically arranged in nests and cords of stellate cells in a pale, myxoid background. Hemorrhage and hemosiderin-laden macrophages can also be visualized in pathology slides. There are also focal aggregates of lymphocytes and plasma cells. The cells have round nuclei with inconspicuous nucleoli, eosinophilic cytoplasm, and indistinct cell borders. Rarely, ossification and mucinous glands may be observed. Cut surfaces demonstrate a slightly mucoid soft tissue that is variegated yellow-tan to bright yellow and red, which is common of myxomas. Final pathologic diagnosis is necessary because, as stated previously, these tumors can look similar to thrombus.9,10
Conclusion
The preceding six cases demonstrated the differentiating characteristics of cardiac myxomas encountered in clinical practice in echocardiography followed by other cardiac testing to confirm their presence. Because patients who present with intracardiac masses may be asymptomatic, it is important for the sonographer to carefully delineate intracardiac structures. While assessing atrial myxomas, sonographers should look for key features that are distinctive to myxomas such as a pedunculated stalk, mobility of the mass and valvular involvement, biatrial involvement, and filamentous strands protruding off of the mass. The value of noninvasive imaging in detecting and defining myxomas is evident with these case studies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
