Abstract
Primary cardiac tumor remains exceedingly rare, accounting for 0.5 cases per million of the population annually. We report a case of cardiac tumor, which was initially misdiagnosed as heart failure secondary to acute coronary syndrome. The diagnosis was revised later after routine echocardiography in the ward. A 39-year-old gentleman who presented to the emergency department with cardiac failure and ischemic changes on the electrocardiogram was initially worked-up for acute coronary syndrome. However, echocardiography was performed later in the ward to evaluate the cardiac ejection fraction, revealing a massive left atrial mass measuring 6 cm × 4 cm. Severe mitral regurgitation was noted through the echocardiogram owing to the mass prolapse into the mitral annulus. The initial presentation of cardiac tumors can be ambiguous. Studies have shown that echocardiography as the noninvasive cardiac imaging approach remains the gold standard of diagnostic tool. Hence, echocardiography should be performed as a routine assessment of cardiac failure. Meanwhile, the incorporation of point of care ultrasound in the available center may assist the physician to diagnose accurately in the emergency department setting by excluding other potential differential diagnoses. Delay in diagnosis and initiation of treatment may result in progression to chronic heart failure and dangerous complication, i.e. hemodynamic sequelae and systemic embolism of the clot. In our experience, we managed to reach an accurate diagnosis, timely referral and appropriate intervention despite the lack of point of care ultrasound.
Introduction
The cardiac tumor represents a rare entity of cancer in the population, of which primary cardiac tumor is a sporadic type of tumor, accounting for 0.3–0.7% of all cardiac tumors. 1 The majority (75%) of the primary cardiac tumors are benign myxomas, with an annual incidence of 0.5 cases per million in the population.2,3 Myxomas predominately affect females of middle age, and are commonly situated at the left atrium with septum involvement, followed by the right atrium and exceedingly rarely with biatrial or ventricular involvement.2,4–6
Hemodynamic symptoms such as dyspnea are the most frequent presentation, followed by palpation and syncopal episode. Extracardiac manifestations such as stroke and peripheral embolism are not uncommon presentations.2,4 Left atrial myxoma is more commonly complicated with systemic emboli, affecting the central nervous system, retinal area, renal area, and lower limbs. 7 Rare presentations such as constitutional symptoms and paraneoplastic syndromes had been reported. This article emphasizes the importance of echocardiography as the routine imaging strategy in the assessment of heart failure. We report a case of cardiac tumor, initially misdiagnosed as heart failure secondary acute coronary syndrome owing to the ischemic-linked electrocardiogram changes in a young gentleman. The diagnosis was eventually revised to heart failure secondary to severe mitral regurgitation with underlying cardiac tumor after the echocardiography was performed.
Case presentation
A 39-year-old gentleman with no known history of medical illness presented with worsening dyspnea over the previous three days with reduced effort tolerance and bilateral lower limb edema over the past three months. Otherwise, he was afebrile, had no focal neurological weakness, chest pain, constitutional symptoms, and no significant cardiovascular risk factors. On arrival to the emergency department, he was normotensive with a heart rate of 112 beats/ min in sinus rhythm and comfortable under oxygen supplementation by the nasal prong. The electrocardiogram showed persistent T-wave inversion in lead III and V1–V6 chest leads (Figure 1). The peak creatinine kinase (CK) was raised, 430 U/l, and the troponin T was raised, 72 ng/l. The chest X-ray showed cardiomegaly with bilateral pleural effusion. He was then admitted to the ward for observation and therapeutic intervention for acute coronary syndrome.
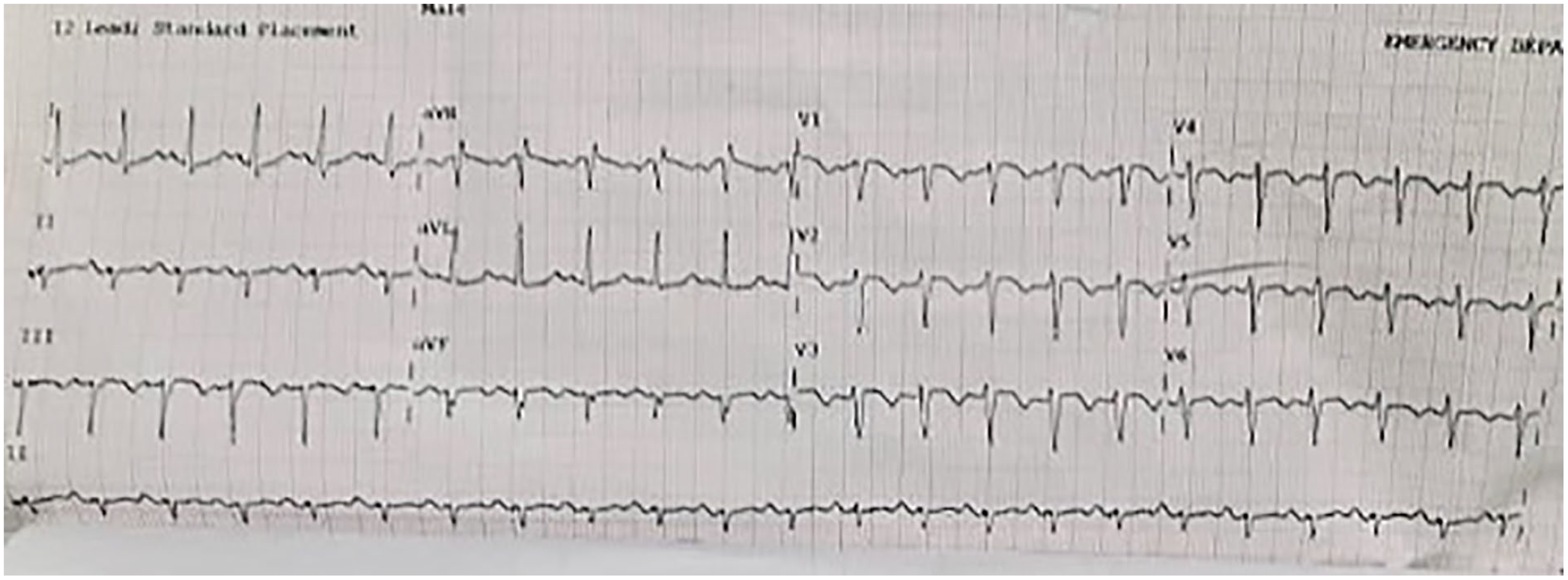
Electrocardiogram shows T-wave inversion in lead III and V1–V6 chest leads.
General clinical re-assessment was carried out upon admission to the ward and revealed a grade 3 systolic murmur on auscultation. Transthoracic echocardiography (ECHO) was performed and revealed the presence of reduced left ventricular ejection fraction of 40% with no pericardial effusion. An isoechoic left atrium mass was found, measuring 5.2 cm × 3.4 cm with evidence of hemodynamic impairment on mitral flow (Figure 2). A coronary angiogram was performed to exclude ischemic heart disease, which showed no significant occlusion. Cardiac magnetic resonance (CMR) imaging revealed a huge left atrial (LA) mass attached to the interatrial septum measuring 5.8 cm × 3.5 cm × 4.6 cm (Figure 3). The CMR with gadolinium enhancement showed heterogeneous enhancement of LA mass prolapsed into the mitral annulus during systole, causing severe mitral regurgitation (MR) (Figures 4 and 5).
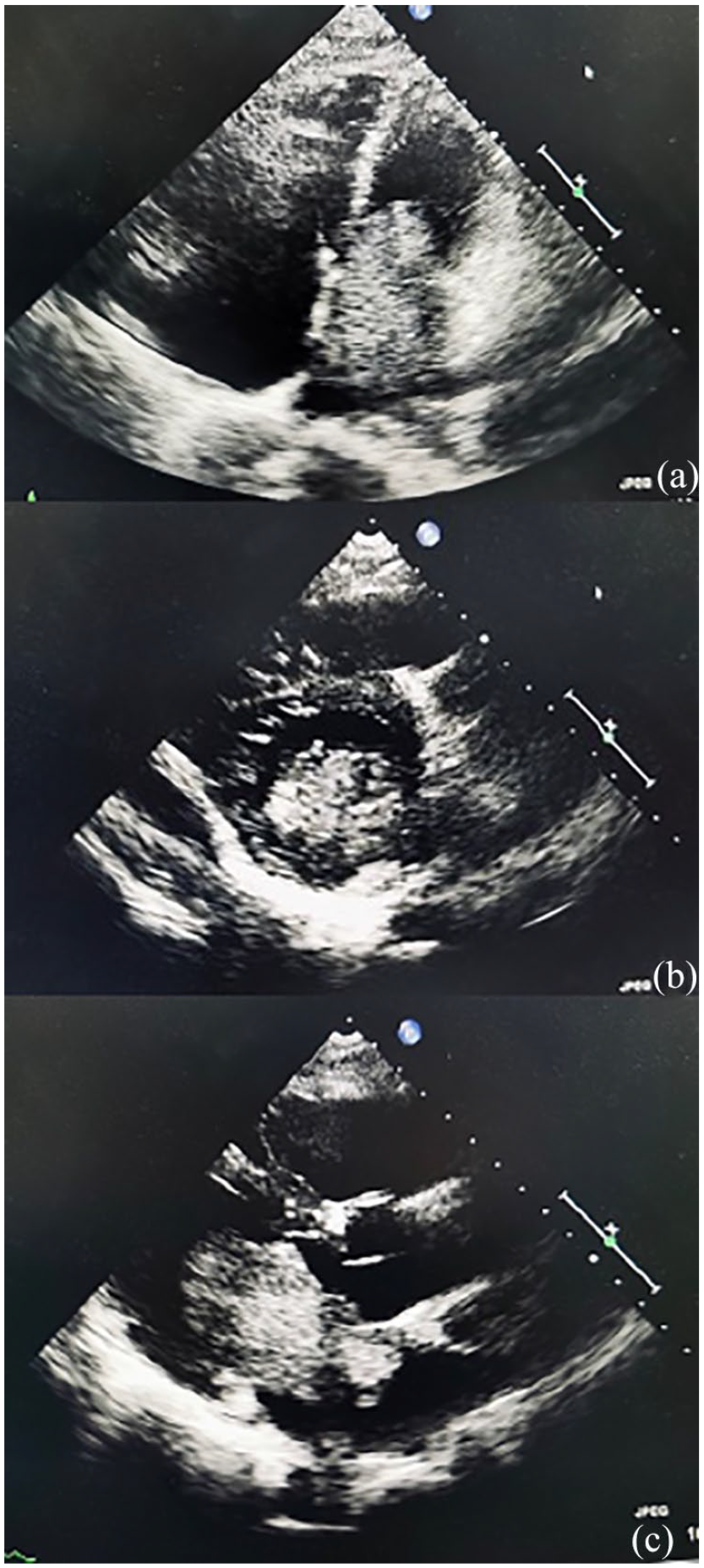
Echocardiography. (a) Apical four-chamber view shows a large left atrial mass with regular surface prolapses into the left ventricle. (b) Parasternal short-axis view shows a mass visible at the left atrium. (c) Parasternal long-axis view shows a left atrial mass protruding into left ventricle during systole.
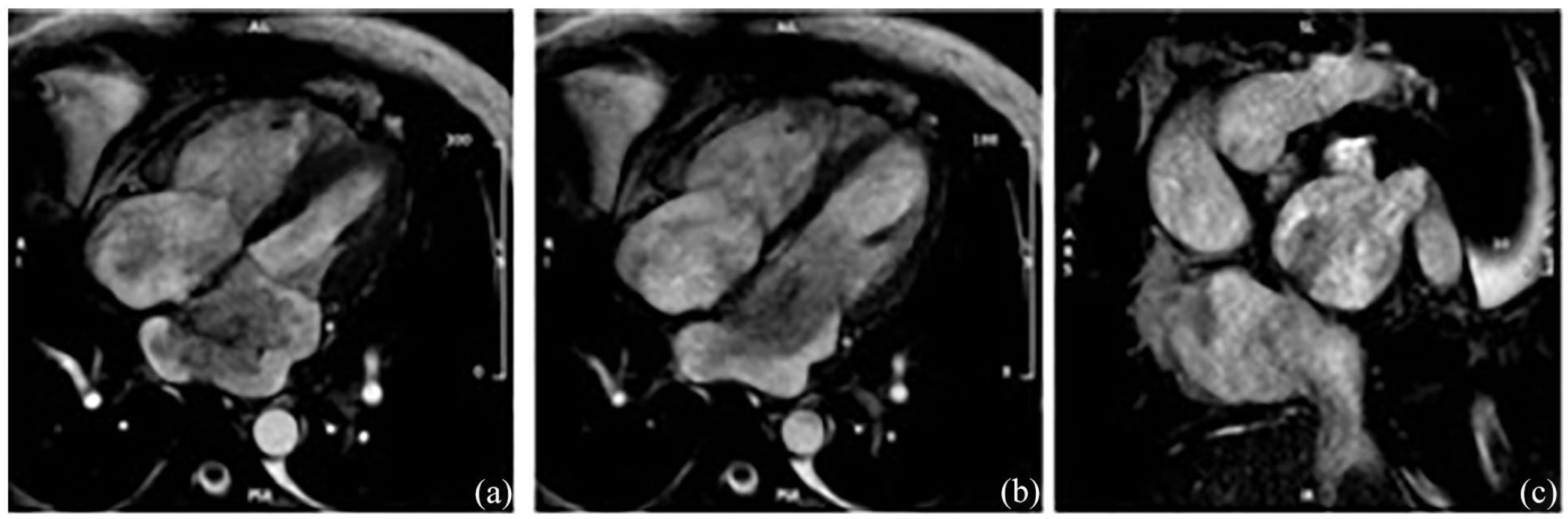
Cardiac magnetic resonance. (a) and (b) Horizontal long-axis view in atrial diastole and systole showing the frond-like mass with a stalk attached to the interatrial septum. It prolapsed into the mitral annulus in systole. (c) Horizontal short-axis view of the left atrium demonstrating the stalk and the mass attachment to the septum.

Cardiac magnetic resonance. Horizontal long-axis view during perfusion (a), early gadolinium (b), and late gadolinium (c) phase. The mass depicts heterogenous, progressing and peripheral enhancement.

Cardiac magnetic resonance. (a) and (b) Vertical long-axis view in atrial diastole and systole showing the frond-like mass prolapsing into the mitral annulus in systole. Early (c) and late gadolinium (d) phase shows peripheral gradual heterogenous enhancement of the left atrial (LA) mass.
After extensive consultation with radiologist and cardiothoracic surgeon, he underwent surgical resection of LA mass for diagnostic and therapeutic purposes. LA myxoma measuring 6 cm × 4 cm × 2 cm was removed intraoperatively. Post-operatively, the patient developed an episode of fast atrial fibrillation which was resolved with intravenous amiodarone. Transthoracic echocardiogram showed no residual atrial myxoma with mild MR. He was discharged home well at day six post-operatively and remained asymptomatic 2 weeks post-operatively during a clinic visit. Histopathology of the specimen was consistent with benign cardiac myxoma.
Discussion
Noninvasive cardiac imaging such as transthoracic echocardiography (TTE) remains as the gold standard diagnostic tool with 95% sensitivity. 4 TTE enables clear visualization and sufficient information on the location, size, and adjacent structures involvement before surgery. Transesophageal echocardiography (TOE) is an invasive diagnostic tool with higher sensitivity compared to TTE. The emergence of point of care ultrasound (POCUS) as a reliable bedside clinical tool that influences decision making and diagnosis provides timely intervention in the emergency department. 8 Several studies have suggested that the utility of POCUS in the emergency setting be a similar ability to detect cardiac pathology accurately compared to the conventional TTE.9,10 However, the common prerequisite would be an experienced or trained operator of POCUS and the accessibility of the POCUS at the center. In our resource-limited center, POCUS was not performed at the emergency department, possibly owing to the lack of trained POCUS operator or the POCUS was not available at the time of presentation. A misdiagnosis on admission was made that was subsequently revised after the standard of care TTE was performed in the ward. Despite the lack, we managed to reach an accurate diagnosis with the TTE and made a timely referral and intervention. An ongoing prospective randomized controlled trial evaluating the impact of POCUS on the length of hospital stays and diagnosis may provide more valuable evidence in the near future. 11
CMR imaging is often used synergistically with TTE, and provides complementary clinical information on myxomas or can be considered in the event where diagnostic uncertainty arises. 12 Meanwhile, the electrocardiogram and chest-X rays provide little to none diagnostic value. Coronary angiography was often performed to rule out concomitant coronary artery diseases, likewise in our case; an angiogram was done given the persistent ischemic changes over the electrocardiogram. Angiocardiography is an obsolete diagnostic tool, commonly adopted in the pre-echocardiography era. 5 Besides, a study demonstrated that elevated C-reactive protein, erythrocyte blood sedimentation rate, and fluctuation of immunological markers such as complement C3/C4, interleukin (IL)2, and IL6 pre- and post-surgical resection of the tumor. 2 Although most of these serum markers are non-specific enough to be adopted as the diagnostic tool, IL6 was recommended as a possible tumor marker that may predict recurrence of diseases. 13
Surgical excision of myxomas was performed with extracorporeal circulation as soon as possible after diagnosis. The aim of surgery is complete resection of the tumor with no residual tissue to reduce the rate of recurrence. At the time, combined surgery for coronary artery bypass, valvular repair, or replacement surgery was done on the same occasion for complicated cases. Valvular regurgitation and cardiac arrhythmias remain as the most common post-operative complication, followed by delayed sternal wound healing, rarely pericardial effusion and pneumothorax.2,14 Pre-operatively, left-sided cardiac myxoma commonly causes mitral stenosis (66.1%) by obstructing the outflow tract followed by mitral regurgitation (33.9%), postulated to be caused by the dilation of the mitral annulus. All cases with mitral stenosis recovered post-excision of myxoma. In contrary, most of the MR cases remained complicated with regurgitation post-operatively even with additional mitral valve repair or replacement procedure.14,15 Our patient recovered substantially post-operative with residual MR without any further mitral intervention intra-operatively.
The prognosis of benign myxomas is excellent, with approximately 80% of long-term survival at 20 years. The rate of recurrence for myxomas are low, estimated to be around 2–5% at 20 years.2,4,5
Conclusions
Transthoracic echocardiography is the essential first-line imaging modality for the evaluation of cardiac failure. Owing to the rare occurrence of cardiac tumor, a high index of suspicion with the aid of the electrocardiogram is crucial for diagnosis. Prompt intervention prevents cardiac and neurological sequelae. The presence of POCUS in the emergency department setting may reduce the delay in diagnosis and subsequently intervention, but may not be widely available at a resource-limited center. Early diagnosis followed by early surgical intervention offers an excellent prognosis with a low recurrence rate for benign atrial myxomas. Mitral regurgitation remains as the most common post-operative complication which may require further interventions. Serial echocardiography and cardiac rehabilitation should be offered to the patient as the follow-up plan.
Footnotes
Acknowledgements
None.
Authors’ contributions
KSSW, KFN, and NO collected and analyzed the data. KSSW and KFN drafted and revised the manuscript. NO and CYL revised and reviewed the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The data used in the literature review are available from the corresponding author on reasonable request.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Hospital Sultanah Aminah does not require ethical approval for reporting individual cases, as the reported case is sufficiently anonymized with no indefinable personal information.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
