Abstract
Routine transthoracic echocardiography (TTE) provides detailed information about intracardiac heart function and physiology. Although statements about the pericardium and visualization of extracardiac areas such as the liver and pleural space are provided in a report, it is often beyond the standard of practice for the cardiac sonographer and interpreting cardiologist to comment on extraneous thoracic abnormalities. This case study demonstrates the findings of a mediastinal thymoma detected by TTE and cross-correlation with thoracic computed tomographic angiography to further detail the existence and location of this incidental finding. The aim of this case study is to alert sonographers and physicians to this particular pathologic entity that was seen during an echocardiogram and can be further investigated for diagnostic completeness.
Keywords
Introduction
Transthoracic echocardiography (TTE) is an ideal imaging tool in the diagnosis of various cardiac diseases. However, it is rare for the results of TTE to include information on extracardiac findings, as it is typically beyond the standard of practice for cardiac sonographers and interpreting physicians. Limiting factors of TTE include the operating frequency (i.e., low frequency harmonics) resulting in poor resolution when imaging superficial structures in the mediastinum. Therefore, additional testing such as computed tomography (CT) or magnetic resonance imaging may be helpful to verify the presence or absence of mediastinal masses. This case study demonstrates how TTE findings precipitated the use of thoracic computed tomographic angiography to confirm the presence of a mediastinal mass, thus streamlining surgical excision, resulting in a positive outcome for this patient.
Case Report
A 54-year-old patient presented to an ambulatory heart clinic with a history of chest pressure, hypertension, tobacco use, and coronary artery disease with a 100% stenosis of the left anterior descending artery and successful percutaneous coronary intervention with a drug-eluting stent. An anterolateral myocardial infarction resulted from the diseased artery. Previous echocardiograms showed the patient’s left ventricular ejection fraction to be 45% to 50%. Due to the patient’s history of a mildly reduced ejection fraction, recent obstructive coronary artery disease involving the left anterior descending artery with subsequent stenting and chest pain resulted in the request for a transthoracic echocardiogram. Upon initial inspection of the parasternal long axis, the ejection fraction appeared to be 50% to 55%.
The technically difficult nature of this echocardiogram allowed the cardiac sonographer to take initiative to scan a large area outside of the traditional scanning angles and views to find the best scanning approach or “window” for imaging. Due to the challenge of locating optimal scanning approaches, many cardiac sonographers use a variety of scanning angles, including off-axis imaging. While scanning the anterior mediastinum, lateral to the heart, a discernible soft tissue mass was noted from the parasternal short axis orientation of the probe. The mass measured 6.2 cm by 6.9 cm on echocardiogram and was encapsulated with a border (Figures 1 and 2). Color Doppler, with a decreased scale, helped to demonstrate evidence of vascularization within the mass, suggesting that the mass was being fed from the body’s main vessels (Figure 3). Computed tomography (CT) or positron emission tomography (PET) is highly recommended for imaging the details of a mediastinal mass and can differentiate malignant neoplasms from benign masses.1,2 A follow-up chest CT was initiated to better visualize the location and presence of the mass within the mediastinum.
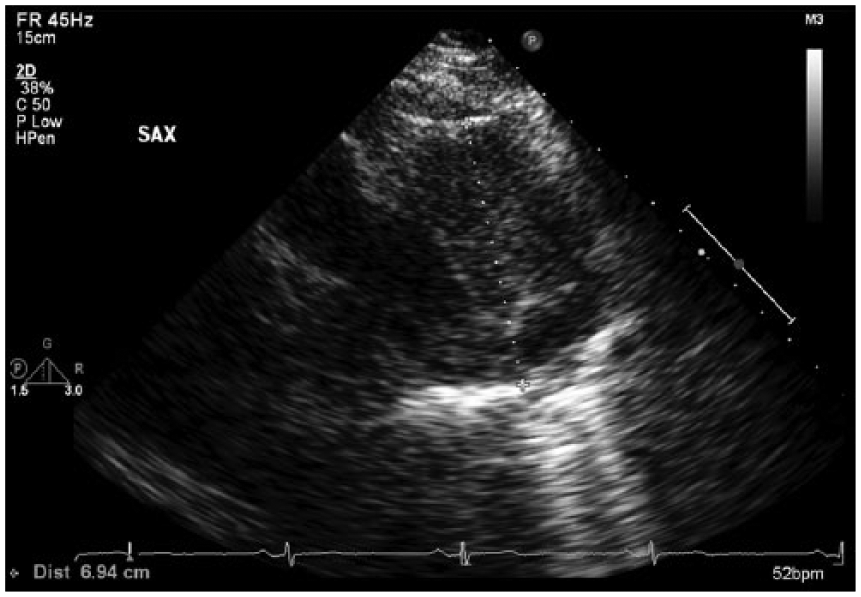
Measurement of the mediastinal mass.
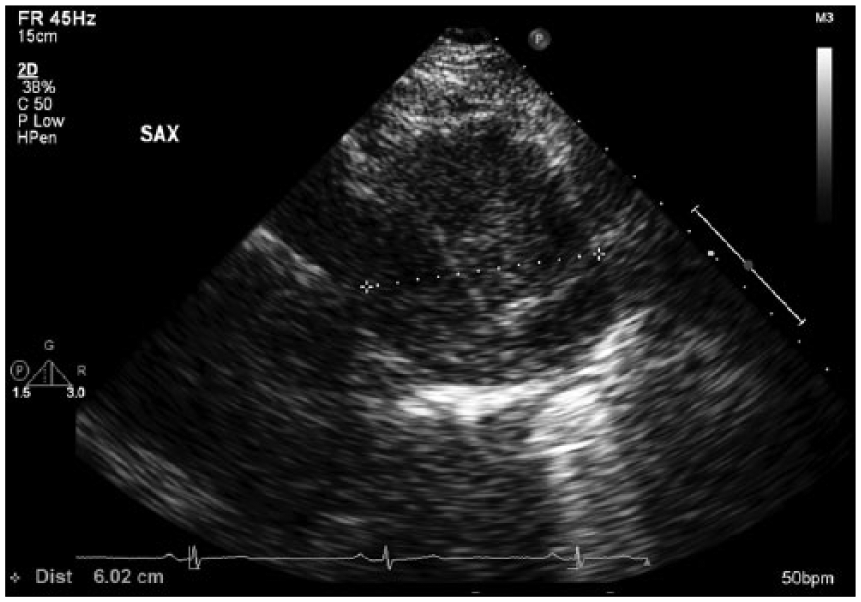
Measurement of the mediastinal mass.
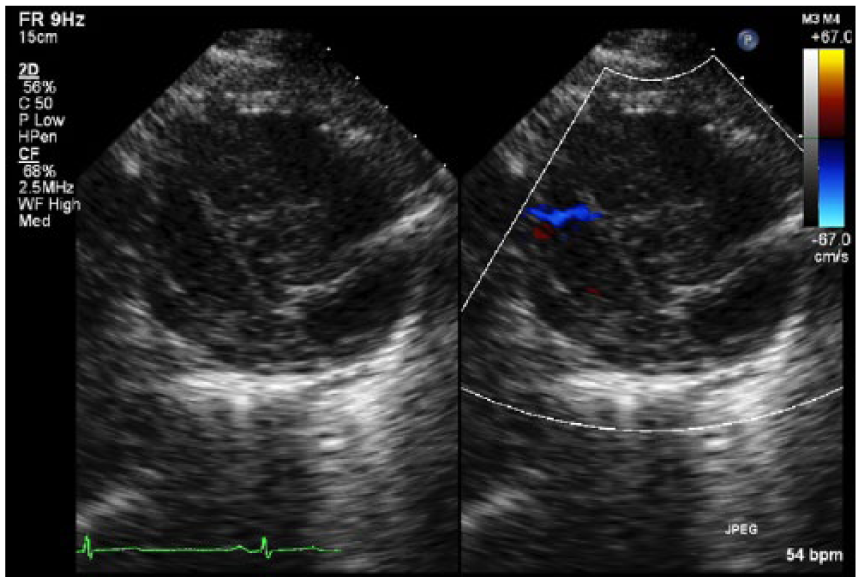
Dual screen imaging showing Doppler color flow within the mass.
A follow-up thoracic CT scan confirmed the existence of an anterior-superior mediastinal mass left of midline (Figures 4 and 5). The mass measured 7.7 cm wide, 8.4 cm antero-posterior, and 7.5 cm cranio-caudal. Other findings in the CT scan were central low attenuation, absence of adenopathy, calcifications within the heterogeneous mass, and visualization of the spleen, which demonstrated innumerable, round, low attenuated lesions, the largest of which being 2.7 cm in diameter. These splenic lesions were thought to represent cysts or hemangiomas. There was no pericardial or pleural effusion present. Effusions can be present when a mass is associated with the pleura. The lungs were clear with the exception of a 5.0 × 1.6 mm linear density along the minor fissure, presumed to be a scar. Without a biopsy or histology of the tissue, the differential diagnosis of the mass was thought to represent that of a thymoma, lymphoma, or teratoma. Based on these findings of the transthoracic echocardiogram and thoracic CT, the cardiologist referred the patient to an oncologist, who in turn referred the patient to a cardiothoracic surgeon. A robotic-assisted resection and thymectomy was the surgical option recommended.
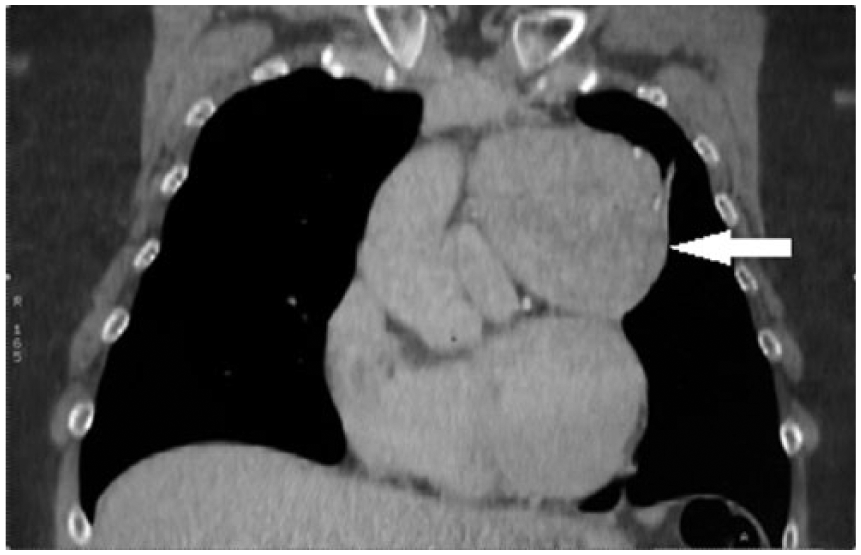
Thoracic computed tomography scan without contrast demonstrating the thymoma adjacent to cardiac structures.
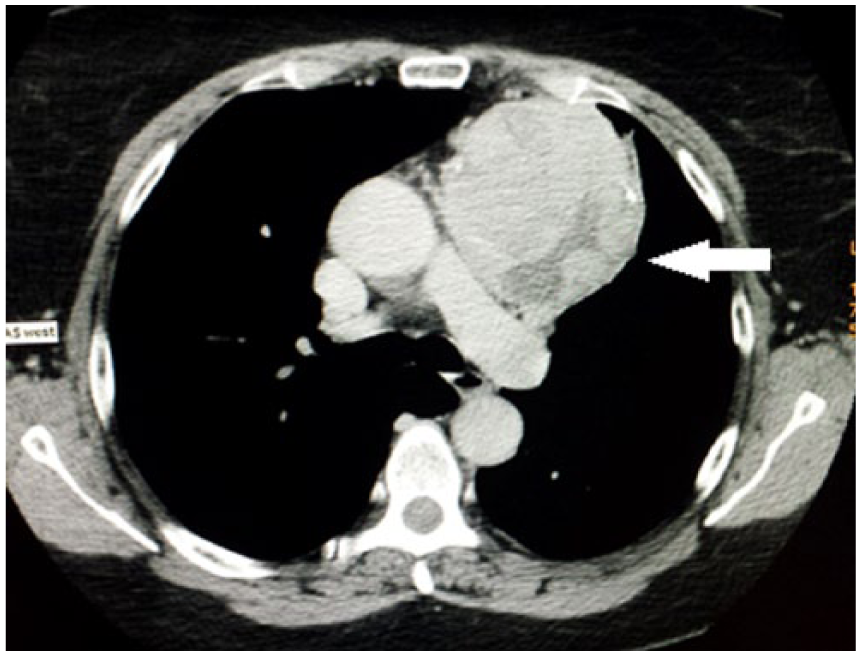
Thoracic computed tomography scan with contrast showing vasculature within the mass.
Upon early visualization in the operating theater, the neoplasm was inspected and seen adjacent to the main pulmonary artery and aorta. Furthermore, the mass had extended into the thymus and invaded the capsule. Upon retraction anteriorly, the mass involved the phrenic nerve. Due to extensive phrenic penetration, attempting to completely separate the mass from the phrenic nerve was unsuccessful. To resume resecting the mass, the phrenic was divided superiorly and inferiorly. The mass was skeletonized from the pericardium, up to the innominate vein, and reflected in the right chest. After a total thymectomy was performed, a specimen of the mass was sent to the pathology lab for further analysis.
Grossly, the specimen labeled “anterior mediastinal mass” consisted of a 263-gram, 9 × 8 × 7 cm aggregate of disrupted, pink-tan soft tissue. The largest fragment was partially surrounded by smooth, pink-tan serosa. The margin of resection was inked black and representative sections were submitted. Cut surfaces of all the fragments demonstrated nodular pink-tan soft tissue with a few scattered bright yellow areas.
Microscopically, the tumor is consistent with a primary thymoma. The tumor cells are arranged in solid sheets with few lymphocytes and have spindled to oval bland nuclei with dispersed chromatin. These histologic features are consistent with type I thymoma according to the World Health Organization’s system of classification. There are focal areas of microscopic transcapsular invasion, which classifies this tumor as stage IIA, according to the modified Masaoka pathologic staging criteria (Figures 6 and 7).
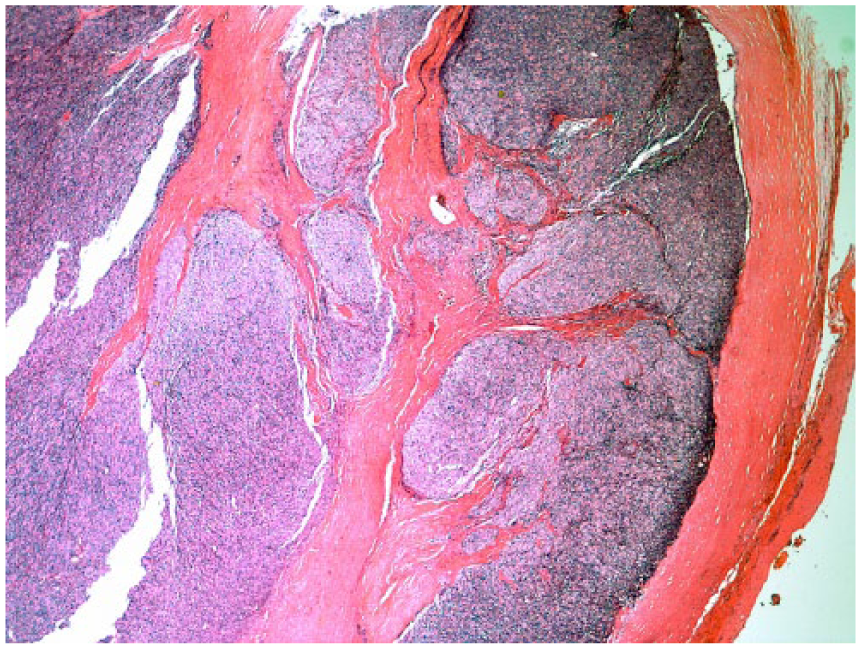
Thymic capsule with extensive infiltration by the tumor, consistent with stage IIA. Hematoxylin and eosin. Original magnification: 20X.
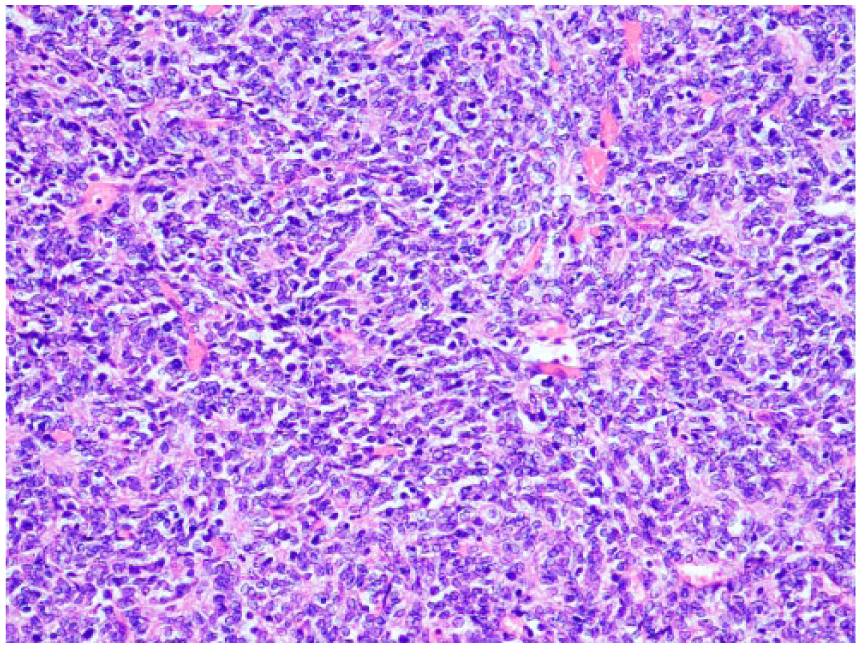
Sheets of spindled to oval cells typical of type IIA thymoma. Hematoxylin and eosin. Original magnification: 200X.
Discussion
Thymomas originate from the thymus and are located in the anterior mediastinum. 3 Although the pathophysiology of the thymus was largely unknown for many years, it is now considered to be central to the lymphatic system. 3 It manufactures T-lymphocytes and processes bodily fluids that are connected to the endocrine system. 3
Thymomas are very rare cancers that have an incidence of 1.5 cases per million people.4,5 They account for only 0.5% of all tumors.4,5 Nonetheless, they are the most frequent tumor of the anterior mediastinum, representing 20% of these tumors.1,6 In the literature, there is a slight male predominance with an estimated 5-year survival rate of 78%. 7 There are no known risk factors and the typical age at presentation is between 40 and 60 years. However, there is an association with myasthenia gravis, which can occur in up to 40% of patients and rarely with other paraneoplastic syndromes. 8
Patients typically have no initial symptoms, and diagnosing this tumor is more often incidental. 1 These masses can be connected with the pleura, pericardium, and thymus gland. The thymoma in this case study measured greater than 8 cm but could have been as large as 12 cm. 9 For surgical and clinical accuracy, a classification system is instituted to further help diagnose the exact details of thymoma.
The Masaoka system is a popular system for categorizing these tumors, and the stages range from I to IV. This system is helpful because although the mass may be a suspected thymoma, cell structure and morphology may be different depending on the typology. 1 An additional factor is whether there is capsular invasion and anatomic involvement with proximal organs; therefore, the system allows surgeons to be better prepared for resection of the thymoma. Generally, if R0 (no residual tumor) resection is achieved for a stage I tumor, then no further adjuvant therapy is needed. For stage II to IV with capsular invasion, if one achieves a R0 resection, one can consider adjuvant radiation to reduce the incidence of local recurrence. For any R1 resection where there is microscopic residual tumor, radiation is indicated in the adjuvant setting. For R2 resection where there is macroscopic residual tumor, one can consider multimodality therapy with radiation and chemotherapy. Locally advanced, recurrent, and advanced disease is generally managed with chemotherapy.
Conclusion
This case study is important as it underscores the potential to overlook this neoplasm when performing an echocardiogram. Two months prior to this transthoracic echocardiogram, a previous echocardiogram was performed at a different facility. There was no mention of a possible echogenic mass found in the proximity of the heart. Although the thymoma was located outside of the normal parasternal views, the mass could be partially visualized from the subcostal view, hinting at an extracardiac mass. Capturing multiple views of the mass is essential in eliminating artifact etiology and demonstrating that the mass can be seen from various scanning “windows.” Proficiency, education, and experience are crucial for sonographers as they evaluate the heart and surrounding chest anatomy. The sonographer has the unique responsibility to provide as inclusive a clinical examination as possible, which affects the accuracy of the diagnostic results as well as detecting other existing abnormalities.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
