Abstract
The craniocaudal (CC) length of the right lobe of the liver (RLL), liver texture, size of the main portal vein, and hemodynamics of the hepatic artery were sonographically evaluated in female diabetic and nondiabetic patients. A one-way analysis of variance, a Tukey post hoc test, and a Kruskal-Wallis with post hoc Dunn’s test were employed. Nonsymptomatic differences in liver anatomy were detected among patients with noncontrolled type 2 diabetes. They exhibited the longest CC length of the RLL (P = .04) as well as an enlarged main portal vein (P = .04). The hepatic artery resistive index was higher among patients with controlled type 2 diabetes (P = .04). These differences were not attributed to fatty infiltration. Patients with noncontrolled type 2 diabetes exhibited significantly higher alanine aminotransferase, lower high-density lipoprotein cholesterol, and higher triglyceride levels than non–type 2 diabetes patients. Longitudinal sonography may provide valuable diagnostic information in the management of type 2 diabetes.
According to the World Health Organization, 1 type 2 diabetes is an epidemic disease, the most common metabolic condition worldwide, and a public health problem. It is produced by several mechanisms including, but not limited to, defects in synthesis, secretion, and/or action of insulin. Hyperglycemia, compensatory hyperinsulinemia, decreased glucose consumption by peripheral tissues, and increased lipolysis have long-term detrimental effects on the anatomy and physiology of the liver.2–4 In the United States, the prevalence of type 2 diabetes is quite similar between the sexes.1,5 Nevertheless, previous studies have shown that a salient risk factor for developing the condition is obesity, which is more prevalent among women. 5 In addition, an atypical menstrual cycle is also a risk factor of type 2 diabetes. 6 For these reasons, this study specifically evaluated liver health among nonsymptomatic women.
It is of clinical relevance to monitor liver health as part of a clinical algorithm in managing type 2 diabetes. Although hepatic physiological disturbances associated with this condition are easily diagnosed through blood testing, it may be too late for the patient when changes in liver anatomy are clinically detected. The liver receives blood from the portal venous system and from the hepatic artery, which plays a crucial role in the metabolism of carbohydrates, by balancing blood sugar levels. 7 Insulin resistance is a key component in the pathophysiology of type 2 diabetes. It has been proposed as an important mechanism leading to hepatic stenosis by increasing triglyceride synthesis and hepatic uptake of fatty acids. It is known that compensatory mechanisms associated with the condition produce an increase in the synthesis of new lipids and fatty acids in the bloodstream that are eventually absorbed by the liver.7,8 Indeed, this is one of many pathways that can lead to nonalcoholic fatty liver disease (NAFLD). 8 If not treated, hepatomegaly, steatohepatitis, cirrhosis, or liver cancer can become endpoints in type 2 diabetes.9–12 Similarly, the hallmark of NAFLD is the accumulation of excess fat in the liver that is not caused by alcohol abuse but is strongly associated with obesity and metabolic syndrome.13–15 NAFLD is also considered an epidemic worldwide, as 30% of the population develops NAFLD. This number increases to 50% to 75% among obese people and up to 78% in patients with type 2 diabetes.1,7–18 In addition, it is a tenet that fat infiltration alters liver size, decreases portal vein velocity, and reduces the hepatic artery resistive index (RI).16,19,20 Unfortunately, data on liver size, portal vein diameter, and hepatic artery RI among females with type 2 diabetes is scarce. Therefore, this study was designed to address a gap in the field by sonographically evaluating the pathophysiological correlates of type 2 diabetes on the liver.
Materials and Methods
Subjects and Recruitment Criteria
Women between 21 and 74 years of age were recruited from two clinical sites: a university-based endocrinology hospital clinic in Puerto Rico and an endocrinology clinic associated with a Puerto Rican school of medicine. Exclusion criteria were (1) previous hepatic disease, (2) chronic kidney disease, (3) morbid obesity, (4) hyperlipidemia, (5) pregnancy, (6) right upper quadrant abdominal trauma, (7) alcoholism, (8) cardiac disease, (9) lack of glycated hemoglobin (HbA1c) lab test results, and (10) lack of consent to obtain the required sonographic measurements. The sample was composed of 20 women with non–type 2 diabetes and 20 women with type 2 diabetes (n = 40). All subjects underwent blood testing to measure HbA1c, hepatic enzymes, and lipid profile (see below for details). The sample of 20 patients with type 2 diabetes was stratified according to plasma levels of HbA1c into controlled (n = 9) and noncontrolled (n = 11) type 2 diabetes. Waist circumference (WC), height, and body weight were the anthropometric measures recorded. An abdominal sonogram was performed to evaluate the liver. The protocol of this study was approved by the Institutional Review Board of the Medical Sciences Campus, University of Puerto Rico, and all participants provided written informed consent prior to recruitment.
Clinical and Laboratory Data
Personal and medical history information was recorded, including age; weight; height; time diagnosed with type 2 diabetes; current diabetes therapy; history of hepatic, renal, and cardiovascular diseases; history of depression; smoking; and alcohol consumption. Body mass index (BMI) was defined as weight (kg) / height (m) 2 . 21 WC was measured in the imaging session at the belly button level, where <35 inches was taken as the reference value for a typical female WC. 22 Results of blood laboratory testing were obtained from the patient’s medical record. In this study, the levels of HbA1c, fasting blood sugar, cholesterol, aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), low- (LDL) and high-density lipoprotein (HDL) cholesterol, and triglycerides were taken into consideration. HbA1c levels were used to determine diabetes status. American Diabetes Association HbA1c reference range values was used to classify the patients into a control group (<5.7% or 39 mmol/mol), controlled type 2 diabetes group (<7% or 53 mmol/mol), and noncontrolled type 2 diabetes group (≥7.0% or 53 mmol/mol). ALT, AST, and ALP levels were used as markers of liver function. Laboratory test results were obtained within 6 months before or after the sonographic imaging sessions.
Sonography and Measurement of the Right Liver Lobe
Sonograms were obtained using a Logiq E9 ultrasound machine (General Electric, Boston, MA) and a C1-6-VN 2D convex probe, and scans were performed by an experienced sonographer (B.L.R.-C.). Sonographic images of the liver, portal venous system, kidneys, and spleen were obtained. The patients were fasting or had taken a light meal more than 4 hours prior to testing. The Logic View Application (General Electric) was used to obtain a panoramic image of the right lobe of the liver (RLL) allowing for the visualization of adjacent structures. Specifically, both the right hemi-diaphragm and the inferior tip of the RLL were typically visualized on a sonogram. Patients were placed in a left anterior oblique position (15°–20°) and with the right arm placed above the head to widen the intercostal spaces. They were also instructed to take deep breaths and to hold their breath for a few seconds. The goal was to reduce shadowing by the ribs, to displace the right hemi-diaphragm inferiorly, and to facilitate visualization of liver parenchyma. Whenever necessary, small back-and-forth rotations of the body wall along its axis were used to slightly displace the RLL medially and inferiorly. The caliper measurement line was traced as high as possible and as close to the diaphragm and as parallel as possible to the anterior wall of the liver from the uppermost right hemi-diaphragm visualized in the sonogram, to the inferior tip of the right lobe.23,24 The diagnosis of hepatomegaly was defined as RLL ≥16.5 cm. 25
The velocity (cm/s), direction of blood flow, and blood vessel diameter (cm) were evaluated with patients in a left anterior oblique position. Hepatic artery velocity (cm/s) and RI were also recorded. All measurements were taken twice for each patient, and the mean values are reported.
Sonograms of the right kidney and the spleen were used to compare the echogencity between those and the liver and to grade hepatic fat. 25 The sonographic appearance of diffuse fatty infiltration was evaluated as follows: (1) mild, a minimally diffuse increase in hepatic echogenicity with normal visualization of the diaphragm and intrahepatic vessel borders; (2) moderate, a moderately diffuse increase in hepatic echogenicity with slightly impaired visualization of the intrahepatic vessels and diaphragm; and (3) severe, the marked increase in echogenicity with poor penetration of posterior segment of RLL and poor or no visualization of hepatic vessels and diaphragm.
Statistical Analysis
Data analyses were conducted according to the patient’s diabetes status: patients with non–type 2 diabetes (n = 20), controlled type 2 diabetes (n = 9), and noncontrolled type 2 diabetes (n = 11). Data are expressed as the mean ± standard error of the mean. Statistical analyses were performed with the latest version of R 3.3.4 (Comprehensive R Archive Network, http://cran.r-project.org) 26 and with XLSTAT-Biomed software (version 2018.5; Addinsoft, New York, NY). Normality of the data was assessed by the Shapiro-Wilk test for normality, and homogeneity of variance was evaluated according to normality results. 27 Normally distributed data were analyzed with a one-way analysis of variance followed by a Tukey post hoc test; otherwise, a Kruskal-Wallis test was followed by multiple pairwise comparisons using a Dunn’s test. Statistical significance was set beforehand at the P ≤ .05 level.
Results
Table 1 shows the age distribution of the female patient cohort according to the study groups: for non–type 2 diabetes patients (48.9 ± 2.4 years), for controlled type 2 diabetes patients (59.3 ± 3.3 years), and for noncontrolled type 2 diabetes patients (52.2 ± 3.4 years). In addition, it shows the values for HbA1c, fasting blood sugar, hepatic enzymes, and lipid profile. As expected, HbA1c levels were higher in female patients with noncontrolled type 2 diabetes than in patients with non–type 2 diabetes and controlled type 2 diabetes, F(2, 37) = 72.2, P < .0001. Similarly, fasting blood sugar levels between the controlled type 2 diabetes and noncontrolled type 2 diabetes groups were significantly different, F(2, 37) = 43.6, P < .0001, from the non–type 2 diabetes group (P < .0001). Differences were also found between non–type 2 diabetes and noncontrolled type 2 diabetes groups for ALT, H(2) = 5.99, P = .03; HDL, H(2) = 5.99, P = .05; and triglyceride levels, H(2) = 5.99, P = .04 (Table 1).
Fasting Blood Sugar, Liver Enzymes, and Lipid Panel by Groups. a
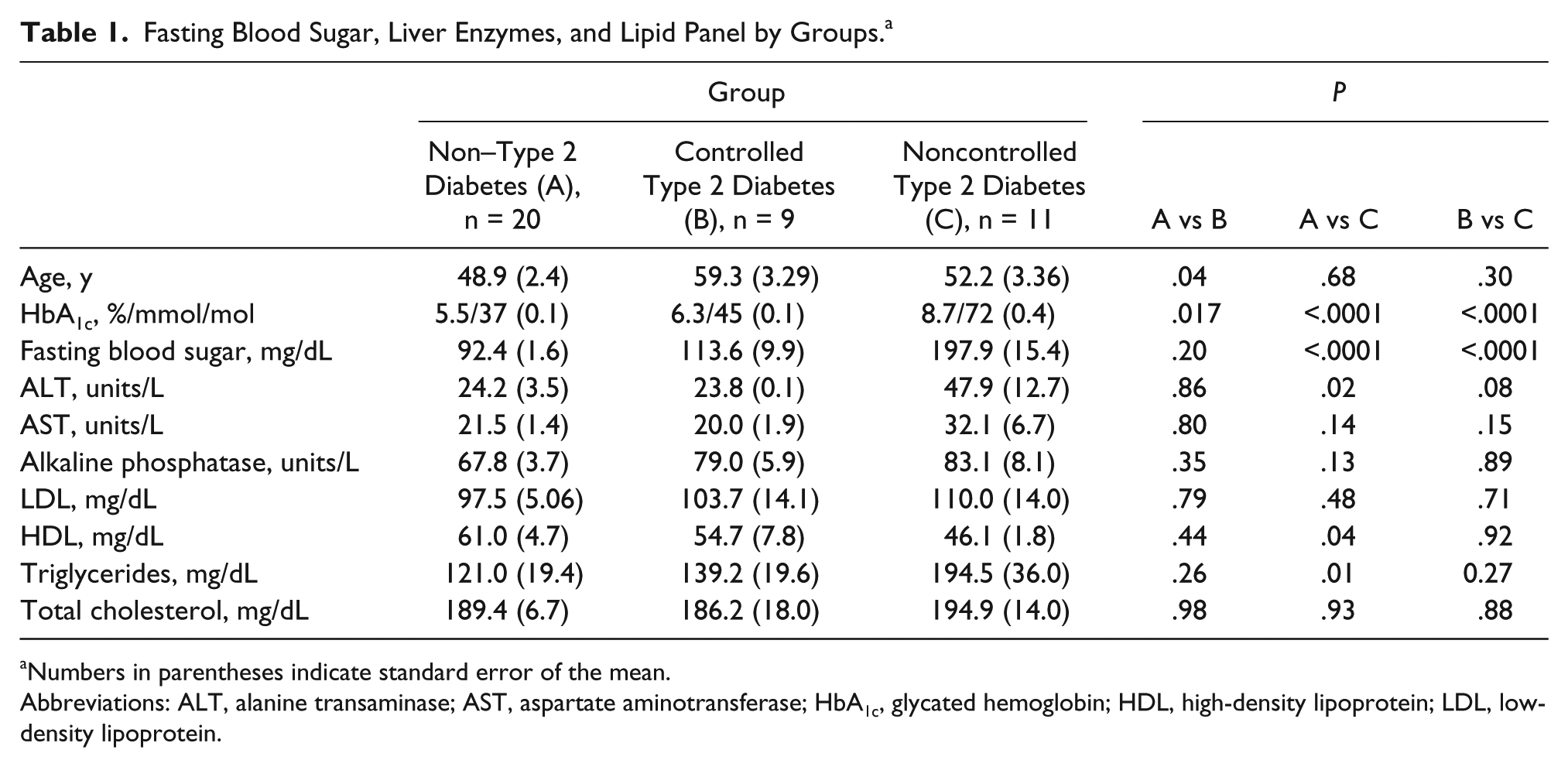
Numbers in parentheses indicate standard error of the mean.
Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Length of the RLL According to HbA1c Levels and Anthropometry of the Patient
The craniocaudal measurement of the RLL was obtained from the uppermost right hemi-diaphragm, visualized on the sonographic panoramic image, to the inferior tip of the right lobe. This was made through a horizontal line, as parallel as possible to the anterior liver wall, which is illustrated in Figure 1A. A significant interaction was found between HbA1c levels and length of the RLL, F(2, 37) = 3.23, P = .05, as well as between the non–type 2 diabetes and noncontrolled type 2 diabetes groups (P = .04; Figure 1B). Plausible relationships between diabetes status and anthropometry were evaluated. When stratified according to HbA1c levels, BMI values were not significantly different between groups. With regard to WC, the noncontrolled type 2 diabetes group had the largest WC, F(2, 37) = 4.25, P = .02, which was statistically significantly different from the non–type 2 diabetes group (P = .02; Figure 1C). Notably, a significant interaction was found between WC and RLL length, F(2, 32) = 3.25, P = .05. Figure 1D shows that such interaction was attributed to significant differences in RLL length between the non–type 2 diabetes and the noncontrolled type 2 diabetes group, with large WC (P = .04).
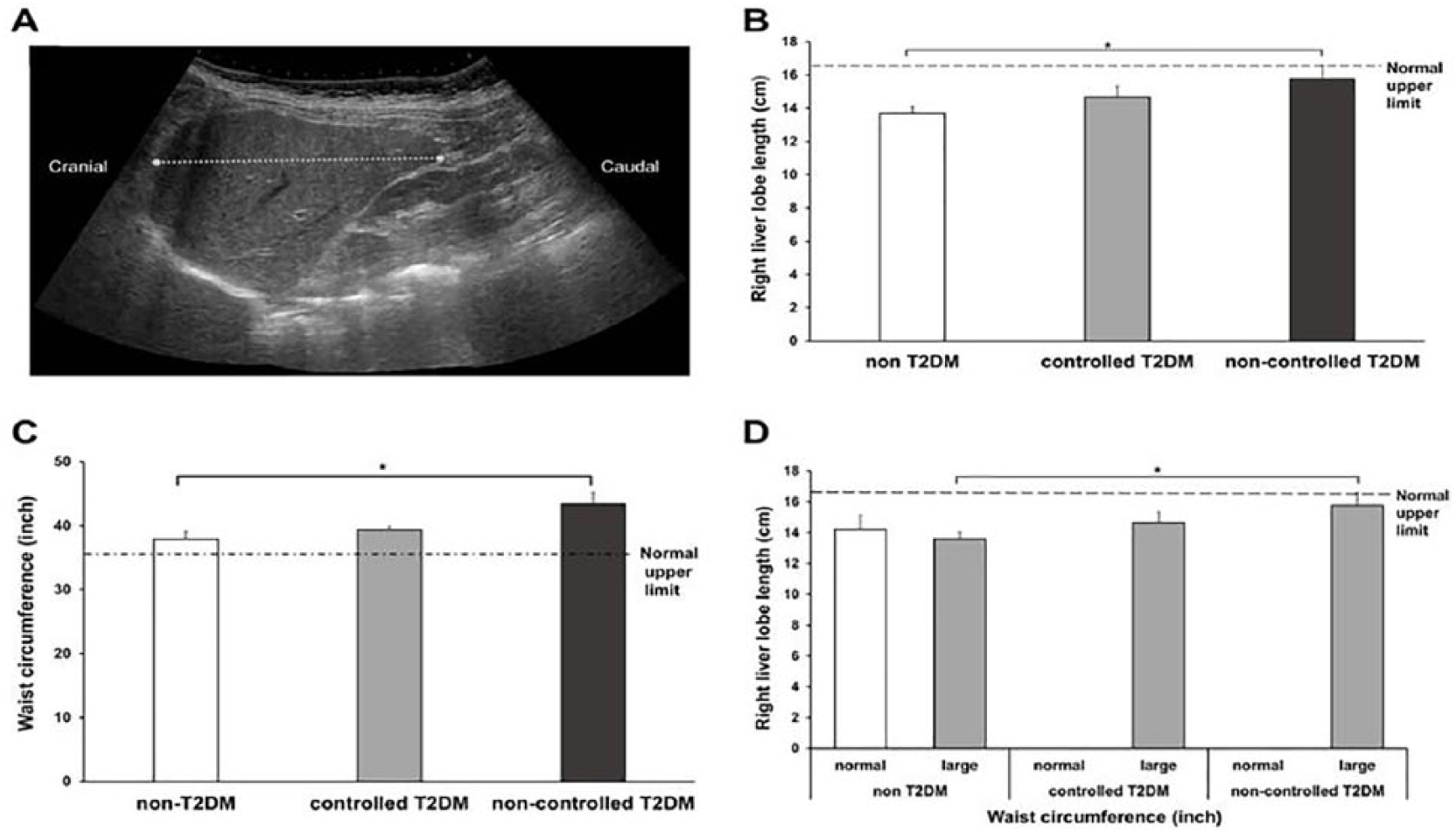
Right liver lobe (RLL) measurement and anthropometric measurements according to glycated hemoglobin (HbA1c) levels. (A) Panoramic sonogram of the RLL. The dashed line represents the craniocaudal (CC) measurement that was taken from the uppermost right hemi-diaphragm to the inferior tip of the RLL, through a horizontal line parallel to the anterior liver wall. (B) Sonographic RLL length according to HbA1c. Classification of HbA1c (% and mmol/mol): non– type 2 diabetes (<5.7%, 39 mmol/mol; n = 20), controlled type 2 diabetes (<7.0%, 53 mmol/mol; n = 9) and noncontrolled type 2 diabetes (≥7.0%, 53 mmol/mol; n = 11). (C) Waist circumference (WC) by groups according to HbA1c levels. (D) RLL length according to WC. Classification of WC (in inches): normal (<35 inches; white bars for panel D, n = 5 in non–type 2 diabetes patients) and large (>35 inches; gray bars for panel D, n = 15 in non–type 2 diabetes patients, n = 9 in controlled type 2 diabetes patients, and n = 11 in noncontrolled type 2 diabetes patients). T2DM indicates type 2 diabetes mellitus. Error bars depict standard error of mean values. *P = .01–.005.
Diameter of the Main Portal Vein and Hemodynamics of the Hepatic Artery
The diameter of the main portal vein (MPV) was related to type 2 diabetes status, F(2, 37) = 3.98, P = .03). Specifically, the MPV diameter was larger in the noncontrolled type 2 diabetes group when compared with the non–type 2 diabetes group (P = .04; Figure 2A). In contrast, a significant interaction was noted between hepatic artery RI and type 2 diabetes status, F(2, 34) = 4.25, P = .02. Figure 2B shows the significant increase in RI between non–type 2 diabetes and controlled type 2 diabetes groups (P = .04).
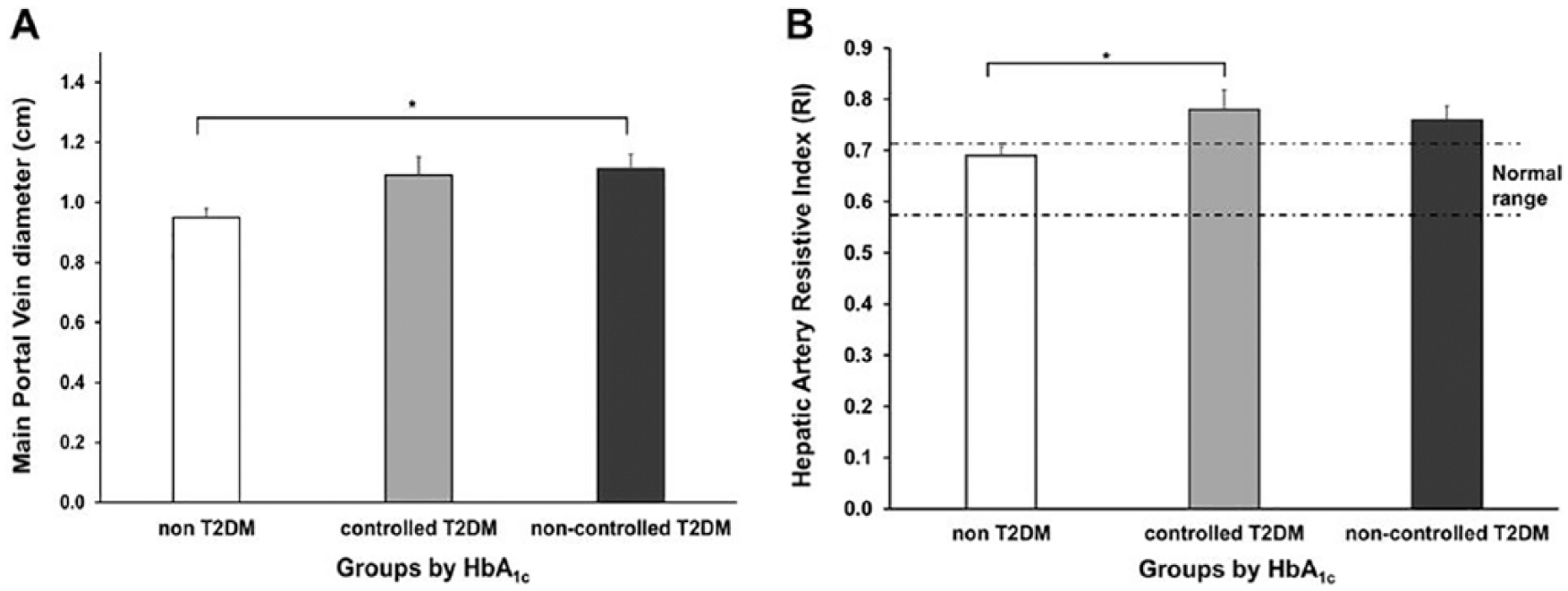
Ultrasonic hemodynamics of the main portal vein and hepatic artery according to glycated hemoglobin (HbA1c). (A) Distribution of main portal vein diameter by type 2 diabetes groups. (B) Distribution of hepatic artery resistive index by type 2 diabetes groups. T2DM indicates type 2 diabetes mellitus. Error bars depict standard error of mean values. *P = .01–.005.
Fatty Liver Grades According to Type 2 Diabetes Status and Nonsymptomatic Hepatomegaly
Table 2 shows the distribution of study groups according to stages of fatty infiltration and in relationship to type 2 diabetes status and nonsymptomatic hepatomegaly. Although no significant interactions were found between these variables, Figure 3 depicts representative sonograms for two non–type 2 diabetes patients and two noncontrolled type 2 diabetes patients. These images demonstrate a qualitative difference in liver texture when compared with the echogencity of the right kidney and spleen.
Fatty Liver Stages by Group.
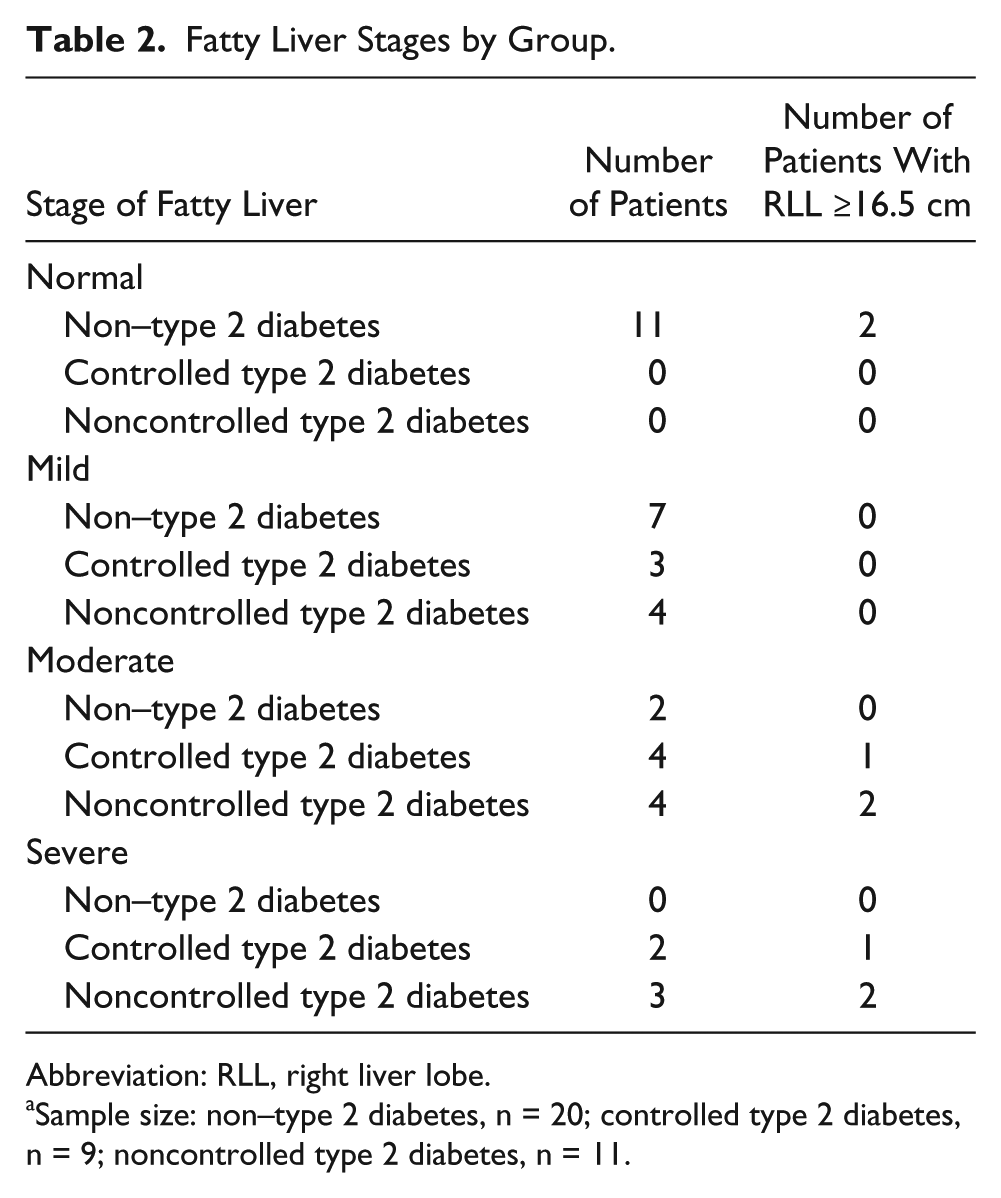
Abbreviation: RLL, right liver lobe.
Sample size: non–type 2 diabetes, n = 20; controlled type 2 diabetes, n = 9; noncontrolled type 2 diabetes, n = 11.
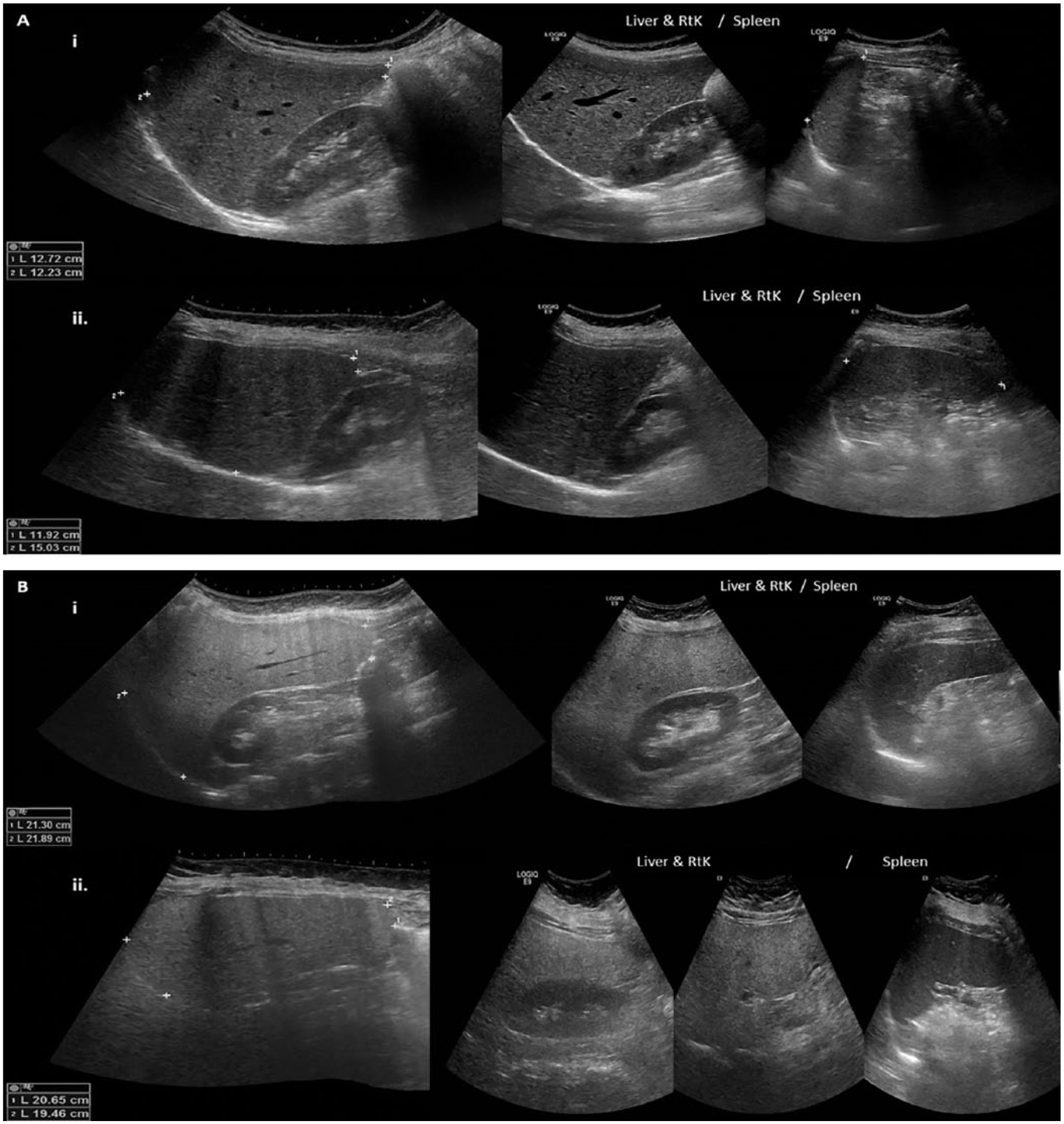
Representative panoramic sonograms of the right liver lobe (RLL), right kidney, and spleen for two non–type 2 diabetes patients (Ai and Aii) and for two noncontrolled type 2 diabetes patients (Bi and Bii). Inserts for each panel show oblique (A) and craniocaudal (B) measurements of the RLL across variations in the anatomy and position of the liver. Whereas Ai and Aii show a normal-appearing liver texture and size of the RLL, Bi and Bii demonstrate a fatty liver and hepatomegaly.
Discussion
Several nonsymptomatic differences in the liver were detected among type 2 diabetes female patients with sonography. First, patients with noncontrolled type 2 diabetes had a longer RLL than non–type 2 diabetes patients. Next, the diameter of the MPV was also larger among noncontrolled type 2 diabetes patients than that of non–type 2 diabetes patients. In addition, the hepatic artery RI was higher among patients with controlled type 2 diabetes when compared with non–type 2 diabetes patients. These differences were not accounted for by fatty infiltration. Finally, the noncontrolled type 2 diabetes patients exhibited higher ALT, lower HDL, and higher triglyceride levels than non–type 2 diabetes female patients.
The finding that the length of the RLL varied according to type 2 diabetes status and WC is of clinical interest. These changes in the length of the RLL were detected with sonography when the measurement line was traced craniocaudally, from the uppermost right hemi-diaphragm to the inferior tip of the right lobe. This was done through a horizontal line as parallel as possible to the anterior liver wall. 23 It may not be possible to trace such a horizontal line in every patient because of an incomplete sonographic view of the RLL. Nevertheless, our goal was to reduce the angle that is produced between the standard oblique measurements of the RLL when extrapolated to a parallel line that is traced along the superior border of the dome of the liver, in relationship to the anatomical plane of the anterior liver wall. 24
In 2013, the American Diabetes Association predicted that by 2050, one of three American adults will have diabetes. 28 This prediction is worrisome given the plethora of subsequent conditions associated with diabetes, including those that affect liver health. In fact, the relationship between type 2 diabetes and fat infiltration by the liver, known as nonalcoholic fatty liver disease (NAFLD), is well established.29–31 Unfortunately, the clinical symptoms of NAFLD are nonspecific until the patient develops liver inflammation or fibrosis. 32 Routine physical examination by palpation is not an effective approach to detect NAFLD. Its diagnosis usually stems from incidental findings from diagnostic imaging studies by sonography, computed tomography, magnetic resonance imaging, or detection of heightened and sustained elevation of liver enzymes over time. Although no single laboratory test is diagnostic for NAFLD, liver function tests and assessment for metabolic syndrome are commonly pursued. Previous studies have shown increased levels of ALT, AST, LDL, HDL, triglycerides, and total cholesterol in NAFLD patients.33,34 ALT has been suggested as a predictor for infiltration of fat in the liver, 32 but it has been shown that normal ALT levels do not rule out liver diseases such as NAFLD and steatohepatitis.30,31 An increase in ALT has been suggested as a marker to identify the risk for developing type 2 diabetes. 33 In the present study, the highest ALT value among the noncontrolled type 2 diabetes group with moderate and severe fat infiltration suggested that elevated ALT correlates with NAFLD. It is of interest to note that ALT levels were not high in the controlled type 2 diabetes group despite mild liver fat infiltration, attesting to the nonlinear relationships between type 2 diabetes, NAFLD, and liver health. Albeit not significant in this patient cohort, a higher proportion of noncontrolled type 2 diabetes patients exhibited RLL sizes greater than 16.5 cm when compared with the other groups, which is indicative of hepatomegaly.
Nonsymptomatic changes in liver texture and the hemodynamics of the hepatic artery were also apparent. Specifically, moderate and severe fat infiltration were noted among type 2 diabetes patients, which suggests qualitative changes in liver appearance that may lead to hepatomegaly if left unattended. Indeed, this seems to be the case for the non-controlled type 2 diabetes group, with RLL size greater than 16.5 cm. Nevertheless, it is important to consider that liver texture may be one of many correlates of hepatomegaly. Similarly, we found that MPV diameter was affected among type 2 diabetes patients, especially among the noncontrolled type 2 diabetes group. This finding is consistent with a previous report showing that the diameter of the portal vein is increased in patients with NAFLD. 19 However, in contrast to a previous report on hepatic artery RI in NAFLD, 20 this study demonstrated a high hepatic artery RI only among women with controlled type 2 diabetes. It is important to note that an elevated hepatic artery RI is related to diffuse distal microvascular disease. 35 A hepatic artery RI above the normal limit was observed in both type 2 diabetes groups, even when classified by fat infiltration stages (RI >0.70), suggesting that heightened hepatic artery RI reflects early liver hemodynamic changes produced by type 2 diabetes.
In patients with type 2 diabetes, insulin deficiency activates lipase in adipose tissue, which leads to an increment in fatty acids in the bloodstream. As a homeostatic mechanism, the liver increases the absorption of these fatty acids. If the synthesis and delivery of free fatty acids exceed their capacity to oxidize and to export them, fatty infiltration or hepatic steatosis of the liver develops, and hepatic tissue function is compromised. In addition, it has been shown that gluconeogenesis and hepatic glycogenolysis is increased in patients with type 2 diabetes, causing hyperglycemia, which in turn causes compensatory hyperinsulinemia.7,8 These compensatory mechanisms induce synthesis of new lipids, contributing to the development of hepatic steatosis or NAFLD, which is well known to affect liver health, 16 as these study results suggest.
Estrogen levels in women are associated with the risk of developing type 2 diabetes and NAFLD. Previous studies have shown that women with atypical menstrual cycles have a higher risk of developing type 2 diabetes. 5 Similarly, it has been shown that estrogen deficiency in postmenopausal women is related to the risk of developing severe NAFLD and liver fibrosis. 36 In fact, estrogen deficiency in postmenopausal women has been related to an increased risk of developing severe NAFLD and fibrosis, which are associated with the length of estrogen deficiency. 36 Whether estrogen has a protective role in liver injury related to NAFLD still needs to be elucidated. In this patient cohort, the average age was greater than 48 years. Therefore, it is of interest for a future study to evaluate liver texture and liver fibrosis by sonography among women of reproductive age with type 2 diabetes.
Commonly, diagnosis and monitoring of chronic liver disease are carried out through image-guided laparoscopy and liver biopsy. Both methods, despite being very useful, have the disadvantage of being invasive, painful, and expensive procedures. It is estimated that the cost of liver biopsy with medical insurance ranges from $1,032 to $ 2,745 US dollars. To reduce the risk of liver biopsy complications, image-guided liver biopsy has been recommended for patients with small livers that are difficult to percuss, obese patients, and those with clinically evident ascites. 37 This study pinpoints a venue for future research on the pathophysiological consequences of type 2 diabetes on the liver, documented with sonography. Sonography is a noninvasive and nonionizing imaging approach that may be useful in long-term monitoring of this disease.
Study Limitations
This was a study of a convenient sample of patients who were evaluated once with sonography. Given the individual trajectories of this disease process, it would be advantageous to conduct a longitudinal study with a larger sample size. To better understand plausible changes in the length of the RLL through time, it would be informative to measure liver stiffness by elastography. As was shown in this study, RLL length is a relatively simple sonographic measurement that can be obtained in a fast-paced clinical environment. Nevertheless, it is of interest for future studies to also evaluate liver volume. Lastly, the endocrine profile of hepatic enzymes and lipids in this patient sample was intriguing, and again, a longitudinal study could better assess plausible relationships between these and liver health.
Conclusion
In agreement with other research, this study demonstrated that sonography was a sufficiently sensitive diagnostic tool to detect nonsymptomatic changes in the liver among type 2 diabetes patients.38,39 From an algorithm standpoint, it is of interest for the future to determine whether sonographic data on the liver, over the course of diabetes treatment, can significantly affect disease management.
Footnotes
Acknowledgements
The authors acknowledge Dr Gory Ballester and the Images Center of the Medical School, UPR Medical Sciences Campus, for providing expert advice and access to the ultrasound equipment. Data reported here were collected as part of the PhD thesis by B.L.R.-C.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: J.C.J. is a Fellow of the Independent-Research Professional Development (I-RPD) Program, Medical Sciences Campus, University of Puerto Rico, sponsored by the National Institute on Minority Health and Health Disparities (NIMHD) and the National Institute of Allergy and Infectious Diseases (NIAID) of the National Institutes of Health under award U54MD007587.
