Abstract
Simultaneous pancreas and kidney transplants are most commonly evaluated with sonography. Thorough understanding of the variety of surgical techniques, postoperative anatomy, and potential complications is necessary for proper diagnosis. A common complication following pancreas transplant surgery is venous thrombosis. In the presented case, sonography was able to demonstrate portosplenic thrombosis, and computed tomography angiography was used as a secondary means of imaging. Following treatment, sonography was used to follow the resolution of the thrombus, which facilitated survival of the pancreas graft.
Keywords
Pancreas transplants are the preferred method of treatment for patients with type 1 diabetes if they are good candidates. It is the only long-term treatment for diabetic patients. Transplantation is often done when diabetes has caused end-stage kidney disease; therefore, pancreatic and renal transplants are performed simultaneously. This specific procedure, referred to as a simultaneous pancreas-kidney transplant (SPK), is performed on over 700 patients annually, but including pancreas transplants alone (PTAs) and pancreas after kidney transplants (PAKs), the number reaches 950 patients annually. 1 Like any transplant, the greatest risk for complications falls within the first few weeks following transplantation. One of the most common causes of posttransplant graft failure is venous thrombosis. 2 It affects 2% to 19% of all pancreas transplants and usually occurs within the first few weeks. 3 Detection of the thrombosis, prior to complete failure of the graft, is necessary to preserve the transplant. Duplex sonography is typically the first type of diagnostic imaging examination ordered.
Case Report
A 36-year-old man previously underwent surgery due to end-stage renal disease (ESRD), secondary to type 1 diabetes mellitus (DM1) that was diagnosed at age 15. He had been dependent on peritoneal dialysis for seven months and was diagnosed with hypertension as well as diabetic retinopathy. The patient was discharged seven days after the operation. Almost two weeks following pancreas and kidney transplant, the patient developed a fever that reached 101°F and was experiencing right lower quadrant pain. It was at this point that his surgeon recommended the patient come to the emergency department for evaluation. The patient did report to the emergency department and subsequently was admitted to the hospital.
Laboratory values indicated an elevated white blood cell count; however, glucose, amylase, lipase, and creatinine were within normal limits. The patient’s urine output was high, with a glomerular filtration rate (GFR) that was greater than 60 mL/min. Computed tomography (CT) of the abdomen and pelvis failed to reveal any notable abnormalities of the transplant kidney. Moderate fat stranding around the pancreas suggested acute pancreatitis or transplant rejection. However, the report stated that without intravenous contrast, CT evaluation of the pancreatic parenchymal enhancement pattern was limited (Figure 1).
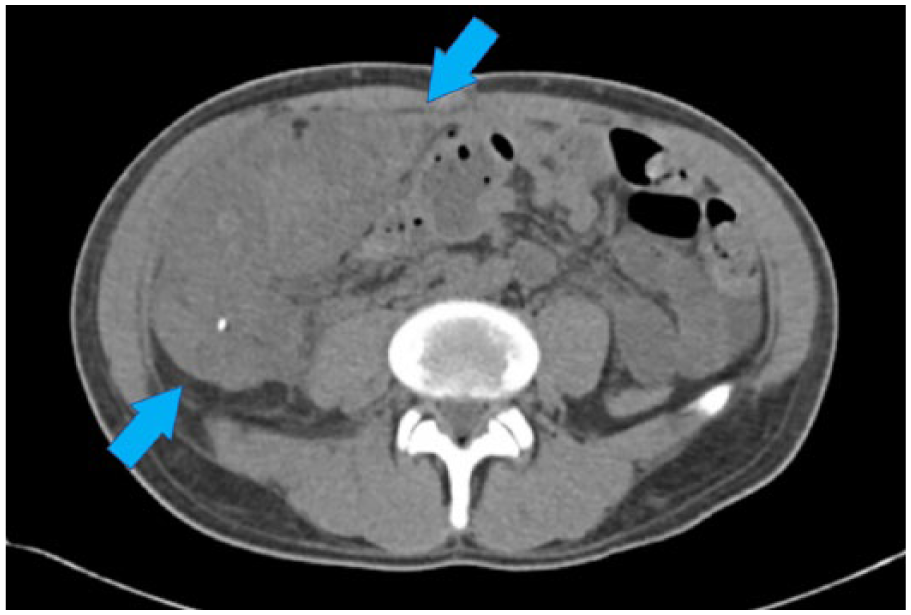
Computed tomography image, without contrast, of the pancreas transplant in the right pelvis.
Following CT, sonographic evaluation of the transplant pancreas and kidney was performed to evaluate vascular flow. Sonography revealed adequate perfusion of the transplant kidney with resistive indices (RIs) ranging from 0.64 to 0.8 (Figure 2). Sonographic evaluation of the pancreas revealed a nonocclusive thrombus within the transplant portal vein with extension into the donor superior mesenteric vein and splenic vein branches (Figure 3).
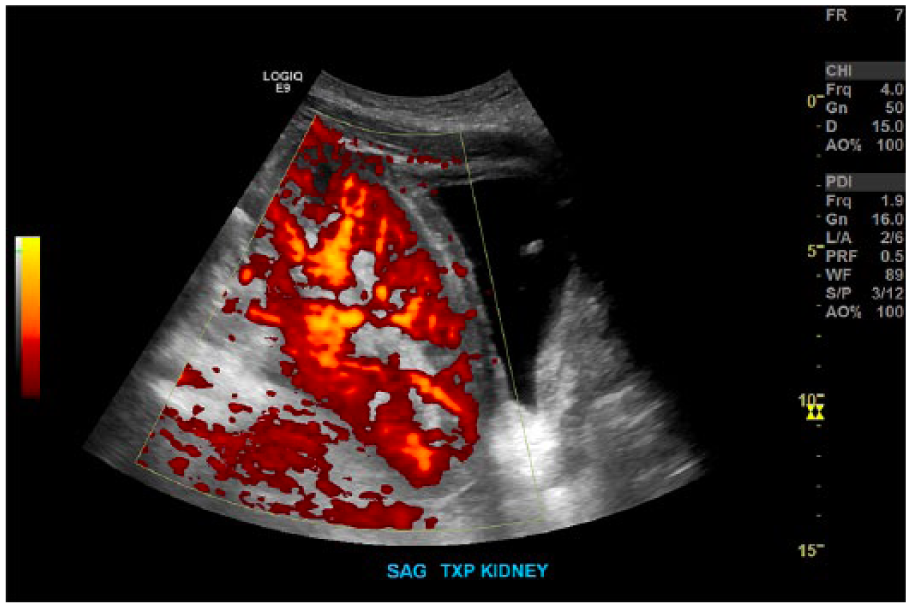
Sagittal image of the transplant kidney showing perfusion of blood flow with power Doppler.
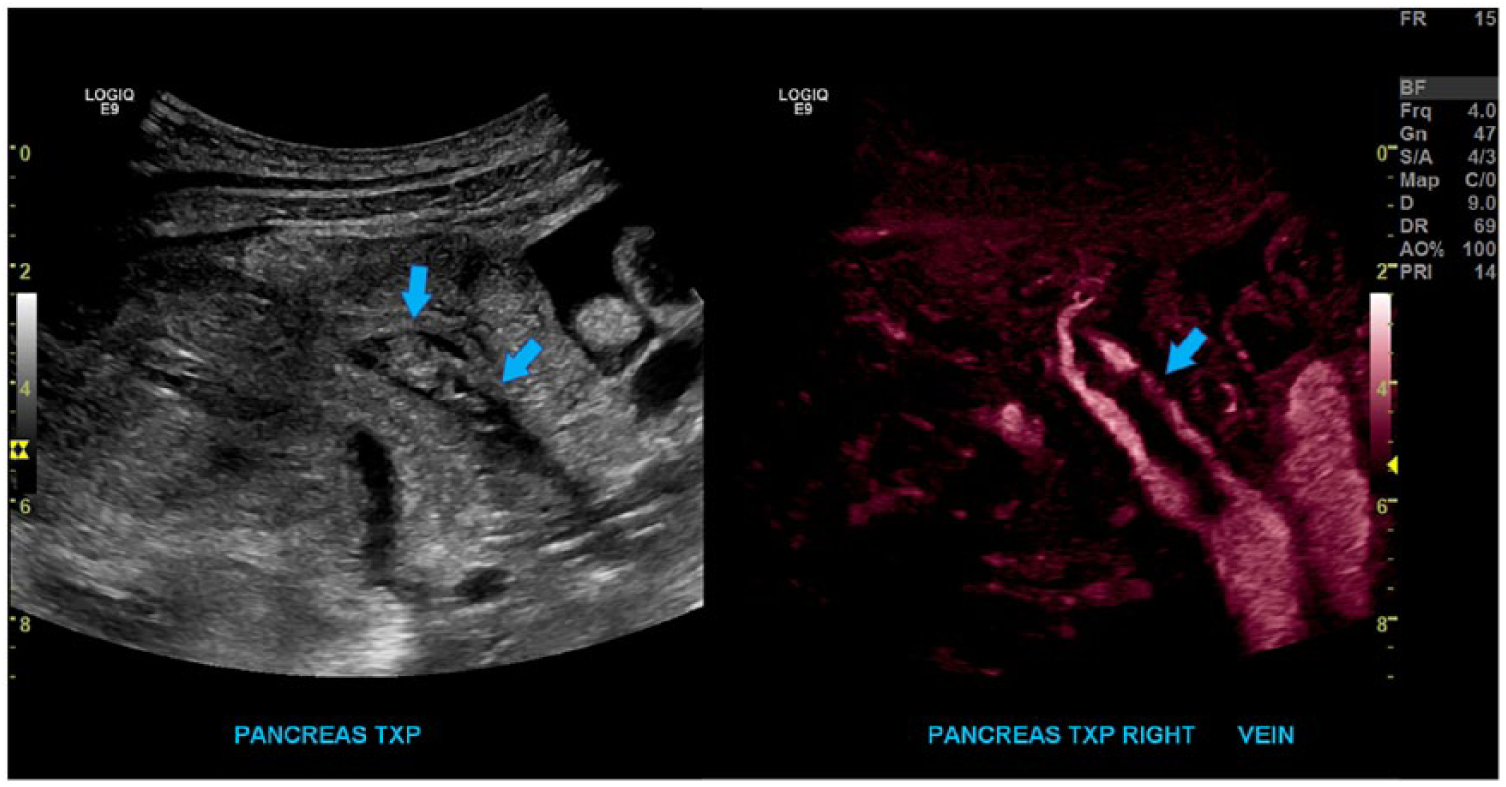
Sagittal images of the transplant main portal vein (arrow) first in gray-scale demonstrating the thrombus, which appears isoechoic to the pancreatic tissue within the anechoic vessel. B-flow imaging is used to indicate that it is nonocclusive.
Computed tomographic angiography (CTA) was obtained, which confirmed the nonocclusive thrombus within the donor portal vein. Moreover, it demonstrated greater extension of the thrombosis to the anastomosis of the vein, as well as thrombus in the distal branches of the transplant pancreatic artery. The patient was then administered a variety of anticoagulants. At that time, there was no indication for surgical thrombectomy.
Following initial treatment, a one-week follow up sonogram was performed to reevaluate the portal vein thrombus. A small residual nonocclusive thrombus was seen in the transplant portal vein, but the previously seen large, nonocclusive thrombus that extended into the donor splenic and superior mesenteric vein branches was no longer visualized. It was concluded that the thrombus had likely resolved (Figures 4 and 5). The patient was discharged from the hospital with plans for continued normal posttransplant monitoring.
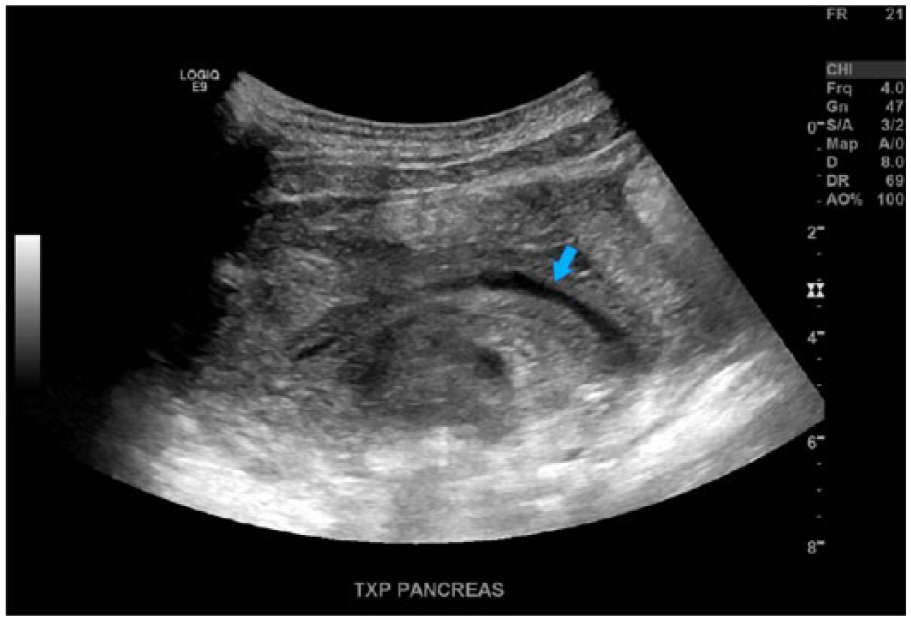
Sagittal image of the transplant main portal vein (arrow) in gray-scale at the previous location of the thrombus.
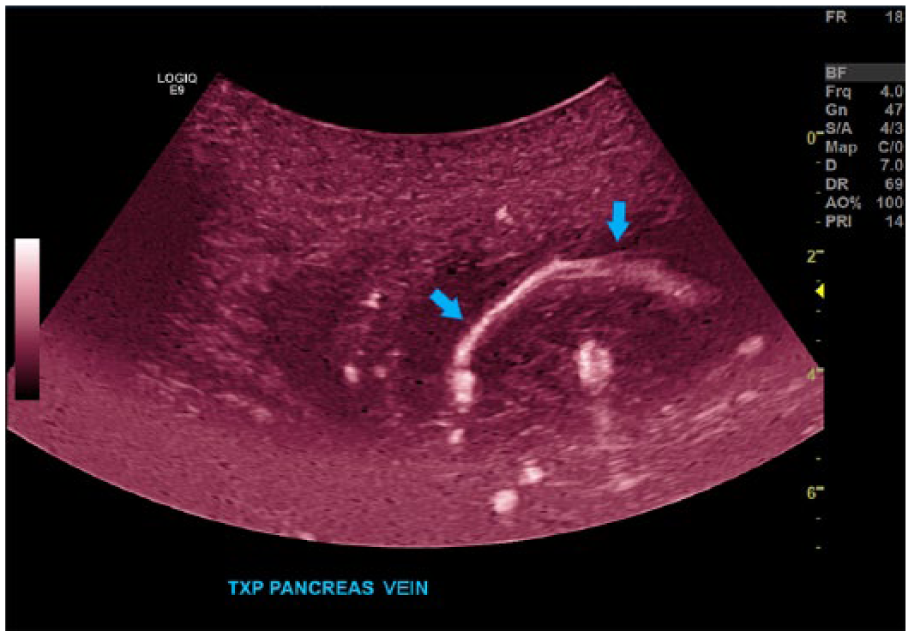
Sagittal image of the transplant main portal vein (arrow), with B-flow after treatment with anticoagulants, demonstrating that the thrombus had resolved.
Discussion
Diabetes is a prevalent disease that is often only the beginning of medical issues for patients. Once diagnosed, these patients face a lifelong struggle of monitoring glucose levels. Type 1 diabetes is caused by a deficiency of insulin secretion, which is the hormone that regulates glucose in the blood. Type 2 diabetes can be due to insulin resistance or to pancreatic failure to secrete enough insulin, and it is usually a combination of both systems. 4 The state of hyperglycemia caused by type 1 or type 2 diabetes may then cause other long-term damage to the eyes, nerves, heart, blood vessels, and kidneys. 4 Because of those secondary effects, pancreas transplants are done for those with insulin-dependent diabetes mellitus to prevent further damage to their organs. 5 Although this means it is primarily performed for type 1 diabetes, there are cases where it is used as a treatment for type 2 diabetes as well. 1 Type 2 diabetes is primarily managed by diet and lifestyle changes, and insulin or medication is used only if diet and exercise alone fail to control the glucose levels. 4 Type 2 diabetes actually used to be considered a contraindication for a pancreas transplant because of the statistical increase in graft failure. 6 However, current research shows that the diagnosis of type 2 diabetes is not the cause of the less favorable outcome but results from other patient characteristics. Sampaio et al. 7 found that older age and a longer pretransplant dialysis time increased the risk of death, and older donor age and higher recipient body mass index (BMI) increased the risk of graft failure for those with type 2 diabetes. Favorable results have been achieved with pancreas transplants to treat type 2 diabetes under strict donor and recipient criteria. Therefore, the rate of SPK transplants done for type 2 diabetes has increased. 8 When a patient has ESRD secondary to DM1, SPK is the ideal method of treatment. 9 In fact, 84% of all pancreas transplants are performed simultaneously with a kidney transplant, rather than a PTA or a PAK. 1 Pancreas transplants are optimal for glycemic control because they not only improve quality of life by eliminating the need for daily glucose monitoring and insulin administration but also prevent life-threatening complications that happen due to hypoglycemic unawareness, which is when a patient does not have overt symptoms but his or her glucose is too low. 5 Although SPK is the preferred treatment, the procedure has high rates of morbidity and mortality due to its complexity and technical failure.10,11 The current one-year survival rate for the procedure is 85%. 1 Diagnosis of postoperative complications is difficult because, unlike other organ transplants, there are no reliable laboratory indicators for the function of the transplant pancreas. 5 To increase the chance of survival, most candidates for the surgery are young, thin, and relatively fit. Recently, there has been a case that reported the success of pancreas transplant for a morbidly obese patient by using minimally invasive surgical techniques. 12
Like stand-alone renal transplants, pancreas transplants are placed in the iliac fossa. When simultaneously done with a kidney, usually the pancreas is placed on the right and the kidney on the left (Figure 6). The pancreas is placed in a superior to inferior manner rather than right to left like the native pancreas. Depending on the type of venous drainage system used, the head can be placed inferiorly (systemic venous drainage) or superiorly (portal venous drainage), with head inferior being the most common. 13
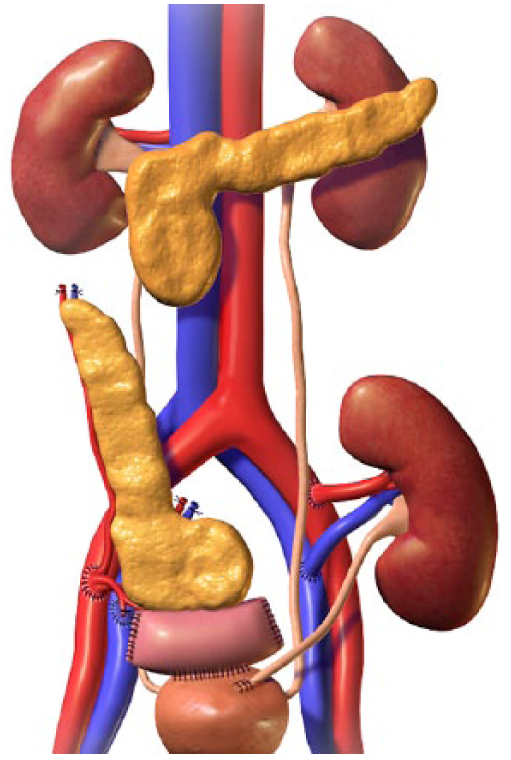
Diagram of simultaneous pancreas and kidney transplant anatomy. 18
Along with the procurement of the donor pancreas, a portion of the duodenum, the iliac artery bifurcation, the splenic artery, the superior mesenteric artery, the portal vein with splenic vein, and the superior mesenteric vein still connected are harvested as well. 13 The iliac artery bifurcation is anastomosed to the splenic and superior mesenteric arteries, creating the Y graft (Figure 7). The donor common iliac artery is anastomosed to either the recipient common or external iliac artery. 13 The donor superior mesenteric artery will then supply the head by way of the inferior pancreaticoduodenal artery, and the splenic artery will supply the body and tail, completing the arterial blood supply (Figure 8). The donor portal vein is anastomosed to either the recipient iliac vein or inferior vena cava in systemic venous drainage, which is the most common, or the recipient superior mesenteric vein in portal venous drainage. 13 There are two options for exocrine drainage. The donor duodenum C-loop may be attached to the recipient urinary bladder with a duodenocystostomy (Figure 9), allowing for future assessment of the transplant pancreas exocrine function by measuring urinary amylase. 14 A more common option is to attach the donor C-loop to the recipient jejunum (Figure 10). This eliminates potential complications associated with the drainage of pancreatic enzymes into the urinary bladder, including cystitis, hematuria, recurrent urinary tract infections, and urethritis.8,13,14 In the case presented, the pancreas was placed in a head-down position, the Y graft was anastomosed to the common iliac artery, the portal vein was anastomosed to the common iliac vein, and a duodenojejunostomy was done for ductal drainage.
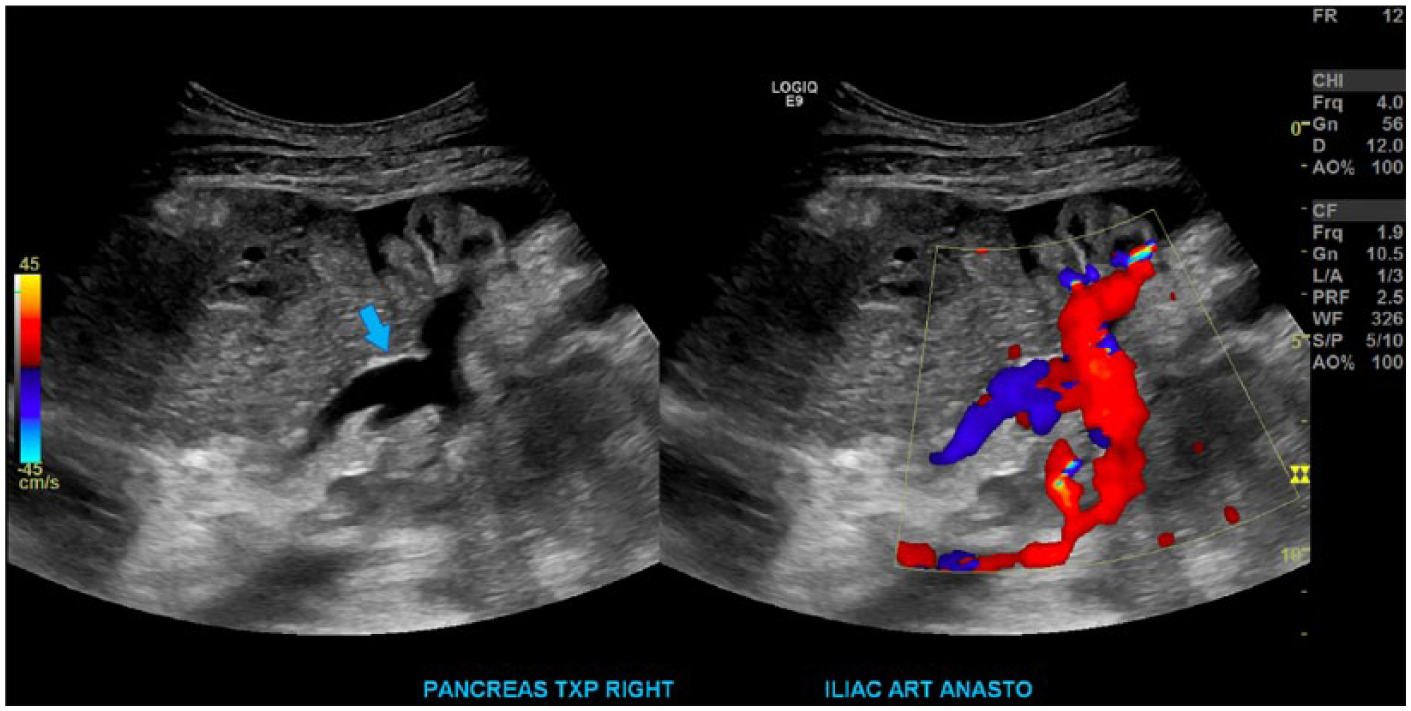
Sagittal images in gray-scale and with color Doppler of the anastomosed Y graft that is created from the donor iliac artery bifurcation (arrow), donor splenic artery, and donor superior mesenteric artery.
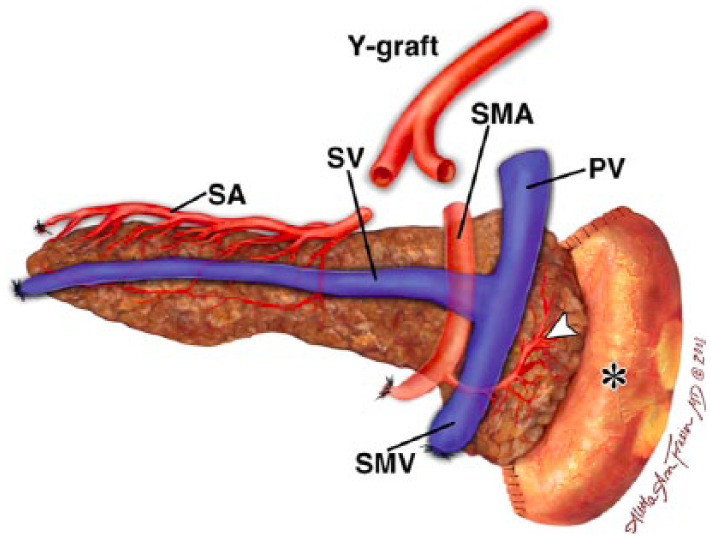
Diagram showing the vasculature of the transplant pancreas. PV, donor portal vein; SA, donor splenic artery; SMA, donor superior mesenteric artery; SMV, donor superior mesenteric vein; SV, donor splenic vein. 19
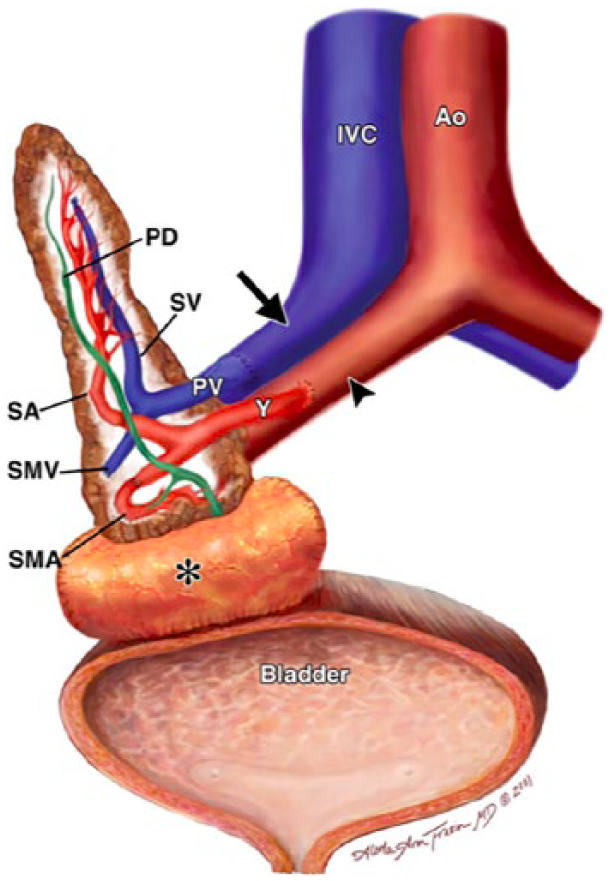
Diagram showing the surgical technique where the donor duodenum C-loop (*) is attached to the recipient urinary bladder. Ao, recipient aorta; IVC, recipient inferior vena cava; PD, pancreatic duct; PV, donor portal vein; SA, donor splenic artery; SMA, donor superior mesenteric artery; SMV, donor superior mesenteric vein; SV, donor splenic vein; Y, Y graft. 19
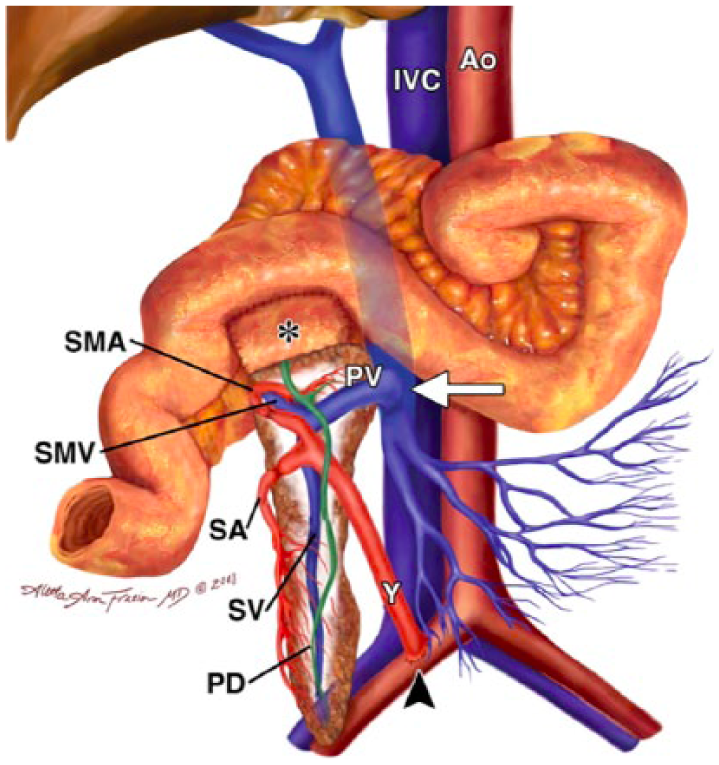
Diagram showing the surgical technique where the donor duodenum C-loop (*) is attached to the recipient jejunum. Ao, recipient aorta; IVC, recipient inferior vena cava; PD, pancreatic duct; PV, donor portal vein; SA, donor splenic artery; SMA, donor superior mesenteric artery; SMV, donor superior mesenteric vein; SV, donor splenic vein; Y, Y graft. 19
As with any surgery, there are many complications that the transplant recipient’s care team is working to prevent. These complications include hemorrhage, arterial or venous thrombosis, pancreatitis, small bowel obstruction, infection, anastomotic leakage, and rejection. 8 Multiple studies show that vascular thrombosis is the major cause of pancreas transplant failure.2,3,5,8 In fact, it is responsible for more than half of all losses within the first three months following surgery, due to a lack of specific symptoms. Thrombosis of the veins associated with pancreas transplant is not evident on physical examination. Glucose levels may fluctuate, but this is not specific to thrombosis; it is often attributed to medication. 15 In a case reported by Shahbazov et al., 15 the patient complained of bilateral leg pain and swelling that were more severe on the side of the graft. That patient’s thrombus extended into the external iliac vein and occluded the internal iliac vein. The symptoms of the patient in the present case were indicative of infection, not thrombus. Undetected thrombus can result in vascular dysfunction that progresses rapidly to transplant failure. 2
There are several means of treatment for venous thrombosis. The first is the use of anticoagulants. However, most pancreas transplant recipients begin with anticoagulants as part of their postsurgical care. 10 In the event of a thrombus developing despite the patient being administered anticoagulants, the only viable option is a thrombectomy. A previous study showed successful pancreas graft recovery following thrombosis treated by surgical thrombectomy in only 45% of patients. 2 Often, thrombus removal is incomplete, preventing full recovery. Therefore, when graft thrombosis happens soon after surgery, a transplant pancreatectomy may be required. 2 If another pancreas is available at that time, an immediate retransplantation may occur.
Several imaging techniques can be used to detect problems within a pancreas transplant. CTA or magnetic resonance (MR) angiography can be used to assess vascular abnormalities and an MR cholangiopancreatography is used to detect ductal abnormalities. 5 However, many facilities will use duplex sonography to monitor the pancreatic graft and assess its vascular patency. 13 Sonography is often the first-line modality for detection of posttransplant complications as well as for monitoring the efficacy of treatment. In fact, the American Institute of Ultrasound in Medicine (AIUM) has published practice parameters for solid organ transplants that include how transplant pancreases should be evaluated as well as indications for the examination. 16 The other imaging modalities are typically used only if sonographic evaluation is limited due to bowel gas or to better assess problems already detected with sonography.
The normal sonographic appearance of a pancreatic transplant is a homogeneous echotexture that is hypoechoic to surrounding fat and the native pancreas (Figure 11).3,5,17 Color Doppler images and Doppler waveforms can be obtained to visualize and assess arterial and venous blood flow. Arterial waveforms normally show a rapid systolic upstroke with continuous diastolic blood flow, while venous waveforms show a monophasic flow (Figures 12 and 13).3,17
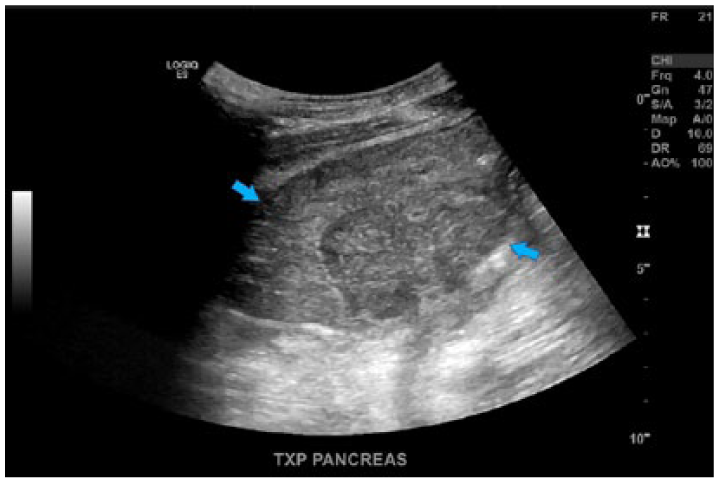
Sagittal image of the transplant pancreas in gray-scale demonstrating the homogeneous appearance.
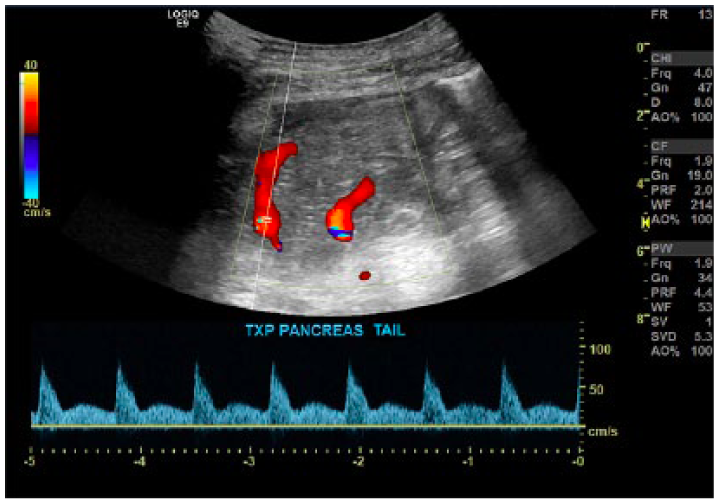
Image depicting color Doppler and normal spectral Doppler waveform of a pancreatic graft artery.
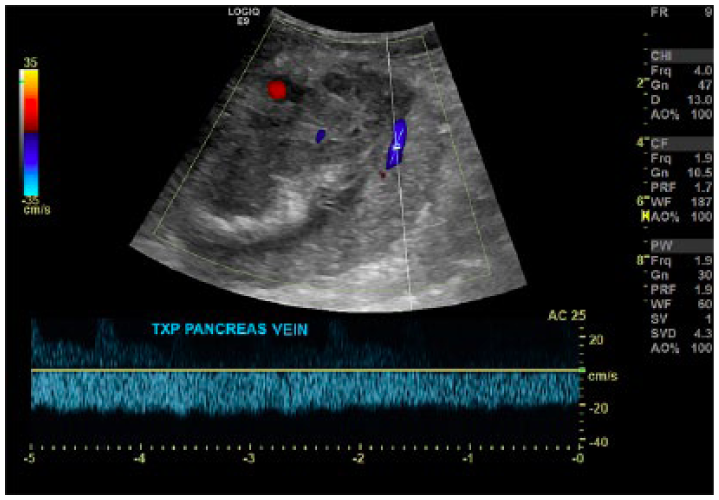
Image depicting color Doppler and normal spectral Doppler waveform of a pancreatic graft vein.
When scanning pancreas transplants for vascular complications, it is important to know the type of anastomoses used to ensure accuracy and that the examination is comprehensive.5,13 If the sonographer is not careful in assessing all the vessels involved, a thrombus could be easily missed. The thrombus may be nonocclusive or occlusive, which would result in different presentations. With proper color Doppler settings, one should see a filling defect outlining the nonocclusive thrombus. However, spectral Doppler would show little variation in the waveform due to flow still being present. If the thrombus is occlusive, there would be absent color and spectral Doppler. The arterial flow, in the presence of venous thrombosis, would also become highly resistive with reversed diastolic flow.3,5,17 In gray-scale imaging, the pancreas may appear enlarged and edematous with a heterogeneous echogenicity (Figure 14). This is similar in appearance to pancreatitis and rejection. 5 If the thrombus is acute, it may be central and anechoic as well as cause the vein to expand. However, chronic thrombus will more likely present as echogenic and sit more laterally in the vessel. Because of the varied appearances of pancreatic transplants, sonographers need to know the surgical anatomy, as well as the varied clinical presentations that could lead to the discovery of a thrombus.
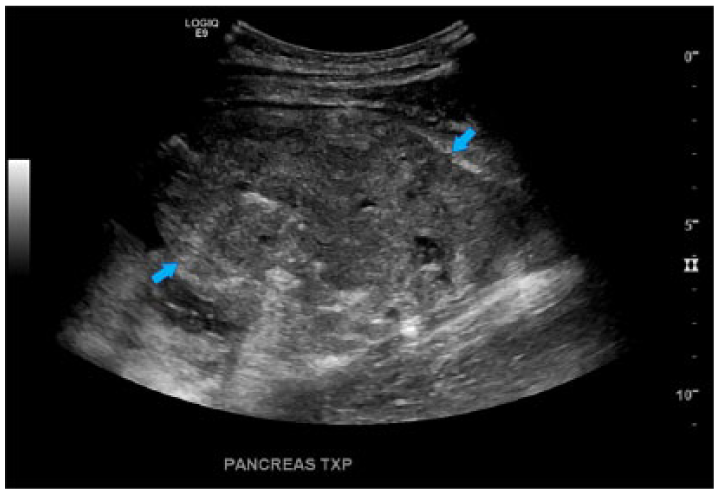
Sagittal image of the transplant pancreas in gray-scale demonstrating the heterogeneous appearance.
Conclusion
Sonography is the primary means of imaging a posttransplant pancreas because it is a portable and efficient way to narrow the differential diagnosis. It also can contribute to the clinical picture by prompting the acquisition of other imaging modalities as needed. Because there are few clinical or laboratory indications of pancreatic transplant thrombosis, physicians rely heavily on sonographic findings to help guide patient management. To obtain the best outcome for these patients, sonographers need to be aware of the variations in surgical technique to fully evaluate the graft. Those examinations should include gray-scale images of the transplant pancreas, duplex imaging of all the donor vessels, and native vessels to which they are anastomosed. Incomplete evaluation or improper image optimization can result in subtle indications of complication being overlooked. Since venous thrombosis is one of the most common posttransplant complications, accounting for more than half of transplant failures, prompt detection and treatment are critical.
Footnotes
Acknowledgements
The author thanks David Karsten, BS, RDMS, and Tony Gregory, RT(R), RDMS, for the images as well as their guidance and support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
