Abstract
The purpose of this study was to retrospectively review sonographic imaging features of estrogen-negative breast cancers and compare tumors with and without human epidermal growth factor type II (HER2) overexpression. Breast sonography findings from a sample of 54 patients with estrogen-negative breast cancer as well as pathological data and HER2 status were reviewed. Sonographic features including size, depth shapes, margin, location, patterns of internal echoes, posterior echoes, orientation, and presence of halo and clinicopathologic data and imaging features were correlated with tumor HER2 status. Based on these 54 patients with estrogen-negative breast cancers, 21 patients were positive for HER2 receptor, and 33 patients were negative for HER2 receptor. Among HER2 positive cancers, irregular shape, microlobulation, indistinct margins, posterior shadowing, a thin halo, heterogeneous internal echoes, and parallel orientation were the most frequent sonographic features. No associations were found between HER-2 status and tumor size, shape, margins, posterior feature, halo, internal echoes, or orientation on sonography.
Introduction
Breast cancer is the most prevalent cancer among women and the predominant cause of death in both developed and developing countries. 1 An early diagnosis of invasive breast cancer has significant impact on the outcome of these patients. Breast sonography is an indispensable diagnostic tool in imaging of breast lesions and is employed for both detection and characterization of breast masses. The potential capability to predict the breast cancer subtype based on sonography may lead to an accelerated diagnosis and management and consequently improve outcomes.
Breast cancer is a heterogeneous disease with an increasing number of identified biological subtypes and various prognostic and survival implications. 2 Immunohistochemistry (IHC) markers including estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor (HER2) as well as clinicopathological variables such as tumor size, nuclear grade, along with lymph nodal stage are traditionally employed for patient prognosis and treatment planning. 2 ER, PR, and HER2 are also the commonly targeted receptors in hormonal therapy of operable breast cancers. The advancements in hormonal therapy justified the need for distinction between ER-positive and ER-negative breast cancers and the state of HER2 receptor.
ER-negative tumors constitute 25% to 30% of all breast cancers and tend to have a more aggressive clinical course and poor response to chemotherapeutic agents. 3 Furthermore, positive ER status predicts for response to hormonal therapy such as anti-estrogen (tamoxifen) treatment, which reduces the risk of tumor recurrence and mortality by more than 30%. 3 As a consequence, patients with ER-negative breast cancer in most cases have shorter disease-free survival and worse prognosis than ER-positive breast cancers. 4
Overexpression of HER2 oncoprotein is detected in nearly 15% to 20% of primary breast carcinomas using IHC staining. HER2-positive breast carcinomas have the worst prognosis among all types of invasive breast cancers, but they show a high rate of response to anti-HER2 targeted therapy (e.g., trastuzumab, lapatinib). Roughly 30% to 40% of ER-negative breast cancers overexpress the human epidermal growth factor receptor type 2. 5 The introduction of trastuzumab (Herceptin) indicated the significance of distinguishing breast cancers with overexpression of HER2. 6 Trastuzumab adjuvant therapy in patients with HER2 positive breast cancer results in improvement of survival rates at 3 years. 7 Conversely, patients with estrogen-negative, progesterone-negative, and HER-2-negative receptors (triple negative) tumors present a therapeutic challenge for the clinician. 8
For the aforementioned reasons, distinction of imaging features of ER-negative breast cancers with and without HER2 overexpression could potentially play a role in pretreatment planning and prognosis estimation. 9 Therefore, the purpose of this study was to investigate whether pretreatment sonographic imaging features are associated with the status of HER2 and overexpression in ER-negative breast cancers.
Materials and Methods
Patient Population and Data Collection
This study was approved by the institutional review board, and informed consent from the patients was waived. Using a breast cancer registry pathology database, 54 women who had ER-negative, PR-negative breast cancer that had gone under pre- and postoperative staging and clinicopathologic examination from January 2016 to July 2017 were identified. Patients with recurrent breast cancer were not included in our study. The diagnosis of suspected breast cancer was made based on mammographic and sonographic findings and was confirmed by core-needle biopsy in all of the participants.
All breast sonograms were performed by two radiologists, which included bilateral whole breasts and axillary regions with the patient in the supine position using linear 7.5- to 13-MHz transducers on a GE E8 (GE Medical Systems, Piscataway, NJ, USA) ultrasound unit. Digital sonograms and cines were retrospectively reviewed and interpreted by an attending radiologist (20 years of experience in breast radiology) who was blinded to the pathologic data. Digital sonograms were assessed, and lesions were described by the mass shape (oval, round, and irregular), margins (circumscribed, indistinct, angular, microlobulated, and spiculated), posterior feature (shadow, enhancement, and combined), halo (thin and thick), and orientation in relation to skin surface (parallel and anti-parallel) according to ACR BI-RADS sonographic descriptors.
Clinicopathologic Data
Pathology reports included tumor type; ER, PR, and HER2 status; tumor size; and tumor nuclear grade, which were derived from medical files. Tissue samples with less than 1% positive nuclear staining for ER categorized as negative. A HER2 staining score of 0 and 1+ were recorded as negative, and 3+ positive scores were recorded as positive. Fluorescence in situ hybridization (FISH) was used to define HER2 positivity in equivocal (2+) cases.
Statistical Analysis
Chi-square test and an univariate analysis of variance (ANOVA) test were used to assess the association of HER2 status with imaging features. Statistical Package for Social Sciences version 17 (SPSS Inc., Chicago, IL, USA) was employed for statistical analysis.
Results
Fifty-four patients with ER-negative, PR-negative breast cancers who had mass lesions in retrieved digital sonograms were included in this study. Twenty-one patients were positive for HER2 receptor (37.5%), and 33 patients (58.9%) were negative for HER2 receptor. The patients’ median age was 49 years (range, 27–84 years). All patients were of a similar ethnicity.
The mean tumor size at diagnosis was 18 mm (range, 5–53 mm). Localization of the tumor was predominantly in the upper outer quadrants (39.3%). Forty-two patients (75.0%) had invasive ductal carcinoma (IDC), 12 (21.4%) had invasive ductal carcinoma associated with in situ ductal carcinoma (IDC+DCIS), 1 (1.8%) had in situ ductal carcinoma (DCIS), and 1 (1.8%) had invasive lobular carcinoma (ILC). These were grade 2 tumors in 28.6% of cases and grade 3 tumors in 64.3% of cases.
Among HER2-positive cancers, an irregular shape (57.1%), microlobulation (33.3%), indistinct margin (33.3%), posterior shadowing (47.6%), thin halo (66.7%), heterogeneous internal echoes (63.6%), and parallel orientation (52.4%) were the most frequent sonographic features (see Figure 1). Among HER2-negative breast cancers, an irregular shape (37.5%), microlobulation (69.6%), combined posterior features (30.3%), thin halo (62.5%), heterogeneous internal echoes (57.1%), parallel (50%), and anti-parallel orientation (50%) were the most frequent sonographic features (see Figure 2). A significant association was noted between HER2 status and mean age (P = .959), mass lesion size (P = .811), shape (P = .173), margin (P = .900), posterior feature (P = .098), halo (P = .231), and orientation (P = .201) (see Table 1).
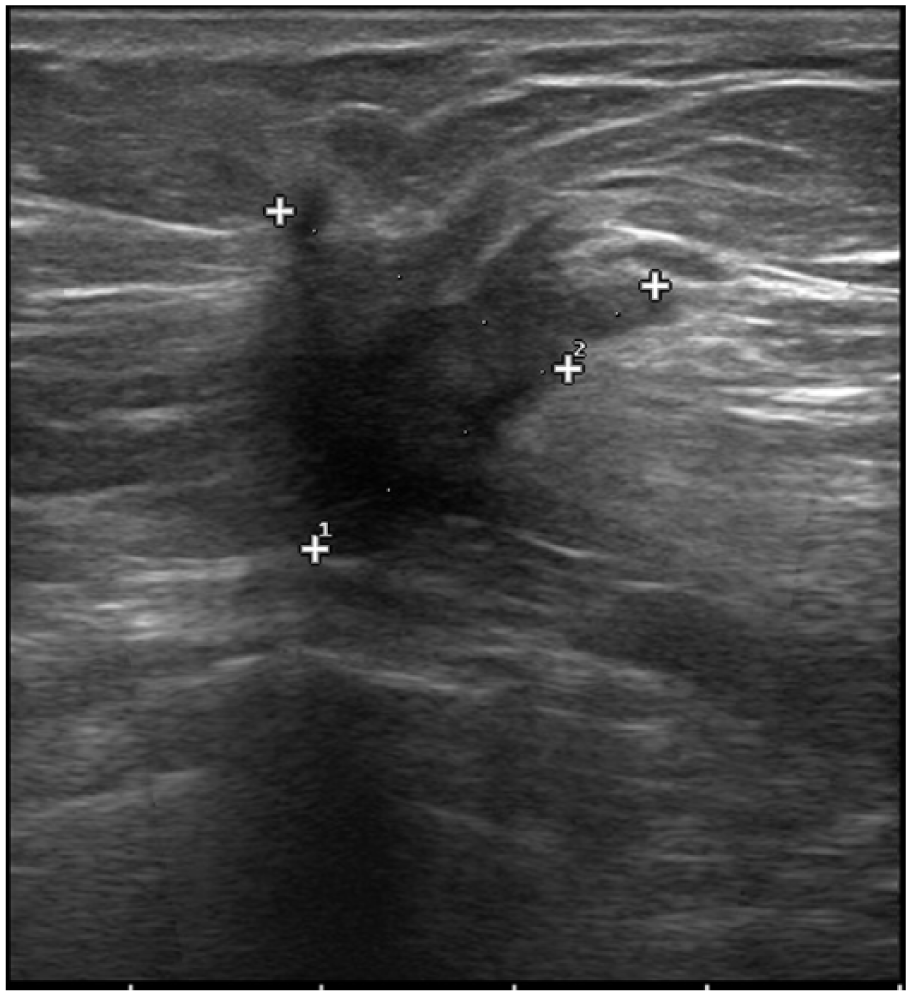
Sonographic image shows an irregular hypoechoic mass with spiculated margins, thin halo, and posterior shadowing in a postmenopausal woman. The tumor was found to be ER-negative, PR-negative, and HER2-positive in IHC examination.
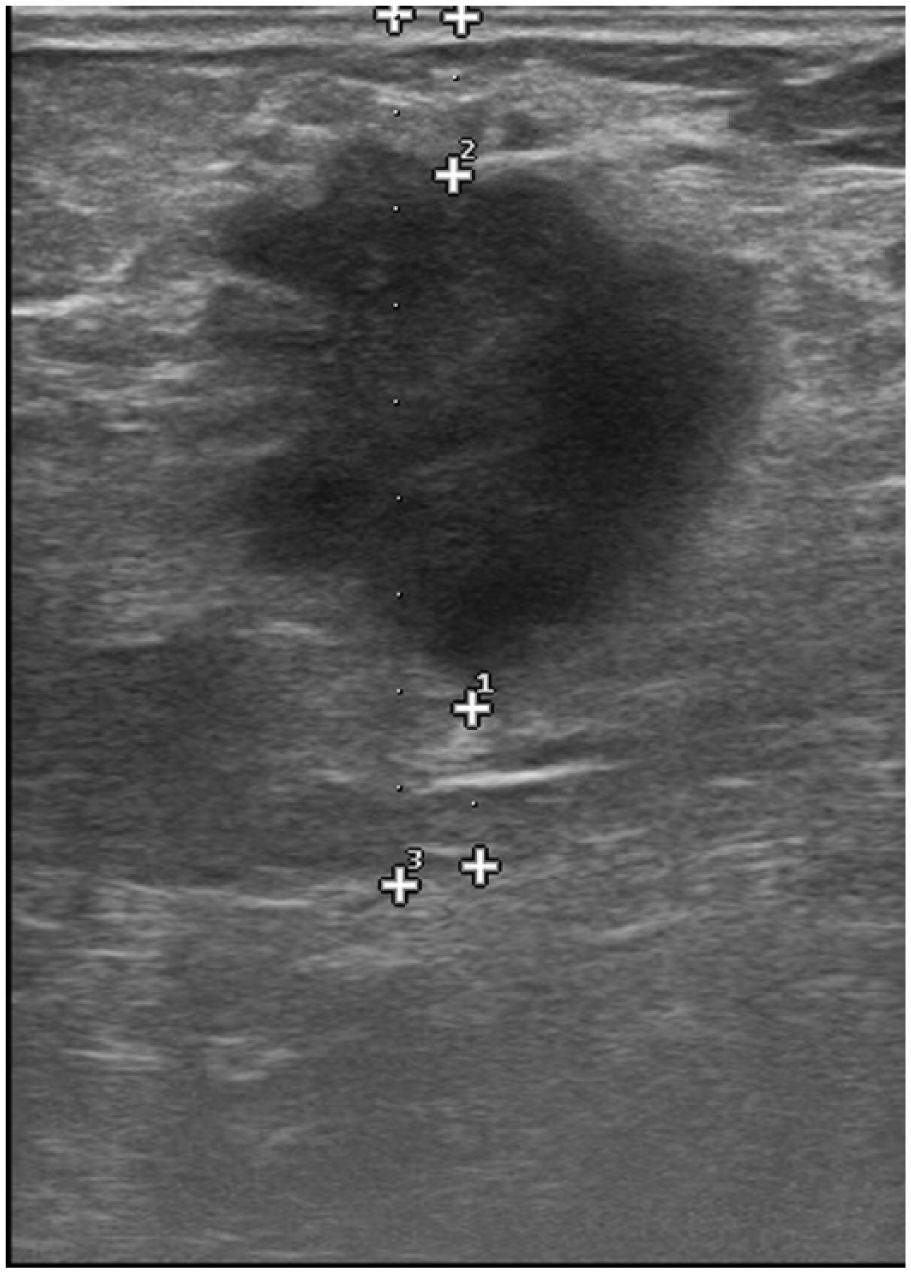
Sonographic image shows a lobulated hypoechoic mass with lobulated margins, thin halo, and posterior enhancement in a 43-year-old woman. The tumor was found to be ER-negative, PR-negative, and HER2-negative in IHC examination.
Sonographic Features of ER-Negative Breast Cancers.
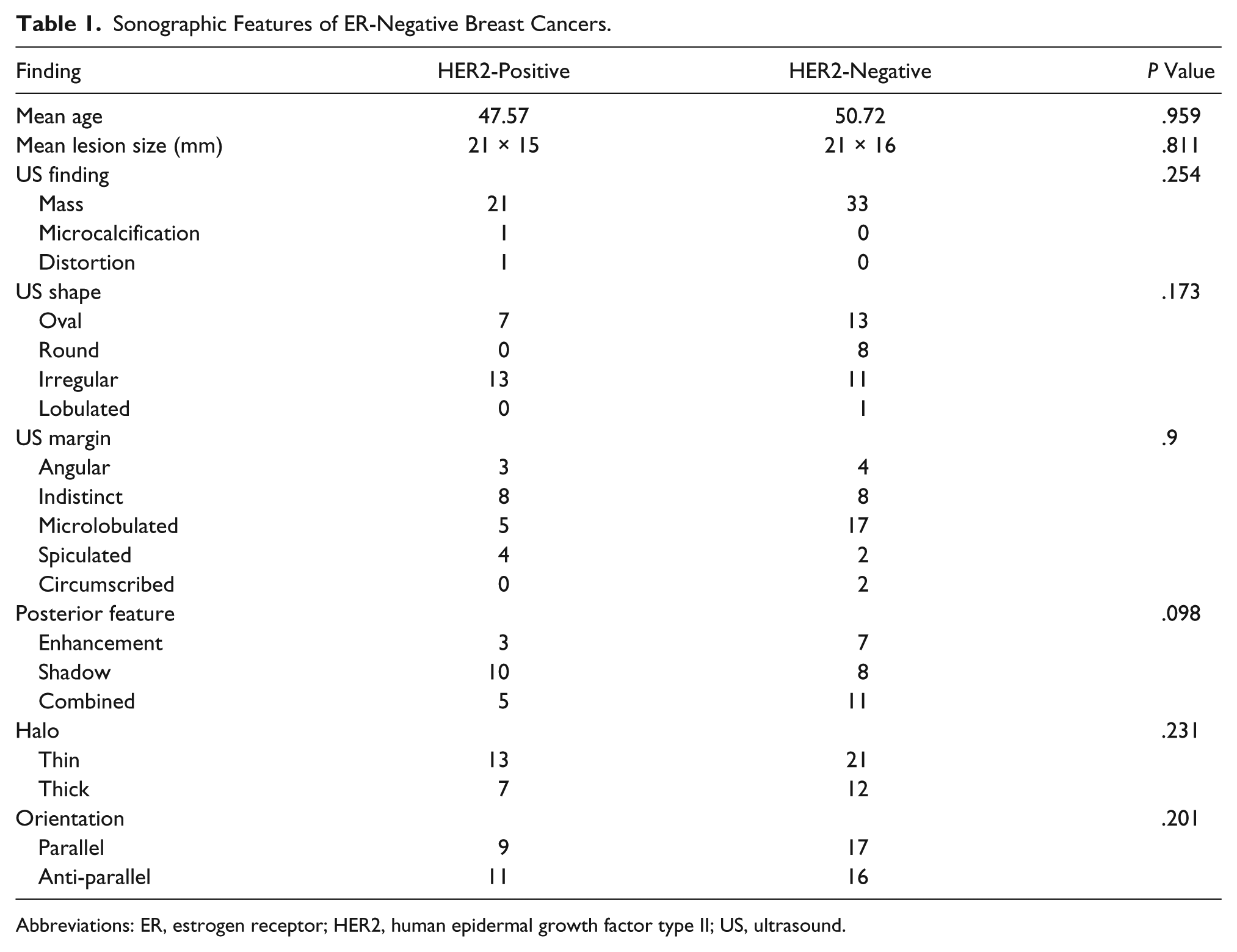
Abbreviations: ER, estrogen receptor; HER2, human epidermal growth factor type II; US, ultrasound.
Discussion
Overall, the reported incidence of ER-negative breast cancers in the United States is about 30%, and roughly one-fourth of these tumors show overexpression of HER2 receptor in IHC. 10 Yet, the prevalence of HER2-positive tumors reported in the literature as the result of different ethnic groups and regions is inconsistent. In this study, the proportion of HER2-positive breast cancers was higher, at 37.5%. Moreover, in the course of breast carcinogenesis, overexpression of HER2 was found to be an early event. 11 Therefore, the relatively high ratio of HER2-positive cancers in this cohort could be due to larger ratio of diagnosed higher stage invasive breast cancers as well as a lack of state-run breast cancer screening programs in the study region. It has been previously reported that dense breast tissue of young patients could harbor a microenvironment that is more prone to the development of HER2-positive tumors.12,13 However, no association was found between HER2-positive, ER-negative breast cancers, and the age of the patients.
Several studies in respect to association of imaging features and molecular phenotypes of breast cancers have been published in recent years. In a similar study, Wang et al. 14 suggested that spiculated tumor margins are associated with HER2 positivity in ER-negative breast cancers. However, in a meta-analysis by Elias et al., 9 85 articles were reviewed, and spiculated margins were not among the 11 imaging features that are correlated with HER2 overexpression. A recent study by Liu et al. 15 related spiculated margins on mammography with HER2-negativity, which is contradictory to Wang et al.’s 14 findings. It should be noted that Wang et al. 14 as well as the present study were limited to ER-negative breast cancers, but the two latter mentioned studies did not include ER status in their comparison of imaging features. Circumscribed margins on a breast sonogram is suggested to relate to a lesser likelihood of HER2 overexpression. This observation might have been caused by an overrepresentation of triple-negative (ER-negative, PR-negative, HER2-negative) breast cancers in studies that selected patients with regard to receptor expression (most of these studies excluded ER-positive breast cancers). 9 In the present study, no significant difference was detected between HER2-positive and -negative tumors in terms of lesion margins. Similar to other studies, there was no association between HER2 status and posterior feature, halo, or orientation of the lesion on the breast sonogram.
This study had a several limitations, such as the pre-experimental research design and convenient sample. Retrospective analysis was preformed on archived data of breast cancer patients, which is potentially associated with selection bias. Evaluation of mass shapes and margins is subjective, and there is interobserver variability even among experienced breast radiologists. Additionally, the small number patients lowered the statistical power of these findings.
Conclusion
Identification of distinguishing sonographic features between HER2-negative and HER2-positive breasts would enable the sonographer to have a better diagnostic perspective in the evaluation of breast lesions. It would also lead to improved management of breast cancer patients. The results of this study suggest that routine gray-scale sonographic findings might not be useful in differentiating HER2-positive and HER2-negative tumors among ER-negative breast cancers. However, there are emerging advanced sonographic techniques in breast imaging, such as elastography, contrast-enhanced sonography, and molecular ultrasound imaging, that will provide for future studies in this field. A more rigorous study of this association would be facilitated by the inclusion of a control group of patients for comparison.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
