Abstract
This pilot study evaluated associations between carotid wall echogenicity, cardiovascular disease (CVD) risk factors, and three markers of smoking heaviness in a cohort of active smokers. Common carotid artery (CCA) gray-scale median (GSM) values were measured from sonographic images. Univariable correlations and exploratory multivariable models were used to determine associations between CCA GSM, CVD risk factors, and measures of smoking heaviness. CCA GSM was measured in 162 smokers and was correlated inversely with cigarettes smoked/d (r = −0.16, P = .048); pack-years (r = −0.204, P = .009); CVD risk factors such as age, male sex, waist circumference, and low-density lipoprotein cholesterol (all P ≤ .03); and positively with high-density lipoprotein cholesterol (P < .001). Associations between CCA GSM and smoking heaviness markers were not statistically significant after adjustment for traditional risk factors. The results from this pilot study demonstrate the feasibility of measuring the GSM value of the CCA far wall and its association with measures of smoking heaviness and traditional CVD risk factors among current smokers.
Introduction
Atherosclerosis is a chronic systemic disease that begins early in life with injury to the arterial lumen resulting in inflammation, increased lipid accumulation, and hyperplasia of medial smooth muscle fibers.1–4 Progression of atherosclerotic vascular disease is dependent on genetic and environmental risk factors, such as hyperlipidemia, hypertension, and cigarette smoking.2,4,5
Smoking is a powerful risk factor for cardiovascular disease (CVD) that is associated with an increased risk for coronary heart disease and stroke.6–9 Smoking promotes atherogenesis and increased CVD risk by several mechanisms that contribute to arterial injury, plaque formation, and plaque vulnerability.8,9 Imaging markers indicative of late stage atherosclerosis, such as increased coronary artery calcification, 8 carotid artery plaque formation, and carotid wall intima-media thickness (IMT), 10 have been associated with smoking. Inflammation is an important mechanism for increased CVD risk among smokers. Atherosclerotic plaques of smokers have a greater content of inflammatory cells than observed in nonsmokers. 11 Smokers have increased macrophage recruitment to the arterial wall, 11 increased leukocyte counts, and higher levels of C-reactive protein and fibrinogen.8,12
It has been suggested that early atherosclerotic changes in arterial composition (i.e., increased lipid accumulation and inflammatory changes) can be seen with gray-scale sonography, evidenced as a more hypoechoic or echolucent intima-media complex that will be manifest prior to changes in wall thickness.1–3 Thus, measuring carotid artery wall echogenicity may prove to be an early in vivo marker for assessing atherosclerosis risk and monitoring treatment.1,2
Carotid wall echogenicity is a novel, sensitive predictor of CVD risk that is related to but independent of carotid IMT.3,13,14 To our knowledge, the relationship between smoking heaviness (cigarettes smoked/d and pack-years) and common carotid artery wall echogenicity has not been explored previously in a smoking cohort. Others 15 have demonstrated a nonlinear, V-shaped relationship between carotid plaque echogenicity (but not arterial wall echogenicity) and current smoking. The advantage to looking at arterial wall echogenicity versus plaque echogenicity is that it can assess all individuals instead of being limited to only evaluating individuals with plaque present. 3 Understanding the relationships among carotid wall echogenicity, smoking heaviness, and other CVD risk factors may suggest mechanisms for understanding the effects of smoking on arterial injury, how it interacts with other risk factors, and assessing the arterial health of smokers. A pilot study was conducted to assess the relationship among common carotid artery (CCA) gray-scale median (GSM) values, three markers of smoking heaviness, and other CVD risk factors in current smokers.
Materials and Methods
Participants
This research used baseline, pretreatment data from smokers who participated in a randomized, double-blind, placebo-controlled smoking cessation trial. 16 The institutional review board at the University of Wisconsin School of Medicine and Public Health approved this study, and all participants provided written informed consent. Major inclusion and exclusion criteria were based on participation in the clinical trial and included age ≥18 years old, current smoking of ≥10 cigarettes/d for the previous 6 months, expired carbon monoxide (CO) level of >9 ppm, and stated motivation to try to quit smoking. Details of other exclusion criteria are described in the primary analysis of this cohort. 16
Participants were 162 smokers from Madison, Wisconsin, who were selected based on having images acquired with the same ultrasound equipment preset (e.g., CV preset, linear gray-scale map [map L] maximum dynamic range 70 dB, baseline lab values available, and carotid intima media measurements).
These images were used to assess CCA GSM, a measure of carotid wall echogenicity. Participants provided a fasting blood sample to assess total cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglycerides, glucose, high-sensitivity C-reactive protein (hsCRP), and leukocyte count using standard techniques. Measures of smoking heaviness were cigarettes smoked/d, pack-years (cigarettes smoked/d × years smoking), and exhaled carbon monoxide (CO). Tobacco dependence was assessed with the Fagerstrom Test for Cigarette Dependence (FTCD). 17
Carotid Ultrasonography and Measurement of CCA GSM
Digital images of the far wall of the right CCA were acquired as described previously that were performed on the same diagnostic ultrasound imaging system (CV 70, Siemens Medical Solutions, Mountain View, CA, USA), same linear transducer L10-5, and using the same imaging preset. 18 Prior to the start of the study and throughout the study, quality assurance measures were performed on a Gammex Small Parts (Grey Scale) Phantom (404GS-LE 0.7) (Gammex, a Sun Nuclear Company, Melbourne, FL, USA). Quality assurance measures were performed to assess and confirm lateral and axial resolution as well as vertical and horizontal calibration measurements (see Figure 1A–1D, demonstrating phantom measurements made on the CV70 system as part of our routine QA program).
Phantom images taken for quality assurance to measure (A) axial and (B) lateral resolution and confirm calibration in the (C) vertical and (D) horizontal planes.
After acquisition, images were transferred to a DICOM server (Freeland Systems LLC, Alpharetta, GA, USA) at a core ultrasound laboratory. CCA GSM was measured using dedicated plaque texture analysis software (LifeQ Medical, Cyprus). All images were digitized into bitmap images. Normalization was performed utilizing the software normalization module with blood assigned a gray-scale value of 0 and the adventitia a gray-scale value of 190 and then were standardized to a standard pixel density of 20.00 per millimeter.19–22
After images were normalized and standardized, the carotid arterial wall was segmented for analysis. The distal common carotid artery was identified by locating the carotid bulb and then placing an online ruler tool (Microsoft Windows Ruler) scaled to 1.0 cm adjacent to the proximal edge of the carotid bulb and on the leading edge of the arterial wall adventitia. The arterial wall was segmented for gray-scale analysis by manually tracing the intima-media complex of the far wall for a distance of 1.0 cm (Figure 2). The cursor was placed on the intima-blood interface of the far wall and the leading edge of the adventitia to define the segmented arterial wall region of interest. LifeQ software was used to crop the segmented area and analyze the gray-scale characteristics. The measure we were interested in for this study is the gray-scale median value. The GSM value was used instead of the mean value because the gray-scale values are skewed (see Figure 3). In the “features extraction module,” the cropped segmented arterial wall is demonstrated as well as the colorized version depicting the range of gray-scale values (see Figure 4). Once the trace was completed, all data and images were automatically saved digitally and uploaded into a secure study database for statistical analysis.
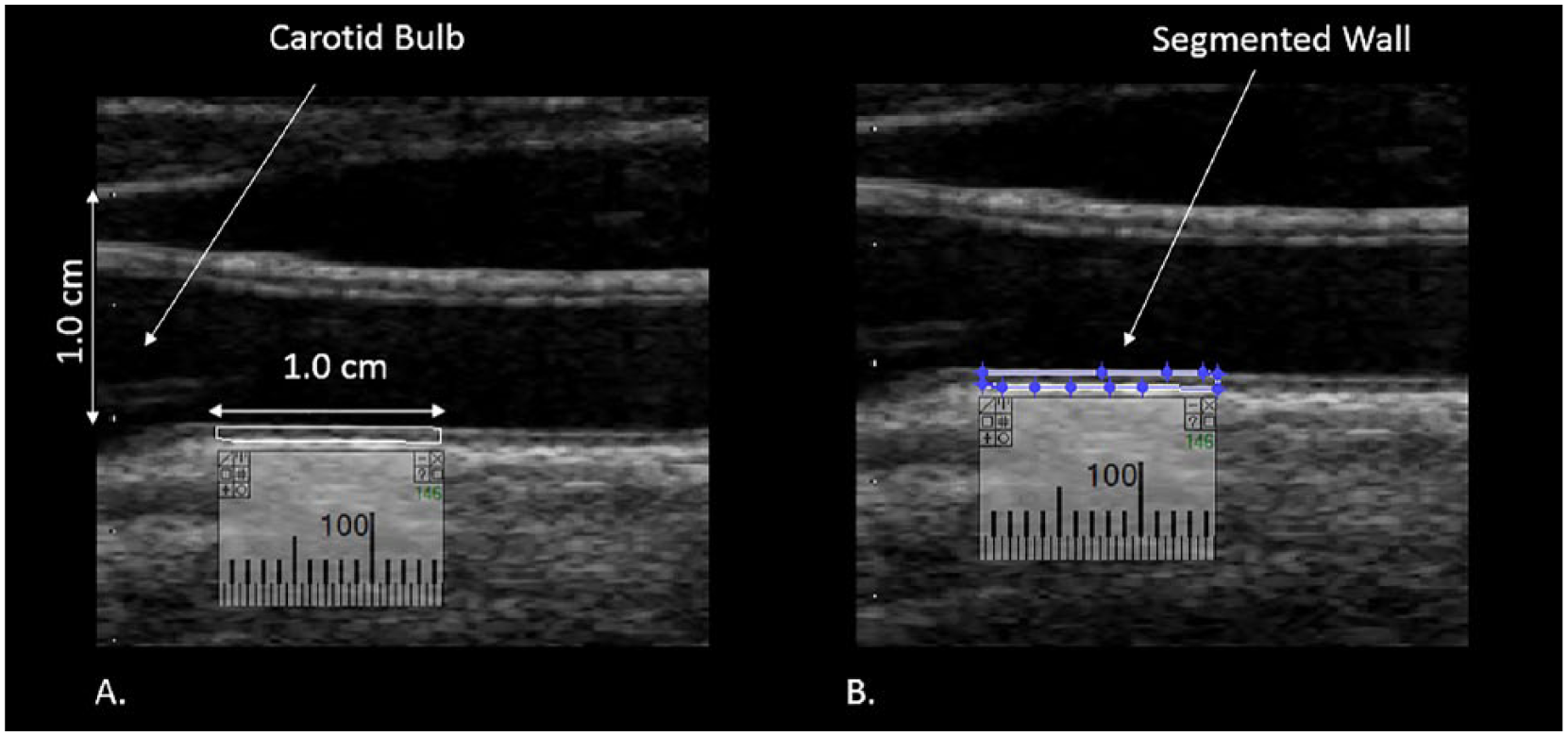
Distal 1.0 cm of the common carotid artery measured to obtain the gray-scale median (GSM) value. (A) Calibration of 1.0 cm and the segment of the wall to be measured. (B) Segmentation of the distal 1.0 cm of the common carotid artery for GSM measurement.
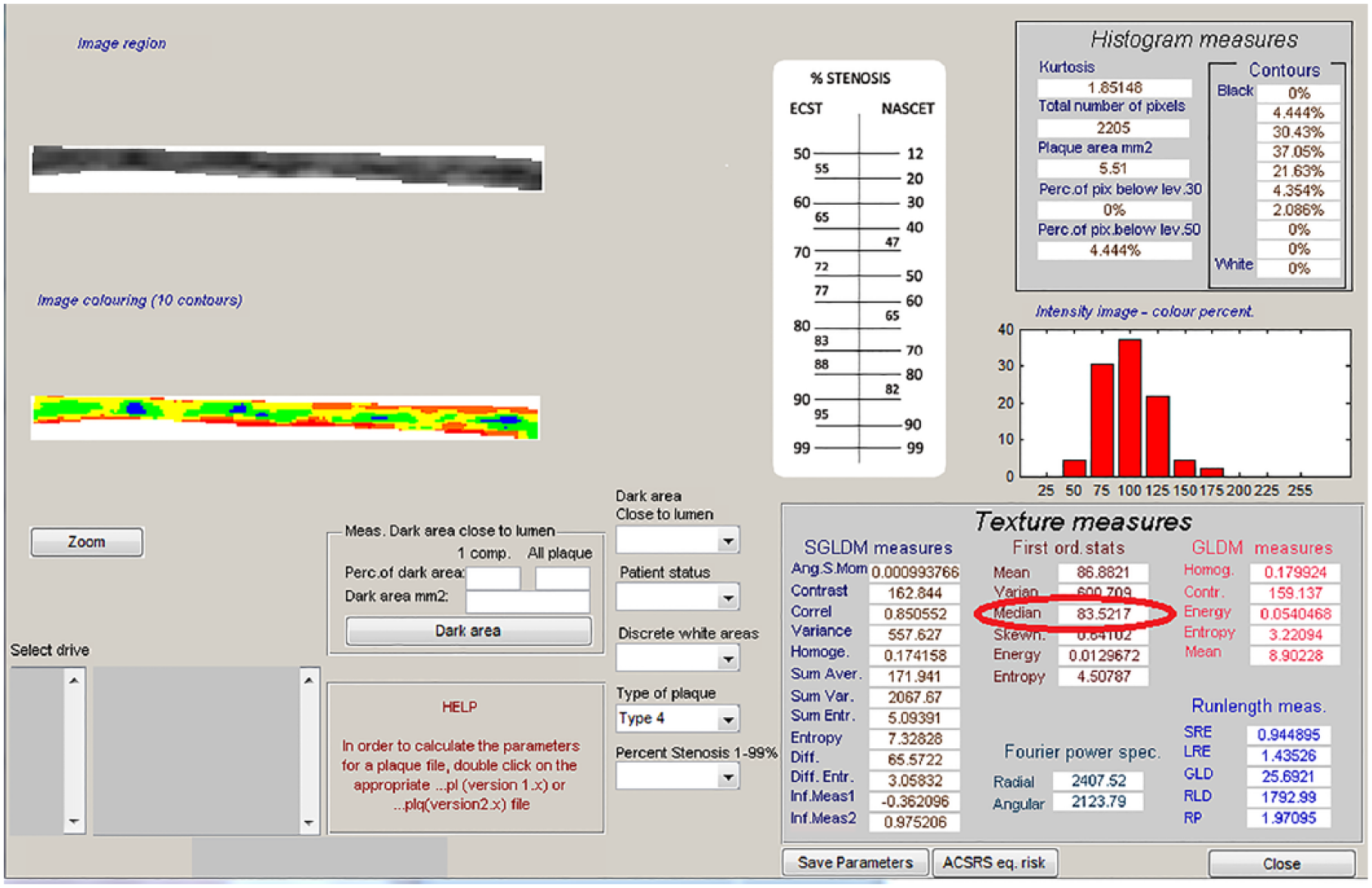
Output from the software demonstrating the measurement of the gray-scale median (red circle).
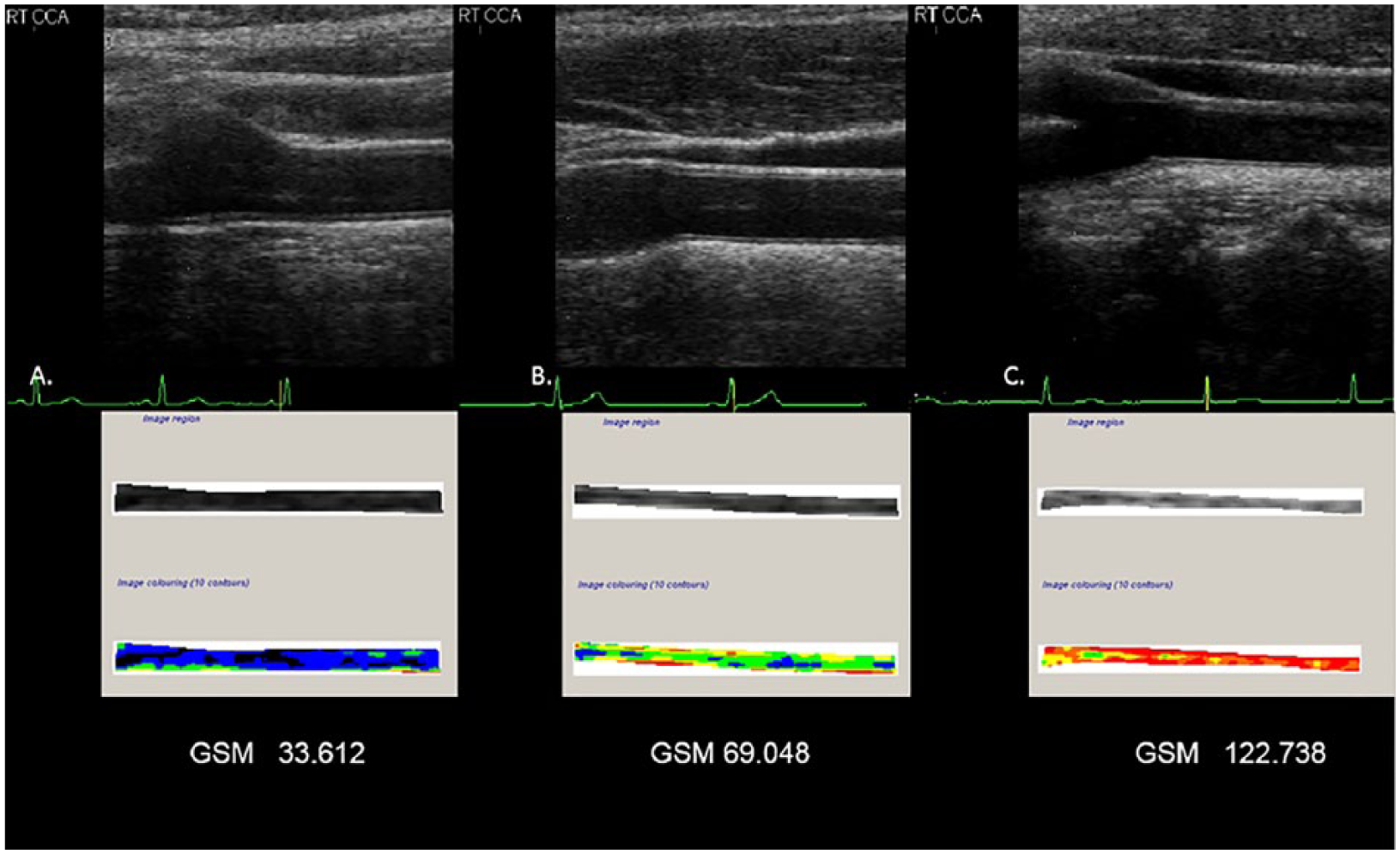
Sonographic image and corresponding gray-scale analysis of the arterial wall. (A) The normalized and standardized B-mode image and associated tracing of the far wall of a common carotid artery for gray-scale median (GSM). This artery is more echolucent. (B) The normalized and standardized B-mode image and associated tracing of the far wall of a common carotid artery for GSM. This artery is representative of the mean GSM value of the study cohort. (C) The normalized and standardized B-mode image and associated tracing of the far wall of a carotid artery for GSM. This artery is less echolucent. Image colorization (10 contours) depicts the far wall of a common carotid artery based on the local GSM value. Values of 0 to 25 units are displayed as black, 26 to 50 as blue, 51 to 75 as green, 76 to 100 as yellow, 101 to 125 as orange, and 126 to 255 as red. 40 .
Intra-reader measurement reproducibility for far wall CCA GSM measurements was excellent (r = 0.99 for 26 blinded paired readings, mean [standard deviation] absolute delta = 1.95 [1.22]). The intraclass correlation coefficient was 0.99. Inter-reader measurement reproducibility also was excellent (r = 0.98 for blinded paired readings, absolute delta = 2.29 [1.78] units with an intraclass correlation coefficient of 0.97).
Statistical Analysis
Statistical analyses were performed using SPSS (USA IBM Corporation, Armonk, NY, USA). Descriptive statistics were computed, and sex differences in GSM values were examined using Student’s t test. Pearson correlations were used to examine the relations between CCA GSM and smoking markers and CVD risk factors. We then conducted multivariable linear regressions with CCA GSM as the dependent variable for each smoking heaviness marker, adjusting for age, sex, and race (coded as white vs. nonwhite). Finally, we conducted multivariable linear regression to examine the impact of nonsmoking CVD risk factors (systolic blood pressure, total cholesterol, HDL-C, waist circumference, hsCRP, hemoglobin A1C, and use of antihypertensive and lipid-lowering medications) on CCA GSM. Because this was a pilot study based on image availability, no a priori power analysis was performed.
Results
Participant Characteristics
Participants had a mean age of 44.9 (11.4 SD) years old, and 61.7% were females (Table 1). They smoked 20.7 (7.5 SD) cigarettes/d and reported 27.5 (17.4 SD) pack-years of smoking. Their CO was 26.4 (13.3 SD) ppm, and their FTCD score was 5.2 (2.1 SD). These measures provide a description of the smoking burden on participants in this pilot study. Compared to definitions previously used to characterize an individual as a “light smoker” (<15 cigarettes/d), our participants smoked more cigarettes per day and would be considered “moderate” smokers. 23 Exhaled CO measures previously have been reported as 3.6 (2.15) ppm in healthy nonsmokers, 5.2 (3.4) ppm in passive smokers, and 17.13 (8.5) in ppm healthy smokers. 24 A FTCD score of 0 to 3 represents an individual with low dependence on nicotine, a score of 4 to 6 is associated with moderate dependence on nicotine, and a score of 7 to 10 is considered to be highly dependent on nicotine.17,24,25 For white and black Americans, a waist circumference greater than 35 inches for women and greater than 40 inches for men is indicative of high risk. 26
Baseline Participant (n = 162) Characteristics.
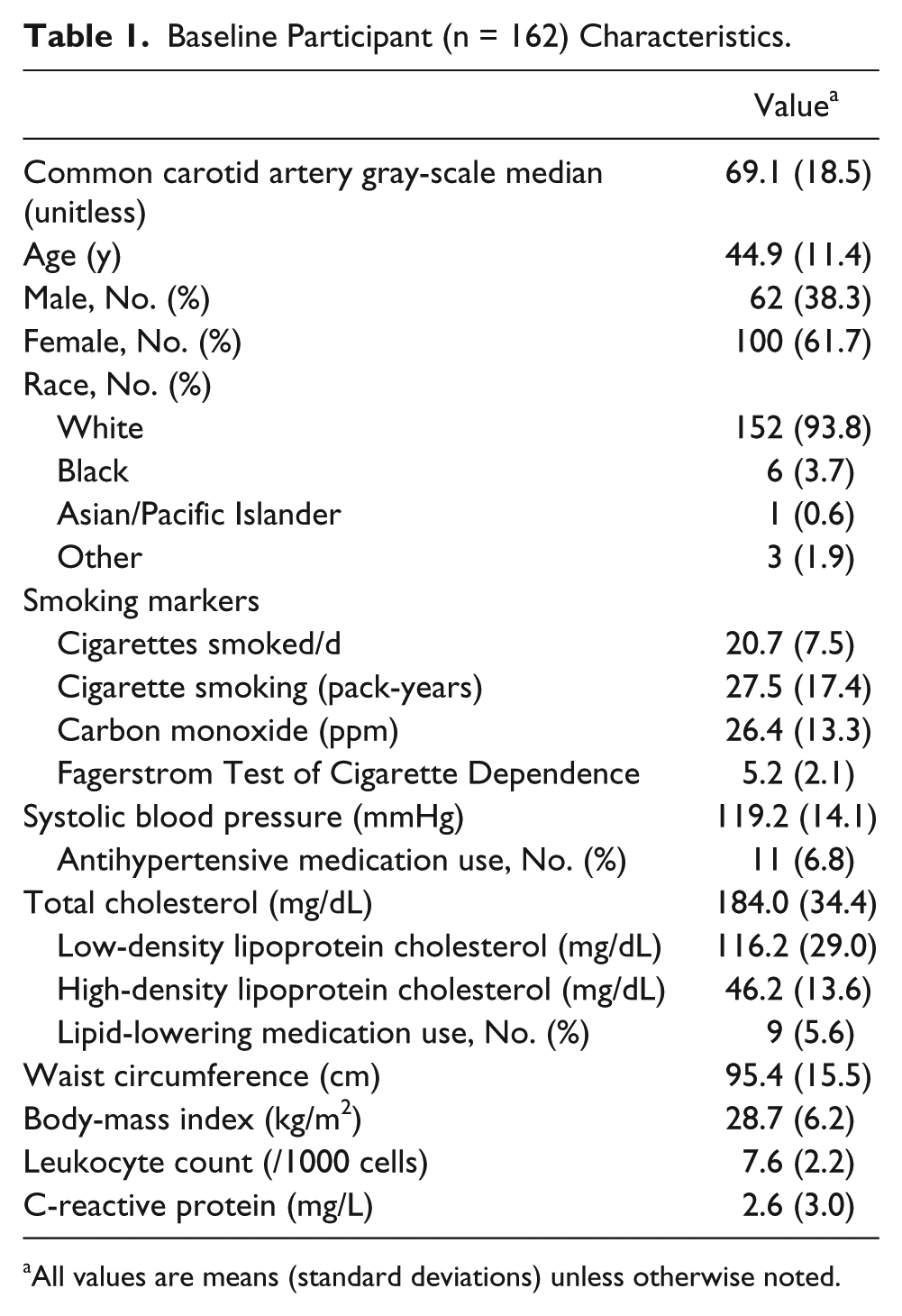
All values are means (standard deviations) unless otherwise noted.
Correlates of CCA GSM
The mean right CCA GSM was 69.1 (18.5) units. Mean CCA GSM was lower in men than in women (63.0 [16.5] vs. 72.9 [18.8], t = −3.40, P = .001). CCA GSM was correlated inversely with cigarettes/d (r = −0.16, P = .048) and pack-years (r = −0.20, P = .01) but not with CO or FTCD score (all P > .05). CCA GSM was inversely associated with CVD risk factors as expected: age (r = −0.26, P = .001), waist circumference (r = −0.38, P < .001), hemoglobin A1C (r = −0.18, P = .02), total cholesterol (r = −0.17, P = .03), and systolic blood pressure (r = −0.16, P = .047). HDL-C was positively correlated with CCA GSM (r = 0.33, P < .001). Leukocyte count, hsCRP, and heart rate were not correlated significantly with CCA GSM (all P > .05).
Multivariable Models of CCA GSM
Covariate models are used to determine if participant characteristics such as age and sex are confounders (variables that may affect the relationship being studied if not controlled for). 27 Multivariable models are used to assess relationships between a number of independent variables (in this study, CVD risk factors and measures of smoking heaviness) and an outcome, or dependent variable (in this study, GSM value). 28
In the covariate-only model, age (β = −0.22, P = .005) and sex (β = 0.23, P = .004) were significant predictors of CCA GSM, but race was not (β = −0.002, P = .98). After adjusting for age, sex, and race, none of the smoking heaviness markers predicted CCA GSM with statistical significance: cigarettes smoked/d (β = −0.08, P = .29), pack-years of smoking (β = −0.04, P = .68), and CO (β = 0.06, P = .41).
In the CVD risk factor multivariable model, we initially included systolic blood pressure, total cholesterol, HDL-C, waist circumference, hsCRP, hemoglobin A1C, and use of antihypertensive and lipid-lowering medications. Using a backward model-building procedure, the final model (adjusted R2 = 22.2%) adjusted for age (β = −0.20, P = .01), sex (β = 0.09, P = .29), and race (β = 0.01, P = .89) and included total cholesterol (β = −0.16, P = .04), HDL-C (β = 0.21, P = .01), and waist circumference (β = −0.20, P = .03) as the statistically significant predictors of CCA GSM.
Discussion
In this pilot study, we demonstrated the feasibility of measuring GSM and findings differences related to smoking burden in a cohort of active smokers. We observed inverse univariate associations between CCA GSM, a measure of carotid wall echogenicity, and two markers of smoking heaviness. Lower GSM values were associated with more cigarettes smoked per day and more years of smoking (pack-years). However, after adjusting for age, sex, and race/ethnicity, these markers of smoking heaviness no longer were associated independently with CCA GSM. Age, waist circumference, and HDL-C were the most consistent CVD risk factors associated with carotid wall echogenicity, supporting findings in previous studies that GSM values are associated with CVD risk factors such as age, dyslipidemia, and smoking.3,13,14,29 In this pilot study, the multivariable models were underpowered; however, the effects were in the correct direction, although the sizes were not very large relative to those of sex and age.
Pack-years, a marker of smoking heaviness, was a univariate associate with CCA GSM, but this association was no longer present in multivariable models that included age, likely because age and pack-years are collinear. Since pack-years will increase with age, it is difficult to determine the independent influences of age and pack-years in multivariable models, 30 especially with our relatively small sample size and wide range of ages. However, we also observed that CCA GSM values decreased with the number of cigarettes smoked per day, indicating that smoking may influence the echogenicity of the carotid arterial wall and that a larger sample size is needed to more thoroughly evaluate the independent contributions of smoking heaviness parameters and other CVD risk factors.
The GSM value is a measure of ultrasound echogenicity (brightness/darkness) of the carotid arterial wall.3,13,14,29,31 Gray-scale sonography interacts with tissues, and the reflected strength of the signal is indicative of tissue composition. 32 Therefore, it is believed that the shades of gray are representative of the tissue present in the arterial wall.3,13,14,33 Based on histopathological examination, lipids and hemorrhage appear as darker shades of gray (hypoechoic) and fibrous and calcific tissue as brighter shades of gray and white (hyperechoic) when imaged with ultrasound.20,34,35 Inflammation has been associated with both diffuse hypoechogenicity 36 and focal (discrete) white areas without acoustic shadowing.19,20 Abnormal levels of CVD risk factors are associated with altered arterial wall tissue composition (i.e., higher lipid content) and have different patterns of echogenicity measured by the GSM value.3,13,14,29 Lower CCA GSM values have been associated with CVD risk factors (older age, higher body mass index, lower HDL-C, higher LDL-C, oxidative stress, inflammation,3,13 and increased risk for all-cause and CVD mortality 14 ).
In a cross-sectional study of HIV-infected and noninfected women, current smoking was associated paradoxically with higher rather than lower CCA GSM. 33 This differs from our finding of lower GSM values being associated with markers of smoking heaviness; however, all participants in our study were smokers, so there are no comparisons to nonsmokers. Furthermore, our study examined relationships to markers of smoking heaviness (cigarettes smoked/d and pack-years).
This is the first study that we are aware of to report associations of smoking heaviness (cigarettes smoked/d and pack-years) with CCA echogenicity in a smoking cohort. However, these relations may have less impact on CCA echogenicity than gender and age. We also identified relationships between increasing age, total cholesterol, waist circumference, and low HDL-C with CCA GSM among active smokers, suggesting that being an overweight and dyslipidemic smoker may contribute to carotid wall injury. Indeed, secretion of adipocytokines from adipose tissue adversely influences cellular components of the arterial wall and has been postulated to be associated with echolucent plaques37–39 and thus could also be associated with carotid wall echogenicity, smoking heaviness, and central adiposity.
Limitations
This was a relatively small pilot study, a cross-sectional analysis restricted to current smokers intending to make a quit attempt. The novel associations we identified need to be reproduced in a larger, more racially diverse sample of smokers, and carotid GSM measures and their associates among smokers need to be compared to data from nonsmokers and former smokers. Measuring nontraditional CVD risk markers, including markers of oxidant stress and additional inflammatory markers, also may shed light on the pathobiology of arterial wall injury among smokers.
These images were acquired with the same preset (controlling for transducer, dynamic range, and gray-scale map); however, sonographers were allowed to optimize the time-gain compensation controls (TGC) and the overall gain. Ideally with this software, the TGC curve should be set to compensate for image depth but have the potentiometers (i.e., slide pods) at the same vertical setting in the arterial lumen.19,21,22,40,41 Since this was not controlled for, we are not sure what effect this may have on the results of this study.
Conclusions
This pilot study demonstrated the feasibility of measuring the GSM of the CCA far wall and preliminary associations with measures of smoking heaviness and traditional CVD risk factors. The independent associations identified in this study need to be explored in a larger cohort, longitudinally, and with additional biomarkers to determine the mechanisms underlying arterial injury with active smoking and how they change with cessation.
Footnotes
Authors’ Note
Clinical trial registration: https://clinicaltrials.gov/ct2/show/NCT01116986?term=NCT01116986&rank=1. Unique identifier: NCT01553084.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C. Mitchell: Davies Publishing, Inc., authorship for two echocardiography textbooks, currently under review, may have future royalties. Elsevier, Wolters-Kluwer, author textbook chapters, may have future royalties. J.H. Stein: Wisconsin Alumni Research Foundation patent related to carotid wall thickness and vascular age.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant 5R01HL109031 from the National Heart, Lung, and Blood Institute and a grant K05CA139871 from the National Cancer Institute to the University of Wisconsin Center for Tobacco Research and Intervention.
